Abstract
Hypopituitarism, resulting from a partial or complete deficiency of anterior or posterior pituitary hormones, is associated with increased cardiovascular (CV) morbidity and mortality. This heterogeneous endocrinological disorder may arise from various etiologies, including genetic mutations, pituitary tumors, traumatic brain injury, and autoimmune diseases. Hypopituitarism often results in multiple endocrine deficits that contribute to metabolic dysregulation characterized by insulin resistance, dyslipidemia, and increased visceral adiposity, all known risk factors for CV disease (CVD). Additionally, the presence of chronic inflammation and endothelial dysfunction further increases the risk of CVD in these patients. While standard hormone replacement therapy (HRT) is crucial for restoring hormonal balance, it can sometimes have adverse metabolic effects that can exacerbate atherosclerosis and CVD. Emerging evidence suggests that optimizing HRT regimens and addressing specific hormone deficiencies, such as growth hormone and cortisol, may reduce these risks and improve CV outcomes. This review comprehensively analyzes the etiology, pathophysiological mechanisms underlying CV risk in anterior pituitary dysfunction, and treatment strategies to mitigate CV morbidity and mortality in patients with hypopituitarism.
Keywords
Introduction
Pituitary hormone deficiency is a condition characterized by varying severity, encompassing a diverse group of patients with a wide spectrum of hormone insufficiencies, ranging from a single pituitary hormone deficiency to complete loss of pituitary function, known as panhypopituitarism. 1 Partial or complete deficiency of anterior or posterior pituitary hormones leads to growth hormone (GH) deficiency, hypogonadotropic hypogonadism (HH), central hypothyroidism, adrenocorticotropic hormone (ACTH) deficiency, and consequent secondary adrenal insufficiency (AI), or arginine vasopressin (AVP) deficiency (central diabetes insipidus), depending on the hormones affected. 2
The pituitary hormones play a pivotal role in the regulation of metabolic homeostasis and the physiological function of the cardiovascular (CV) system. 1 One of the initial studies on panhypopituitarism and mortality revealed that life expectancy decreased because of CV diseases (CVDs). 3 Thereafter, studies confirmed that patients with hypopituitarism have increased CV morbidity and mortality. 1 The majority of patients with hypopituitarism are exposed to the most common conventional risk factors for CVD, such as obesity, insulin resistance (IR), central fat distribution, and dyslipidemia. 4 Furthermore, endothelial dysfunction and chronic inflammation may contribute to the high CV risk in patients with hypopituitarism. 4 Cases of cardiomyopathy associated with hypopituitarism have been reported, and GH deficiency, AI, and hypothyroidism in isolation can lead to heart failure (HF), a rare but serious complication of untreated hypopituitarism. 5
In addition, conventional hormone replacement therapy (HRT), which is the cornerstone of treatment for hypopituitarism, may also contribute to an unhealthy metabolic state that promotes atherosclerosis and CVD. 1 Based on the evidence to date, it seems most likely that inadequate HRT, either under or overtreatment, adversely affects CV risk factors and creates an unfavorable dysmetabolic milieu that leads to increased CV risk and consequently the development of CVD. GH treatment may improve lipid profiles and reduce CV mortality, it can also increase IR and the risk of diabetes and this may contribute to the development of CVDs in some patients.6,7 Thyroid hormone overdosing may lead to increased heart rate and arrhythmias (eg, atrial fibrillation), and consequently higher CV risk. 8 Under-replacement with thyroid hormones may increase total fat mass, body mass index (BMI), waist and hip circumference, low-density lipoprotein (LDL) cholesterol (LDL-C) and triglyceride (TG) levels, and also may lower high-density lipoprotein (HDL) cholesterol (HDL-C) levels, and this may contribute to atherosclerosis and poor cardiac function.8–10 Even a modest excess of replacement glucocorticoid (GC) therapy in hypopituitarism is associated with an increased risk of CVDs attributable to classic CV risk factors such as obesity, glucose intolerance, high blood pressure, and dyslipidemia. 11 The impact of estrogen or testosterone replacement therapy on the CV system may differ depending on the time of onset, but it is clear that earlier onset of secondary hypogonadism, and lack of treatment, lead to a poor metabolic profile characterized by elevated LDL-C and decreased HDL-C levels, with impaired glucose metabolism, increased arterial stiffness, and consequent hypertension.12–14 Data on overtreatment with estrogen or testosterone replacement therapy in terms of CV risk are scarce.
Although morbidity and mortality rates have improved in patients with hypopituitarism with contemporary HRT and adequate treatment of the causes, these patients remain at increased risk for complications, and close monitoring and individualized adjustments of therapy are crucial. 15
In this review article, we summarize the evidence on CV risk in patients with complete and partial anterior pituitary dysfunction, as well as the potential additional CV burden of the modern therapeutic approach, aiming the need for timely introduction and patient-oriented optimization of HRT, in term of ameliorating CV risk in patients with hypopituitarism.
Epidemiology and Etiology
Hypopituitarism is rare and likely to be underdiagnosed, with an unknown but rising prevalence and incidence. It occurs in individuals due to various congenital or acquired conditions, as shown in Table 1.
Etiology of Hypopituitarism.

Abbreviations: ALMS1, Alstrom syndrome 1; CHD7, chromodomain helicase DNA binding protein 7; CTLA-4, cytotoxic T-lymphocyte-associated protein 4; FGF8, fibroblast growth factor 8; FGFR1, fibroblast growth factor receptor 1; GH1, growth hormone 1; GLI2, GLI family zinc finger 2; HESX1, homeobox expressed in ES cells 1; IgG4, immunoglobulin G4; KAL1, Kallmann syndrome 1; LHX3, LIM homeobox 3; LHX4, LIM homeobox 4; NR0B1, nuclear receptor subfamily 0 group B member 1; OTX2, orthodenticle homeobox 2; PAX-6, paired box 6; PCSK9, proprotein convertase subtilisin/kexin type 1; PD-1, programmed cell death protein 1; PD-L1, programmed cell death ligand 1; POMC, pro-opiomelanocortin; POU1F1, POU domain class 1 transcription factor 1; PROKR2, prokineticin receptor 2; PROP1, prophet of pituitary-specific transcriptional factor 1; SHH, sonic hedgehog; SOD, septo-optic dysplasia; SOX2, sex-determining region Y protein-related high mobility group box gene 2; SOX3, sex-determining region Y protein-related high mobility group box gene 3; TBX-19, T-box transcription factor 19; TCF7L1, transcription factor 7-like 1.
Epidemiologic data on hypopituitarism are not entirely reliable, given the wide variety of clinical presentations and the complexity of testing for transient and partial hypopituitarism. 2 Nevertheless, the prevalence of hypopituitarism in adults was reported at 45.5/100,000, and the average annual incidence rate was 4.21/100,000, with no significant variations in incidence trends and no differences between genders. 16 A congenital background of multiple pituitary hormone deficiency is found in about one in 4,000–10,000 live births and frequently presents with GH deficiency. 17 Increased detection of pituitary adenomas, a common cause of hypopituitarism in adults, due to the use of high-resolution imaging and the rise of newer causes, including immune checkpoint inhibitors (ICIs) and traumatic brain injury, will likely lead to increased hypopituitarism detection, while radiation-associated hypopituitarism rates will probably decrease. 1
There are many identified causes of hypopituitarism, however, the clinical impact is variable and determined by the age of onset, the gender of the patient, the underlying cause, and the pattern of hormone deficiencies. Luteinizing hormone (LH)/follicle-stimulating hormone (FSH) deficiency has been reported to be most prevalent in patients with hypopituitarism, and about half of the patients have a deficiency of more than two pituitary hormones.16,18 GH is typically the first pituitary hormone to become deficient in response to tumor pressure, pituitary surgery, radiotherapy, or any other trauma. 19 However, GH deficiency is probably underreported because many centers do not routinely test and report GH deficiency.
In childhood, congenital hypopituitarism is the most frequent cause of hypopituitarism, caused by various genetic mutations (Table 1) and/or developmental disorders of the pituitary gland. 17 Interestingly, congenital hypopituitarism can also occur as a syndromic form caused by mutations in genes involved in the early development of the pituitary gland. 17 Other causes are tumor-related, in children, most frequently caused by craniopharyngiomas, while pituitary adenomas are rare. 20 Annually, 0.5 to 2.5 cases of newly diagnosed craniopharyngiomas per one million people are observed, with more than one-third occurring in childhood. 21
In adults, on the other hand, the main cause of hypopituitarism is nonfunctional pituitary adenomas (NFPAs), which along with treatment by radiotherapy and surgery, account for about 70% of cases. 22 The prevalence of NFPAs ranges from 7 to 22 per 100,000. 23 In the study by Seejore et al, 24 37.6% of patients developed deficits in 3 or more anterior pituitary axes after surgical treatment of NFPA. Furthermore, radiotherapy is still considered a major cause of hypopituitarism, although, with newer techniques, the rates are lower, with up to 30% of new hypopituitarism after gamma knife radiosurgery. 25 Other causes include hypophysitis, traumatic brain injury, subarachnoid hemorrhage, and less frequently, primary empty sella syndrome and Sheehans's syndrome. 2 In high-income countries, ICIs are the most frequent cause of hypophysitis, 26 and patients taking ICI are most commonly diagnosed with ACTH deficiency but also can develop panhypopituitarism. 27 Hypopituitarism prevalence after traumatic brain injury and subarachnoid hemorrhage were reported to be 27.5% and 47%, respectively. 28 Primary empty sella syndrome in the pooled analysis showed the relative frequency of hypopituitarism in 52% of patients, although the reported prevalence varies from 8% to 60% in different studies.29,30 The prevalence of Sheehan's syndrome varies by population, appearing more frequently in developing countries, while approximately 6% of hypopituitarism patients in developed nations are attributed to this condition.16,31 In patients undergoing surgery for pituitary lesions, metastases to the pituitary are uncommon, detected in about 1.8% of cases, 32 most frequently breast cancer in women, and lung cancer in men. 33
Hypopituitarism and Mortality due to CVD
The literature suggests that the risk of premature mortality may be increased in the population with hypopituitarism. 34 Jasim et al 34 showed in their metaanalysis that hypopituitarism was associated with an overall increased mortality (weighted standardized mortality ratio [SMR] of 1.55; 95% confidence interval [CI]: 1.14-2.11, P = .000) compared to the general population. In addition, female sex and younger age, hypogonadism, the presence of craniopharyngioma, and a history of craniotomy or brain irradiation were shown to be associated with increased all-cause mortality.34–36 In another metaanalysis (n = 19,153 hypopituitary adults, six studies, follow-up of >99,000 person-years), hypopituitarism was associated with an overall increased mortality (weighted SMR 1.99) in adults, and earlier onset of hypopituitarism was associated with a higher SMR. 36 Furthermore, women with hypopituitarism had a higher SMR compared to men (2.53 vs 1.71), and GH replacement improved the mortality risk, which was comparable to the background population (SMR with GH replacement 1.15 vs SMR without GH replacement 2.40). 36
Initial studies have shown that the life expectancy of patients with hypopituitarism is reduced highly due to CVD, and that GH deficiency could be a major factor.3,36,37 The incidence of CVD was significantly higher among the hypopituitary patients (incidence ratio, 3.7; 95% CI: 1.2-11.3), and the consumption of cardioactive drugs was also significantly higher (P = .002). 38 Bülow et al 39 in their trial showed that the risk of cardio- and cerebrovascular mortality (SMR 3.21; 95% CI: 1.29-6.61) was also enhanced. In another study, increased mortality from cerebrovascular disease (SMR 3.39; 95% CI: 2.27-4.99) was the main contributor to the increased overall CV mortality (SMR 1.75; 95% CI: 1.40-2.19), but the increase in mortality from cardiac diseases was much smaller (SMR 1.41; 95% CI: 1.04-1.88), as well as mortality from ischemic heart disease (SMR 1.18; 95% CI: 0.83-1.66). 40 The relative risk for cerebrovascular death was independent of the time interval since diagnosis of pituitary insufficiency, but was greater in subjects diagnosed at an earlier age (<55 years). 40 The hazard function for CVD death was independent of gender, time after diagnosis, age at diagnosis, degree of hypopituitarism, diabetes mellitus (DM), and hypertension. 3 In a more recent nationwide study of 2,795 NFPA patients, hypopituitarism was found in 54% of patients, and CV morbidity was higher in females with hypopituitarism, and standard incidence ratios for cerebral and myocardial infarctions were higher in hypopituitary females with NFPAs versus males (P < .001). 41 Although very important, data on the prevalence of individual CV clinical entities are scarce.
The mortality rate due to craniopharyngioma in children is low, but the long-term morbidity is high and significant, which is due to chronic pituitary hormone deficiency and CVD.21,42 Furthermore, patients with Sheehan's syndrome are particularly vulnerable to CVD and higher mortality due to early onset and prolonged hypopituitarism.31,43
In recent years, the question has arisen as to whether GH replacement therapy (GHRT) improves mortality rates, however, the available data to date suggest that it does play a significant role in normalizing mortality in patients with hypopituitarism, 44 and this must be emphasized.
Pathophysiology of Hypopituitarism and CV Risk Factors
The majority of CV risk in hypopituitarism can be attributed to a cluster of classic CV risk factors such as obesity, glucose intolerance, dyslipidemia, and hypertension resulting from under- or over-replacement with hormones, 4 as shown in Figure 1. However, some experimental studies also indicate the direct effect of hormones on the myocardium and vasculature, albeit the exact pathophysiological mechanisms are not yet fully understood. 45 The vast majority of studies have focused on the effects of hypopituitarism in multiple pituitary hormone deficiencies or exclusively on GH deficiency and GH replacement, while the effects on other pituitary deficiencies and replacement treatments are unfortunately poorly understood. In most publications, authors only report the number or proportion of patients with one or more deficiencies and their impact on CV risk.

Pathophysiology of Cardiovascular Disease in Hypopituitarism.
In this section, we specifically review published data on the pathophysiological mechanisms underlying the dysfunction of specific pituitary axes and the impact on CV risk. By understanding these mechanisms, we emphasize the need for a patient-oriented optimization of HRT to mitigate CV risk in patients with hypopituitarism.
GH Deficiency
GH deficiency is highly prevalent among patients with hypopituitarism, as hypopituitarism typically develops sequentially, with GH secretion being one of the first to be impaired. 19 Given the significant effects of GH on metabolism, a deficiency of this hormone has numerous implications for the functioning of the organism, including an impact on the CV system. GH deficiency is linked to multiple CV risk factors, and individuals with this condition may be regarded at high CV risk. 46
As early as 1990, Rosén and Bengtsson 3 reported that mortality due to CVD was higher in patients with hypopituitarism compared with age- and sex-matched individuals and stated that GH deficiency might be a contributing risk factor. Van Bunderen and Olsson 32 published a metaanalysis on mortality in adult patients with GH deficiency in 2023. The analysis showed that the SMR was increased in studies that included patients without GH replacement (1.82, 95% CI: 1.19-2.46). This ratio was also higher in females than in males. 44 However, it is important to highlight some inherent limitations of studies analyzing mortality in patients with GH deficiency or hypopituitarism in general, most notably the potential for selection bias and the influence of varying underlying etiologies and treatments of hypopituitarism on mortality. Additionally, GH deficiency was not consistently assessed in all studies, especially not in earlier studies. 44
Although all pathophysiological mechanisms of the impact of GH deficiency on CV morbidity and mortality are not fully clarified, the influence of GH deficiency on particular CV risk factors is undeniable. The analysis of 2531 patients with GH deficiency in the hypopituitary control and complications study, revealed a metabolic syndrome prevalence of 42.3%. 47 It was also shown that the prevalence was higher than that of the general population in the United States and higher in the United States compared to Europe. 47 Similarly, one year later, the KIMS (Pfizer International Metabolic Database) analysis of 2,479 patients with GH deficiency reported that metabolic syndrome was highly prevalent in these patients. 48 Since the components of metabolic syndrome are independent risk factors for CVD, 49 it is expected that patients with GH deficiency are at higher risk due to the overall higher prevalence of metabolic syndrome among them. However, specific features of metabolic syndrome can exist and be evaluated separately.
The influence of GH on glucose homeostasis is well established. 50 The metabolic effects of excess GH in acromegaly include glucose intolerance and DM. 51 On the other hand, GH deficiency may be linked to impaired glucose metabolism, 52 although this should be observed separately in children and adults. In children with isolated GH deficiency, fasting hypoglycemia frequently occurs due to reduced glucose production in the liver. 53 Conversely, the KIMS analysis of 6,050 patients with hypopituitarism and adult-onset GH deficiency found a higher prevalence of DM in patients with GH deficiency. 54 Furthermore, IR has been demonstrated in adults with GH deficiency using the euglycemic hyperinsulinemic clamp, which is considered the gold standard for evaluating insulin sensitivity. 55 Greater accumulation of visceral fat and increased release of free fatty acid from visceral fat have been linked to IR in adults with GH deficiency,53,54 although not all studies are consistent.56,57 The complexity of this topic is also illustrated by the study by Garmes et al, 57 who investigated insulin sensitivity using the euglycemic hyperinsulinemic clamp in adult patients with childhood- and adulthood-onset GH deficiency and compared them with control subjects. It was found that patients with childhood-onset GH deficiency were more sensitive to insulin than healthy controls despite having a higher percentage of fat mass. 57 In contrast, patients with adult-onset GH deficiency exhibited insulin sensitivity similar to the controls. 57 This suggests that GH deficiency in childhood may alter the metabolic characteristics of tissues involved in glucose metabolism.57 Another important factor in CVD is the alteration of the lipid profile. An unfavorable lipid profile has been observed in patients with GH deficiency.58,59 Abdu et al 58 compared lipid tests between patients with hypopituitarism including GH deficiency and healthy control subjects. The concentrations of total cholesterol (TC) and LDL-C were increased in patients with GH deficiency (statistically significant only in males), while HDL-C and apolipoprotein A-I were decreased (statistically significant only in females). 58 The ratio TC to HDL-C and TG concentrations were significantly higher in patients with GH deficiency, whereas no differences were observed for apolipoprotein B and lipoprotein (a) between patients and healthy controls. 58 Similarly, other studies also demonstrated a more atherogenic lipid profile in patients with GH deficiency than in controls.59–61
Unlike insulin-like growth factor-1 (IGF-1), GH exerts a pronounced catabolic effect on adipose tissue and provides a substantial amount of metabolic energy required to support the growth of other tissues. 62 In contrast, GH deficiency increases adiposity.62,63 It has been shown that individuals with GH deficiency have a lower lean body mass and a higher fat mass. 62 Excess fat accumulates mainly in the abdominal area, particularly in the visceral compartment, which is recognized as a CV risk factor. 59 Interestingly, in these patients, the enlarged fat deposits are due to increased adipocyte volume. 63
In addition to the so-called traditional CV risk factors, nontraditional ones have also been investigated in patients with GH deficiency. 46 It was shown that they have elevated levels of C-reactive protein, as well as interleukin (IL)-6 and tumor necrosis factor-alpha (TNF-α), indicating an underlying pro-inflammatory state.46,52,64,65 Alterations in adipokine secretion, coagulation system, endothelial function, and even oxidative stress observed in patients with GH deficiency are additional risk factors for CVD, as highlighted in the comprehensive reviews by Gazzaruso et al 46 and Ratku et al. 66
GH affects the vascular endothelium, plays an important role in heart development during fetal growth, and contributes to the maintenance of normal heart structure and function. 67 Accordingly, GH deficiency may induce vascular and cardiac morphological alterations, detectable via different imaging modalities, which is important for the clinical assessment and monitoring of these patients. GH deficiency is linked to a higher prevalence of atherosclerosis, coronary artery disease, and stroke. 67 Patients with GH deficiency have been found to have increased intima-media thickness of blood vessels, which is one of the earliest morphological changes in the arterial wall associated with atherosclerosis,67,68 and is a marker for the risk of future coronary and cerebrovascular complications. 69 In addition, flow-mediated dilatation has been shown to be impaired, indicating endothelial dysfunction in adult and pediatric patients.70–72
In addition, GH deficiency in children leads to a marked reduction in cardiac dimensions. 73 When comparing patients with childhood- and adulthood-onset GH deficiency, it was also found that the patients with childhood-onset GH deficiency had smaller heart dimensions but a comparable ventricular ejection fraction. 74 However, adult patients with GH deficiency may represent a heterogeneous group that varies according to age of onset. 74 Furthermore, older patients, for instance, were not found to have differences in cardiac dimensions determined by echocardiography compared to controls, but their cardiac performance was impaired. 75 Nevertheless, earlier studies on cardiac morphology and function in patients with GH deficiency reported inconsistent findings and were predominantly conducted using conventional echocardiography. 66 Recently, in their metaanalysis assessing cardiac function and morphology using magnetic resonance imaging in adults with GH deficiency, Bioletto et al 76 found significant changes in the left and right ventricles in patients with GH deficiency compared to controls. For the left ventricle, a reduction in end-diastolic volume, stroke volume, and ventricular mass was also demonstrated in patients with GH deficiency. In addition, a reduction in end-diastolic volume and a trend towards a lower stroke volume were detected for the right ventricle. 76
Central Hypothyroidism
Consequences of the CV system in central hypothyroidism have not been widely investigated in comparison with primary; however, there is no reason to expect a difference.
It has been known for decades that triiodothyronine (T3) controls the inotropic and lusitropic properties of the myocardium, cardiac growth, myocardial contractility, and vascular function and, consequently, overt hypothyroidism has been shown to lead to CVD due to impaired left ventricular diastolic function, subtle systolic dysfunction, but also an increased risk of atherosclerosis and myocardial infarction, presumably due to associated hypercholesterolemia and hypertension. 77 Furthermore, vascular abnormalities such as increased systemic vascular resistance and altered endothelial-mediated vasorelaxation and vascular compliance have also been reported. 78
There are several studies demonstrating the effect of inadequate thyroid replacement in central hypothyroidism, independent of other pituitary hormone replacement medications, on unfavorable CV risk profiles. 79 Central hypothyroidism itself does not severely reduce life expectancy, but the quality of life can be severely affected by the hypothyroid state at any age. In a study by Klose et al of 208 GH-deficient patients starting GH replacement at baseline, patients were divided into groups with sufficient (TSH) and TSH-deficient at the beginning of the study. 9 Results showed that lower free levothyroxine (T4) was associated with higher body mass index (BMI) (r = −0.15; P = .03) and higher waist (r = −0.19; P = .03) and hip circumference (r = −0.20; P = .05), but lower HDL-C (r = 0.28; P < .001), independent of age, gender, and IGF-1. 10 No significant correlations were observed between fT4 and the other body composition or lipid markers. 10 Furthermore, subgroup analyses showed that TSH-deficient patients with the lowest fT4 had higher BMI (P = .02), total fat mass (P = .03), TC (P = .05), TG (P < .01) and waist circumference (P = .01) and lower HDL-C levels (P = .03) compared to TSH-sufficient patients, while patients with higher fT4 had higher TG levels (P < .01) but otherwise did not differ from TSH-sufficient patients. 9 Another study of patients with central hypothyroidism (n = 1,080) from the KIMS database demonstrated that patients with a low substitution dose (≤1.18 μg levothyroxine/kg/day) had higher weight, BMI and waist circumference, while those on a high substitution dose (≥ 1.58 μg levothyroxine/kg/day) had lower weight, BMI, waist circumference and IGF-1 than TSH-sufficient patients. 10
Interestingly, experimental data have shown that functional TSH receptors promote in cardiomyocytes brain natriuretic peptide secretion to some extent, suggesting a direct effect of TSH on cardiac function. 80 Furthermore, the potential direct relationship between TSH and lipids, unrelated to circulating thyroid hormones, has been demonstrated. 81
Secondary AI
There is extensive research showing that the excess risk of CVD and increased mortality in secondary AI is attributed to overtreatment with GCs, which is not surprising given the pathophysiologic mechanism of the GC's effects on the CV system. It has been demonstrated that GCs have direct effects on the heart and blood vessels, mediated by both glucocorticoid receptors (GRs) and mineralocorticoid receptors (MRs) and modified by the local metabolism of GCs by the enzymes 11 β-hydroxysteroid dehydrogenase (11βHSD). 82 In addition to the effects of cortisol via activation of ubiquitously expressed GRs, MRs in the myocardium and vascular smooth muscles are also occupied by cortisol and compete with much lower concentrations of aldosterone. 11 This leads to diverse effects of GCs on the CV system, namely on vascular development, remodeling, tone, and inflammation. 82 Furthermore, although the vascular endothelium has the activity of enzyme 11βHSD type 2, which inactivates active cortisol to inactive cortisone, it can become saturated at times of increased cortisol exposure, and the protective effect may be lost. 82
The negative effect of GCs excess in Cushing's syndrome or as a part of anti-inflammatory treatment on CV is well established. However, there are strong data demonstrating that even a modest excess of GC associated with replacement treatment in secondary AI is associated with increased CVD and excess CV mortality attributable to classic CV risk factors such as obesity, glucose intolerance, and dyslipidemia. 11 The high bioavailability and short half-life (∼90 min) of nonselective GR/MR agonists make it impossible to replicate physiological circulating cortisol levels, 83 leading to peaks in supra-physiological secretion and chronic over-replacement. Traditional substitution with daily hydrocortisone (HC) doses of 25–30 mg/day has been shown to lead to an adverse metabolic profile. However, when the dose was reduced to the equivalent of HC 20 mg/day or less, which was demonstrated in a large Scandinavian study (n = 2,424 patients with hypopituitarism), metabolic endpoints did not differ from patients not requiring GCs. 84 Patients with secondary AI on GC replacement had larger waist circumference compared to ACTH-sufficient patients, while patients on a daily HC equivalent dose of < 20 mg did not differ from ACTH-sufficient patients in terms of BMI, waist circumference, and lipid profile. 85 Furthermore, all new cases of DM, stroke, and myocardial infarction in this study occurred in the GC-treated groups. 84 In addition, in a prospective randomized controlled study on men with secondary AI Behan et al demonstrated that a lower HC replacement dose was associated with a lower arterial stiffness index and possibly a more physiological nocturnal blood pressure profile, which may confer reduced vascular risk at lower replacement doses. 85 It is noteworthy that the nonphysiological timing of HC replacement also increased morbidity and mortality. 84
In addition to the negative effects of long-term over-replacement with GCs on the CV system, under-replacement also needs to be avoided, as part of the vascular mortality may be attributed to the adrenal crisis. 86 Over the years, case reports of severe CV complications, namely left ventricular dysfunction, cardiomyopathy, Takotsubo cardiomyopathy, and lethal arrhythmias due to prolonged QT interval associated with secondary AI have been reported.87–89 These complications occurred during acute stress and resolved shortly after treatment with HC.87–89 In a large study (n = 6,821 patients with AI, secondary AI in 3948 of the patients), AI patients were compared with 67,564 individually-matched controls and were found to have increased mortality (hazard ratio [HR] for secondary AI 1.52 [1.40-1.64]), with CVD being the leading cause of death (HR 1.54 [1.32-1.80]), specifically ischemic heart disease. 90 However, a substantial proportion of patients died from infection (HR 4.00 [2.15-7.46]), which was also demonstrated in a large prospective postmarketing surveillance study on adult GH replacement (KIMS database), 91 and by Burman et al. 92 These studies raise the question of whether some of the CV mortality reported in trials on secondary AI in general, may be overestimated, that is, adrenal crisis and sudden death were misclassified as CV causes of death.
Interestingly, GCs and mineralocorticoids (MCs) have both been shown to have a direct effect on cardiac electrical activity. 93 Preclinical studies in adrenalectomized mice suggest that GC signaling plays an important role in regulating left ventricular function, while MC signaling has a predominant effect on modulating cardiac electrical activity, 94 specifically chronic GC deficiency was found to be related to left ventricular dysfunction and MC deficiency to arrhythmia. 94 Given these recent findings, it is important to bear in mind that the key clinical difference between primary and secondary AI is the intact renin-angiotensin-aldosterone system (RAAS) in secondary AI. Whether this has a clinical effect on CV outcomes in patients is not known. However, it is a fact that patients with secondary AI have an increased risk of adrenal crisis, which is about half of that what is seen in primary AI. 86 Further studies on the effects of an intact RASS in secondary AI on morbidity and mortality are needed.
In addition, patients with secondary AI and concomitant GH deficiency may be more susceptible to metabolic adverse effects of standard GC replacement treatment. It has been shown that hypopituitary patients on GC replacement therapy and untreated GH deficiency may have increased sensitivity to GCs due to an upregulation of 11βHSD type 1 in the absence of GH and with reduced circulating IGF-1 levels, implying that patients receiving cortisone acetate may have higher physiologic tissue exposure to glucocorticoids in the GH deficient state. 95
Secondary Hypogonadism in Men
Hypogonadism, characterized by low testosterone levels, is increasingly recognized as a significant factor in the pathophysiology of CVD, leading to increased CV morbidity and mortality. 45 Both primary and secondary testosterone deficiencies are linked to an increased risk of CVD, though the underlying mechanisms differ. Primary hypogonadism is directly related to low testosterone and its metabolic effects. In contrast, secondary hypogonadism often involves additional risk factors such as obesity, metabolic syndrome, and systemic disease that increase the risk of CVD.96,97
Published data show that testosterone enhances myocardial function through direct and indirect effects on myocardiocytes.98–100 Consequently, hypogonadism may increase susceptibility to functional damage, particularly after the onset of coronary artery disease. 101 Accordingly, testosterone deficiency has been shown to directly impair cardiac function, particularly diastolic function, that is impaired relaxation of the ventricles, which can lead to HF with preserved ejection fraction (HFpEF). 102
The relationship between testosterone and cardiac function is further demonstrated by studies in men with type 2 DM, in which lower testosterone levels were associated with worsening diastolic dysfunction independently of other CVD. 102
In addition to its role in diastolic function, testosterone also exerts a protective effect on the heart by preventing fibrosis, which can impair its function. 103 The animal study showed that testosterone inhibits the migration, proliferation, and differentiation of cardiac fibroblasts into myofibroblasts, thus reducing the risk of myocardial fibrosis. 104 This antifibrotic effect is critical in preventing HF, particularly HFpEF, which is often caused by fibrosis in cardiac tissue. 98
Moreover, the antioxidative properties of testosterone are of crucial importance for the mitigation of oxidative stress, which contributes significantly to the development of CVD. 105 Oxidative stress leads to activation of fibroblasts, increased collagen production, and myocardial stiffness, all of which contribute to the progression of heart disease. Testosterone's ability to modulate oxidative pathways and reduce collagen production helps to maintain myocardial flexibility and prevent cardiac tissue stiffening. Clinical trials have demonstrated that testosterone replacement therapy enhances myocardial function and improves exercise capacity, skeletal muscle performance, and IR in patients with chronic HF. 98
Hypogonadism is closely associated with adverse changes in lipid metabolism, which significantly contribute to the development of atherosclerosis, a key driver of CVD. Men with testosterone deficiency often have elevated TC and LDL-C levels, while HDL-C levels decrease. 106 These lipid abnormalities create a pro-atherogenic environment that promotes the accumulation of cholesterol in arterial walls and leads to plaque formation. 106 Furthermore, low testosterone impairs HDL function, particularly its cholesterol efflux capacity, which is essential for removing cholesterol from macrophages in arterial walls. 107 Impaired HDL function increases the risk of foam cell formation and the development of atherosclerotic plaques, both of which are critical in the pathogenesis of coronary artery disease. 107
Testosterone deficiency also promotes subclinical atherosclerosis, as evidenced by increased intima-media thickness of the carotid arteries and aorta108,109 and elevated coronary artery calcium scores. 109 These markers are strongly predictive of future CV events such as myocardial infarction and stroke. The increased CV risk associated with low testosterone levels underscores the need for early detection and management of hypogonadism to prevent the progression of atherosclerosis and related complications. 109
Testosterone deficiency is associated with a pro-inflammatory state, which exacerbates the development of atherosclerosis and other CVD.109,110 Hypogonadal men have elevated levels of pro-inflammatory cytokines such as IL-6, TNF-α, and high-sensitivity C-reactive protein (hsCRP), all of which are known to promote endothelial dysfunction and accelerate the atherosclerotic process.109,111
Endothelial dysfunction, characterized by impaired nitric oxide synthesis and increased vascular inflammation, is a precursor to atherosclerosis. Testosterone has been shown to benefit endothelial function by increasing nitric oxide bioavailability, thus improving vasodilation and reducing vascular stiffness. 112 Low testosterone levels, however, impair these protective mechanisms and lead to increased vascular tone, inflammation, and the development of CVD.101,113 The study by Sonmez et al 114 highlights that young, untreated male patients with congenital HH exhibit significant markers of endothelial dysfunction, IR, and inflammation, even before testosterone replacement therapy. Specifically, these patients showed elevated levels of asymmetric dimethylarginine, a marker of endothelial dysfunction, and increased insulin and HOMA-IR (homeostatic model assessment for IR) levels, indicating IR, decreased HDL-C, and increased LDL-C and TG compared to healthy controls. 114
One of the lesser-known CV risks associated with testosterone deficiency is prolongation of the QT interval,115,116 which increases the risk of life-threatening arrhythmias such as torsade de pointes. 117 The study by Pecori Giraldi et al 116 has shown that hypogonadal men, regardless of whether they have primary or secondary causes, have a significantly higher prevalence of prolonged QT intervals compared to healthy controls. Interestingly, testosterone replacement therapy was found to normalize QT intervals in hypogonadal men, supporting the hypothesis that testosterone plays a crucial role in regulating cardiac repolarization and protecting against arrhythmias. 116 This finding suggests that QT interval monitoring should be part of the CV assessment in hypogonadal patients, particularly those at risk for arrhythmias. The ability of testosterone replacement therapy to correct prolonged QT intervals highlights its potential as a protective therapy against arrhythmia-related complications in hypogonadal men. 116
Metabolic syndrome, which is characterized by a cluster of conditions including IR, central obesity, dyslipidemia, and hypertension, is strongly linked to hypogonadism. 97 Testosterone deficiency exacerbates IR, leading to hyperglycemia, hyperinsulinemia, and an increased risk of developing type 2 DM, all of which contribute to the CV risk.114,118
Studies have demonstrated significantly higher fasting plasma glucose levels, elevated fasting insulin, and increased IR in men with idiopathic HH compared to healthy controls.114,118,119 These findings underscore the metabolic disturbances associated with testosterone deficiency, which increase the risk of CVDs. 118
Secondary Hypogonadism in Women
Secondary hypogonadism in women is a complex condition characterized by a deficiency of sex hormones, primarily estrogen, due to dysfunction in the hypothalamic-pituitary-gonadal (HPG) axis. This deficiency significantly impacts CV health, increases the risk of CVD, and diminishes the natural survival advantage typically seen in women over men. 45 The most common causes of death in women with secondary hypogonadism are CV and cerebrovascular diseases, which are often linked to metabolic syndrome and the side effects of treatments such as cranial irradiation. 45
In hypopituitarism, the lack of GH and estrogen plays a central role in increasing CV risk. 45 GH deficiency contributes to an unfavorable metabolic profile, including increased visceral adiposity, dyslipidemia, and IR. 45 These metabolic disturbances are associated with a higher prevalence of atherosclerosis, endothelial dysfunction, and increased arterial stiffness. 52 Additionally, estrogen deficiency further exacerbates these effects and leads to a greater incidence of CVD compared to the general population. 120
The role of estrogen in CV health is well documented. It contributes to the regulation of lipid profiles, enhances endothelial function, and has anti-inflammatory properties. In women with secondary hypogonadism, estrogen deficiency leads to adverse lipid profiles characterized by elevated LDL-C and decreased HDL-C levels.12,13 This imbalance, along with increased arterial stiffness and impaired glucose metabolism, significantly raises the risk of atherosclerosis and other CV complications. 12
The risk of CVD is particularly high in women with obesity-related secondary hypogonadism. 121 Obesity leads to reduced LH levels and impaired gonadotropin-releasing hormone pulsatility due to increased hypothalamic leptin resistance and IR. These hormonal disruptions contribute to an adverse lipid profile and chronic low-grade inflammation, which further increases the risk of CV events such as coronary artery disease and stroke. 121
Young women with primary ovarian insufficiency or those with conditions such as Sheehan's syndrome face unique CV risk due to the early onset of estrogen deficiency. 31 Early estrogen loss disrupts the balance between vasodilator and vasoconstrictor factors, leading to increased arterial stiffness and endothelial dysfunction. This early estrogen deficiency, coupled with metabolic disturbances such as IR and central adiposity, elevates the risk of developing metabolic syndrome and CV events at a young age.31,122
Women with HH due to hypothalamic or pituitary dysfunction also exhibit distinct CV risks. 123 In these women gonadotropin secretion is impaired, resulting in severe estrogen deficiency and a higher risk of CVD.124,125 This condition is often accompanied by additional factors such as IR and visceral adiposity, compounding the CV risk.124,125
Overall, the pathophysiology of CV disease in women with secondary hypogonadism involves a complex interplay of hormonal deficiencies, metabolic disturbances, and inflammatory processes.
Prolactin (PRL) Deficiency
Based on the current evidence, there is a causal relationship between elevated PRL levels and CVD. 126 Evidence also suggests that PRL is involved in obesity, endothelial function, glucose metabolism, and lipid metabolism, and may even be associated with increased CV mortality. 127
Unlike hyperprolactinemia, the clinical significance of PRL deficiency remains poorly understood. Initial research showed that in patients consulting for sexual dysfunction, PRL levels in the lowest quartile levels were associated with metabolic syndrome. 128 Recently, Krysiak et al 129 assessed the cardiometabolic profile of patients with low PRL levels in their study. Women of reproductive age with hypoprolactinemia had higher levels of glycated hemoglobin, 2-hour postchallenge glucose, uric acid, TG, hsCRP, and fibrinogen, lower levels of total testosterone, free androgen index, and HDL-C, as well as reduced insulin sensitivity. 129 The results of this study suggest that PRL deficiency may also increase CV and metabolic risk. This lack of data indicates that future studies must also address this issue in more detail.
Clinical Assessment of CV Diseases in Patients With Hypopituitarism
There are no recommendations for the assessment of CV status in these patients supported by high-quality evidence. Guidelines for the management of GH deficiency in adults and in patients transitioning from pediatric to adult care recommend monitoring fasting lipids, blood pressure, and heart rate at 6- to 12-month intervals and considering more detailed investigations (electrocardiogram, echocardiography, and carotid ultrasonography) when clinically indicated. 130 However, this statement is based on expert opinion. 130
Ratku et al 66 also suggested close follow-up of CV status and advised maintaining a low threshold for conducting comprehensive diagnostic evaluations, with the potential use of various imaging methods (echocardiography, cardiac magnetic resonance imaging, and carotid ultrasonography) for assessing these patients.
This proactive strategy is crucial for the early detection and management of CV risk factors and can potentially mitigate the long-term CV complications associated with GH deficiency, secondary hypothyroidism, AI, and hypogonadism.
Impact of Hypopituitarism Treatment on CV Risk
HRT aimed at addressing deficiencies in GH, thyroid hormones, testosterone, estrogens, and cortisol is expected to improve CV outcomes by regulating metabolic and inflammatory processes. However, potential side effects and inadequate dosing might contribute to residual risks. Therefore, effective management of these therapies is crucial to reduce the increased CV risk. To reduce the risk, it is essential to promote regular monitoring, personalized treatment plans, patient education, management of coexisting diseases, encouraging a healthy lifestyle, adjusting treatment to changing circumstances, and preparing patients for emergency situations. 131
In this section, we specifically focus on the effects of different treatment options on CV risk, analyzing their impact on CV health and potential risk modification (as shown in Table 2), emphasizing the importance of patient-oriented optimization of HRT to mitigate CV risk in patients with hypopituitarism.
Hormone Replacement Options and Their Role in CVD Risk.
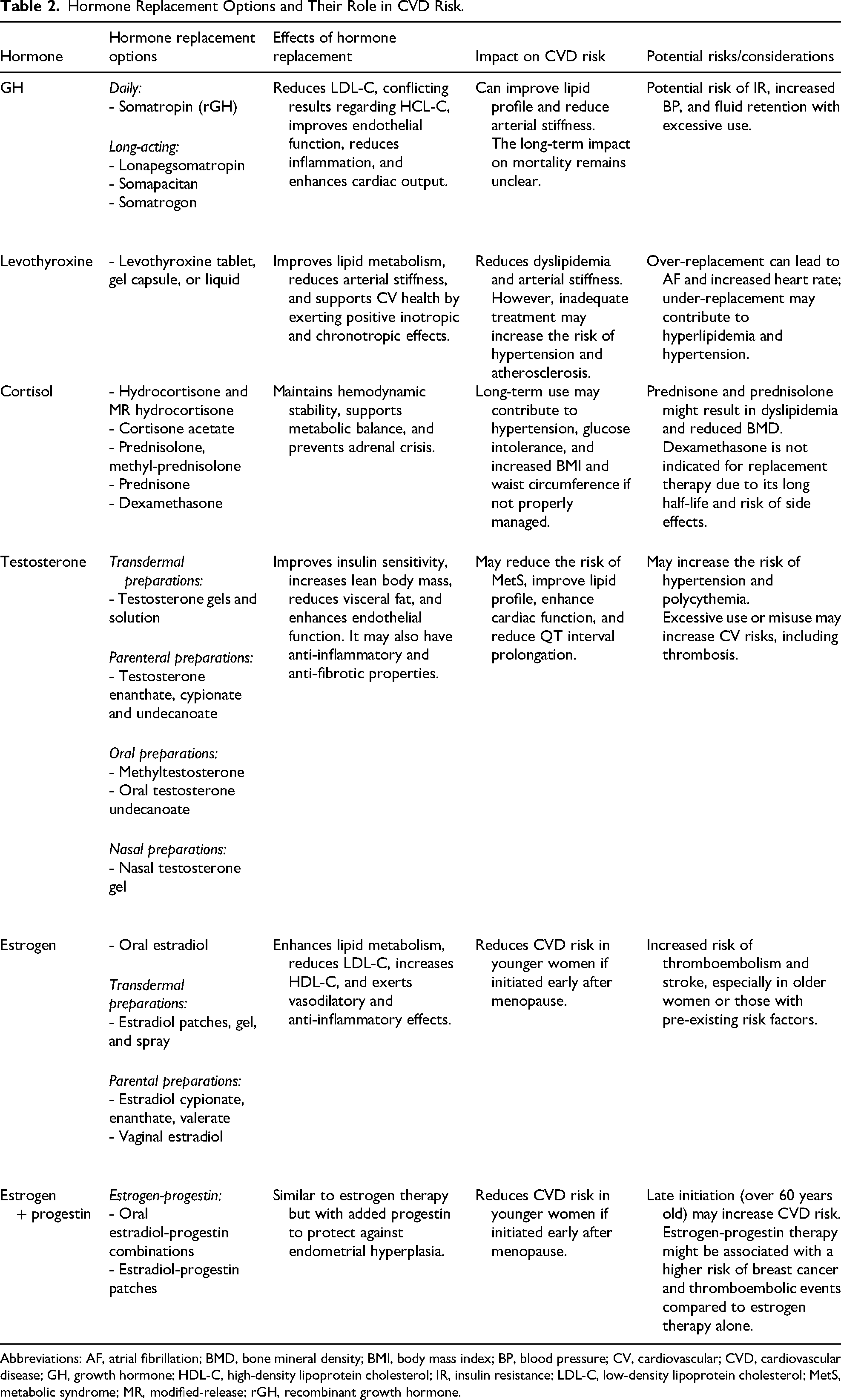
Abbreviations: AF, atrial fibrillation; BMD, bone mineral density; BMI, body mass index; BP, blood pressure; CV, cardiovascular; CVD, cardiovascular disease; GH, growth hormone; HDL-C, high-density lipoprotein cholesterol; IR, insulin resistance; LDL-C, low-density lipoprotein cholesterol; MetS, metabolic syndrome; MR, modified-release; rGH, recombinant growth hormone.
GH Replacement Treatment
Conventional GH replacement therapy involves once-daily GH application. Once-weekly formulations such as somapacitan, somatrogon, and TransCon GH (lonapegsomatropin), a recombinant human sustained-release GH prodrug, are being developed to ensure their efficacy and enhanced compliance. 132 Somapacitan is the first approved once-weekly reversible albumin-binding GH derivative (long-acting GH—LAGH), which was shown to decrease truncal fat, improve visceral fat and lean body mass, increase IGF-1 standard deviation score, and exert neutral effects on glucose metabolism. It is well-tolerated, adverse event rates are comparable to daily GH, and it decreases the treatment load of once-daily GH injections. 133
The effects of GH replacement therapy on CV risk were analyzed using many single parameters, such as lipid levels, glycated hemoglobin, insulin levels, blood pressure, hsCRP, serum adhesion molecules (eg, intercellular adhesion molecule-1 [ICAM-1] and vascular cell adhesion molecule-1 [VCAM-1]) and several markers of organ damage, such as cardiac function and endothelial dysfunction estimated from carotid artery intima-media thickness, and flow-mediated dilatation of the brachial artery.134,135
It is well-known that GH deficiency leads to dyslipidemia with high TC, LDL-C, and low HDL-C levels.46,136 Many studies that have analyzed the influence of GH replacement therapy on dyslipidemia have reported a significant decrease in LDL-C levels (around −7%), with conflicting results for TC and HDL-C. 6 GH replacement in women with Sheehan's syndrome decreases hsCRP regardless of improvement in serum lipids and lean body mass.31,137
Since GH inhibits insulin-mediated glucose uptake in skeletal muscle, several studies have analyzed the effects of GH replacement therapy on glucose metabolism and IR.6,7 Some studies suggest that GH replacement therapy modestly decreases insulin sensitivity. 7 However, the metaanalysis showed no significant effect of GH replacement therapy on plasma glucose levels or glycated hemoglobin, even with long-term therapy. 6 Likewise, blood pressure does not seem to be affected by GH treatment, neither in the short- nor in the long-term observation.6 Endothelial function seems to be impaired in GH deficiency. Studies have shown a decreased flow-mediated dilation in patients with hypopituitarism compared to healthy subjects, and GH replacement therapy for 6 months improved the flow-mediated dilation. 138 Also, serum levels of ICAM-1 and E-selectin were higher in hypopituitary patients with GH deficiency than in healthy controls and correlated negatively with IGF-1 levels.70,139
Treatment of Secondary Hypothyroidism
Both insufficient and excessive replacement therapy is associated with CV disorders, such as arrhythmias, atherosclerotic vascular disease, and HF.8,140
Replacement treatment can only be started after the normal function of the hypothalamic-pituitary-adrenal axis is confirmed or after the correct HC replacement has been started. 2 The standard replacement therapy for secondary hypothyroidism is T4. 141 T4 has a long half-life but difficulties in gastrointestinal absorption. Novel formulations enable its administration without food restriction. 141 Rarely, T3 is used for short-term replacement therapy. It is better absorbed by the gastrointestinal tract, but its long-term effects on arrhythmia risk are unknown. 140 Hyperthyroidism has been associated with both QTc prolongation and short QTc intervals, and hypothyroidism with QTc prolongation and an increased risk of torsades de pointes. 142 Few data are available on the effects of thyroid hormones on atrial and ventricular myocytes and nodal and His-Purkinje cells. 140 Hypothyroidism alters the lipid profile, including levels of TC, LDL-C, HDL-C, TG, and levels of apolipoprotein A and apolipoprotein B. 143 When hypothyroid patients are treated with T4 therapy, a significant decrease in TC, LDL, and TG values is usually seen. 144 However, T4 substitution in patients with secondary deficiency appears insufficient in preventing hyperlipidemia. 145 Hypothyroidism has been linked to diastolic hypertension, increased plasma homocysteine levels, endothelial dysfunction, and higher carotid intima-media thickness.79,146,147 Thyroid hormones exert positive inotropic and chronotropic effects and reduce systemic vascular resistance by increasing endothelial nitric oxide production and calcium reuptake within the arterioles. 148
Glucocorticoid Replacement Treatment
The most commonly used replacement therapy for secondary AI worldwide is immediate-release oral HC. The usual dosage in adults is 15–20 mg in divided doses, working to replicate a healthy 24-hour cortisol circadian rhythm. 11 Extended use of doses > 20 mg HC or equivalent per day must be avoided as they have been associated with adverse metabolic outcomes and increased mortality in patients with hypopituitarism. 149
Dexamethasone is disadvantageous in the context of replacement therapy because of its long-term metabolic and CV consequences. 150 Other options for GC replacement are prednisolone, methyl-prednisolone, once- and twice-daily modified-release HC (MRHC), cortisone acetate, and an immediate-release oral HC granule formulation. 141 In a study of patients with primary and secondary AI a similar cardiometabolic risk was reported in patients treated with HC (mean dose 20.5 mg daily) and prednisolone (mean dose 3.7 mg daily). 151 However, prednisolone-treated patients had significantly higher LDL-C levels than patients treated with HC. 152 The once-daily MRHC formulation (Plenadren) comprises an immediate-release coating and an extended-release core. It enables more physiologic cortisol secretion and reduces 24-hour exposure to cortisol. 153 The glucocorticoid replacement option, which is used exceptionally but can be the only option for poorly controlled patients, is continuous subcutaneous hydrocortisone infusion (CSHI), which enables a reduced daily dose of glucocorticoids and a reduced number of hospitalizations. 154
Treatment of Secondary Hypogonadism in Men
Testosterone replacement therapy for men with secondary hypogonadism follows the same principles as for those with primary hypogonadism. The most commonly used formulations include intramuscular depot injections of testosterone undecanoate, testosterone enanthate, cypionate, and transdermal gels. 141 Less common formulations, such as oral testosterone undecanoate, buccal testosterone, and testosterone pellets, are also options for testosterone replacement therapy. 141 There are not a lot of studies comparing different modes of testosterone replacement. The study from the HEAT-Registry found that long-acting testosterone undecanoate, when compared to transdermal formulation, leads to a higher rate of hematocrit levels >50%. This effect is especially prevalent in obese older men with functional hypogonadism. 155
Studies have found a beneficial effect of testosterone administration on insulin sensitivity, glycated hemoglobin levels, and fasting blood glucose levels. 156 Many trials, including testosterone trials (TTrials), have indicated that testosterone treatment results in lower TC and LDL-C levels, although the effects on HDL-C are unknown. 157 However, the testosterone in older men with mobility limitations (TOMs) trial had to be stopped prematurely due to an increased incidence of CV events in the intervention group. 158 The participants in this study had a high risk of CV events at baseline (approximately one-third had diabetes and obesity, more than half had hyperlipidemia, more than three-quarters had hypertension, and nearly half had pre-existing heart disease). 158 The participants who experienced CVD had lower serum total testosterone levels at baseline, which might indicate a greater underlying disease burden. 158 Similarly, a retrospective cohort study showed a significant increase in the rate of nonfatal myocardial infarction postprescription of testosterone, compared to preprescription, with a significantly pronounced effect in men aged over 75 years. 159 In men aged <65 years, the excess risk was limited to those with a history of heart disease. 159 One study showed an inverse association between endogenous total testosterone and CVD in men over 70 years and no association in younger men. 160 The metaanalysis found no significant association between testosterone therapy and CV events and mortality. Due to the heterogeneous baseline population characteristics in the studies analyzed, the quality of the evidence was rated low. 161 A recent clinical trial revealed that in men with hypogonadism and preexisting or a high risk of CVD, testosterone replacement therapy was noninferior to placebo concerning the incidence of major adverse cardiac events. 162
Treatment of Secondary Hypogonadism in Women
Mortality in patients with hypopituitarism is higher in women than in men. 36 Moreover, DM, hypertension, and abdominal adiposity are more prevalent among hypopituitary women than in men. 41 There can be many reasons for this, but the most obvious distinguishing factor between men and women is testosterone and estrogen replacement therapy. Some argue that estrogen replacement may be insufficient in women of fertile age. 163 Untreated female hypogonadism is associated with an increased risk of CV and cerebrovascular disease and an increased SMR; SMR of 2.09 in females with untreated hypogonadism and SMR of 0.94 in treated hypogonadism. 164
HRT can mitigate some of the CV risks by restoring estrogen levels. For hypogonadal women of fertile age without contraindications, estrogen-progesterone replacement therapy is beneficial.164–166 The data on estrogen-progesterone replacement therapy after fertile age are less uniform. The metaanalysis found that the risk of coronary artery disease was reduced by 32% in early menopausal women treated with HRT who had been in menopause for < 10 years or in women who were younger than 60 years. 167 However, no such beneficial effects were evident for women aged 60 or older.14,167 Furthermore, increasing doses of HRT in young hypogonadal women have been shown to reduce carotid intima-media thickness and improve lipid profile, suggesting a cardioprotective effect when initiated early. 168 This effect contrasts with findings in postmenopausal women, in whom HRT can increase CVD risk. 168 The data imply that the impact of hormone therapy on the CV system could differ depending on the timing of initiation, HRT formulation, dose, and risk factor profile of the patient.14,169 Some observational data suggest a lower risk of VTE for transdermal formulations. 170 With respect to the risk of myocardial infarction and stroke, clinical trial data are lacking. 171
Women with secondary hypogonadism and simultaneous secondary adrenal insufficiency have severe androgen deficiency. 171 Also, both estrogen and GC replacement may suppress endogenous androgen secretion in women with hypopituitarism.172,173 Studies that have analyzed an association between androgen levels and the risk of CV events have reached inconsistent conclusions.174–176 Furthermore, there is minimal data on the manner of androgen replacement and the health consequences of such treatment. 171 An article by Renke and Tostes 177 analyzed CV safety and the benefits of testosterone implant therapy in postmenopausal women. They conclude that this topic remains controversial as no well-defined criteria for diagnosing androgen deficiency and monitoring treatment exist. Also, data on the long-term impact on CV health are lacking.
Conclusion
There is convincing evidence that pituitary insufficiency, regardless of whether it involves one or multiple pituitary hormones, is associated with increased mortality, mainly due to CVD, particularly in females and young patients. More precisely, patients with hypopituitarism, especially those with insufficient levels of GH and ACTH, are more susceptible to developing CVD.
The mechanisms for this phenomenon are complex and multifactorial. Common CV risk factors and HRT may further complicate CV health management in these patients. In the majority of patients, it is due to the cluster of classic CV risk factors such as visceral obesity, impaired glucose metabolism, dyslipidemia, hypertension, and adverse changes in body composition, resulting from under- or over-replacement with standard HRT. In addition, nontraditional CV risk factors such as increased oxidative stress, pro-inflammatory state, and endothelial dysfunction may contribute. By recognizing their shared influence across different pituitary axes and treatment strategies, our goal was to provide a comprehensive perspective on their impact on CV risk and underscore the importance of a tailored therapeutic approach.
In patients with hypopituitarism, HRT should be considered as a treatment that modifies CV risk, but it should be optimal and personalized. Mortality rates in hypopituitary patients appear to improve after GH replacement at levels comparable to the general population, although female patients benefit less than males. Furthermore, testosterone replacement therapy in men and estrogen-progesterone in fertile women, as well as optimal dosing of thyroid hormones, have positive effects on the CV system. On the other hand, overtreatment with glucocorticoids, leading to an unfavorable metabolic profile, remains a problem in the majority of patients, even though progress has been made in the field of HC administration in a more physiological pattern. Despite these advances, CV morbidity remains the main burden, and early diagnosis and successful treatment of hypopituitarism remains a challenge and the only way to normalize the CV burden of pituitary disease.
Recent developments in personalized hormone replacement have demonstrated a reduction in certain CV risks, resulting in better patient outcomes. Tailored management of CV risk factors in hypopituitary patients is crucial for minimizing long-term CV morbidity and mortality. Thus, continued research is necessary to refine treatment strategies and enhance CV outcomes in these patients.
Currently, no conclusive evidence shows that treatment of hypopituitarism completely reduces CV risk, primarily because long-term, prospective, and controlled clinical trials necessary to establish such outcomes have not been conducted. While various studies may suggest potential benefits, these findings remain inconclusive without robust data from extended, well-designed trials that could more definitively confirm the impact of hypopituitarism treatment on CV risk mitigation. Therefore, continued research is necessary to refine treatment strategies and enhance CV outcomes in these patients.
Footnotes
Author Contributions
Ivana Kraljević contributed to the conception and design, wrote the manuscript, contributed to the research data discussion, and critical review of the manuscript. Mirsala Solak contributed to the conception and design, wrote the manuscript, and contributed to the research data discussion, and critical review of the manuscript. Ante Mandić contributed to the conception and design, wrote the manuscript, contributed to the research data discussion, and critical review of the manuscript. Maja Raičević contributed to the conception and design and wrote the initial draft of the manuscript. Katarina Mlekuš Kozamernik contributed to the conception and design, wrote the initial draft of the manuscript, and contributed to the research data discussion. Anida Divanović Slato contributed to the conception and design and wrote the initial draft of the manuscript. Zlata Kovačević contributed to the conception and design, and wrote the initial draft of the manuscript. Emir Muzurović contributed to the conception and design, wrote the manuscript, contributed to the research data discussion, and critical review of the manuscript. All authors reviewed and approved the final version of the manuscript.
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
