Abstract
Background
Cardiovascular disease remains the first major reason for death in global, and hypertension is one very usual risk factor for this disease. Chemokine (CXC motif) ligand 13 (CXCL13) exhibits a crucial role in the development and angiogenesis of a variety of diseases. However, the impacts of CXCL13 in the amelioration of hypertension and the associated regulatory mechanisms have rarely reported.
Methods
The hypertensive rat model was induced by Ang II treatment. The protein and mRNA expressions were measured through western blot and RT-qPCR. The systolic blood pressure was monitored. The ET-1 level was examined through the commercial kit. The thickness of vascular elasticity layer was observed through hematoxylin eosin staining. The levels of angiotensin-converting enzyme (ACE), thiobarbituric acid-reactive substances (TBARS), and brain natriuretic peptide (BNP) were inspected through the commercial kits.
Results
CXCL13 exhibited high protein and mRNA expressions in hypertensive rat model. Knockdown of CXCL13 improved vascular remodeling and elevated blood pressure in hypertensive rat model. Moreover, suppression of CXCL13 protected heart in hypertensive rat model through declining the levels of ACE, TBARS, and serum BNP. Next, it was demonstrated that CXCL13 modulated the PF4V1/NF-κB pathway. Eventually, through rescue assays, it was proved that CXCL13 ameliorated hypertension through affecting PF4V1.
Conclusion
This study manifested that knockdown of CXCL13 improved vascular remodeling, reduced blood pressure, and protected the heart by affecting the PF4V1/NF-κB signaling pathway in a hypertensive rat model. This discovery may offer novel opinions in the treatment of hypertension.
Introduction
Hypertension badly influences the health of people, and it is the primary reason for cardiovascular disease. 1 In the world, more than 1 billion people have been diagnosed as hypertension, and the prevalence of it is aggrandized by over 10% in each year. 2 Hypertension can aggravate mechanical stress, thereby stimulating cardiomyocyte apoptosis, hypertrophy, and vascular remodeling. 3 Subsequently, heart will change its structure, shape, size, and function to result into cardiac dysfunction. 4 Therefore, looking for serviceable bio-targets to ameliorate hypertension has become pivotal.
Chemokines exist key functions in multiple cellular processes, and they contribute to cell homing, angiogenesis and other activities.5,6 Chemokine (CXC motif) ligand 13 (CXCL13) has been clarified to exhibit crucial roles in the development of a variety of diseases and angiogenesis. 7 For instance, CXCL13 facilitates the generation of pro-inflammatory cytokines and NF-κB activation to aggravate chronic pain in mouse model of complex regional pain syndrome type-I. 8 In addition, CXCL13 recruits M2 macrophage and restrains inflammation to accelerate thermogenesis in mice. 9 In systemic sclerosis, Fli1 deficiency-mediated CXCL13 strengthens vasculopathy, tissue fibrosis, and immune activation. 10 Moreover, CXCL13/CXCR5 axis can promote cell homing and angiogenesis in the development of rheumatoid arthritis. 11 Importantly, in idiopathic pulmonary arterial hypertension, CXCL13 is closely associated with inflammation and vascular remodeling, and CXCL13 was elevated in serum, suggesting that CXCL13 owns potentially pathogenic role in this disease. 12 However, the detailed roles of CXCL13 in hypertension progression remain vague.
In this work, it was purposed to investigate the influences and associated pathways of CXCL13 in the treatment of hypertension. Our results uncovered that knockdown of CXCL13 improved vascular remodeling, reduced blood pressure, and protected the heart by affecting the PF4V1/NF-κB signaling pathway in a hypertensive rat model. This study insinuated that CXCL13 may be a helpful bio-target for ameliorating hypertension.
Materials and Methods
Rat Model
Wistar-Kyoto male rats (n = 24) were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd (Beijing, China). Under free food and water at 12/12 h light/dark cycle, rats were kept for 1 week. This study was approved by the Animal Care and Use Committee of Chaohu Hospital affiliated to Anhui Medical University and executed in line with the Care and Use of Laboratory Animals of the National Institutes of Health.
After anesthesia by sodium pentobarbital (2%), Angiotensin II (Ang II, 1.5 mg/kg/day, Sigma-Aldrich, MO, USA) under one osmotic minipump (Alzet model 2004, MA, USA) was subcutaneously injected into rats for 14 days to establish the hypertensive rat model. The systolic blood pressure of rats was determined through using a tail-cuff instrument (BP-98A, Softron, Japan) at 0, 3, 6, 9, 12, and 15 days.
Rats were randomly divided into four groups: control group (n = 6), Ang II group (n = 6), Ang II + si-CXCL13 group (n = 6), and Ang II + si-CXCL13 + si-PF4V1 group (n = 6). The small interfering RNAs (siRNAs) targeting CXCL13 (si-CXCL13) or PF4V1 (si-PF4V1) were obtained from GenePharma (Shanghai, China). The recombinant adenovirus with si-CXCL13 or si-PF4V1 was injected into rats by tail intravenous injection.
Western Blot
The proteins extracted from aorta were made through using RIPA lysis buffer. Then, proteins were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis for separation. The polyvinylidene difluoride membrane (Sigma, St Louis, Missouri, USA) was put with the transferred proteins. The primary antibodies were added for one-night incubation, and the secondary antibody (1/1000, ab6721, Abcam, Shanghai, China) was added for another 2 h incubation. At last, the enhanced chemiluminescence kit (Thermo Fisher Scientific, USA) was employed for determining the blots.
The primary antibodies: CXCL13 (1/500, ab190662, Abcam, Shanghai, China), myosin light chain phosphatase (MLCP, 1/1000, 3672, Cell Signaling, Shanghai, China), Endothelin A (ETA,1/1000, ab178454), eNOS (1/1000, ab300071), Platelet Factor 4 Variant 1 (PF4V1, 1/200, PF4V1, Shanghai Huzhen company, Shanghai, China), p-p65 (1/1000, ab76302), p65 (1/1000, ab16502), p-IkBα (1/1000, ab92700), IkBα (1/1000, ab32518), and GAPDH (1/500, ab8245).
RT-qPCR
The RNAs from aorta were extracted through using the Trizol reagent (Thermo Fisher Scientifc, Waltham, MA, USA). The cDNA from RNAs was generated through the PrimeScript™ RT Reagent Kit (Takara, Dalian, China). Then, the SYBR Green PCR kit (Toyobo, Japan) was adopted for performing qPCR. The relative mRNA expression of CXCL13 was examined through the 2−ΔΔCt method.
The primers were shown:
CXCL13:
F, 5’-CTGCTCGGAATCTTAGTGT-3’,
R, 5’-GGTAATGCGTCTGCTTCT-3’;
GAPDH :
F, 5’-GGCACAGTCAAGGCTGAATG-3’,
R, 5’-ATGGTGGTGAAGACGCCAGTA-3’.
Detection of ET-1
Serum ET-1 level was measured by ET-1 ELISA kit (ab133030, Abcam, Shanghai, China).
HE Staining
The 4-µm thick sections of paraffin-embedding vascular or heart tissues were made. Then, dyeing through hematoxylin (5 min) and eosin (30 s) was proceeded for these sections. Eventually, the thickness of vascular elasticity layer was assessed through the light microscope (Olympus Corporation, Tokyo, Japan).
Detection of Angiotensin-Converting Enzyme, Thiobarbituric Acid-Reactive Substances and Brain Natriuretic Peptide
The cardiac angiotensin-converting enzyme (ACE) activity, thiobarbituric acid-reactive substances (TBARS) content, and serum brain natriuretic peptide (BNP) level were inspected through the ACE assay kit (ab239703, Abcam, Shanghai, China), OxiTek TBARS assay kit (Zeptometrix, USA), and BNP ELISA kit (ab108816, Abcam, Shanghai, China).
Statistical Analysis
The data were exhibited as mean ± standard deviation, and statistical analysis was proceeded by using GraphPad Prism software 9.0 (GraphPad Software, La Jolla, CA). The differences were executed under the Student's t-test or one-way analysis of variance. P < 0.05 was accepted as statistically significant.
Results
CXCL13 Exhibited High Expression in Hypertensive rat Model
The CXCL13 protein expression was elevated after Ang II treatment (Figure 1A). Furthermore, CXCL13 mRNA expression was also risen after Ang II treatment (Figure 1B). In short, CXCL13 exhibited high expression in hypertensive rat model.

CXCL13 Exhibited High Expression in Hypertensive Rat Model. (A) The Protein Expression of CXCL13 was Measured through Western Blot in the Control and Ang II groups. (B) The mRNA Expression of CXCL13 was Examined Through RT-qPCR in the Control and Ang II Groups. ***P < 0.001.
Knockdown of CXCL13 Improved Vascular Remodeling and the Elevated Blood Pressure in Hypertensive rat Model
The elevated CXCL13 protein expression triggered by Ang II treatment was attenuated after suppressing CXCL13 (Figure 2A). In addition, the systolic blood pressure was raised after Ang II treatment, but this impact was descended after silencing CXCL13 (Figure 2B). The ET-1 level was increased after Ang II treatment, but this phenomenon was offset after inhibiting CXCL13 (Figure 2C). The protein expressions of MLCP and eNOS were lessened as well as ETA was ascended after Ang II treatment, but these changes were counteracted after repressing CXCL13 (Figure 2D). Through hematoxylin eosin (HE) staining, the thickness of vascular elasticity layer was aggrandized after Ang II treatment, but this influence was neutralized after restraining CXCL13 (Figure 2E). To sum up, knockdown of CXCL13 improved vascular remodeling and the elevated blood pressure in hypertensive rat model.
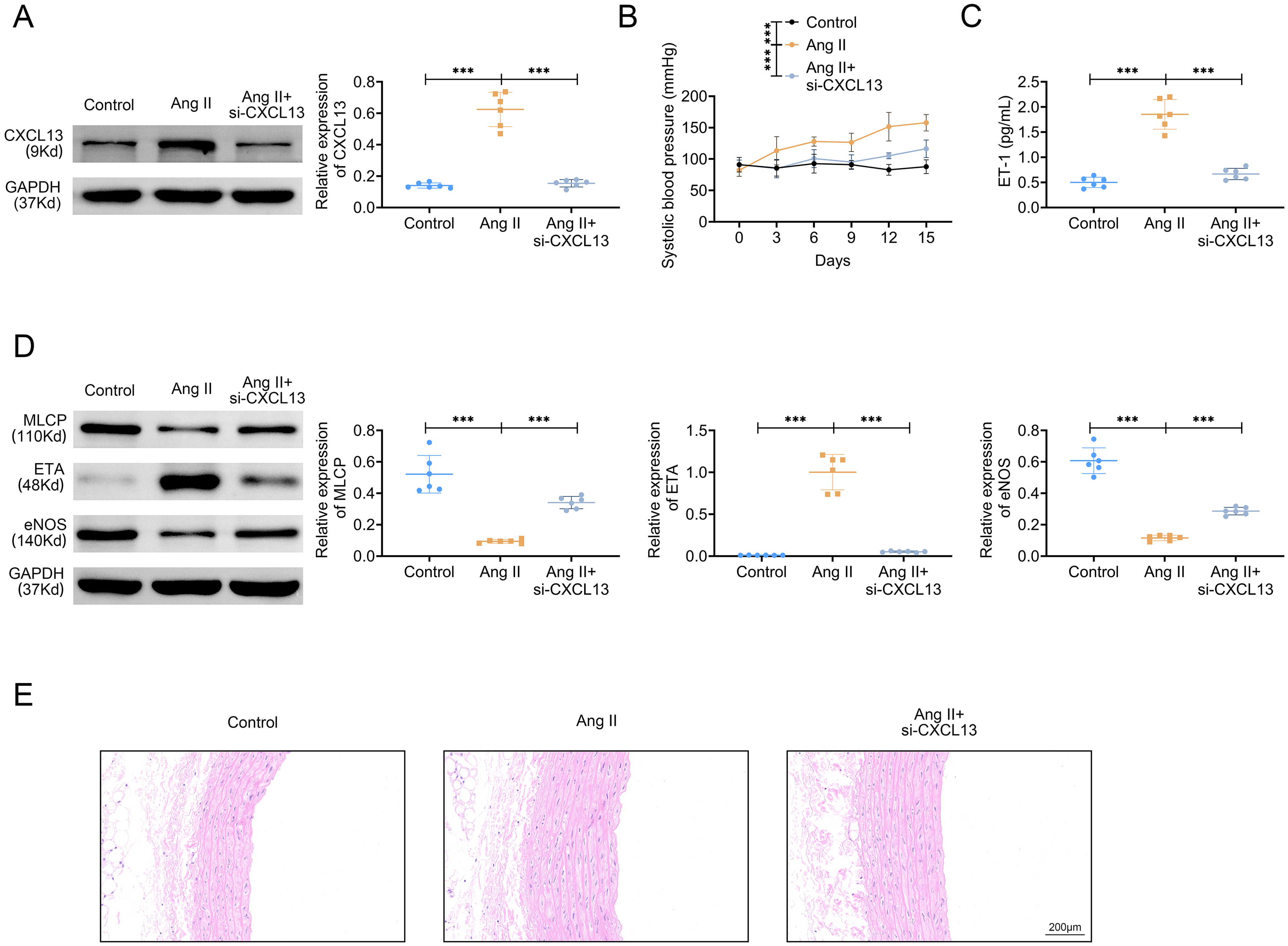
Knockdown of CXCL13 Improved Vascular Remodeling and the Elevated Blood Pressure in Hypertensive Rat Model. Groups were Separated into the Control, Ang II and Ang II + si-CXCL13 Group. (A) The Protein Expression of CXCL13 was Confirmed Through Western Blot. (B) The Systolic Blood Pressure was Monitored. (C) The ET-1 Level was Examined Through the Commercial kit. (D) The Protein Expressions of MCLP, ETA, and eNOS Were Tested Through Western Blot. (E) The Thickness of Vascular Elasticity Layer was Observed Through HE Staining. ***P < 0.001.
Suppression of CXCL13 Protected Heart in Hypertensive rat Model
The damage of heart tissues was strengthened after Ang II treatment, but this phenomenon was offset after CXCL13 knockdown (Figure 3A). Next, it was elucidated that the levels of ACE, TBARS, and serum BNP were all strengthened after Ang II treatment, but these impacts were offset after CXCL13 inhibition (Figure 3B), indicating that suppression of CXCL13 protected heart in hypertensive rat model.

Suppression of CXCL13 Protected Heart in Hypertensive Rat Model. Groups were Separated into The Control, Ang II and Ang II + si-CXCL13 group. (A) The Damage of Heart Tissues was Examined Through HE Staining. (B) The Levels of ACE, TBARS, and BNP were Inspected Through the Commercial Kits. **P < 0.01, ***P < 0.001. ACE, Angiotensin-Converting Enzyme; TBARS, Thiobarbituric Acid-Reactive Substances; BNP, Brain Natriuretic Peptide.
CXCL13 Modulated the PF4V1/NF-κB Pathway
The protein expressions of PF4V1 and IkBα were declined as well as p-p65/p65 and p-IkBα were uplifted after Ang II treatment, but these changes were counteracted after CXCL13 silencing (Figure 4). In general, CXCL13 modulated the PF4V1/NF-κB pathway.

CXCL13 Modulated the PF4V1/NF-κB Pathway. Groups were Separated into The Control, Ang II and Ang II + si-CXCL13 group. The Protein Expressions of PF4V1, p-p65, p65, p-IkBα, and IkBα were Assessed Through Western Blot. **P < 0.01, ***P < 0.001.
CXCL13 Ameliorated Hypertension Through Affecting PF4V1
As displayed in Figure 5A, based on Ang II treatment, the thickness of vascular elasticity layer was attenuated after CXCL13 suppression, but this influence was reversed after PF4V1 inhibition. Furthermore, the damage of heart tissues was relieved after Ang II treatment, but this change was rescued after CXCL13 knockdown (Figure 5B). Under the condition of Ang II treatment, PF4V1 and IkBα were up-regulated as well as p-p65/p65 and p-IkBα were down-regulated after CXCL13 knockdown, but these impacts were further neutralized after PF4V1 inhibition (Figure 5C). Moreover, the systolic blood pressure was cut down after CXCL13 suppression, but this influence was counteracted after PF4V1 silencing (Figure 5D). The ET-1 level was lessened after CXCL13 down-regulation, but this phenomenon was rescued after PF4V1 repression (Figure 5E). The protein expressions of MLCP and eNOS were risen as well as ETA was weakened CXCL13 inhibition, but these affects were reversed after CXCL13 silencing (Figure 5F). Besides, the levels of ACE, TBARS and serum BNP were all weakened after CXCL13 repression, but these changes were recovered after CXCL13 inhibition (Figure 5G). Taken together, CXCL13 ameliorated hypertension through affecting PF4V1.
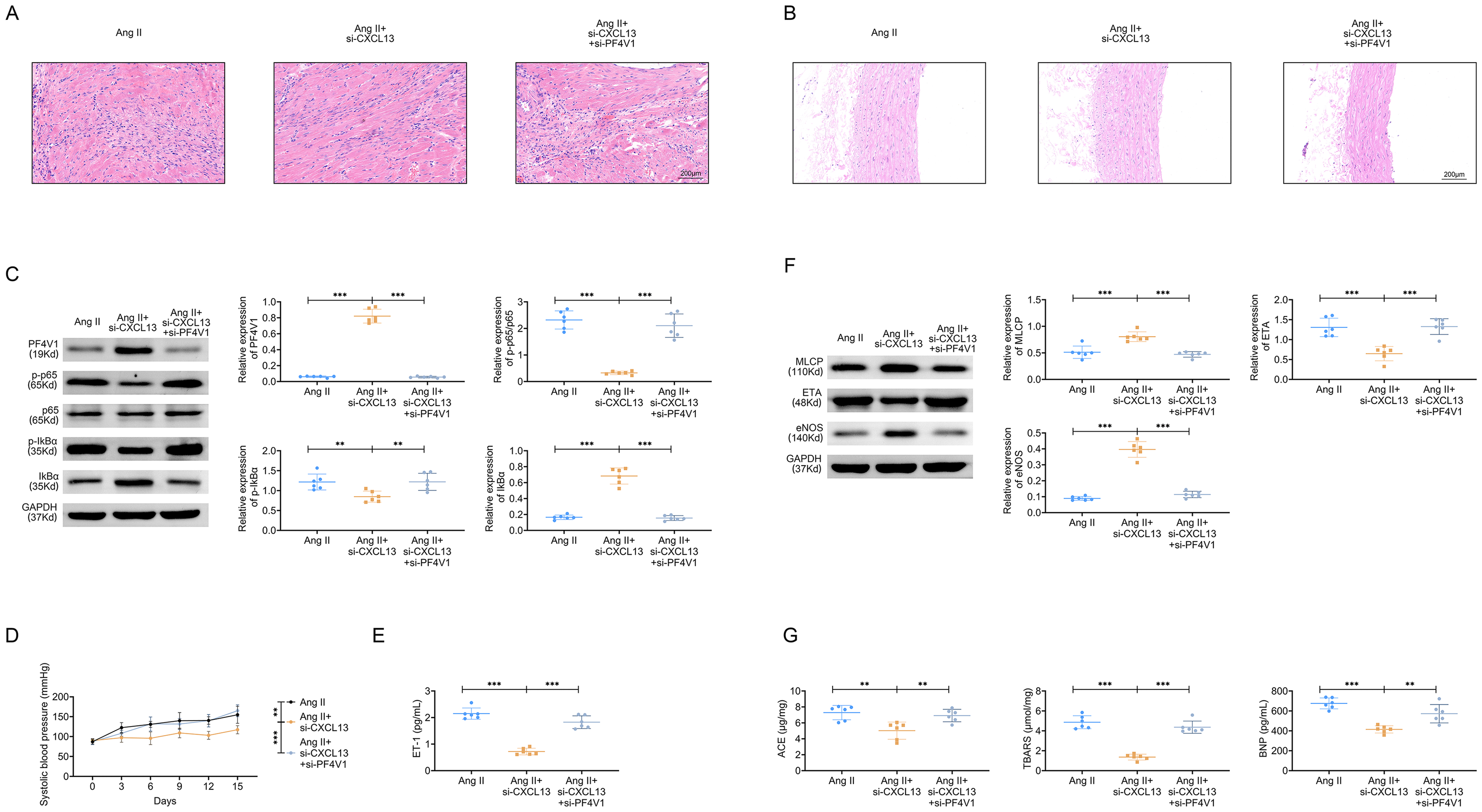
CXCL13 Ameliorated Hypertension Through Affecting PF4V1. Groups were Separated Into The Ang II, Ang II + si-CXCL13, and Ang II + si-CXCL13 + si-PF4V1 group. (A) The Damage of Heart Tissues was Examined Through HE Staining. (B) The Thickness of Vascular Elasticity Layer was Observed through HE Staining. (C) The Protein Expressions of PF4V1, p-p65, p65, p-IkBα, and IkBα were Determined through Western Blot. (D) The Systolic Blood Pressure was Monitored. (E) The ET-1 Level was Measured Through the Commercial kit. (F) The Protein Expressions of MCLP, ETA, and eNOS Were Evaluated Through Western Blot. (G) The Levels of ACE, TBARS, and BNP were Inspected Through the Commercial Kits. **P < 0.01, ***P < 0.001. ACE, Angiotensin-Converting Enzyme; TBARS, Thiobarbituric Acid-Reactive Substances; BNP, Brain Natriuretic Peptide.
Discussion
CXCL13 has been certified to own regulatory roles in the development of a variety of diseases and angiogenesis. 7 However, the impacts of CXCL13 in the amelioration of hypertension and the associated regulatory mechanisms keep dimness. In this study, it was illustrated that CXCL13 exhibited high protein and mRNA expressions in hypertensive rat model.
Vascular remodeling exerts important functions in the pathogenesis and development of hypertension. 13 In hypertension, inflammation, fibrosis and cell growth can result into vascular remodeling. 14 Many researchers have focused on the modulation of vascular remodeling in hypertension progression. For example, Resolvin E1/ChemR23 can exhibit protective impacts on vascular remodeling in Ang II-evoked hypertensive mice. 15 Moreover, oviductal glycoprotein 1 interacts with MYH9 to trigger vascular remodeling, thereby aggravating hypertension. 16 Besides, in spontaneously hypertensive rats, neferine can ameliorate vascular remodeling through affecting diversiform pathways. 17 In addition, deficiency of caveolin-1 suppresses vascular remodeling stimulated by Ang II. 18 Similar to these previous researches, in this work, it was also manifested that knockdown of CXCL13 improved vascular remodeling and elevated blood pressure in hypertensive rat model. Moreover, suppression of CXCL13 protected heart in hypertensive rat model through declining the levels of ACE, TBARS, and serum BNP.
The NF-κB pathway has been testified to be one pivotal pathway in hypertension progression and has gained many attentions. For example, leonurine restrains the NF-κB pathway to ameliorate cardiac injury and dysfunction mediated by Ang II. 19 Additionally, corynoline retards the NF-κB pathway to alleviate hypertensive heart failure stimulated by Ang II. 20 Moreover, USP7 inhibitor-P22077 can modulate the NF-κB/NLRP3 pathway to protect Ang II-evoked hypertensive mice through inhibiting cardiac hypertrophy and remodeling. 21 Importantly, PF4V1 (another name: CXCL4L1) is one chemokine that can suppress neovascularization to participate into multiple diseases.22,23 Inhibition of PF4V1 leads to the reduced level of IκBα. 24 Interestingly, it was proved that CXCL13 can stimulate CXCR5-dependent NF-κB. 8 However, the regulatory functions of CXCL13 on the PF4V1/NF-κB pathway remain unknown. In this study, it was also demonstrated that CXCL13 modulated the PF4V1/NF-κB pathway through ascending PF4V1 and IκBα protein expressions as well as declining p-p65/p65 and p-IκBα protein expressions. At last, through rescue assays, it was testified that inhibition of PF4V1 can rescue the changes of vascular remodeling, blood pressure, and heart mediated by CXCL13 knockdown in hypertension.
In conclusion, it was firstly manifested that knockdown of CXCL13 improved vascular remodeling, reduced blood pressure and protected the heart by affecting the PF4V1/NF-κB signaling pathway in a hypertensive rat model. This work also owns some limitations, so that more experiments about CXCL13 in hypertension progression will be further executed in the future.
Footnotes
Ethical Considerations
Ethical approval was obtained from the Ethics Committee of Chaohu Hospital affiliated to Anhui Medical University.
Author Contributions
All authors contributed to the study conception and design. Material preparation and the experiments were performed by JM. Data collection and analysis were performed by XS. The first draft of the manuscript was written by XH and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
