Abstract
Cervical artery dissection (CAD) is an important etiology of stroke in young adults. Its etiology is incompletely understood. Here, we report a young woman who presented with acute ischemic stroke in the setting of internal carotid artery (ICA) dissection and essential thrombocythemia (ET). We present a review of previous cases with comorbidity of CAD and ET and discuss the pathophysiological implications of this co-occurrence. In particular, we speculate that ET may increase the susceptibility of cervical vessels to spontaneous dissection, for example, by disturbing the microcirculation within the vessel wall.
Introduction
Cervical artery dissection (CAD) is an important etiology of stroke in young adults, accounting for about 20% of cases. 1 Apart from exogenous triggers such as trauma or infection, 2 a number of intrinsic predisposing factors have been reported, including inherited connective tissue disorders, migraine, low plasma levels of α1-antitrypsin, 3 and increased plasma homocysteine. 4 However, its etiology is still incompletely understood. Here, we report an otherwise healthy young woman, who presented with acute ischemic stroke in the setting of spontaneous internal carotid artery (ICA) dissection and essential thrombocythemia (ET), a chronic myeloproliferative disorder characterized by an increased frequency of arterial and venous thrombosis and microvascular disturbances.
Case Presentation
The patient is a 43-year-old Caucasian female. Past medical history was unremarkable, and except for nicotine abuse (10 pack years) there were no known vascular risk factors. She was on no regular medication. There was no recent history of head or neck trauma or chiropractic manipulation. For the 2 weeks prior to admission, she had recurrent transient numbness and tingling of the left arm and hand without other symptoms. After the acute onset of severe left-sided brachiofacial weakness, she presented to the emergency room (ER) of our hospital. She did not complain of head or neck pain neither during the prior 2 weeks nor at the time of admission.
Initial neurological evaluation revealed left brachiofacial hemiparesis (grade 2/5), accompanied by multimodal left-sided hemineglect. There were no cranial nerve palsies or Horner’s syndrome. Cranial computed tomography (CT) displayed multiple acute and subacute infarcts in the territory of the right middle cerebral artery (Figure 1A). Extracranial CT angiography (CTA) showed occlusion of the right ICA, extending over a distance of 15 cm (Figure 1C and D). In addition, it revealed thrombotic material in the right subclavian artery (SA) in direct proximity to the right vertebral artery (VA; Figure 1D); of note, no thrombotic material was detected in the brachiocephalic trunk. The patient was transferred to the stroke unit.
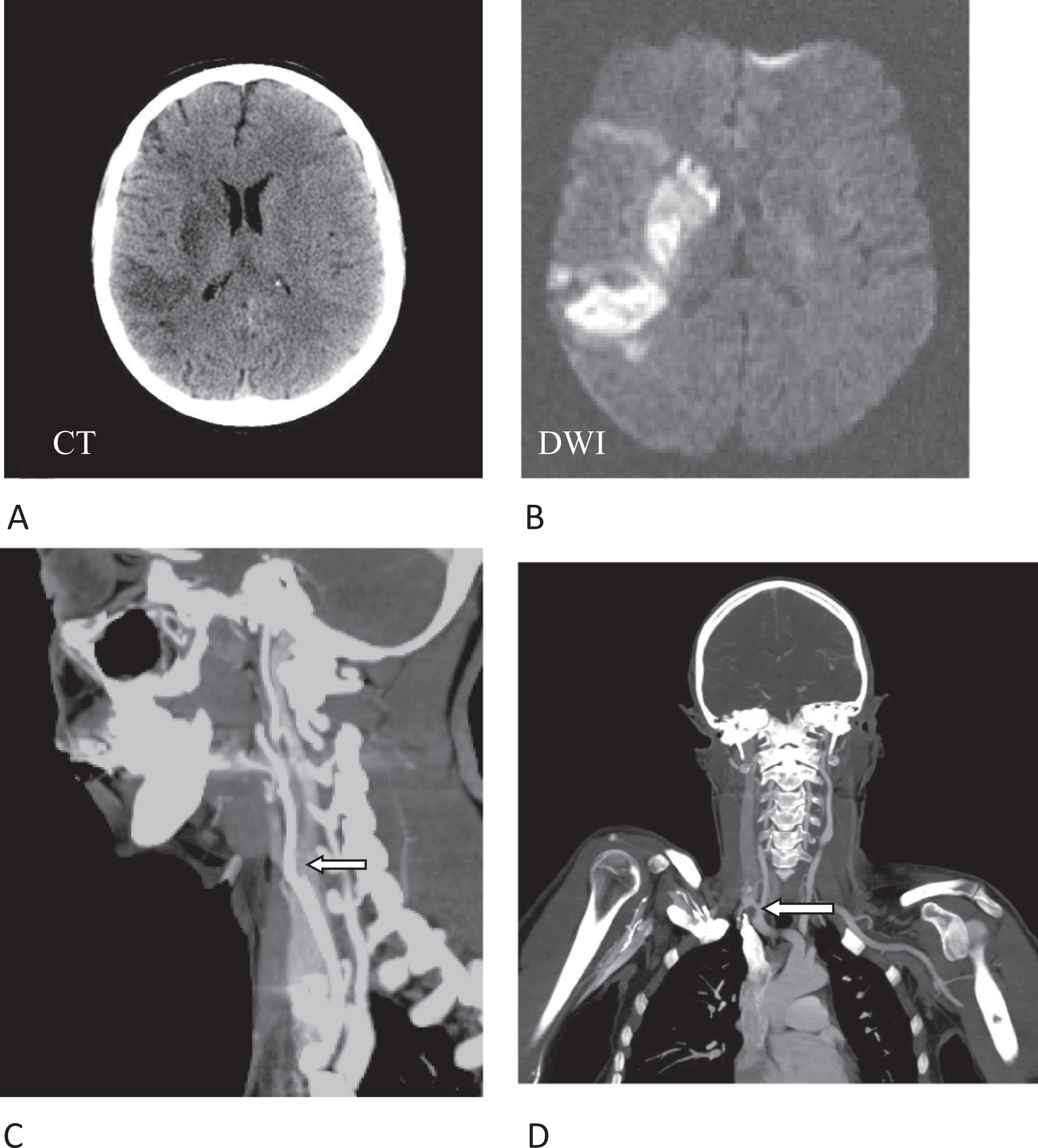
Cranial CT, showing multiple right-hemispheric infarcts (A). Diffusion-weighted (DWI) cMRI, showing acute ischemic stroke in the right MCA territory (B). CTA shows occlusion of the right ICA (arrow) directly at the bifurcation (C). In addition, thrombotic material is detected in the right SA in proximity to the right VA (D, arrow). CT indicates computed tomography; CTA, CTangiography; DWI; diffusion-weighted imaging; ICA, internal carotid artery; MCA, middle cerebral artery; cMRI, cranial magnetic resonance imaging.
Under intravenous anticoagulation with heparin (partial thromboplastin time [PTT] 50-60 seconds), which was started because of the thrombotic material in the right SA, and hypertensive therapy, the right-hemispheric syndrome was slowly regressive. Diffusion-weighted cranial magnetic resonance imaging (MRI) showed multiple right-hemispheric infarcts (Figure 1B); of note, no lesions were detected in the posterior circulation in spite of the SA thrombus near the right VA. Magentic resonance (MR) angiography of cervical vessels, which was performed 5 days after admission, revealed intramural hematoma of the occluded right ICA, thus establishing a diagnosis of ICA dissection (Figure 2).
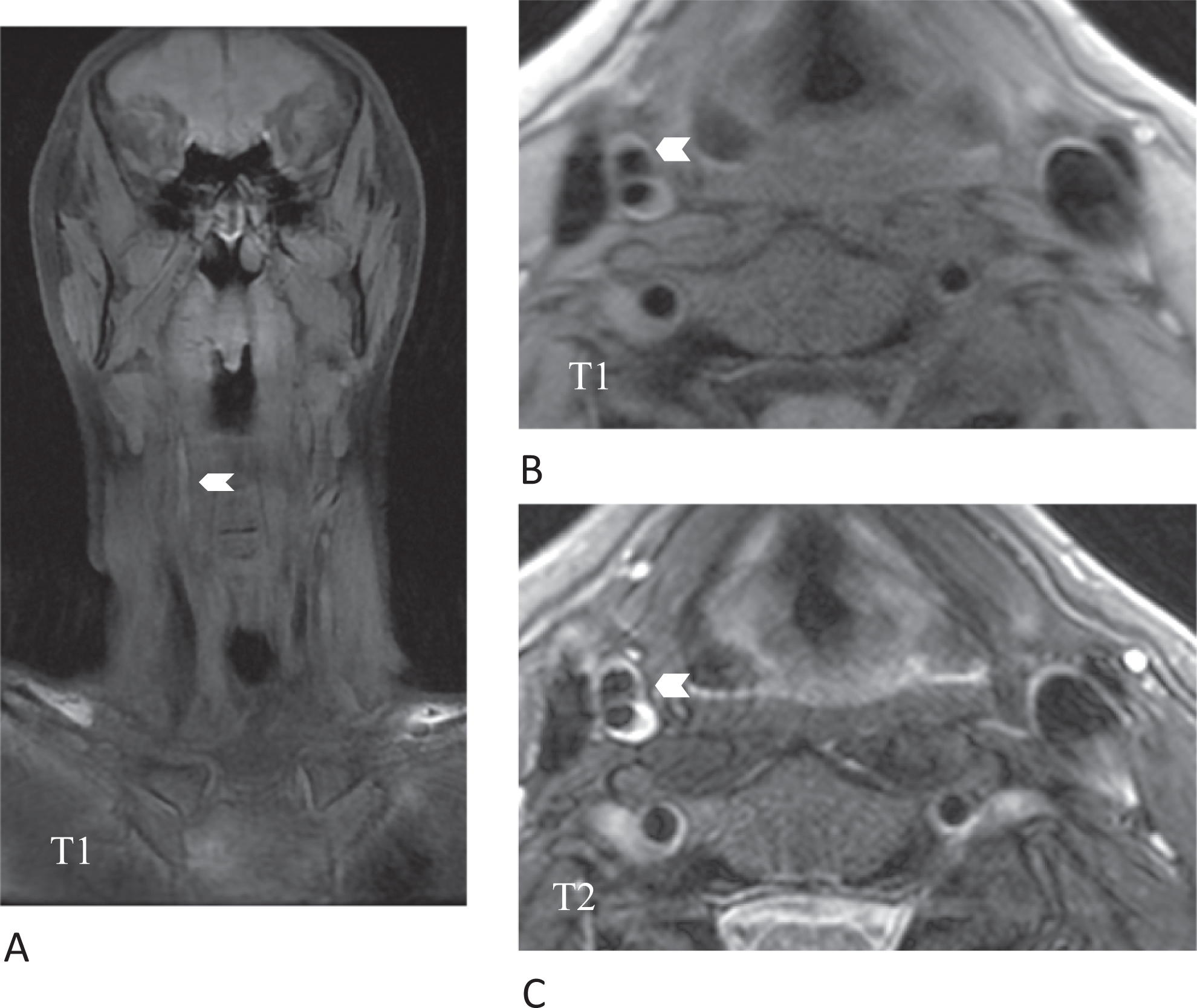
Cervical MRI: coronal (A) and axial fat-saturated T1-weighted (B) and axial T2-weighted images (C). The chevron points to a iso- to hyperintense area on T1-weighted images which is hyperintense on corresponding T2-weighted images, consistent with a subacute intramural hematoma in the left internal carotid artery. MRI indicates magnetic resonance imaging.
Dermatological and opthalmological examination were without signs suggestive of vasculitis. Transesophageal echocardiography showed patent foramen ovale (PFO) with hypermobile atrial septum of <15 mm amplitude, therefore considered nonrelevant. Holter monitoring showed regular sinus rhythm, without evidence of atrial fibrillation. Extensive laboratory tests including serum chemistry, liver function tests, coagulation studies, vasculitis parameters, iron studies, and serological investigations showed unremarkable results. However, while erythrocytes and leukocytes were within the normal range, thrombocytes were found to be mildly increased at 550 G/L (reference range 150-440 G/L). During the course of the hospital stay, thrombocytes continued to rise to a maximum of 700 G/L. In the absence of explanations for reactive thrombocytosis (normal iron studies, no signs of inflammation), bone marrow biopsy was performed, which established a diagnosis of ET. In addition, molecular genetic testing of the JAK2 gene revealed presence of the V617F mutation, which has been described as a diagnostic marker for ET. 5
For treatment of dissection, intravenous (IV) heparin was changed to oral anticoagulation (international normalized ratio [INR] 2.0-3.0), and 4 weeks after admission the patient was discharged to rehabilitation with mildly persistent left hemineglect. During the following months, thrombocytes continued to persist at levels between 500 and 600 G/L, and cytoreductive therapy with hydroxy urea was begun. As soon as thrombocytes had reached normal levels, oral anticoagulation was stopped and aspirin was begun. Under this regimen, there were no further thrombotic events (follow-up 10 months).
Discussion
Here, we present a young female with a right-hemispheric stroke due to ICA occlusion secondary to spontaneous dissection in whom extensive diagnostic workup revealed presence of thrombotic material in the right SA and, in particular, established a diagnosis of ET. Our observation raises the question whether this co-occurrence of dissection and ET reflects a causal relationship or is merely by chance.
Dissection of cervical vessels does not necessarily lead to occlusion of the affected artery: older series reported occlusion in only a minority of cases, 6 while more recent studies described a frequency of approximately 50%.7,8 Most likely, in our patient the initial dissection was followed by a prolonged thrombotic process finally leading to occlusion; this scenario is reflected both by the clinical history of recurrent neurological symptoms over a 2-week period as well as the imaging finding of multiple infarcts of different age. Further evidence for this concept is provided by the presence of thrombotic material in the SA; in this context, the presence of PFO most likely can be disregarded from a pathophysiological point of view, especially since it was not associated with atrial septal aneurysm. Essential thrombocythemia is known to cause a prothrombotic state,9,10 and a few cases of ischemic stroke due to ET-associated large-vessel occlusion have been reported. 11 Therefore, it is likely that ET promoted occlusion of the dissected ICA in our patient, thereby complicating the clinical course.
Beyond this, one may speculate that ET does increase the susceptibility of cervical vessels to (spontaneous) dissection. A possible biological explanation would be that ET, due to prothrombotic changes, causes disturbance of the microcirculation within the vasa vasorum, thereby—together with other factors such as nicotine abuse—increasing the vulnerability of the vessel wall. Further vascular damage may be caused by activation of leukocytes and release of, for example, elastase or alkaline phosphatase, which have been shown to occur in ET. 9 However, these assumptions are very speculative, in particular since the larger published CAD series only rarely reported comorbidity with ET. In a series of patients with multiple dissections, only one patient with bilateral vertebral and unilateral carotid artery dissection was reported to have ET, but no further clinical details are available. 12 Furthermore, D’Ambrosio et al. 13 reported the case of a single patient with co-occurrence of intrapetrous ICA dissection and ET; however, in contrast to our case, diagnosis of ET was established with a latency of several months after the initial vascular event. Furthermore, there is one case of spontaneous coronary artery dissection in a patient with polycythemia vera, a disease genetically related to ET. 14 Finally, an association between systemic mastocytosis, that is, another hematological disease, with CAD has been described in two cases. 15
In conclusion, while assumptions about a causal relationship between ET and dissection remain highly speculative, it is evident that ET was an aggravating pathophysiological factor in our patient. Thus, from a practical point of view, our observation deserves attention because the diagnosis of ET, which might otherwise have been missed, was established during careful diagnostic workup of ischemic stroke. This emphasizes the clinical lesson that in patients with ischemic stroke, and in particular with dissections, ET should be considered, even if platelet counts are only mildly elevated; it is particularly important not to miss the diagnosis of ET, because the prothrombotic changes associated with ET may increase the risk of stroke recurrence, which emphasizes the significance of adequate (cytoreductive) therapy of ET in addition to routine secondary prophylaxis of stroke.
Finally, in order to clarify the relationship between ET and dissections of cervical vessels, further systematic studies in larger patient cohorts are needed.
Footnotes
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
