Abstract
Background
Potassium-binders patiromer and sodium zirconium cyclosilicate (SZC) are approved to treat hyperkalaemia, which is frequently observed in chronic kidney disease (CKD). Elevated blood pressure (BP) is common in CKD, due in part to impaired sodium excretion. The effect of patiromer, which exchanges calcium for potassium and SZC, which exchanges sodium or hydrogen for potassium, on BP was assessed in a CKD rat model.
Methods
Thirty-six Sprague Dawley rats with 5/6 nephrectomy were randomised to three groups (n = 12/group) to receive 4 g/kg/day patiromer or SZC, or vehicle treatment, for 8 weeks. BP was determined by radiotelemetry and urinary protein and electrolytes were measured.
Results
At Week 8, systolic BP (sBP) increased in all groups; however, patiromer led to a lower mean (standard deviation) sBP than vehicle or SZC (141 [2.9] vs 158 [5.2] or 162 [6.1] mm Hg, respectively, both p < 0.001), with no difference in sBP between vehicle and SZC (p = 0.08). Similar results were observed for diastolic BP. Serum potassium levels fell with SZC (p < 0.02), but not vehicle or patiromer. Urine potassium decreased with both patiromer and SZC versus vehicle (p < 0.01); urine sodium increased with SZC (p < 0.01); and urine calcium increased with patiromer (p < 0.01). Urine phosphorus decreased with patiromer (p < 0.01) but increased with SZC (p < 0.01). Patiromer resulted in less proteinuria than vehicle or SZC (both p < 0.017).
Conclusions
After 8 weeks, treatment with patiromer resulted in lower BP in rats than vehicle or SZC. Further studies are needed to determine the mechanism of the differential effect of potassium binders on rat BP.
Introduction
Hyperkalaemia (HK), defined as a serum potassium level >5.0 mEq/L, is frequently observed in patients with chronic kidney disease (CKD) and may be exacerbated in response to therapy with renin-angiotensin aldosterone system inhibitors (RAASi). The frequency and severity of HK increases as kidney function declines. 1 Chronic HK can lead to damaging effects on the kidney and cardiovascular system due, in part, to increased secretion of aldosterone, vascular inflammation, renal and myocardial fibrosis, and ventricular hypertrophy. 2 Increased hospitalisation and adverse clinical outcomes are reported in patients with HK. 3 Two potassium binders for the treatment of HK have been developed and approved, both of which increase faecal excretion of potassium.4,5 Sodium zirconium cyclosilicate (SZC) exchanges sodium or hydrogen for potassium,5,6 while patiromer exchanges calcium for potassium. 4
Elevated blood pressure (BP) is frequently reported in patients with CKD, likely as a consequence, in part, to impaired sodium excretion. 7 Reducing sodium intake in pre-dialysis CKD patients has been shown to reduce hypertension and enhance the anti-proteinuric action of RAASi. 8 Although the impact of a low-sodium diet is less clear in end-stage kidney disease patients undergoing dialysis, excess sodium is a direct cause of fluid overload and hypertension in these patients, resulting in left ventricular hypertrophy and increased mortality.7,9 In several clinical studies in which patiromer was used to treat HK in patients with CKD, decreases in BP were observed during treatment.10,11
To study the potential for a differential effect of patiromer with SZC on BP, we utilised a CKD rat model that develops elevated BP. The hypertensive 5/6 nephrectomy (5/6 Nx) model in rats models the progressive renal failure seen after the loss of renal mass in humans and is a widely used model to study CKD.12,13
Methods
Materials and Methods
The study was conducted at Charles River Laboratories (Ashland, OH) in accordance with the Guide for the Care and Use of Laboratory Animals from the National Research Council, 14 with all investigations adhering to the ARRIVE2 Guidelines. 15 The study protocol was reviewed and approved by the Testing Facility Institutional Animal Care and Use Committee (IACUC) before the initiation of the study.
Animals
Thirty-six male Sprague Dawley (SDW) rats (Charles River Laboratories, Raleigh, NC) ≥10 weeks old, weighing 275–400 g were used in this study. All procedures were conducted in conformity with the US Ethical Guidelines for the Use of Animals in Research and approved by the Testing Facility IACUC (Charles River Laboratories, Ashland, OH). Animals received the Envigo rodent diet (Teklad Diet 04498, Envigo, Indianapolis, IN), which included 0.5% Na, 0.9% K, 0.2% Ca, and 17.7% protein. Animals underwent a surgical removal of 2/3 of the left kidney, and after 1 week of recovery, each animal had the right kidney removed 16 and HD-S10 radiotelemetry transmitter implanted using the abdominal aorta approach. 17 The pressure sensor catheter was inserted into the abdominal aorta and advanced cranially. The body of the transmitter was then slipped into the abdominal cavity.
Study Design
After 2 weeks of recovery, and after a baseline measurement, animals were randomised into three groups (n = 12) based on baseline serum creatinine and systolic blood pressure data. Patiromer, SZC, or vehicle (0.3% xanthan gum) was administered daily via oral gavage for 8 weeks. In the US, SZC and patiromer are usually prescribed at a maintenance dose of 10 g/day and 8.4 g/day, respectively. In this study, the same dose of 4 g/kg/day was administered for both patiromer and SZC. Patiromer and SZC were administered with 0.4 g/mL dosing suspensions (in 0.3% xanthan gum) at 10 mL/kg dosing volume, based on the most recent body weight measurement. The study groups and treatments were not blinded, however, the automated radiotelemetry recording of blood pressure provided unbiased data analyses.
Blood Pressure Measurements by Radiotelemetry
The radiotelemetry system (Data Sciences International, St Paul, MN) consisted of small animal radiotelemetry transmitters (HD-S10) with arterial pressure and body temperature receivers (RMC-1). Arterial pressure waveforms were sampled at 500 Hz. Temperature data were sampled at 50 Hz. Data were acquired continuously and logged every 120 s. Telemetry data were collected continuously for at least 24 h from baseline (pre-test period) and once weekly from Weeks 1 to 8. BP parameters were averaged to 2-h time intervals and reported as the mean of the 24-h period for descriptive statistical analysis.
Blood and Urine Sample Collection
At baseline and Weeks 1, 4 and 8 of treatment, blood samples were collected via venipuncture from the jugular vein for measurement of serum potassium and creatinine levels using a clinical chemistry analyser (Advia 1800, Siemens, Malvern, PA). At Weeks 1, 4 and 8, animals were housed overnight in individual metabolic cages for a 24-h urine collection. Total urine volume was recorded and samples were analysed for protein, potassium, sodium, calcium and phosphorus. Urine electrolytes were analysed using a urine chemistry analyser (NOVUS, Siemens, Malvern, PA). Urine protein was measured semi-quantitatively by dipsticks and was graded as negative (designated as 0 for statistical analysis), 1+ (30 mg/dL), 2+ (100 mg/dL), 3+ (300 mg/dL) or 4+ (1000 mg/dL).
Statistical Analysis
Total urine volume and protein, serum potassium, and BP (sBP and dBP) were analysed for statistical significance using one-way analysis of variance (ANOVA) when comparing data among groups. If analysis by ANOVA was significant, each set of two groups were analysed with the student's t-test (two-sample, assuming unequal variances). The significance was corrected by the Bonferroni method (a p-value <0.017 [0.05 divided by 3 comparisons] was considered as a statistically significant difference). The student's t-test (two-sample, assuming unequal variances) was applied for data analysis against the baseline. All values are mean ± standard deviation unless otherwise stated.
Additional statistical analyses for the telemetry data were conducted using SAS Software (SAS Institute Inc, Cary, NC). Raw values and the change from baseline data corresponding to each parameter of interest were statistically analysed using a repeated measures analysis of variance model, with group, time, and group × time interaction as fixed factors, and animal as the subject repeated across time. The corrected Akaike's information criterion was used in order to select the model with one of the four covariance structures: compound symmetry, heterogeneous compound symmetry, first-order autoregressive, and heterogeneous first-order autoregressive.
Results
Baseline Characteristics
All 36 rats across the three groups completed the entire study. The baseline characteristics including blood pressure and serum creatinine of the 12 rats in each group showed no significant differences between groups (Supplemental Table 1). There was no difference in body weight between treatment groups throughout the study (Supplemental Table 2).
Blood Pressure
Significant interaction effects (group × time, p < 0.001) were observed for both sBP and dBP, showing significant BP differences between groups over time in the study (Supplemental Figure 1). Systolic blood pressure increased significantly from baseline to Week 8 in all rats (vehicle, patiromer, and SZC treated) following 5/6 Nx (p < 0.001) (Figure 1a). At Week 8, patiromer-treated rats had significantly lower sBP compared with rats in the vehicle- and SZC-treated groups (p < 0.001), whereas there was no significant difference between the sBP observed between vehicle- and SZC-treated rats (p = 0.08). The increase in sBP observed for patiromer-treated rats (9.0 ± 2.9 mm Hg) was significantly less than that observed for vehicle- and SZC-treated rats (21.8 ± 2.2 mm Hg and 27.0 ± 3.8 mm Hg, respectively; both p < 0.001), while mean sBP change from baseline in SZC-treated rats was significantly higher versus the vehicle group (p < 0.001) (Supplemental Figure 1a). Similar results were observed with diastolic BP (dBP) (Supplemental Figure 1b). At Week 8, patiromer-treated rats had significantly lower dBP as well as significantly less dBP increase from baseline compared with rats in the vehicle- and SZC-treated groups (p < 0.001).

No statistically significant difference was observed for heart rates between treatment groups, except for a higher rate at Week 6 for patiromer compared with placebo (Table 1).
LS Mean (SE) Heart Rate (Beat/minute) in 5/6 Nx Rats Treated with Vehicle, Patiromer, or SZC.

Data are presented as LS mean (SE). (n = 12/group).
Abbreviations: LS, least square; SE, standard error; SZC, sodium zirconium cyclosilicate; W, week. *p < 0.05 patiromer versus vehicle (Statistical test: LS mean analysis in SAS).
Serum Chemistry
Serum potassium levels from baseline to Week 8 did not change in vehicle-treated rats (5.6 ± 0.26 mEq/L to 5.5 ± 0.21 mEq/L) or in patiromer-treated rats (5.6 ± 0.42 mEq/L to 5.5 ± 0.29 mEq/L) and fell in SZC-treated rats (5.3 ± 0.36 mEq/L to 4.9 ± 0.33 mEq/L (p = 0.02). Normokalaemia in rats has been reported as 5.1–6.6 mEq/L. 18 There was no difference in serum creatinine levels among the three treatment groups during the study (p > 0.05) (data not shown).
Urine Electrolyte Excretion and Urinary Protein
Compared with vehicle treatment, 24-hour urine potassium excretion was lower with patiromer (p < 0.01 patiromer vs vehicle at Week 8) and SZC (p < 0.01 SZC vs vehicle at Weeks 1, 4, and 8) (Table 2). Urine sodium was higher following SZC (p < 0.01 vs vehicle at Weeks 1, 4, and 8), and urine calcium was higher with patiromer (p < 0.01 vs vehicle at Weeks 1, 4, and 8). Patiromer-treated rats had lower urine phosphorus levels during the study (p < 0.01 vs vehicle and SZC groups at Weeks 1, 4, and 8), whereas SZC treatment resulted in higher urine phosphorus (p < 0.01 vs vehicle and patiromer groups at Week 8).
24-h Urine Potassium, Sodium, Calcium, and Phosphorus Excretion in 5/6 Nx Rats Treated with Vehicle, Patiromer, or SZC (n = 12/group).

Data are presented as mean ± SD. (n = 12/group).
Abbreviations: SD, standard deviation; SZC, sodium zirconium cyclosilicate. ap < 0.01 patiromer versus vehicle.
p < 0.01 SZC versus vehicle.
p < 0.01 patiromer versus SZC (Statistical test: Student t-test and adjustment by the Bonferroni method).
Patiromer-treated rats had lower levels of proteinuria versus vehicle- and SZC-treated rats when measured semi-quantitatively by the dipstick method (p < 0.017 vs vehicle group at Weeks 1 and 8, and p < 0.017 vs SZC group at Weeks 1, 4, and 8) (Figure 2). There were no significant differences in urinary volume among treatment groups at Weeks 1, 4, and 8 (data not shown).
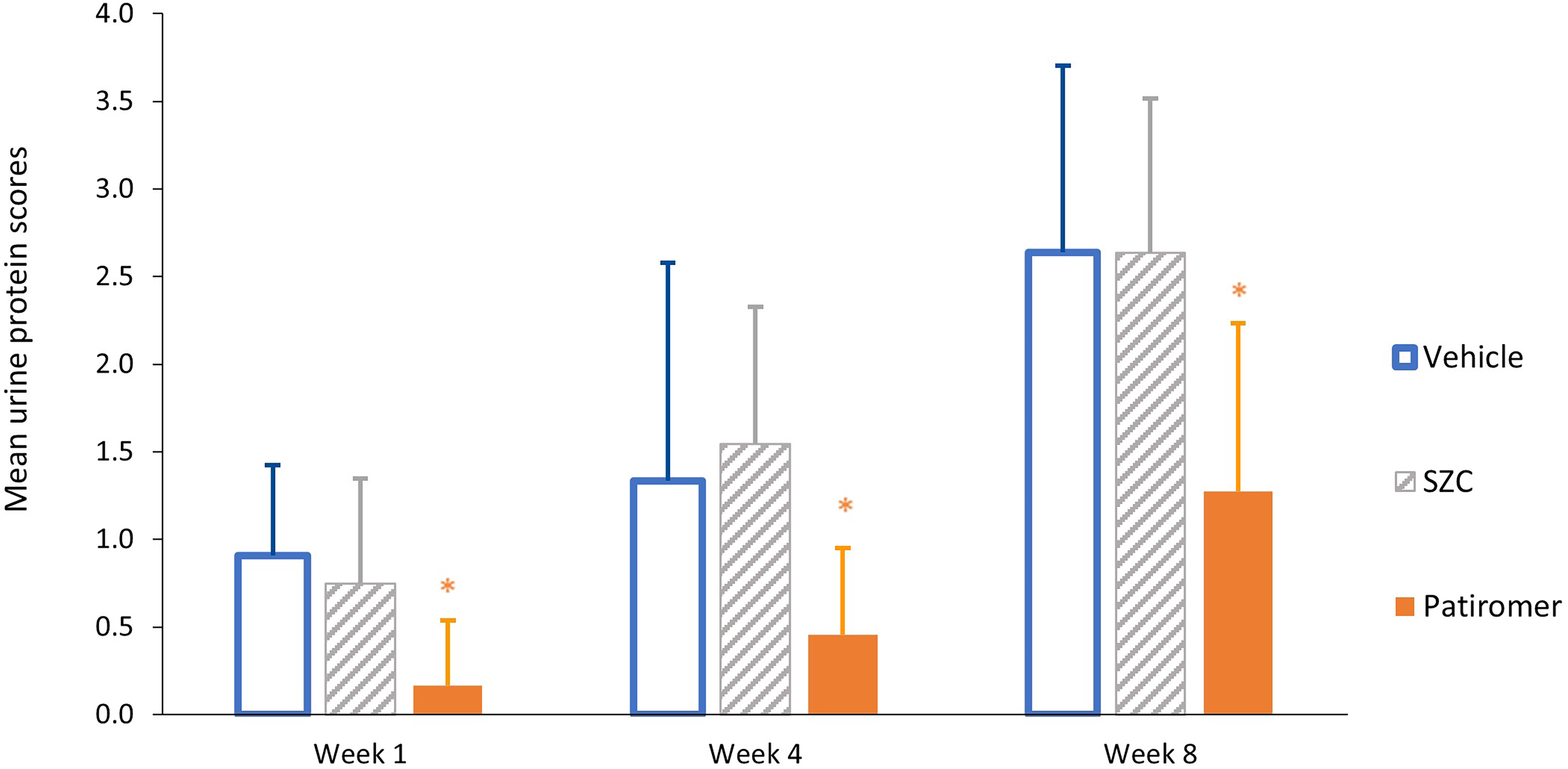
Proteinuria in rats treated with vehicle, patiromer, and SZC (n = 12/group).
Discussion
Patiromer and SZC are both approved potassium binders for the treatment of HK. In contrast with SZC, which uses sodium and hydrogen to exchange with potassium, patiromer uses calcium as the counter-exchange ion.4,5
In this study, we used the surgical 5/6 Nx as a model of CKD to study the effect of patiromer and SZC on BP. This experimental model is characterised by the development of hypertension and renal damage (both histologically and by increased protein excretion), which can be exacerbated with a high-salt diet.19,20 Results obtained with the 5/6 Nx model have also demonstrated clinical relevance.21,22
The method of nephrectomy has also been shown to be a critical modulator of hypertension and glomerular injury in rat models. 23 Mass reduction by infarction resulted in significant increases in BP, proteinuria, and glomerulosclerosis, whereas renal mass reduction by surgical excision resulted in an increased salt sensitivity of BP compared with those caused by infarction. 23 Additionally, within hypertensive rat models, significant differences in BP observations have been reported depending on the method of BP monitoring used.24,25 Use of overall mean BP values obtained from groups of rats via continuously monitored radiotelemetry is a useful method to overcome individual and diurnal variations in BP otherwise observed through tail cuff BP measurements.20,24
We found that SDW rats with 5/6 Nx treated with patiromer had a significantly smaller increase in sBP compared with those treated with vehicle or SZC after 8 weeks of treatment. The mean sBP increase from baseline in patiromer-treated rats was 9 mm Hg at Week 8, whereas mean sBP increased by 22 mm Hg and 27 mm Hg in vehicle and SZC groups, respectively. Similar results were observed for dBP, with a significantly smaller increase in dBP observed with patiromer than for vehicle or SZC. The higher heart rate observed with patiromer at Week 6 compared with vehicle was considered to be patiomer-related based on the concurrent reduced sBP (compared with vehicle and SZC) during this period.
Although the study did not investigate the mechanisms by which patiromer blunts the increase in BP, there are at least two potential explanations for this effect. First, non-specific binding of sodium to patiromer may occur in the gastrointestinal (GI) tract and lead to decreased sodium absorption. Clinical studies have shown that patiromer treatment reduced urinary sodium excretion in healthy volunteers. 26 Enhanced faecal Na+ excretion with patiromer treatment may help prevent the rise in BP in this model of CKD. In this study, significantly increased urine sodium compared with vehicle was observed at Weeks 1, 4, and 8 with SZC administration in SDW rats. The increase in urine sodium with SZC suggests that some of the sodium released when SZC binds to potassium is absorbed, potentially leading to an increase in BP. Second, the smaller increase in BP with patiromer could be related to a patiromer-induced reduction of aldosterone levels. In a rat HK model, patiromer reduced aldosterone levels in a dose-dependent manner. 27 Reduction of aldosterone following treatment with patiromer has also been observed in clinical studies. 11 Further studies are needed to determine the mechanism underlying the differential effects of patiromer and SZC on BP.
Increased urine calcium was observed with patiromer treatment, suggesting intestinal absorption of at least some of the calcium released when patiromer binds potassium. 26 The calcium released from patiromer may also bind to phosphate in the GI tract, which is supported by the decreased urinary phosphorus excretion observed in patiromer-treated healthy adults. 26 Findings from clinical studies have also suggested that calcium released in the GI tract binds phosphate, leading to the formation of insoluble salts that are excreted in the stool. 26 The binding of intestinal phosphate by the calcium released when patiromer binds potassium may help mitigate hyperphosphatemia, which is observed in many patients with CKD. 28
Although urine protein level was measured only semi-quantitatively by the dipstick method, results showed that patiromer-treated rats had significantly lower levels of proteinuria versus vehicle- and SZC-treated rats. Proteinuria is a marker for CKD progression. BP control in patients with CKD is essential to avoid progression of renal deterioration. 29
Study Limitations
The study was limited by the cohort size and use of parametric test considering number of observations. Both the patiromer and SZC doses used in the study were at least 8-fold higher than human doses. The dose of patiromer used in this study (4 g/kg/day) was within the range of doses (1.5-4.5 g/kg/day) that have previously been shown to be effective in reducing serum potassium in a rat HK model. 27 The extent of urinary potassium, sodium, calcium, and phosphorus changes observed are likely to be influenced by the higher dosing; however, the changes in BP observed with patiromer in this study are consistent with those observed in clinical trials.
Conclusion
Patiromer-treated rats exhibited significantly lower BP compared with vehicle- and SZC-treated SDW rats with 5/6 Nx after 8 weeks of treatment. Additional studies are needed to determine the mechanisms underlying the differential effect of these two potassium binders on BP.
Supplemental Material
sj-docx-1-cpt-10.1177_10742484241227580 - Supplemental material for Effects of Patiromer and Sodium Zirconium Cyclosilicate on Blood Pressure in Rats with Chronic Kidney Disease
Supplemental material, sj-docx-1-cpt-10.1177_10742484241227580 for Effects of Patiromer and Sodium Zirconium Cyclosilicate on Blood Pressure in Rats with Chronic Kidney Disease by Lingyun Li, MD, PhD, Jeff Budden, BSc, Carol Moreno Quinn, PhD, and David Bushinsky, MD in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
sj-docx-2-cpt-10.1177_10742484241227580 - Supplemental material for Effects of Patiromer and Sodium Zirconium Cyclosilicate on Blood Pressure in Rats with Chronic Kidney Disease
Supplemental material, sj-docx-2-cpt-10.1177_10742484241227580 for Effects of Patiromer and Sodium Zirconium Cyclosilicate on Blood Pressure in Rats with Chronic Kidney Disease by Lingyun Li, MD, PhD, Jeff Budden, BSc, Carol Moreno Quinn, PhD, and David Bushinsky, MD in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
All authors participated in the conception, design, and implementation of the study. All authors were involved in the interpretation of analysed data and the decision to submit for publication. Medical writing support was provided by Nicola Illingworth, AXON Communications, United Kingdom and funded by Vifor Fresenius Medical Care Renal Pharma Ld.
Data Availability
Data available on reasonable request.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Jeff Budden is an employee and shareholder of CSL Vifor. David Bushinsky received grants from the National Institutes of Health and the Renal Research Institute, received consultancy fees and honoraria from Amgen, CSL Vifor/Fresenius, Relypsa, Sanifit, Sanofi/Genzyme, and Tricida, received honoraria or payment for talks as a visiting Professor at several universities, received payment for speaking at the Harvard Nephrology Board review course and an American Society of Nephrology course, and is a shareholder of Amgen and Tricida. Lingyun Li is an employee and shareholder of CSL Vifor. Carol Moreno Quinn is a former employee and shareholder of CSL Vifor.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Vifor Fresenius Medical Care Renal Pharma Ltd.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
