Abstract
We aimed to determine plasma thrombin-activatable fibrinolysis inhibitor (TAFI) antigen levels in women with previous gestational diabetes mellitus (GDM) and to evaluate the possible association of plasma TAFI with glucose intolerance and markers of subclinical atherosclerosis. This cross-sectional study was performed in 111 women with previous GDM and 60 controls. Glucose intolerance was evaluated. Homeostasis model assessment score was calculated. Circulating lipids, interleukin-6, matrix metalloproteinase-1, fibrinogen, plasminogen activator inhibitor-1, and TAFI antigen levels were assayed. Carotid intima media thickness (IMT) was measured. Women with previous GDM had increased levels of atherosclerosis markers and carotid IMT. On the other hand, plasma TAFI antigen levels were similar (P = .395). Thrombin-activatable fibrinolysis inhibitor was not associated with the indices of insulin resistance, glucose intolerance, markers of atherosclerosis, and carotid IMT. Our data demonstrated that plasma TAFI was not altered in women with previous GDM. TAFI was not associated with glucose intolerance and subclinical atherosclerosis.
Introduction
The coagulation cascade is initiated by tissue factor and factor FVIIa and generates thrombin, which promotes the conversion of fibrinogen to fibrin. Conversion of plasminogen to plasmin by tissue-type plasminogen activator (t-PA) leads to plasmin-mediated fibrinolysis. Fibrinolysis induces the solubilization of fibrin clot and results in clot lysis and fibrin degradation products. There are several inhibitors of fibrinolytic system. Plasminogen activator inhibitor-1 (PAI-1) is the major inhibitor of fibrinolytic system and it downregulates the fibrinolytic system primarily by inhibiting t-PA. 1 Inhibition of plasminogen activation by PAI-1 impairs fibrinolysis and promotes thrombosis. 2 It is well-established that hypofibrinolysis promotes atherosclerosis in insulin-resistant participants. Increased concentrations of fibrinolytic system proteins have been proposed to be predictors of cardiovascular events. 3 Plasminogen activator inhibitor-1 is elevated in a variety of clinical situations that are associated with insulin resistance and increased risk of ischemic cardiovascular events. 2
Women with previous gestational diabetes mellitus (GDM) are at high risk for developing diabetes, cardiovascular disorders, hypertension, dyslipidemia, and metabolic syndrome.4,5 It has been shown that patients with previous GDM have increased carotid intima media thickness (IMT), a noninvasive assessment for early atherosclerosis. 6 Previously, we 7 and others 8 demonstrated increased carotid IMT in Turkish women with previous GDM. Previous GDM has also been associated with subclinical inflammation and fibrinolytic system disturbance. 9 Several studies have demonstrated elevated plasma PAI-1 antigen levels in women with previous GDM.7,9,10
Thrombin-activatable fibrinolysis inhibitor (TAFI) is a novel fibrinolysis inhibitor isolated from human plasma. Thrombin-activatable fibrinolysis inhibitor antigen is a glycoprotein synthesized by the liver that can be activated by thrombin-catalyzed proteolysis to a carboxypeptidase B-like enzyme that inhibits fibrinolysis. Thrombin-activatable fibrinolysis inhibitor cleaves the C-terminal lysine residues of fibrin and subsequently blocks the activation of plasminogen by fibrin. Thrombin-activatable fibrinolysis inhibitor has also suppressor effect on conversion of inactive plasminogen to plasmin.11,12
There is very limited data in literature concerning the effect of glucose intolerance on plasma TAFI levels. Studies of TAFI levels in diabetes have revealed controversial results. While some investigators proposed a strong relationship between TAFI and diabetes,13–15 several recent reports indicated that TAFI levels were not affected by glucose intolerance and/or insulin resistance.16,17 In a recent study, we showed that GDM had no additional effect on TAFI antigen levels beyond pregnancy. 18 Here, first time in literature, we attempted to determine TAFI antigen levels in women with previous GDM. Furthermore, we intend to evaluate the possible association of plasma TAFI antigen levels with glucose intolerance and the markers of subclinical atherosclerosis in women with previous GDM.
Materials and Methods
A total of 130 consecutive women with previous GDM were included in this cross-sectional study. After exclusion of 19 women (6 with newly diagnosed hypertension, 5 with body mass index [BMI] >35 kg/m2, and 8 using medications listed in the exclusion criteria), finally, 111 women were enrolled. In women with previous GDM, GDM had been diagnosed by screening with a 50-g 1-hour glucose challenge test at 24 to 28 weeks of gestation, followed by a 100-g 3-hour oral glucose tolerance test (OGTT) at Dokuz Eylul University Hospital between January 2002 and January 2008. The diagnosis had been established according to criteria of American Diabetes Association (ADA) for GDM 19 (equal to or exceeding 95, 180, 155, and 140 mg/dL, with at least 2 elevated values considered abnormal). Sixty age-matched healthy women were included as control group. The control group was generated from healthy women who had experienced a normal pregnancy during the same period. In all control participants, GDM had been excluded by a 50-g screening test between 24 and 28 weeks of gestation at Dokuz Eylul University.
Informed consent was obtained in all cases, and the study was approved by the local ethics committee of Dokuz Eylul University.
Height (m) and weight (kg) were measured under fasting conditions with participants in light clothing and without shoes. Body mass index was calculated as body weight divided by square height. Waist circumference was measured from the mid-level between iliac crest and the lowest rib.
Blood was taken from the cannulated antecubital vein between 8:00
Glucose levels were measured by a colorimetric method with the Roche/Hitachi D/P Modular System Autoanalyzer (Roche Diagnostics, Basel, Switzerland). Triglycerides, total cholesterol, and high-density lipoprotein (HDL) cholesterol were measured by Roche/Hitachi D/P Modular System Autoanalyzer (Roche Diagnostics, Basel, Switzerland). Low-density lipoprotein (LDL) cholesterol was calculated by the Friedewald’s equation method. 20 Insulin levels were measured by a chemiluminescent method using an automatic immunoanalyzer (Roche Diagnostics, Mannheim, Germany). Homeostasis model assessment (HOMA) score, an estimate of insulin resistance, was calculated as fasting serum insulin (mU/L) × fasting plasma glucose (mmol/L)/22.5.
Plasma fibrinogen was analyzed by the Clauss method. 21 Interleukin 6 (IL-6) levels were measured with an ELISA kit (Invitrogen, Grand Island, New York). Experiments were performed according to the manufacturer’s instructions. Intra- and inter-assay coefficients of variations for IL-6 were 7.2% and 5.4%. Plasminogen activator inhibitor-1 antigen was measured by ELISA (Bender Medical Systems, Vienna, Austria). Intra-assay and inter-assay coefficients of variations were 4.7% and 5.0%, respectively. Serum matrix metalloproteinase-1 (pro-MMP-1) levels were measured using an ELISA kit (R&D Systems, Minneapolis, Minnesota). Intra- and inter-assay coefficients of variations for pro-MMP-1 were 6.1% and 7.5%. Plasma TAFI antigen assay was performed with ELISA (American Diagnostica, Stamford, Connecticut). Intra-assay and inter-assay coefficients of variation were 5.8% and 8% for TAFI. The lower limit of detection was less than 5% with a standard range of 0% to 150%.
Ultrasounds for carotid IMT were performed after 10-hour overnight fasting between 08.00
Sample size determination used a significance level of .05 for a 2-tailed test with 80% power. The sample size was calculated using Power and Precision software (Biostat, Englewood, New Jersey). Variable distributions were assessed by the Kolmogorov-Smirnov normality test. Independent sample t-test was used to compare variables of patients. One-way ANOVA with Bonferroni correction was applied for the comparison of groups. Categorical variables were compared by the chi-square test. Correlation analyses were performed using Pearson’s coefficients. Regression analysis was employed to assess correlations between studied parameters. Statistical analysis was performed using Statistical Package of Social Science (SPSS Inc, Chicago, Illinois), version 15.0 for Windows. Data were expressed as mean ± standard deviation (SD). A P value less than .05 was accepted as statistically significant.
Results
Table 1 summarizes characteristics of women with previous GDM and age-matched controls. There was no significant difference in terms of the duration of postpartum follow-up. The proportion of smokers was similar. As expected, women with previous GDM had elevated BMI and waist circumference and increased glucose levels on OGTT. Homeostasis model assessment score was significantly higher in women with previous GDM than controls. Women with previous GDM had more atherogenic lipid profiles. Women with previous GDM had significantly increased IL-6, pro-MMP-1 levels, and carotid IMT measurements. Women with previous GDM also had higher plasma fibrinogen (4.03 ± 0.85 g/L vs 3.49 ± 0.65 g/L, P < .001) and PAI-1 levels (138.36 ± 43.47 ng/mL vs 124.51 ± 36.71 ng/mL, P = .037) than controls (Figure 1a and b). On the other hand, there was no significant difference in TAFI levels between the 2 groups (93.81 ± 27.11 % vs 97.83 ± 33.43%, P = .395; Figure 1c).
Characteristics of Patients With Previous GDM and Age-Matched Healthy Controls
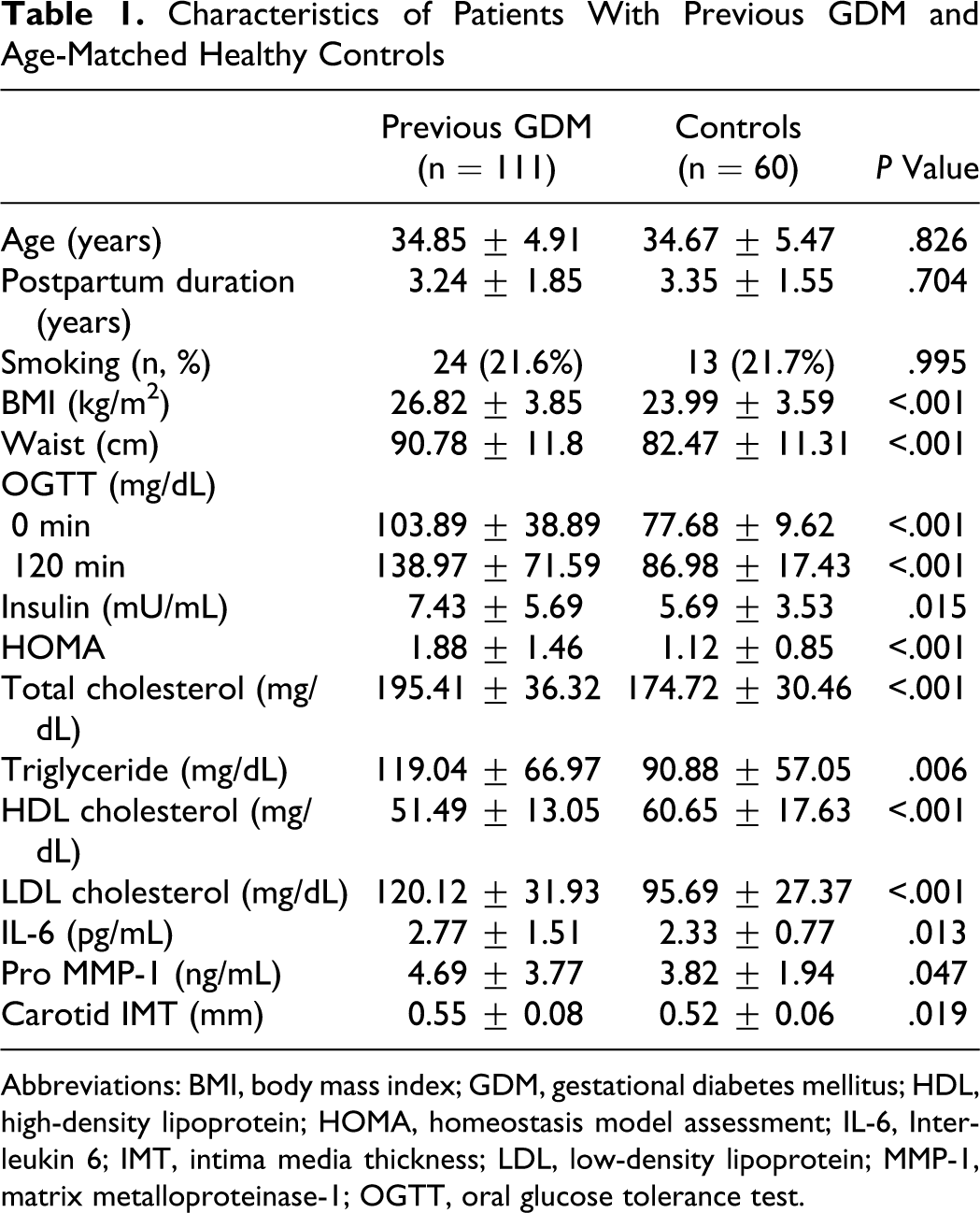
Abbreviations: BMI, body mass index; GDM, gestational diabetes mellitus; HDL, high-density lipoprotein; HOMA, homeostasis model assessment; IL-6, Interleukin 6; IMT, intima media thickness; LDL, low-density lipoprotein; MMP-1, matrix metalloproteinase-1; OGTT, oral glucose tolerance test.
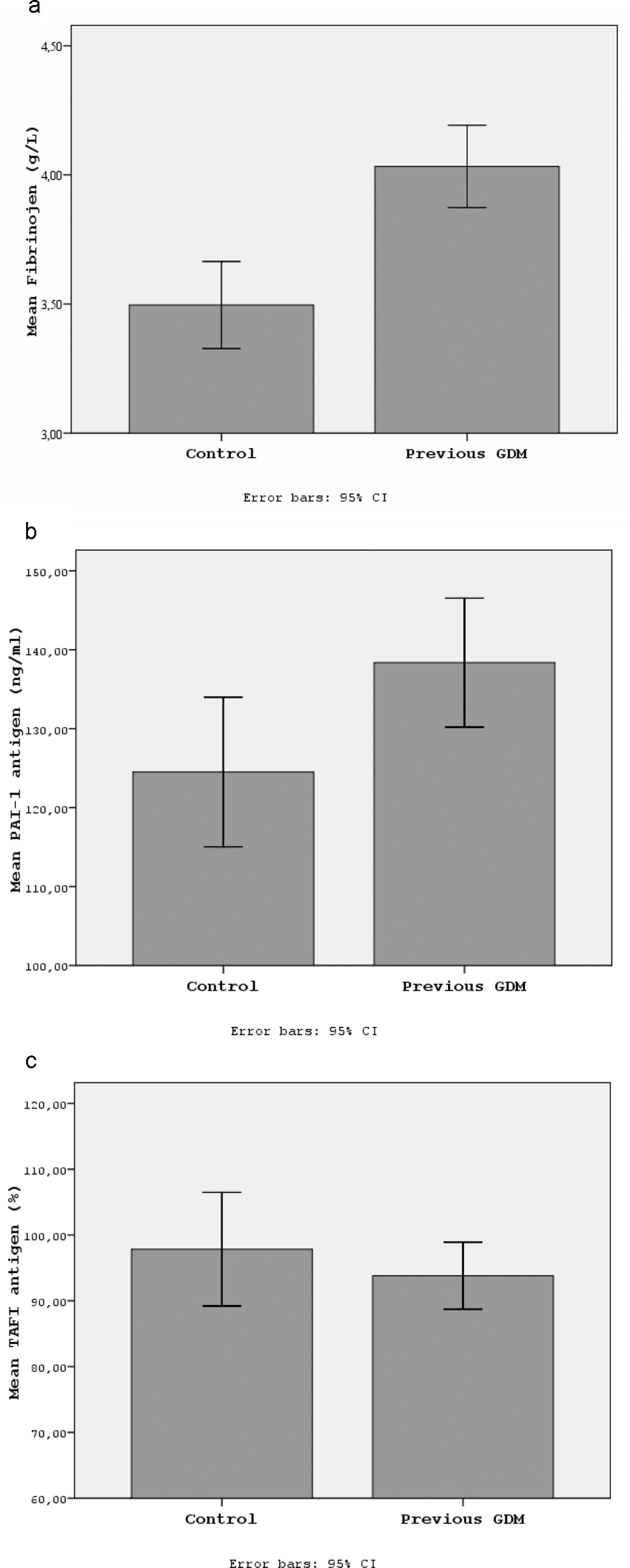
The comparison of the fibrinolytic markers in women with previous gestational diabetes mellitus (GDM) and healthy controls (a) Fibrinogen, (b) plasminogen activator inhibitor-1 (PAI-1), and (c) thrombin-activatable fibrinolysis inhibitor (TAFI).
Among 111 women with previous GDM, 17 (15.32 %) women were diagnosed with postpartum diabetes. While 56 (50.45 %) women had normal glucose tolerance, 38 (34.23 %) women had either IFG and/or IGT (Table 2 ). Women with diabetes or IFG/IGT had elevated BMI than women with normal glucose tolerance. However, probably due to small number of participants developing diabetes, the difference between women with diabetes and those with normal glucose tolerance was not statistically significant (P = .413 for diabetes vs normal glucose tolerance, P = .036 for IFG/IGT vs normal glucose tolerance). Women developing postpartum diabetes or IFG/IGT had higher HOMA scores (P = .071 for diabetes vs normal glucose tolerance, P = .008 for IFG/IGT vs normal glucose tolerance). Lipid levels, except HDL cholesterol (P = .03), were similar (P = .614 for total cholesterol, P = .127 for triglyceride, P = .887 for LDL cholesterol).
The Comparison of Women With Postpartum Diabetes, IFG/IGT, and Normal Glucose Tolerance a
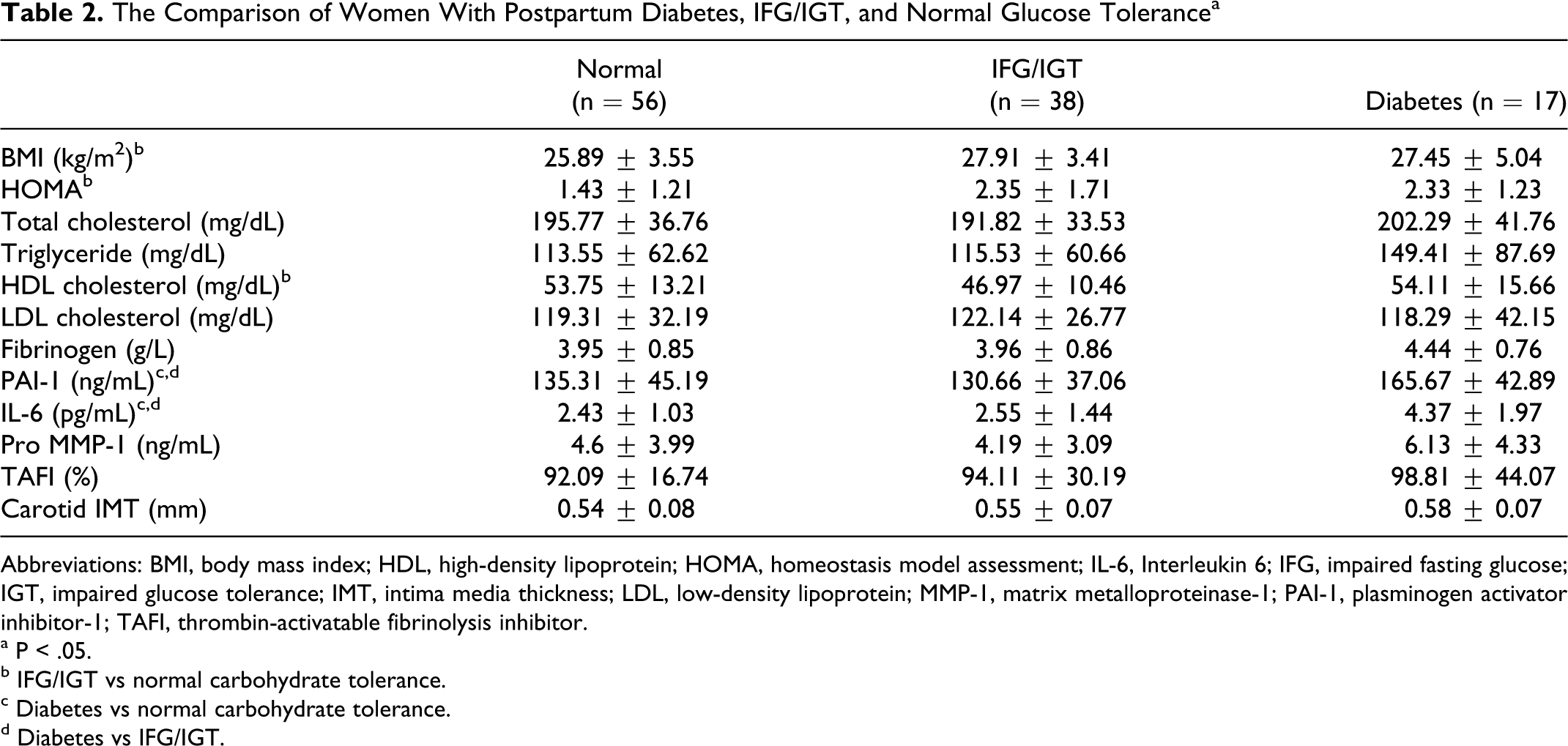
Abbreviations: BMI, body mass index; HDL, high-density lipoprotein; HOMA, homeostasis model assessment; IL-6, Interleukin 6; IFG, impaired fasting glucose; IGT, impaired glucose tolerance; IMT, intima media thickness; LDL, low-density lipoprotein; MMP-1, matrix metalloproteinase-1; PAI-1, plasminogen activator inhibitor-1; TAFI, thrombin-activatable fibrinolysis inhibitor.
a P < .05.
b IFG/IGT vs normal carbohydrate tolerance.
c Diabetes vs normal carbohydrate tolerance.
d Diabetes vs IFG/IGT.
In women with previous GDM, there was no significant difference in plasma TAFI levels when subgroups were analyzed in terms of postpartum carbohydrate intolerance (P = .671, Table 2). In the same way, TAFI levels of women with previous GDM developing diabetes were similar to that of healthy controls (98.81% ± 44.07% vs 97.83 ± 33.43 %, P = .922). On the other hand, women with previous GDM developing diabetes had significantly elevated levels of fibrinogen (4.44 ± 0.76 g/L vs 3.49 ± 0.65 g/L, P < .001), PAI-1 (165.67 ± 42.89 ng/mL vs 124.51 ± 36.71 ng/ml, P < .001), IL-6 (4.37 ± 1.97 pg/mL vs 2.33 ± 0.77 pg/mL, P < .001), pro-MMP-1 (6.13 ± 4.33 ng/mL vs 3.82 ± 1.94 ng/mL, P = .046) and carotid IMT (0.58 ± 0.07 mm vs 0.52 ± 0.06 mm, P = .002) compared to healthy controls. In women with previous GDM, women developing diabetes had significantly increased levels of IL-6 (P < .001 vs normal glucose tolerance, P < .001 vs IFG/IGT) and PAI-1 (P = .032 vs normal glucose tolerance, P = .016 vs IFG/IGT). Although women with postpartum diabetes had slightly elevated fibrinogen and pro-MMP-1 levels and carotid IMT measurements, these differences were not statistically significant (P = .1 for fibrinogen, P = .203 for pro-MMP-1, and P = .155 for carotid IMT).
In order to determine the impact of previous GDM per se independent of obesity and glucose intolerance on studied parameters, further analysis was performed matching the participants and controls for BMI and excluding women who had glucose intolerance. The characteristics of these matched participants and controls are presented in Table 3 . There was no significant difference between the participants and controls for any of the studied parameters, except plasma fibrinogen levels which remained significantly higher in women with previous GDM with normal glucose tolerance than age- and BMI-matched healthy controls.
The Comparison of Women With Normoglycemic Previous GDM and Age- and BMI-Matched Healthy Controls
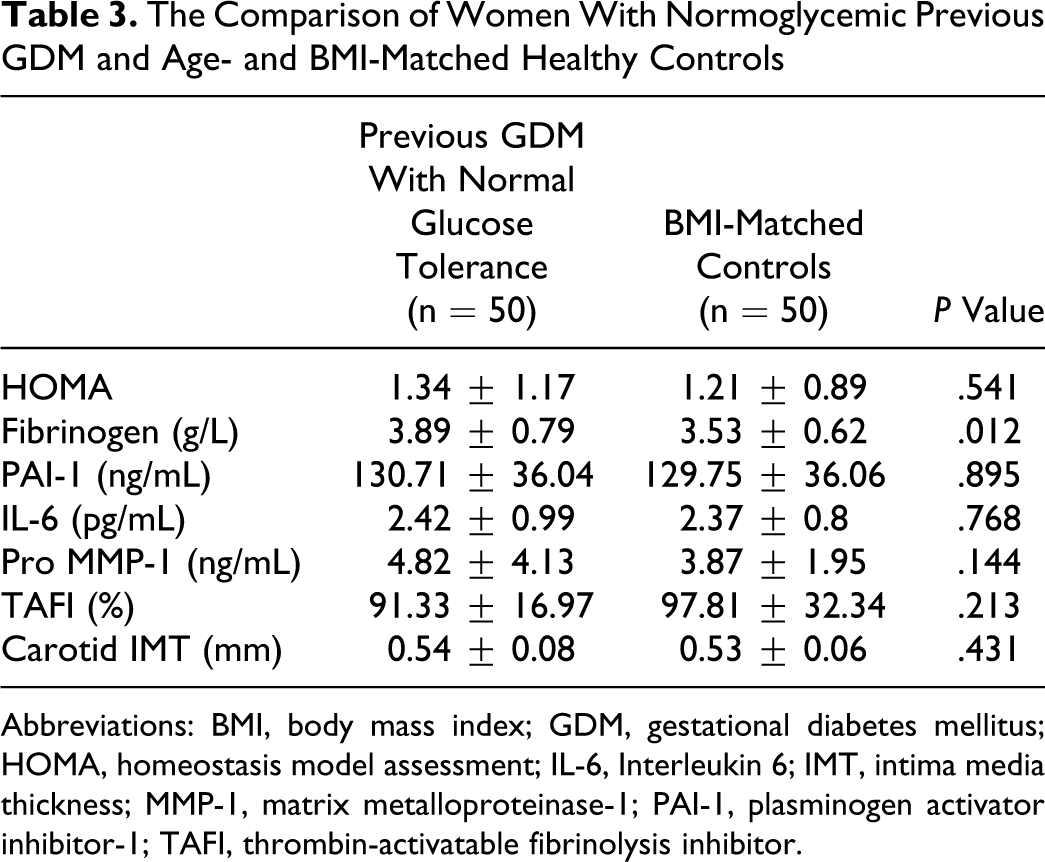
Abbreviations: BMI, body mass index; GDM, gestational diabetes mellitus; HOMA, homeostasis model assessment; IL-6, Interleukin 6; IMT, intima media thickness; MMP-1, matrix metalloproteinase-1; PAI-1, plasminogen activator inhibitor-1; TAFI, thrombin-activatable fibrinolysis inhibitor.
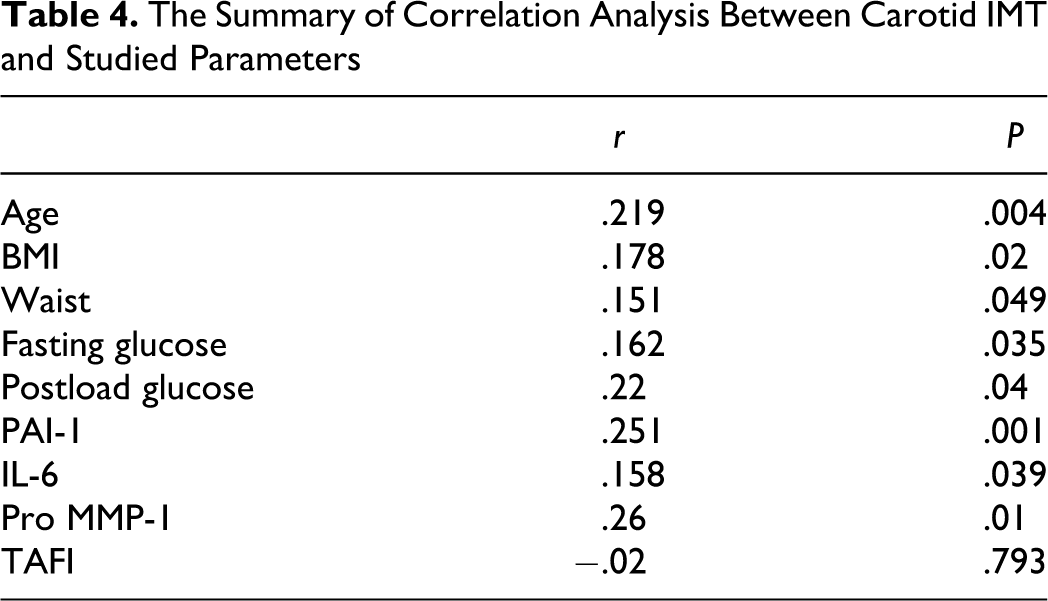
There was no significant correlation between plasma TAFI and studied parameters. We did not observe any significant association between the investigated parameters and the duration of postpartum follow-up. On the other hand, carotid IMT was found to be affected by various factors including age, BMI, waist circumference, fasting and post-load glucose, and PAI-1, IL-6, and pro-MMP-1 levels (Table 4 ). In multiple regression analysis, age, plasma PAI-1 and serum pro-MMP-1 levels were found to be independently associated with carotid IMT measurements (corrected for smoking and postpartum duration, model r 2 : .202; β = .169, P = .039 for age; β = .214, P = .004 for PAI-1; β = .217, P = .003 for pro-MMP-1).
The Summary of Correlation Analysis Between Carotid IMT and Studied Parameters
Discussion
Endothelial dysfunction is proposed to be the initiating factor in the development of atherosclerosis. 22 Subclinical inflammation and hypofibrinolysis are important factors causing alterations in the endothelial system. Plasminogen activator inhibitor-1 is produced by the endothelium, and increased PAI-1 antigen levels have been clearly associated with endothelial dysfunction. The circulating levels of PAI-1 have been found to be associated with atherosclerosis. 2 In this present study, in accordance with the previous studies,2,3,7 we demonstrated that circulating levels of fibrinogen and PAI-1 were related to glucose intolerance and subclinical atherosclerosis. Women with previous GDM had increased levels of circulating atherosclerosis markers and carotid IMT, which were substantially related to underlying obesity and glucose intolerance. On the other hand, we failed to demonstrate any alteration in TAFI antigen levels either in women with gestational diabetes or in a subgroup of women developing postpartum glucose intolerance. Moreover, there was no significant association between plasma TAFI levels and indices of insulin resistance and the markers of subclinical atherosclerosis.
The abnormalities of atherothrombotic risk factors observed in our study were clearly related to underlying obesity and not previous GDM per se. The impact of BMI on atherothrombotic risk factor clustering in women with previous GDM has also been reported in previous studies.4–6,8 It is clear from our BMI-matched subgroups that obesity is the driving factor for atherothrombotic risk factor clustering in women with previous GDM. The single exception for this was the increased plasma fibrinogen levels in women with previous GDM compared to BMI-matched healthy controls. Having established that women with previous GDM have abnormalities in the measures of atherothrombotic risk factors that are related to obesity, the question arises concerning the factors associated with obesity that may be driving this phenotype. Obesity is a major risk factor for development of GDM. It is also known that obesity is associated with postpartum carbohydrate intolerance and metabolic syndrome in women with previous GDM. Despite excluding women with postpartum carbohydrate intolerance in BMI-matched subgroups analysis, it is still likely that women in the previous GDM group are at increased risk for developing diabetes and cardiovascular disorders in the future. Hence, we should consider that multifactorial abnormalities in addition to underlying obesity may take a part in the pathogenesis of cardiovascular risk in women with previous GDM.
It has been shown that plasma TAFI concentrations are stable within individuals, while they may vary widely between individuals due to genetic determination.23,24 Thrombin-activatable fibrinolysis inhibitor is mainly synthesized by the liver. 11 The production of TAFI by the impaired endothelium has been shown in diabetic participants. 13 This finding raised the question whether TAFI might take a part in the development of atherosclerosis in diabetes. Increased TAFI levels observed in several studies including patients with diabetes suggested this idea.13,14 However, it still remains unclear whether diabetes leads to increased plasma TAFI levels. Similar to our present data, our previous study showed that pregnant women with GDM had no increase in plasma TAFI antigen levels when compared to healthy pregnant women without GDM. 18 In patients with type-1 diabetes, no significant alteration in TAFI antigen levels was reported.16,25 Moreover, another study reported lower fasting and postprandial TAFI levels in type 2 diabetic patients and a negative correlation between TAFI and glycemic parameters. 26 In a very recent study, Verkleij et al. 17 found no differences in TAFI parameters after hyperglycemic clamping of healthy volunteers and between tightly and poorly regulated patients with type 2 diabetes in vivo. They also reported that TAFI purified from poorly regulated and tightly regulated patients with type 2 diabetes migrated similarly on SDS-PAGE, indicating little or no glycation of the protein. Despite the deleterious effects of glycation of TAFI in vitro on its function, TAFI was neither affected by hyperglycemic clamping nor by long-term hyperglycemia in patients with type 2 diabetes. As the authors mentioned, this was in contrast to PAI-1, which was affected.
We also observed no significant relationship between plasma TAFI and studied parameters, namely the markers of subclinical atherosclerosis and insulin resistance. The role of TAFI in cardiovascular disorders is currently controversial. There is inadequate data to establish that high plasma TAFI levels lead to any cardiovascular event. But there is some evidence concerning the possible relationship of TAFI with atherosclerotic disorders. In a previous study, Silveria et al. 27 showed elevated plasma levels of TAFI in men with symptomatic coronary artery disease. High plasma levels of TAFI were found in patients with stable angina pectoris and angiographically verified coronary artery disease. 28 Thrombin-activatable fibrinolysis inhibitor is also reported to be overexpressed after ischemic stroke, and TAFI antigen and activity have been proposed as a risk factor for stroke. 29 On the other hand, in a recent large-scale study, Prospective Epidemiological Study of Myocardial Infarction (PRIME) Study, Morange et al. 30 concluded that the relation of plasma TAFI to coronary heart disease risk was not clear. A further analysis of the PRIME data suggested that very elevated TAFI antigen levels may be protective against coronary events. 31 In addition, several recent studies suggested that the amount of activated TAFI was associated with cardiovascular events, but not of the total TAFI.32,33
The effect of insulin resistance on TAFI levels is also unclear. Although some studies reported a possible association between TAFI levels and the indices of insulin resistance,13,34 very recently, Erdogan et al. 35 showed that plasma TAFI levels were not altered in patients with polycystic ovary syndrome, a prototype of the insulin-resistant state. In another study by our group, 36 similarly, we found no significant alteration in TAFI antigen levels in normotensive type 2 diabetic participants without diabetic complications. Insulin-sensitizing treatment neither with metformin nor with rosiglitazone did not cause any decrease in plasma TAFI antigen levels. In the same way, Aso et al. 37 found no change in the circulating levels of TAFI in patients with type diabetes after 12-week treatment with another insulin-sensitizing drug, pioglitazone.
Our study had several limitations. First of all, we measured total circulating TAFI antigen levels, not activated TAFI. Second, we did not investigate genetic polymorphisms in TAFI, which might be of clinical importance as TAFI gene polymorphisms have been strongly related to plasma TAFI levels.23,30 Third, there were a small number of participants developing diabetes in the postpartum period.
In conclusion, our data suggest that plasma TAFI antigen levels are not altered in women with previous GDM. Plasma TAFI antigen is not related to insulin resistance and the markers of atherosclerosis, which might suggest that TAFI is not involved in the pathogenesis of atherosclerosis in women with previous GDM. The genetic determination of TAFI levels may be predominant rather than disease-specific effects.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Dokuz Eylul University Research Program.
