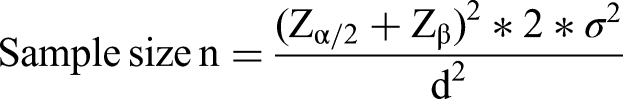Abstract
Introduction
Human immunodeficiency virus (HIV) infection has been associated with an increased risk of hypercoagulability. 1 The incidence of thrombotic complication in HIV-infected patients is estimated at 2.6 per 1000 person-years.2,3 Deep vein thrombosis (DVT) and pulmonary embolism (PE) account for about two-third of all thrombotic events in them. 4 The risk of deep vein thrombosis in patients with HIV infection has been estimated to be 2–10 fold higher than in the general population. 5 Other thrombotic complications such as coronary artery events (myocardial infarction) have also been reported in HIV-infected persons. 6
The interplay of the components of Virchow triad has been reported to play a role in the pathogenesis of thrombotic events in HIV infection. Venous stasis due to comorbidities, prolonged hospitalization and bed rest; vascular endothelial injury associated with endothelial dysfunction, dyslipidaemia and hypercoagulability due to presence of underlying HIV associated neoplasms, opportunistic infections, acquired deficiency of natural anticoagulant protein (protein C, protein S, antithrombin, antiphospholipid syndrome have all been implicated in the pathogenesis of thrombosis in HIV infection. 1 More recently in the era of highly active antiretroviral therapy (HAART), the use of antiretroviral agents has also been associated with hypercoagulability and therefore increased risk of thrombosis independent of the traditional risk factors. 7 With increased availability, access and use of HAART, thrombotic complications may increase in HIV-infected persons on therapy. Specifically, the use of protease inhibitors (PI) have been linked to thrombotic complications.7-9
PAI-1 is a regulating protein of fibrinolytic activities. It inhibits the effect of plasminogen activators, thereby inhibiting plasmin formation and down-regulating the breakdown of fibrin clots. 10 (Elevated levels of PAI-1 is associated with increased risk of cardiovascular complications in various clinical patients including diabetics and HIV-infected patients.11,12. There are paucity of studies in our environment on PAI levels in HIV-positive patients on therapy hence this study was be focused to compare PAI-1 levels in HIV-infected patients on PI-based HAART regimen with those not on any PI-based HAART therapy. It is anticipated that the outcome of the study may influence the choice of HAART combination use in patients with an increased risk of thrombosis. The study aims to evaluate and compare PAI-1 levels a marker of thrombotic risk in HIV-infected persons on PI-based HAART regimen with those on non-PI-based therapy and to correlate PAI-1 levels with their haematological parameters and CD4 counts.
Methodology
This was a comparative cross sectional study conducted at the University of Calabar Teaching Hospital (UCTH), Calabar, Cross River State. UCTH is a tertiary health institution located in Calabar Municipal and serves Cross River state and its neighboring states (Akwa Ibom, Ebonyi) as well as Cameroun. The hospital has a unit called “Special Clinic” that manages patient with HIV infection. The unit is under the care of a consultant Family Physician. Resident doctors in family medicine and medical officers are involved in running the unit. Family Health International (FHI) 360 is the non-governmental organization that provides technical support and oversees the unit activities. The unit has about 3000 HIV patients under its care.
Study Population
The study population consisted of three groups:
Group I: HIV-positive patients on protease inhibitor based HAART therapy. Group II: HIV-Positive patients on non-Protease inhibitor HAART Regimen. Group III: This comprised of controls. There were two subgroups of controls.
Apparently healthy HIV-negative subjects HIV-positive patient yet to commence therapy.
Sample Size
Sample size was calculated using the formula for comparison of two mean.
13
Using the standard deviation value of PAI-1 levels in HIV-infected patients on PI based HAART regimen. The mean and standard deviation values of PAI-1 in HIV-infected population in a study by Pirs et al 14 was used for sample size calculation.
σ = Standard deviation of PAI-1in HIV-infected patients on HAART Regimen – 5.84 IU/ml. The study was hypothesized to detect a mean difference of 5IU/ml.
n = 797.93/25
n = 32
Using an attrition of 10% attrition = 3.2, the estimated sample size was 35.2 for each group.
A total of 125 subjects were recruited. These include:
Forty-five (45) HIV infected patients on PI-based HAART regimen Forty-two (42) HIV-infected patients on non-PI-based HAART regimen Thirty eight controls including 18 HIV-infected therapy naïve subjects and 20 HIV-negative apparently healthy controls.
Using the clinic data, the HIV-infected patients on therapy were pooled into two groups based on the HAART combination. Stratified random sampling method was used to recruit the subjects using their medical record information accessed from the “Special Care Unit”. The selected subjects were invited, counseled on the study and consent obtained from them. For the HIV-infected therapy naïve participants, they were recruited consecutively through the outpatient clinic. The HIV-negative controls were recruited from the blood donor clinic.
Exclusion Criteria
Patients on fibrinolytic therapy (eg tranexamic acid), thrombolytic and antithrombotic agent were excluded. Patients with evidence of hepatic damage were excluded because the liver is a source of PAI-1. Patients who are critically ill including symptomatic anaemic subjects were excluded to minimize the risk of harm (from blood sampling or exertion) related to research activity.
Study Instrument
A questionnaire was used to collect data on the subjects personal information and history of disease/treatment including sex, age, year of diagnosis, duration on therapy, antiretroviral therapy combination, viral load, CD4 count. Medical history was obtained from patient directly and complemented with information that was obtained from their medical record. History of hypertension, diabetes mellitus, dyslipidaemia, and opportunistic infections was documented. A venous blood sample was collected for estimation of PAI-1 levels and blood counts.
Blood Sampling and Storage
From each subject, five millilitres (5 ml) of venous blood was collected from the ante-cubital vein with minimal stasis. Two and a half millilitres (2.5 ml) of whole blood was dispensed into a commercially prepared ethylene di-amine tetra-acetic acid (EDTA) bottle for basic haematological parameters, and 2.25 ml of whole blood for PAI-1 level estimation was dispensed into a plain sample bottle that contains 0.25 ml of 0.109 M sodium citrate (3.2%). This was to obtain a blood: citrate ratio of 9:1. The sample was centrifuged at 2000 rpm for 15 min. Thereafter, platelet-poor citrated plasma was extracted into a plain bottle and stored at −20 °C until it was analyzed.
Test Procedures
Haematological parameters: The Full blood count which included haemoglobin, white cell count, platelet counts and their indices were evaluated from the EDTA sample, using automated blood cell counter (Sysmex Haematology Autoanalyser model KN21). The basic principles underlying this technique are electronic impedance and light scatter. This was done in the main haematology laboratory, UCTH.
Plasminogen activator inhibitor 1 Assay: Assaypro enzyme-linked immunoassay kit was used for the estimation of PAI-1 levels.
CD4 count estimation: CD4 count was estimated with a FACS machine.
Statistical Analysis
The data was analyzed with the IBM Statistical Package for Social Sciences version 23 (Chicago). Sociodemographic parameters including age group, sex, educational status and marital status were summarized as simple frequency, proportions and percentages. Test for Normality was done on the continuous variables. The skewed variable (PAI-1) was summarized as median and interquartile range. Normally distributed variables (blood counts including CD4 count) were summarized as mean, standard deviation and ranges. Chi square test was used to compare categorical variables between the groups. Mann Whitney U test was used to compare median PAI-1 levels between the groups. Analysis of variance (ANOVA) was used to compare blood counts across the groups and the student t-test was used to compare blood counts between two groups. Pearson's correlation coefficient was used to correlate PAI-1 levels with blood count. P value was set at 0.05.
Result
A total of 125 subjects including 45 HIV positive subjects on PI-based highly active antiretroviral therapy (HAART), 42 HIV-positive patients on Non-PI based HAART, 18 HIV-positive treatment naïve and 20 HIV-negative subjects participated.
Table 1 shows the sociodemographic parameters of the subjects on PI-based and non-PI-based HAART regimen that participated in the study. The subjects in the age group of 30 −39years and 40–49 years were the highest proportion of participants accounting for 14 (31.1%) each of the patients on PI-based therapy. The peak age group of patients on Non-PI HAART regimen was 30–39 years, accounting for 13 (31.0%). The differences in age group distribution was not statistically significant (p = 0.440)
Sociodemographic Parameters of the Study Population

The patients on PI-based HAART include 34 (75.6%) females and those on non-PI-based HAART included 33 (78.6%) females. There was no significant difference in the gender distribution of those on PI and non-PI-based HAART therapy (p = 0.738). Thirteen (28.9%) of those on PI-based HAART were single, 29 (64.4%) were married as against 26 (61.9%) of the non-PI-based HAART that were single and 13 (31.0) married. The differences in proportions was statistically significant (p = 0.006).
The educational status of the study population is contained in Table 2. There was no significant difference in the educational status between patients on PI and Non-PI-based HAART regimen (p = 0.439).
Haematological Parameters of the Study Populations and the Controls.

Control Population
The controls include 18 HIV-positive patients yet to commence therapy (therapy naïve) and 20 apparently healthy controls. HIV-positive therapy naïve patients consist of 9 males and 9 (50%) females while the HIV-negative controls comprised 10 (50.0%) males and 10 (50.0%) females.
Duration on Therapy
The median duration on therapy for the patient on PI-based therapy was 8 years (interquartile range 4.0-13.0 years) while for those on Non-PI-based HAART, the median duration on therapy was 6 years (interquartile range 3.8-12.0 years). The differences in the median duration of therapy between both groups was not statistically significant (p = `0.324).
Plasminogen Activator Inhibitor 1 (PAI-1)
Figure 1 is a boxplot of the distribution of PAI-1 in study population. The median PAI-1 in patients on PI-based HAART was 6.1 ng/mL with an interquartile range of 2.6–22.5 ng/mL. Patients on non-PI-based HAART had a median of 2.8 ng/mL and interquartile range of 1.0–8.0 ng/mL. The therapy naïve group had a median of 2.0 and interquartile range of 1.0–3.9 ng/mL while the apparently healthy controls had a median of 2.5 ng/mL. The differences in median PAI-1 across the group was statistically significant (p = 0.004). The difference in median PAI-1 level between patients on PI-based HAART and those on Non-PI-based HAART, though high was not statistically significant (p = 0.052). The difference in median PAI-1 between patients on PI-based HAART and therapy naïve patients as well as healthy controls were statistically significant (p = 0.002 and p = 0.003 respectively). There were no significant difference between PAI-1 levels in patients on non-PI-based HAART regimen and those that are therapy naïve (p = 0.778) as well as apparently healthy controls (p = 0.786).
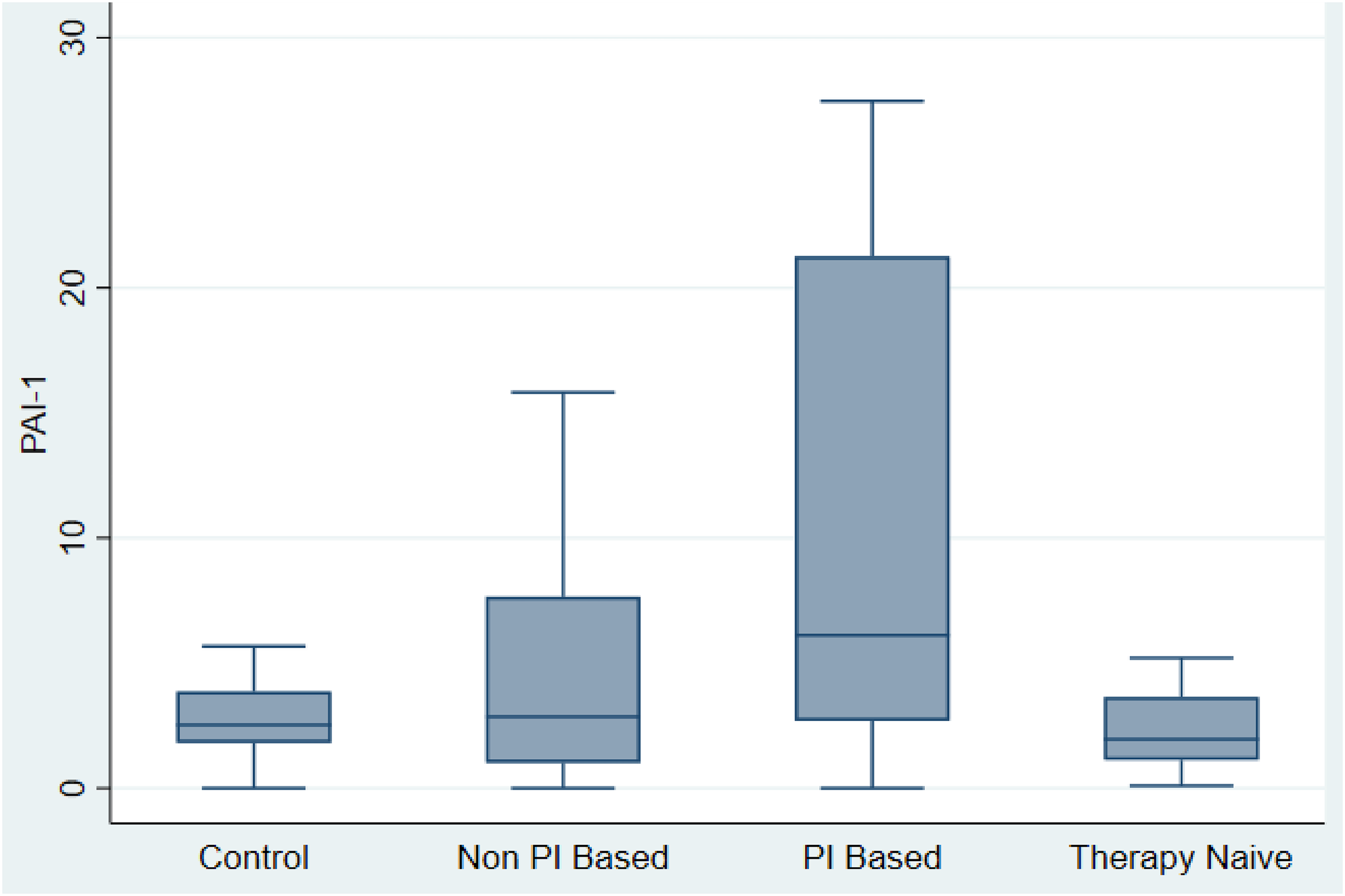
Boxplot of PAI-1 in the study Population.
Haematological Parameters of the Study Population and Controls
Table 2 shows the haematological of the study subjects and control populations. The mean haemoglobin was highest in those on non-PI-based HAART regimen (13.4 ± 2.3 g/L) and lowest in therapy naïve HIV-positive subjects (12.2 ± 1.7 g/L) however, the difference in mean across the group was not statistically significant (p = 0.132).
Mean corpuscular volume (MCV) was highest in patients on the Non-PI-based HAART regimen (93.4 ± 8.4fL) followed by those on PI-based HAART regimen (91.1 ± 10.1fL) and least in therapy naïve patients (84.8 ± 5.7fL). The difference in mean across the groups was statistically significant (p = 0.005). The average of mean corpuscular haemoglobin (MCH) and the mean corpuscular haemoglobin concentrations were significantly different across the groups p = 0.002 and p = 0.029 respectively.
White blood cell count was highest in patients on PI-based HAART regimen (5.9 ± 2.5 × 109/L) followed by those on non-PI-based combinations (5.6 ± 1.7 × 109/L). The difference in mean across the groups was statistically significant (p = 0.024). There was a statistically significant difference in the mean of lymphocytes and neutrophils across the groups (p = 0.040 and p = 0.033) respectively. There was no significant difference in monocyte counts across the groups (p = 0.702). The platelet count was highest in patients on PI-based HAART regimen however the difference in mean across the groups was not statistically significant (p = 0.234).
The CD4 count was highest in the apparently healthy control population with a mean of 1146.3 ± 220.1 cells/L, followed by patients on PI-based HAART regimen (552.0 ± 325.0 cells/µL) and then those on non-PI-based combinations (535.8 ± 353.2 cells/µL). The differences in mean was statistically significant (p < 0.001)
Table 3 shows haematological parameters between patients on PI-based combinations and those on non-PI-based combinations. There were no significant differences in all the haematological parameters between the two groups (p >0.05).
Comparison of Haematological Parameters Between Patients on PI Based HAART Combination and Non-PI Based HAART Combination.

Table 4 shows the mean, standard deviation, ranges and comparison of haematological parameters between patients on PI-based combinations and HIV-positive therapy naïve patients
Comparison of Haematological Parameters Between Patients on PI Based HAART Combination and HIV Positive Therapy Naïve Patients.

The total WBC count and neutrophil count were higher in patients on PI-based combinations (p = 0.039 and p = 0.030 respectively). Monocyte and lymphocyte counts did not differ significantly between them (p > 0.05). Similarly, platelet count did not differ significantly between them (p = 0.290). The CD4 count was significantly lower in the HIV-positive therapy naïve population (p = 0.017).
Table 5 shows the mean, standard deviation, ranges and comparison of haematological parameters between patients on PI-based combinations and apparently healthy controls
Comparison of Haematological Parameters Between Patients on PI Based HAART Combination and Apparently Healthy Controls
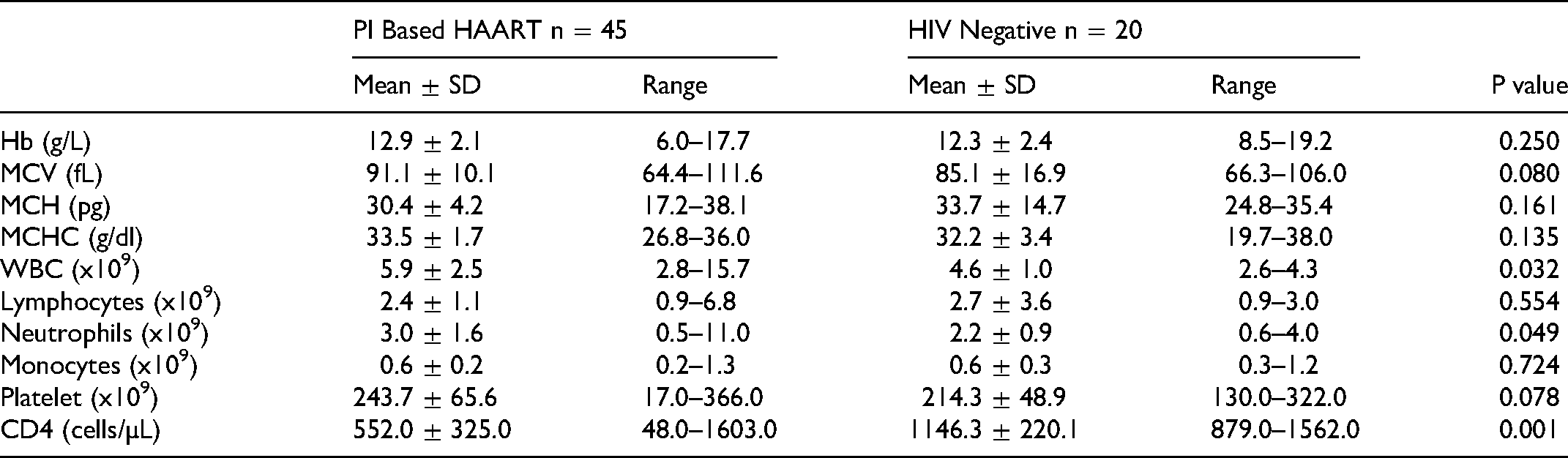
Table 6 shows the mean, standard deviation, ranges and comparison of haematological parameters between patients on non-PI-based combinations and HIV-positive therapy naïve patients. The red cell indices (MCV, MCH and MCHC) were significantly higher in patients on non-PI-based combinations compared to therapy naïve patients (p < 0.05). Similarly, the Non-PI-based group had significantly higher WBC and lymphocytes (p = 0.038 and p = 0.029 respectively). The CD4 count was also significantly higher in the therapy naïve group (p = 0.038).
Haematological Parameters of Patients on Non-PI Based HAART and HIV Negative Healthy Controls

Table 7 compares the mean haematological parameters between patients on non-PI-based therapy and HIV-negative healthy controls. The haemoglobin, WBC and platelet do not differ significantly between patients on non-PI-based antiretroviral combination and healthy controls (p > 0.05). The red cell index (MCV) and lymphocyte counts were significantly higher in patients on non-PI-based combinations (p = 0.012 and p = 0.032 respectively). The CD4 count was significantly higher in the HIV-negative controls compared to patients on non-PI-based antiretroviral therapy (p < 0.001).
Haematological Parameters of Patients on non-PI based HAART and HIV Negative Healthy Controls

Correlations Between PAI – 1 and Haematological Parameters in Patients on PI and Non-PI Based HAART
Table 8 shows the correlation between PAI-1 and haematological parameters in patients on PI-based HAART regimen and those on non-PI-based HAART combination therapy. There was a negative correlation between PAI-1 and haemoglobin in patients on PI and non-PI based-HAART combinations however, these were not statistically significant (r = -0.150, p = 0.331 and r = -0.157 and p = 0.321 respectively).
Correlations Between PAI – 1 and Haematological Parameters in Patients on PI and Non-PI Based Antiretroviral Combination Therapies
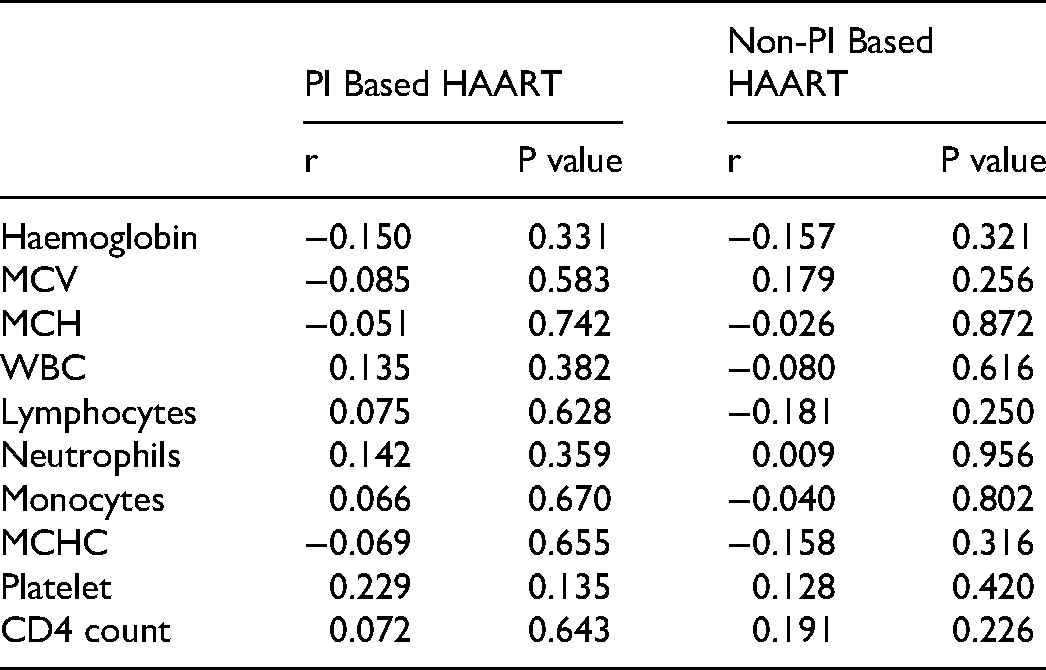
There was a positive correlation between PAI-1 and WBC count in the patients on PI-based HAART combination therapy (r = 0.135, p = 0.382) but there was no significant correlation in patients on non-PI-based HAART regimen (r = -0.080, p = 0.616).
There were positive correlations between PAI-1 and platelet counts in patients on PI-based and non-PI-based HAART combination therapies however, these were also not statistically significant (r = 0.229, p = 0.135 and r = 0.128 and p = 0.420 respectively).
PAI-1 correlated positively with CD4 count in patients on PI-based and non-PI-based HAART regimen but these were not statistically significant (r = 0.072, p = 0.643 and r = 0.191 and p = 0.226 respectively).
Discussion
The use of antiretroviral agents especially protease inhibitor based combination therapies have been associated with alteration in haemostatic parameters which may tilt the haemostatic balance towards hypercoagulability. There is paucity of literature in this regard in our environment. Hence this study was focused to evaluate fibrinolytic activity using plasminogen activator inhibitor 1 (PAI-1) as a marker in patients on PI-based HAART combination therapies.
The findings of the study include elevated PAI-1 level in patients on PI-based HAART compared those on non-PI based HAART though this did not reach statistical significance. Patients on PI-based HAART had significantly elevated PAI-1 level compared to therapy naïve and apparently healthy controls. This finding suggests that use of protease inhibitor-based HAART combination may have an inhibitory effect on fibrinolysis due to the associated elevation of antifibrinolytic molecule plasminogen activator inhibitor-1 in circulation. The hypofibrinolytic state may thus predispose to hypercoagulability. This is consistent with the findings of Koppel et al, 15 and Knudsen et al 12 in their separate studies. However unlike Knudsen who found an association between PAI-1 and risk of myocardial ischaemia, the subjects in the index study did not report any ischaemic event.
The risk of thromboembolic event may be predicted by duration of use of the agent and the presence of other traditional risk factors/comorbidities for thrombosis. Young et al, 16 evaluated changes in tissue plasminogen activator and PAI-1 in patients on PI-based HAART, and observed that while there was a reduction of tPA in the short term, there was no change in the PAI-1 levels. This suggests that the use of medication for a considerable duration is required to bring about the clinically observable effect associated with elevated PAI-1 levels in the patients. Padilla et al 17 reported that a change in PAI-1 level is detectable at about 12 weeks after commencing therapy. The minimum duration of use of PI based agent as reported by the study subjects in the index study was 13 weeks. The relatively short duration of exposure may account for inability to establish thrombotic event in the study population.
PAI-1 level did not differ significantly between patients on non-PI-based HAART and the controls (including therapy naïve and healthy controls). There is a paucity of study in the literature on PAI-1 levels in patients on non-PI-based HAART regimen. Pirs et al 14 reported no significant difference in PAI-1 levels between HIV-positive patients on therapy and therapy naïve subjects in their study. However, they were not specific with regard to the composition of HIV therapy combinations used by subjects in their study. While patients on non-PI-based HAART had higher median PAI-1 levels, it did not reach statistical significance in the index study. They also reported a significant difference in PAI-1 levels between patients on therapy and HIV-negative controls, as well as between therapy naïve and HIV-negative controls. This was contrary to the findings of the index study. In this study, PAI-1 levels in patients on the non-PI-based combination and those of therapy naïve patients did not differ significantly from the controls.
Haematological parameters did not differ significantly between the patients on PI-based HAART and those on Non-PI-based combinations. The haematocrit, white blood cell count, and platelets counts were within reference values in both groups and their mean CD4 counts were above 500cells/µL. This suggests that the effect of the PI-based combinations on the haematological parameters including CD4 counts is optimal and comparable to those of the non-PI-based HAART regimen. Protease inhibitors have not been reported to have significant effect on haemtological parameters, rather they are associated more with metabolic adverse events.18,19
Compared to the HIV-positive therapy naïve group, patients on the PI-based HAART regimen had no significant difference in haemoglobin levels and platelet count but the mean white blood cell count and CD4 counts were significantly higher in patients the on PI-based combination group. Similarly, the haemoglobin and platelet count in patients on non-PI based combinations did not differ significantly from those of therapy-naïve HIV-positive patients while white blood cell count and CD4 counts were significantly higher in the patients on non-PI-based HAART combinations. The lack of significant difference in haematocrit and platelet counts may be attributed to early diagnosis and treatment of infected persons before they develop significant immunosuppression. Increased awareness, as well as increased opportunities for counselling and testing and the recent policy of diagnosis and treatment, makes it possible for patients to receive appropriate care early in the disease process before the onset of clinically significant morbidity associated with the disease.20,21 The relatively higher CD4 and white cell count may be attributed to immune recovery following the commencement of HAART. 22
Patients on PI-based HAART therapy were found to have similar haemoglobin levels compared to HIV-negative controls but they had a significantly higher white blood cells and a significantly reduced CD4 count compared to the controls. The relatively higher white cell count may be attributed to inflammatory response associated with immune reconstitution in the HIV-positive patients on therapy. The difference is CD4 counts between them may be due to the relative depletion of their immune cells (CD4) occasioned by the disease process.23-25
HIV-negative controls when compared to patients on non-PI-based HAART were found to have significantly higher lymphocyte counts including the CD4 count. Understandably the HIV-negative participants are more immunocompetent thus the higher lymphocyte and CD4 counts observed in them. 23-25
There was no significant correlation between haematological parameters, CD4 counts and PAI-1 levels in the two major study groups (PI-based HAART and non-PI-based HAART groups). There is a paucity of literature in this regard. Future studies in this domain will shed more light on the relationship between PAI-1 and other haematological parameters especially, in the context of protease inhibitor-based therapy.
None of the study participants had a thromboembolic event thus the association of the changes in PAI-1 level with the thromboembolic event, could not be evaluated. However a number of studies on PAI-1 levels in HIV patients on HAART have found significant associations.7,26,27
The study has some strengths and limitations. This is the first study of PAI-1 in HIV-positive patients in our environment and may serve as possible strength of the study. However, limitations include the inability to establish any association between altered PAI-1 levels and thrombosis in the study population. There are heterogeneous combinations of HAART regimen in the study population, and the altered PAI −1 level may not be attributed to a specific agent in the regimen. The study could not establish any association between haematological parameters and thrombotic events. This was largely because none of the participants had any thrombotic event. However, Jackson et al reported lower haemoglobin concentration and elevated white blood cell counts to be associated with deep vein thrombosis in HIV-positive patients. 28
In conclusion, PAI-1 level is significantly increased in patients on PI-based HAART therapy. There was no significant difference in haematological parameters between patients on PI-based HAART and those on non-PI based HAART combination therapies. There were no significant correlation between PAI-1 level and haematological parameters in patients on antiretroviral therapy.
Recommendations
Further studies on the association of PI based HAART regimen and thrombosis is needed. A case control study may be desirable due to low incidence of thrombosis observed in the study population.
Caution should be exercise in the use of PI-based HAART in patients with history of thrombosis or other markers of hypercoagulability while PAI-1 assay may be useful in predicting risk of thrombosis or recurrence.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval to report this study was obtained from the University of Calabar Teaching Hospital Institutional Review Committee (UCTH/NREC/33/605). Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.