Abstract
Background:
Heart failure (HF) is an important adverse outcome of diabetes mellitus (DM) with high rates of mortality and HF-related hospitalizations. The risk of HF is 2 times higher in patients with DM compared to those without DM. Due to under-recognition and underdiagnoses, HF is often a neglected outcome in the diabetic population. There is a dearth of data regarding the true prevalence of HF and the management protocols for diabetic patients at risk of HF in the UAE and the Middle East. This lacuna in the information has led to the inception of this “call to action” paper, which identifies the gaps in the true prevalence of HF and describes the importance of early diagnosis and appropriate management of HF in the Middle East.
Methodology:
An advisory board meeting was convened and a group of key opinion leaders and experts in cardiology and endocrinology assembled to describe the prevalence, diagnosis, and management of HF in diabetes patients and to present a “call to action” in the UAE and Middle East scenario. After the group discussion, key expert opinions were formulated and “call to action” recommendations were proposed.
Conclusion:
This “call to action” is mainly based on the available evidence from the literature and the experts’ clinical experience. Based on the new evidence from various cardiovascular outcome trials, the “call to action” highlights a series of collaborative learning regarding the role of newer antidiabetic therapies like sodium–glucose cotransporter-2 inhibitors in the prevention and management of HF. This “call to action” intends to serve as a guide for physicians, including primary healthcare providers, in their management of diabetic patients with HF.
Keywords
Introduction
Heart failure (HF) has a significant impact on public health in Middle East countries. Although the true prevalence of diagnosed HF in the Middle East is not known, the extrapolation from known databases suggests that an estimated 1.35 million patients are undergoing treatment for HF. 1 This excludes the prevalence of subclinical HF and patients who have not received HF treatment and are not being followed up. HF is a common cardiovascular (CV) complication in patients with diabetes mellitus (DM), and the risk of HF is 2 times higher in patients with DM compared to those without DM. 2 HF in diabetic patients is caused by multiple factors with hyperglycemia and insulin resistance playing pivotal roles. 3 A study conducted on outpatients in an HF clinic revealed an 18% prevalence of HF in the newly diagnosed diabetes population, and the total prevalence of diabetes in these HF outpatients was 34%. 4 Diabetes also serves as an independent risk factor for mortality in patients with HF. 5 In contrast to patients with normal glucose tolerance, mortality is approximately threefold higher (29%) in previously diagnosed diabetic patients. 4 Crude mortality analysis in hospitalized HF (HHF) patients reported a much higher 1-year mortality rate in patients with diabetes as compared to patients without diabetes (31% vs 23%). 5 Moreover, after 3 years, 50% of all diabetic HF patients died. 5,6 Thus, among HF patients, those with diabetes are at a higher risk of hospitalization due to HF and mortality. 6
The increased occurrence of HF in diabetic patients is attributed to diabetic cardiomyopathy. 7 Diabetic cardiomyopathy is a challenging diagnosis that can occur in the absence of coronary artery disease (CAD) and has been reported in up to 68% of patients with type 2 diabetes mellitus (T2DM) of even 5-year duration. 8 Diabetic cardiomyopathy could manifest as systolic and/or diastolic left ventricular dysfunction. Diastolic dysfunction, representing the early stage of diabetic cardiomyopathy, is frequently observed in T2DM patients and is associated with a poor prognosis. 7,9 Even in well-controlled asymptomatic diabetic patients, 47% were diagnosed with diastolic dysfunction. 10 This reiterates the need for early diagnosis and management of HF in patients with diabetes.
Further, antidiabetic drugs like glitazones and certain dipeptidyl peptidase-4 inhibitors, such as saxagliptin, have shown to increase the risk of HF including new-onset and progressive HF in diabetes patients. 11 -14 Insulin and sulfonylureas may increase the risk of CV death mostly due to hypoglycemia and other conflicting mechanisms that need further validation. 15 -17
Several mechanisms explain the pathophysiology of HF in patients with DM. 3 Metabolic disturbances, such as hyperglycemia, dyslipidemia, accumulation of advanced glycated end products, obesity, and inflammation, can lead to oxidative stress, impaired calcium homeostasis, autonomic neuropathy, and activation of the renin–angiotensin system (RAS) system. These negative effects in turn can cause endothelial dysfunction, atherosclerosis, cardiac fibrosis, and cardiac dysfunction, thereby triggering HF (Figure 1). 3
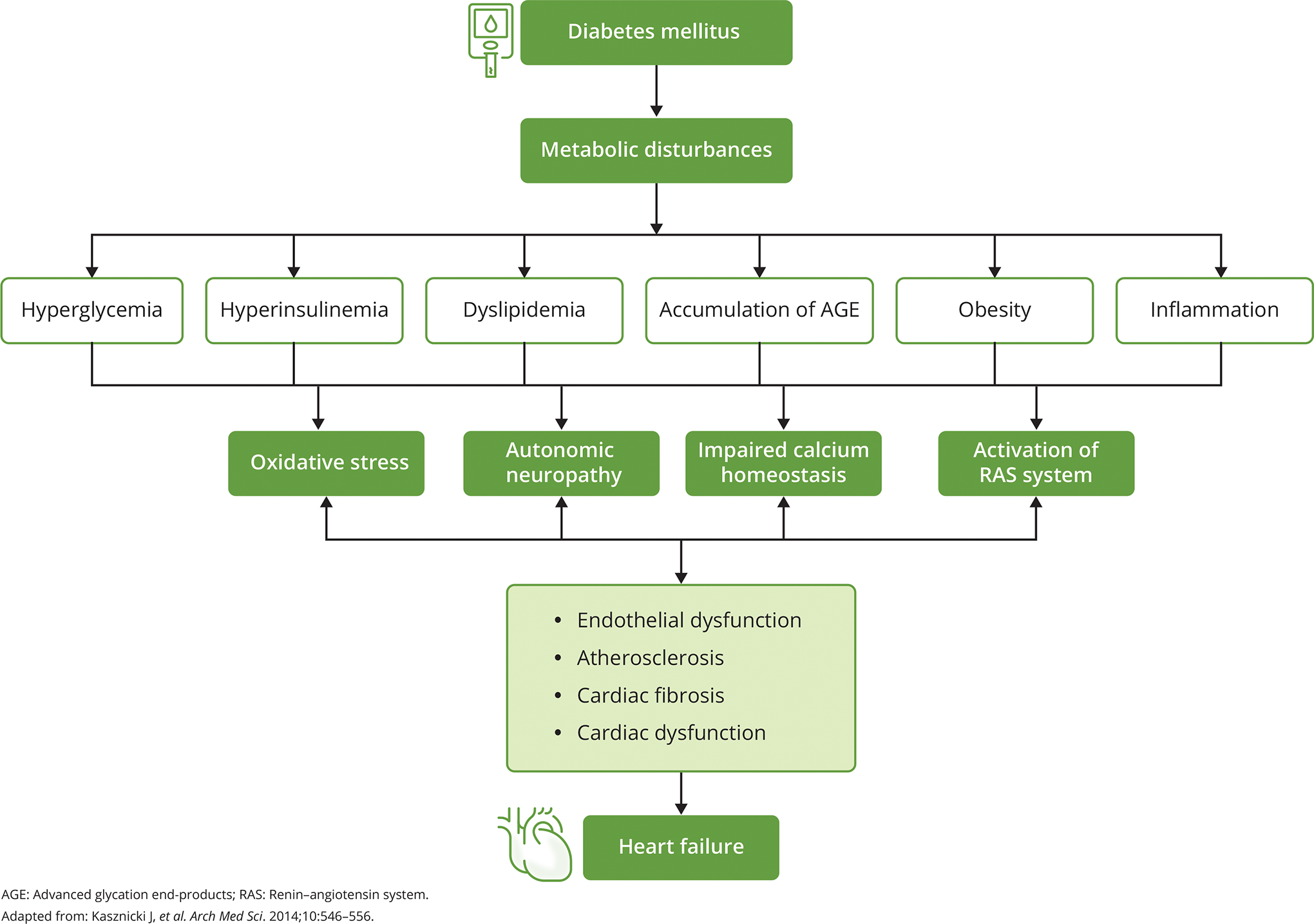
Pathophysiology of heart failure in diabetic patients. 3
As seen in global data, the prevalence of HF in diabetic patients is reported to be around 9% to 22%, which is 4 times higher than that reported in the general population. 18 According to the Framingham study, DM is an independent risk factor for HF, increasing the risk by twofold in males and fivefold in females. 2 A recent meta-analysis by Einarson et al reported a CV disease (CVD) prevalence of 32.2% and HF rate of 14.9% in patients with DM. However, the study has possibly underestimated the prevalence of HF as this registry included only patients with confirmed hospitalized HF. 19 According to the Gulf CARE registry, the prevalence of DM was around 50% in patients with HF. 20 Further, the UAE and the Gulf countries have shown the highest prevalence of DM in the younger population. 21 Therefore, an epidemic of HF in this population is quite likely, warranting the need for early diagnosis. Though the prevalence of HF in diabetes is reported in different populations, there is a lack of studies reporting the true prevalence in the Gulf region. Hence, more studies are warranted in the Gulf region to understand the true prevalence of HF in the diabetic population. Further, patients with both comorbidities are at a higher risk of adverse outcomes, including higher mortality, thereby posing a clinical challenge to the treating physician. In addition, the early stages of HF diabetic cardiomyopathy are particularly challenging from a diagnostic standpoint because these patients generally visit a diabetes clinic and/or a primary care setting for diagnosis and treatment. Moreover, the initial recognition of diastolic cardiomyopathy due to diabetes requires echocardiography. Access to diagnostic tests is limited in the earlier subclinical or less symptomatic stages, thereby further impacting management in the early stages. Hence, the management of these patients is critical, and there is a lack of management protocol for patients with comorbidities of HF and DM in the UAE scenario. Given these challenges, the key opinion leaders in the fields of cardiology and endocrinology came together at the St. Regis Hotel, Abu Dhabi on 07th February 2020 to describe the prevalence, diagnosis, and management of HF in diabetic patients and to present a “call to action” in the UAE scenario. This “call to action” is mainly based on the available evidence from the literature and the experts’ clinical experiences.
Early Detection of HF in T2DM Patients
Early identification of HF in the diabetes population can reduce the associated negative outcomes of comorbidities. However, identifying the diabetic population at risk of HF is challenging, and diagnostic methods predicting these risks are the need of the hour. According to a meta-analysis by Wang et al, the common clinical variables associated with a significantly increased risk of HF in the diabetic population included a history of CAD, poor glycemic control markers (use of insulin, high fasting glucose, glycated hemoglobin [HbA1c] > 9%), hypertension, and advancing age (5-year increase in age). 22 In addition, according to the 2016 European Society of Cardiology (ESC) guidelines, HF should be evaluated initially based on the presenting symptoms (e.g., orthopnea and paroxysmal nocturnal dyspnea), clinical history (myocardial infarction, radiation exposure, and use of diuretics), physical examination (e.g., bilateral edema, increased jugular venous pressure, displaced apical beat), and resting electrocardiography (ECG). 23 Hence, in routine clinical practice, it is recommended to screen for the above-mentioned independent risk factors along with physical examination, blood tests, and ECG in asymptomatic diabetic patients. 24
ECG is an initial diagnostic modality used in assessing the silent cardiac risk factors in DM. ECG is a quick, noninvasive screening modality that can be used in the early identification of adverse cardiac events in the diabetic population. Patients with T2DM have demonstrated a high prevalence of ECG abnormalities, reaching up to 73%, and the presence of ECG abnormalities predicts the occurrence of future CV events. 25 Studies have elucidated the relationship between clinical outcomes and ECG findings in HF patients and have shown that abnormalities in ECG are almost universally found in these patients, with normal ECG detected in only <2% to 7.5% of HF patients. 26 -28 Thus, ECG serves as a useful tool for predicting HF [29]. Even the ESC guidelines suggest the use of ECG for diagnosis of HF, in addition to clinical history and physical examination. 23
The clinical utilization based on PONTIAC and STOP-HF studies not only led to a better understanding of the pathophysiology of HF in diabetes patients but also encouraged ESC guidelines to incorporate N-terminal prohormone of brain natriuretic peptide (NT-proBNP) and BNP as a relevant marker to identify the risk of HF in diabetes patients, especially in outpatient settings. 23,29,30 In a study on the patient community attending their primary care physicians (PCPs) with symptoms suggestive of new-onset HF, adding BNP to the ECG model was found to increase the robustness in HF prediction (area under the curve [AUC] = 0.88) in the receiver operating characteristic curve analyses as compared to using ECG model alone (AUC = 0.81). 31 The presence of cardiac stress and volume overload leads to the production of NT-proBNP by cardiac ventricles to promote cardioprotection. 32 Evidence from various studies has reflected the role of NT-proBNP in predicting cardiac events, including the risk of HF. The prospective, observational study by Clodi et al compared the standard of care of albuminuria versus NT-proBNP in predicting cardiac events in the diabetic population. The results of their study revealed NT-proBNP as a better predictor of cardiac events (hazard ratio [HR]: 2.314; 95% confidence interval [CI]: 1.914-2.798; P < .001) in comparison to albuminuria (HR: 1.544; 95% CI: 1.007-2.368; P = .047). 32 A similar study was conducted by Jarolim et al to assess the prognostic implications of variations in concentrations of NT-proBNP in patients with T2DM and ischemic heart disease. The results of their study demonstrated baseline NT-proBNP to be associated with the development of major CV events including HF. Further, it was also observed that the risk of CV death or HF was significantly higher in patients with persistently high NT-proBNP (P < .001) or those who had increased NT-proBNP at 6 months (P < .001). There was a strong association between absolute changes in NT-proBNP at 6 months and subsequent outcomes in patients with DM. 33 The individual participant data meta-analysis published by the Natriuretic Peptides Studies Collaboration has also reported NT-proBNP concentration as a strong predictor of first-onset HF, and hence, it is recommended to include NT-proBNP assessment for identifying patients at risk of HF among the diabetic population. 34 The PONTIAC and the STOP-HF studies have particularly led to the incorporation of NT-proBNP in the standard clinical care pathway, which leads to the detection of HF in diabetic patients at risk of HF, that is, the American College of Cardiology/American Heart Association (ACC/AHA) stage A and B HF. 30,31 The ESC guidelines suggest that if ECG is completely normal, then HF is unlikely. Hence, its routine use is recommended to rule out HF. If there is an abnormality in ECG, BNP levels should be measured. If both ECG and BNP are normal, HF is highly unlikely. Echocardiography should be considered only when BNP levels are above the threshold or could not be assessed. 23 However, it should be noted that patients with a normal NT-ProBNP and abnormal ECG may have underlying atherosclerosis due to diabetes and metabolic syndrome, and this should be excluded particularly if there is no evidence of HF. 30,31
Albuminuria (including both micro- and macroalbuminuria) is another factor that is associated with left ventricular systolic and diastolic dysfunction in T2DM patients and is predictive of CV and HF events. Presence of albuminuria should trigger the suspicion of and confirmation of HF. Hence, albumin is an important marker for assessing HF risk in diabetes patients. 35 The data from the HOPE study indicated a 70% increase in CVD and HF in asymptomatic diabetic patients who had microalbuminuria only. The HOPE study demonstrated microalbuminuria in 32.6% of patients with DM. Further, microalbuminuria increased the relative risk of major CV events by 1.83-fold (95% CI: 1.64-2.05) and hospitalization for HF by 3.23-fold (95% CI: 2.54-4.10). 36 Additional indicators of target organ damage, such as chronic kidney disease (CKD) with reduced glomerular filtration rate (GFR) (eGFR < 60 mL/min/1.73 m2), also signify an increased likelihood of LV dysfunction in the form of diabetic cardiomyopathy and HF. Increased risk with lower GFR was also demonstrated in the original UK Prospective Diabetes Study and subsequent studies on T2DM management. With worsening GFR (30-59 mL/min/1.73 m2 to < 30 mL/min/1.73 m2), the risk of HF increases two- to threefold. 37 Further, the study by Nayor et al has reported incident HF in 17.2% of the study patients with CKD. Based on the Cox regression models, CKD increased the risk of HF by 52%. The study also reported microalbuminuria to be associated with the incidence of overall HF, with Cox models demonstrating a twofold higher risk of overall HF in patients with microalbuminuria. 38 Several clinical studies have demonstrated GFR of <60.0 mL/min/1.73 m2 to be a relatively common finding in patients with chronic heart failure (CHF). 39,40
As screening is usually conducted in asymptomatic patients, imaging plays a crucial role in the diagnosis of underlying comorbidities. Echocardiography is the gold standard in the noninvasive assessment of cardiac structure and function. The majority of the risk evidence documented is from echocardiography studies. 24 The earliest echocardiographic findings in diabetic cardiomyopathy is, in fact, a reduction in global longitudinal strain. 41 Indeed, this was the subject of many recently published articles, demonstrating that early treatment with sodium-glucose cotransporter-2 inhibitors (SGLT2i) can modify the global strain within 6 months of initiation of therapy. However, global longitudinal strain is not performed routinely in echocardiographic assessment. The ESC algorithm specifically mentions left atrial volume index and left ventricular mass and specific diastolic parameters, such as early mitral inflow velocity and mitral annular early diastolic velocity (E/e) ratio, performed on routine echocardiogram to demonstrate the earlier echocardiographic effects of diabetic cardiomyopathy (impaired relaxation and diastolic dysfunction). Later, as the disease progresses, left ventricular hypertrophy (LVH) is noted with an increasing range of diastolic dysfunction, and ultimately, the latter stages include progression to cardiomyopathy with reduced ejection fraction. The main finding of HF on an echocardiogram is LVH, and the underlying histopathology includes fibrosis. The objective of these evaluations is to provide early disease-modifying therapies before overt structural changes and progression to clinical HF and worsening of HF with reduced ejection fraction. Various clinical and epidemiological studies have demonstrated LVH in echocardiograms of diabetic patients, including those in the prediabetic stage. 22,24,42,43 The European guidelines recommend echocardiography to be included during the management of patients with T2DM even in the absence of any known CVD. 44 In the practical evaluation of echocardiography, a detailed assessment of diastolic function is mostly not performed; the routine echocardiography tests rely on the assessment of ejection fraction. Assessment of ejection fraction using echocardiography is not useful for the diagnosis of HF with preserved ejection fraction. 45 However, if echocardiography is normal, but BNP levels are elevated, it is diagnosed as HF with preserved ejection fraction. 23
Future directions in the diagnosis of HF
Many other biomarkers are still under development but are not clinically available or utilized. The biomarkers that are currently studied for evaluating cardiac risks in diabetic patients include transforming growth factor (TGF)-β1, procollagen type I propeptide, tumor necrosis factor-α, interleukin-6, isoprostane, leptin, adiponectin, bilirubin, insulin-like growth factor-binding protein 7, TGF-β, and inflammatory markers like polymeric immunoglobulin receptor (PIGR), receptor of advanced glycation end-products (RAGE), growth differentiation factor 15 (GDF-15), and WAP 4 disulfide core domain protein (WAP4C) among others. 46 -49
Management of HF in T2DM Patients
According to the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD), the main goal of diabetes treatment includes the prevention or delaying of disease-related complications, such as atherosclerotic CVD, CKD, and HF, and maintaining the patient’s quality of life through prevention of amputation, blindness, and dialysis, which are largely dependent on glycemic control. In order to achieve these goals, treatment should include glycemic control along with CV risk management. Further, regular follow-ups and a patient-centered treatment approach will improve patient engagement with self-care activities. 50
A number of HF management programs have been developed to combat the challenge of treating HF in outpatient care. According to the Canadian Cardiovascular Society consensus conference guidelines, there should a multidisciplinary outpatient HF clinic to accept referrals and a transition of care or to arrange the transfer of serious cases to tertiary hospitals for improved treatment. 51 Further, the HF clinics should educate patients about specific symptoms, managing risk factors, following home-based exercises, medication review, dose titration, monitoring treatment compliance, and regular follow-ups. 52 Even though treatment guidelines are supporting HF clinics, there are limited data pertaining to these clinics in the Middle East region. A dedicated HF multidisciplinary team (MDT) was established at Cleveland Clinic Abu Dhabi on January 1, 2017. The team comprised HF cardiologists, clinical pharmacists, and HF nurses. A retrospective study conducted by Abdalla et al to assess the impact of HF MDT reported a significant decrease in the 30-day readmission rate (P < .001), number of days for follow-up after discharge (P < .001), length of stay in hospital (P < .05), and the number of patients requiring inotropic support (P < .05). 53 However, the focus of the MDT should be on the upstream management, recognition, and prevention of HF to ensure that advanced HF clinics are not overburdened with a large number of patients. This necessitates screening and early diagnosis of HF in diabetes patients in primary care to ensure timely management.
MDT Approach for the Management of HF
Two important aspects need to be considered while managing HF: the management of HF in diabetes patients in HF/cardiology clinics and the management of undiagnosed diabetic cardiomyopathy patients. As part of the standard workup in patients with HF or CAD, the cardiology team should routinely check HbA1c and other parameters that suggest the presence of diabetes or impaired fasting glucose. Owing to data from the DAPA-HF and EMPagliflozin outcomE tRial in Patients With chrOnic heaRt Failure With Reduced Ejection Fraction (EMPEROR-Reduced) trials, irrespective of history of diabetes, patients with HF and reduced ejection fraction should be considered for the addition of SGLT2i, particularly dapagliflozin. 54,55 In general cardiology clinics, 19% to 26% of diabetes and 22% to 43% of prediabetes patients remain undiagnosed. 56 It is important to note that CV events and renal dysfunction occur during the undiagnosed phase of prediabetes and years before the diagnosis of T2DM. Thus, early detection and risk factor modification are crucial in preventing further complications. 57 Assessment of HbA1c, impaired glucose tolerance, and homeostatic model assessment for insulin resistance (HOMA-IR) is crucial.
In diabetic patients, HF with preserved ejection fraction (EF) is commoner in the early stages, and early treatment with SGLT2i prevents the progression to overt CVD. Results from various cardiovascular outcome trials (CVOTs) have reported less than 10% of patients to have HF with reduced ejection fraction (HFrEF) with EF <40% and clinical CHF. Nevertheless, there was a significant benefit of SGLT2i in both types of patients with HFrEF and HF with preserved EF (HFpEF), in terms of reduced disease progression and HF-related hospitalization. 58 -61
Several international guidelines, including the ESC, EASD, and ADA, are available for the management of diabetes with CVD. Further, the ACC guidelines have also laid out expert consensus decision pathways based on expert opinions to guide clinicians in the management of diabetes with CVD. 37 The 2021 ESC guidelines specifically mention dapagliflozin and empagliflozin in reducing CV-related mortality in patients with HF. 23,62 The compelling evidence in favor of the benefits of SGLT2i in the management of HF even in patients without diabetes has triggered a dramatic change in the therapeutic armamentarium. The 2021 ADA guidelines (Figure 2) strongly recommend the use of SGLT2i in diabetes patients based on the presence of risk factors, independent of the baseline HbA1c levels, individualized glycemic targets, or metformin use. Thus, there is a clear deviation from the traditional glucose-centric approach to a risk factor-centric approach in the management of diabetes. 63 These risk factors include established atherosclerotic CVD or indicators of high risk (age ≥55 years with coronary carotid, or lower extremity artery stenosis >50%, or left ventricular hypertrophy), HF, or CKD. 63

The ADA guidelines on the management of T2DM. 63
A study demonstrated that approximately 60% of the general T2DM population should be treated with SGLT2i. 64 Despite such evidence and the fact that recent guidelines strongly recommend SGLT2i in T2DM patients with CVD or at high risk of CVD, the uptake of this cardio protective drug remains very low. 65 Various studies have shown that the uptake of SGLT2i in patients with T2DM and CVD ranges between 1.4% and 15%. 66 -70 As the primary benefit of SGLT2i is CV risk management, there is an urgent need for change in the therapeutic armamentarium of CVD and HF. 65
Practical Approach for the Implementation of SGLT2i Therapy in HF Management
Practical Guidance of SGLT2i Use
According to the 2008 Food and Drug Administration (FDA) antihyperglycemic guidelines, a need for a new drug with no adverse cardiac events was suggested. Based on this guideline, SGLT2i, such as canagliflozin (Invokana®), dapagliflozin (Farxiga®), empagliflozin (Jardiance), and ertugliflozin (Steglatro™), have been approved by the FDA. 71,72
The SGLT2i exert its antidiabetic action by blocking renal tubular glucose reabsorption. Further, the results of the landmark CVOTs have reported SGLT2i to decrease HF events by 27% to 39% in high-risk patients with T2DM. 73,74 Even though studies have reported favorable outcomes in terms of HF improvement with SGLT2i, their prescription is limited especially by the cardiologist colleagues. 75 Based on the results of clinical trials and updated guidelines, in order to reduce HF-related hospitalization and mortality, cardiologists and those managing diabetes patients should consider prescribing SGLT2i to all patients with diabetes unless contraindicated. In patients who have HFrEF with or without diabetes, the addition of SGLT2i to beta-blocker and angiotensin-converting enzyme inhibitor, angiotensin receptor blocker, and angiotensin receptor-neprilysin inhibitor (ACEI/ARB/ARNI) is strongly recommended to reduce HF hospitalization and mortality (Figure 3). 76

Guideline-based treatment algorithm for patients with HFrEF. 76
According to the current labeling of SGLT2i (empagliflozin and dapagliflozin), the initiation of therapy is not recommended when eGFR is <30 mL/min//1.73 m 2 . However, the meta-analysis of major CVOTs has demonstrated higher HF reductions at lower eGFR levels. The ongoing trials of SGLT2i in HF enrolling patients with eGFR of 20 mL/min/1.73 m2 will shed further light on the GFR cutoff points to be considered in HF patients. 73,75
Clinical Precautions Related to SGLT2i
Several adverse effects of SGLT2i have been reported in different clinical trials, including increased risk of amputation, ketoacidosis, volume depletion, genitourinary infection, and hypoglycemia. However, these adverse effects are mostly mild and can be avoided with some precautions or treated with over-the-counter medications. 77 Real-world clinical evidence with SGLT2i has reported genitourinary tract infection and ketosis to be the common side effects of this class of medications, but none were documented as serious adverse events. 78
Evidence on HF From Recent CVOTs
Evidence from the recent CVOTs, including the EMPA-REG OUTCOME study, Canagliflozin and Renal Events in Diabetes with Established Nephropathy Clinical Evaluation (CREDENCE study), Canagliflozin Cardiovascular Assessment Study (CANVAS trial), DECLARE TIMI58, Dapagliflozin And Prevention of Adverse outcomes in Heart Failure trial (DAPA-HF), EMPEROR-Reduced, EMPagliflozin outcomE tRial in Patients With chrOnic heaRt Failure With Preserved Ejection Fraction (EMPEROR-Preserved), Effect of Sotagliflozin on Cardiovascular Events in Patients With Type 2 Diabetes Post Worsening Heart Failure (SOLOIST-WHF), and Effect of Sotagliflozin on Cardiovascular and Renal Events in Patients With Type 2 Diabetes and Moderate Renal Impairment Who Are at Cardiovascular Risk (SCORED), has demonstrated the CV benefits of antidiabetic agents like SGLT2i in terms of a significant decrease in the major adverse cardiac event primary composite endpoint, reduced CV death, as well as all-cause mortality. 55,61,79 -85 All studies have elucidated the cardioprotective effects of SGLT2i in T2DM patients with established CVD. 86 The largest and most consistent benefit observed across this class of medication was a reduction in the risk of HHF and renal outcomes. 68 Besides, significant benefits of SGLT2i have also been observed in reducing CV mortality and all-cause mortality in T2DM patients [86]. These benefits were also observed in patients with HFrEF and HFpEF, with or without diabetes. 61,87 Based on these CV benefits of SGLT2i, it has been suggested that these medications should be considered as first-line therapy. 86 However, the Vertis-CV trial findings showed ertugliflozin to be a non-inferior placebo in terms of reducing CV events in patients with T2DM and established CVD. 88
While evidence-based management of HFpEF has been relatively limited due to negative RCTs until recently, a major breakthrough in the management of HFpEF with SGLT2i (where diabetic–metabolic inflammatory cardiomyopathy is a major contributor to the prevalence and epidemiology in the region) are the EMPEROR-PRESERVED and DELIVER (based on preliminary results; full results yet to be published) studies which have shown robust outcomes in HFPEF, in both patients with diabetes and without diabetes, evidence that will undoubtedly impact the treatment paradigm. 61,89
Identifying the Key Unmet Need in the Management of Patients With Diabetes and HF: Under Usage of SGLT2i
The CAPTURE study estimating the prevalence of CVD in T2DM patients across 13 countries from 5 continents reported that one-third of patients with T2DM have established CVD. Only 21.9% of the overall T2DM patients were receiving treatment with a glucose-lowering agent with proven CV benefit, and the usage of SGLT2i remained consistently low in patients both with and without CVD. These data suggested that the presence of CVD in T2DM patients did not steer any treatment modification. 90 In this context, the DISCOVER study revealed the over usage of non-HF/CVD risk-modifying therapies like metformin and sulfonylureas in T2DM patients, whereas the use of SGLT2i was very low. 70 These statistics indicate that till date the treatment decisions are largely based on HbA1c levels rather than the risk factors defined by the ADA guidelines. 63
Recommendations and Call to Action
Diagnosis and management of diabetic patients with HF are challenging in the Middle East compared to their Western counterparts due to various reasons. Most recent studies show that cardiac MRI (which is a major diagnostic tool in HF) is still much underused in the Middle East compared to the western world. This could be largely due to the unavailability of the service or scarcity of qualified personnel to operate and interpret it. There is also a marked underuse of natriuretic peptides either for diagnosing or for the follow-up of HF patients. This underuse could be due to the inaccessibility of the kits, the expensiveness of the test, and the lack of knowledge regarding its role in HF diagnosis and management. 91 Due to a lack of law as well as societal and potentially religious concerns around heart transplantation, there is a severe paucity of heart transplantation in the Arab world. 91 Another area that needs development is the establishment of specialized heart failure programs with more reliance on trained nurses and public awareness (which is lacking in the Middle East) to cut the circle of non-compliance and repeated admissions to hospitals. Furthermore, ethnicity is a variable which can predict the prognosis or the progression of the illness. It has been demonstrated that Middle Eastern countries warrant ethnic-specific obesity cut-off points which is a major risk factor for DM as well as HF. 92 Due to the cultural differences between Middle Eastern and Western nations, research has shown that diabetes self-management education (DSME) is less effective when it is culturally adapted. This justifies GCC–specific interventions for the control of T2DM self-management, which would likely increase the effectiveness of the therapies. Moreover, due to the diverse etiology and clinical presentation of HF, it is necessary to set up special disease management programs for diabetic patients reporting a suspected case of HF. Along these lines, we propose a screening and referral approach for diabetic patients with suspected HF, as outlined in Figure 4.

Algorithm for the diagnosis of heart failure. 62
The simple steps for screening HF in the diabetes population in the UAE are presented in Box 1. Although the guidelines provide a detailed algorithm for the diagnosis of HF, those complex steps are not adhered to in routine clinical practice. Therefore, a simple list of mandatory questions should be asked to every single diabetic patient during every single visit to help the early diagnosis of HF in patients with diabetes (Box 2). Further, in order to improve the management of HF in diabetic patients in the UAE, the expert panel has provided a few approaches, which have been described in Box 3.
The summary of expert recommendations for the management of HF in patients with diabetes is mentioned below: Based on the ADA recommendations and emerging data, approximately 60% to 70% of the T2DM population should receive SGLT2i after metformin therapy. The rest of the patients should be investigated for eligibility to receive SGLT2i. The uptake of SGLT2i should be increased accordingly. However, at present, the uptake is around 15% only. Assessment of patient history based on the criteria defined above should be mandatory. All T2DM patients fulfilling the ADA criteria should be put on SGLT2i, and those fulfilling the patient history criteria in HF screening should be investigated further. Patients with T2DM and HF should receive SGLT2i, irrespective of baseline HbA1c value. There is an unmet need for early HF screening in T2DM patients in the UAE. To fulfill this lacuna, education on early screening and appropriate treatment of HF in diabetes patients should be disseminated among the PCPs.
Screening criteria for HF in the diabetes population in the UAE.
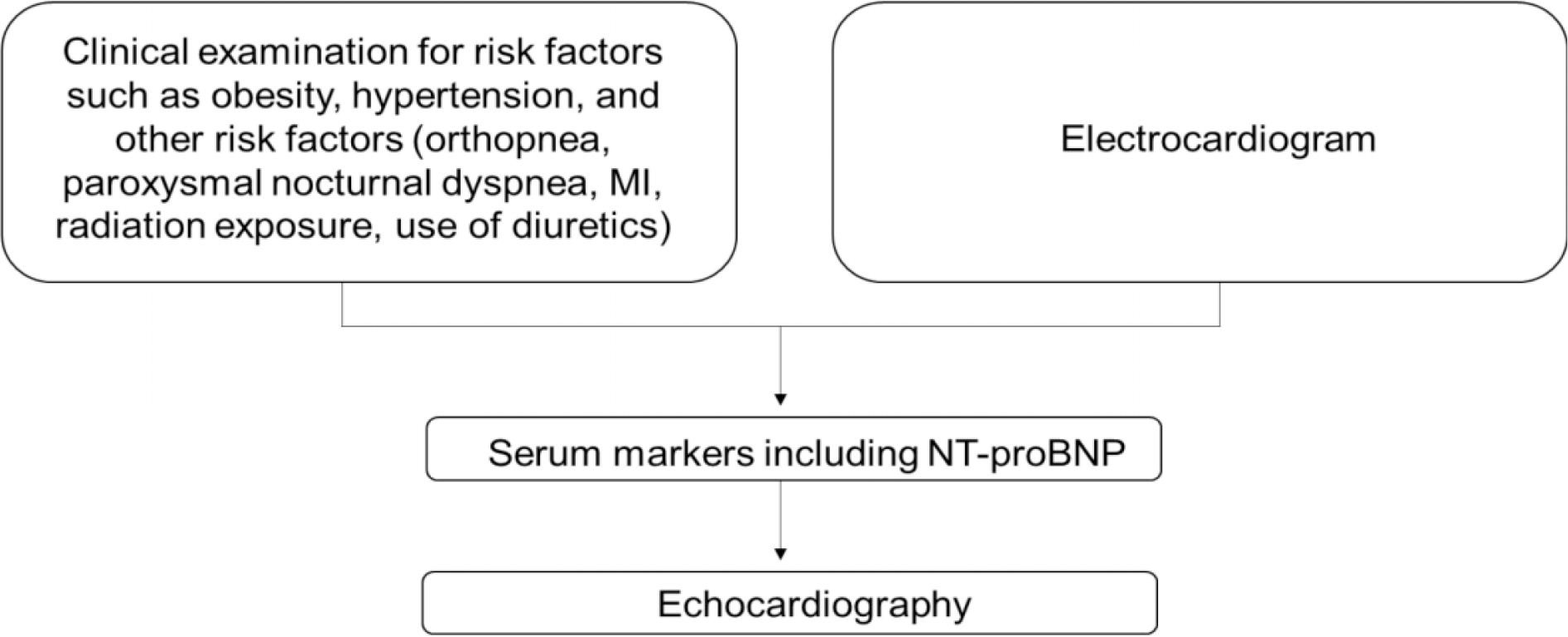
ECG, electrocardiography; MI, myocardial infarction; NT-proBNP, N-terminal prohormone of BNP.
Mandatory questions to be asked to diabetic patients during every visit.
Exertional fatigue Impaired capacity to exercise Ability to go up one flight of stairs Worsening shortness of breath with activity Increased swelling of legs, feet, and ankles Trouble sleeping with nocturnal dyspnea or paroxysmal nocturnal dyspnea Acute symptoms like chest pain and palpitation
Approaches for managing HF in T2DM patients.
Creating primary care structured preventive programs Educational programs for diabetologists and general physicians on HF diagnosis and referral Coordination between diabetologists and cardiologists on mutual diagnosis, agreement, and acceptance of the role of guideline-directed medical therapy, more specifically on the use of RAS blockade and SGLT2i in HF patients Standardization of clinical practice guidelines that apply to the disease itself regardless of specialty Need for developing local guidelines for the management of HF in diabetic patients
RAS, Renin–angiotensin system; SGLT2i, Sodium–glucose cotransporter-2 inhibitors.
The recent recommendations from international guidelines and evidence from different CVOTs demand a “call to action” in the diagnosis and treatment of diabetic patients with HF in the UAE. The UAE model of care includes the majority of patients being initially treated by a nonspecialized PCP. Given the rapid change in the evidence-based guidelines and the need for incorporating new diagnostic pathways, regular educational programs must be conducted for PCPs regarding risk factors, symptoms, dose titrations, referral, monitoring therapeutic compliance, and regular follow-ups. Recognition of the early stages of diabetic cardiomyopathy is not a part of the normal workflow in internal medicine, primary care, and diabetology. Thus, the early recognition of this entity becomes important so that appropriate preventive interventions can be implemented in a broader perspective. A simple screening model (Figure 4) that includes routinely assessed clinical parameters and laboratory investigations will aid the practitioners in identifying and referring the patients to advanced HF centers during regular clinical visits. Further, as CVOTs have demonstrated the benefits of dapagliflozin, registries and other outcome trials that can show the benefits of implementation of preventive therapies, including SGLT2i in high-risk Middle East populations, would be beneficial. This would be a recommendation for CV and diabetes societies and healthcare systems in the UAE. All societies in the UAE related to the fields of endocrinology, diabetology, and cardiology and policymakers/stakeholders should convene twice or thrice every year to discuss and develop guidelines or steps forward for the effective management of T2DM patients with HF or those at risk of developing HF. Additional research in the field of HF screening in diabetic patients is warranted to estimate the true prevalence of HF in the UAE. Further, there is also a need to assess the correlation of NT-proBNP, ECG, and echocardiography in the high-risk diabetes population and its impact on detection and management.
Conclusion
The incidence of HF in diabetes patients is largely undiagnosed. Even in newly diagnosed diabetes patients, the incidence of HF is high. Again, nearly one-third of HF patients have undiagnosed diabetes. Further, mortality is higher in HF patients with diabetes than in nondiabetic HF patients.
However, HF remains an underrated CV event in diabetes patients.
Early diagnosis of HF in diabetes patients is the need of the hour.
Guidelines provide stepwise algorithms for the diagnosis of HF, though they are not adhered to in routine clinical practice, which emphasizes the need for simple diagnosis and treatment algorithms.
Accumulating evidence has established the CV benefits of SGLT2i, including consistent benefits in lowering HHF and HF-related mortality, even in diabetes-independent conditions. This has led to the incorporation of SGLT2i in the HF treatment algorithm by guidelines. However, their use remains suboptimal.
As per the ADA criteria, up to 70% of the T2DM patients are eligible to receive SGLT2i, but till date, the management decisions for T2DM are glucose-centric and not risk factor-centric, which highlights the need for the proper education of the PCPs.
Irrespective of HbA1c levels, all patients with T2DM and HF should be put on SGLT2i.
This “call to action” addresses both the diagnostic and therapeutic gaps of HF in diabetes patients.
Footnotes
Acknowledgments
We thank BioQuest Solutions Pvt. Ltd. for providing editorial support. We also thank AstraZeneca GCC for funding the expert panel meeting, which helped to identify the gaps in the management of heart failure in patients with diabetes mellitus in the UAE and propose steps to address these gaps. The panel included eminent healthcare specialists from the UAE, who came together to identify and address the gaps in heart failure management and shape practices that can support the Ministries of Health and patient communities in the region.
Author Contributions
All authors have contributed equally to the concept or design of the work; the acquisition, analysis, or interpretation of data; the creation of new software used in the work; and the drafting of the work or critical revision of important intellectual content. They also approved the version to be published and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AstraZeneca GCC also funded the editorial support provided by BioQuest Solutions Pvt. Ltd.
