Abstract
Background and aims
Esmolol is a common short-acting drug to control ventricular rate. This study aimed to evaluate the association between use of esmolol and mortality in critically ill patients.
Methods
This is a retrospective cohort study from MIMIC-IV database containing adult patients with a heart rate of over 100 beats/min during the intensive care unit (ICU) stay. Multivariable Cox proportional hazard models and logistic regression were used to explore the association between esmolol and mortality and adjust confounders. A 1:1 nearest neighbor propensity score matching (PSM) was performed to minimize potential cofounding bias. The comparison for secondary outcomes was performed at different points of time using an independent t-test.
Results
A total of 30,332 patients were reviewed and identified as critically ill. There was no significant difference in 28-day mortality between two groups before (HR = 0.90, 95% CI = 0.73-1.12, p = 0.343) and after PSM (HR = 0.84, 95% CI = 0.65-1.08, p = 0.167). Similar results were shown in 90-day mortality before (HR = 0.93, 95% CI = 0.75-1.14, p = 0.484) and after PSM (HR = 0.85, 95% CI = 0.67-1.09, p = 0.193). However, esmolol treatment was associated with higher requirement of vasopressor use before (HR = 2.89, 95% CI = 2.18-3.82, p < 0.001) and after PSM (HR = 2.66, 95% CI = 2.06-3.45, p < 0.001). Esmolol treatment statistically reduced diastolic blood pressure (DBP), mean arterial pressure (MAP), and heart rate (all p < 0.001) and increased fluid balance at 24 hours (p < 0.05) but did not significantly lower SBP (p = 0.721). Patients in esmolol group showed no significant difference in lactate levels and daily urine output when compared with those in non-esmolol group when adjusted for confounders (all p > 0.05).
Conclusion
Esmolol treatment was associated with reduced heart rate and lowered DBP and MAP, which may increase vasopressor use and fluid balance at the timepoint of 24 hours in critically ill patients during ICU stay. However, after adjusting for confounders, esmolol treatment was not associated with 28-day and 90-day mortality.
Introduction
Critically ill patients usually present tachyarrhythmia due to sympathetic activation in various causes including pain, anxiety, exaggerated sympathetic activation, shock, heart failure, primary tachycardia, and so on. 1 Studies have reported that a prolonged higher heart rate (heart rate > 100 beats/min [bpm]) is significantly associated with increased intensive care unit (ICU) mortality, in-hospital mortality, and 90-day mortality.1,2 Heart rate is also a part of ICU prognosis evaluation, which indicates more potential severe diseases.3,4 Therefore, it seems that there is an urgent need to control the fast heart rate though guidelines or recommendations on heart rate management in the ICU setting have been published. 5 Heart rate is normally determined by the pacemaker cells in the sinus node, which could be inhibited by β-1 adrenergic blockers including esmolol.6,7 Esmolol is one of the intravenous, short-acting, titratable β-1 blocker used in critical care settings to play a pivotal role in heart rate reduction.7,8 In a randomized clinical trial including 77 patients with septic shock, the authors found that esmolol significantly reduced heart rate to achieve target levels without increased 28-day mortality. 8 A recent meta-analysis showed a similar result that esmolol treatment reduced heart rate, and this analysis revealed that esmolol may be able to increase the survival rate. 9 Physiologically, β-blocker therapy can reduce sympathetic nervous system activation and catecholamine release. 10 However, studies on the effect of esmolol treatment in critically ill patients with elevated heart rate are scarce. Considering the undetermined effect of esmolol on survival benefits in critically ill patients, we conducted a retrospective study of the MIMIC-IV database to explore esmolol treatment on clinical outcomes including 28-day mortality, 90-day mortality, heart rate control, and the influence of blood pressure, fluid management and lactate levels in critically ill patients.
Methods and Materials
Study Cohort
The data of the study cohort were extracted from the Multiparameter Intelligent Motoring in Intensive Care Database IV (MIMIC-IV), a large critical care database. The MIMIC-IV includes patient's records from Beth Israel Deaconess Medical Center from 2008 to 2019. 11 The database contains common parameters, for example, heart rates, blood pressure, lactate levels, fluid management, urine output, medications, diagnosis code according to ICD-10, and follow-up data as long as 90 days. One of the authors (SY) obtained access (Certification number 35931520) and was responsible for the data extraction.
Inclusion and Exclusion Criteria
Patients (age ≥ 18) with records of ICU stay in MIMIC-IV were screened to meet the inclusion criteria. Those with repeated ICU admission and heart rate lower than 100 bpm were excluded. We included adult patients with a heart rate over 100 bpm during the ICU stay.
Definitions
Controlled heart rate was defined as the mean heart rate within 24 hours of admission to the ICU between 60 and 100 bpm. Normal SBP was defined as SBP between 90 and 130 mmHg, and diastolic blood pressure (DBP) was between 60 and 85 mmHg. Mean arterial pressure (MAP) remained above 65 mmHg.
Outcomes
The primary outcomes were 28-day mortality and 90-day mortality. The secondary outcomes were heart rate control, effects on blood pressure, vasopressor use, lactate levels, urine output, and fluid balance.
Statistical Analysis
A univariable analysis was performed for all eligible patients. Continuous variables were displayed as mean and standard deviation (SD) or median and interquartile range (IQR), depending on whether the normal distribution assumption was satisfied. The Shapiro–Wilk test was performed to test the normality of the variables. Categorical variables were displayed as numbers and percentages (%). Chi-squared tests, Fisher's exact probability test, independent t-test, and Wilcoxon test were used to compare the difference between esmolol group and non-esmolol group appropriately.
To further assess the association between the use of esmolol and the outcomes, we conducted a multivariable Cox proportional hazard model and a logistic regression. The odds ratios and hazard ratios and their corresponding 95% confidence intervals (95% CI) were calculated compared to the non-esmolol group. For confounders, we adjusted all characteristics in Table 1 in analysis for all outcomes. In addition, to minimize the bias of potential confounders, a 1:1 nearest neighbor propensity score matching (PSM) was performed to examine the association between the use of esmolol and 28-day mortality. We generated a propensity score with all the covariates in Table 1 to estimate patients’ probability of using the esmolol. The comparison for secondary outcomes between the esmolol group and the non-esmolol group was performed at different points of time. The differences for each point of time were compared using an independent t-test. All continuous outcomes were described as mean ± SD in the figures.
Baseline Characteristics and Outcomes Between Esmolol Group and Non-Esmolol Group.
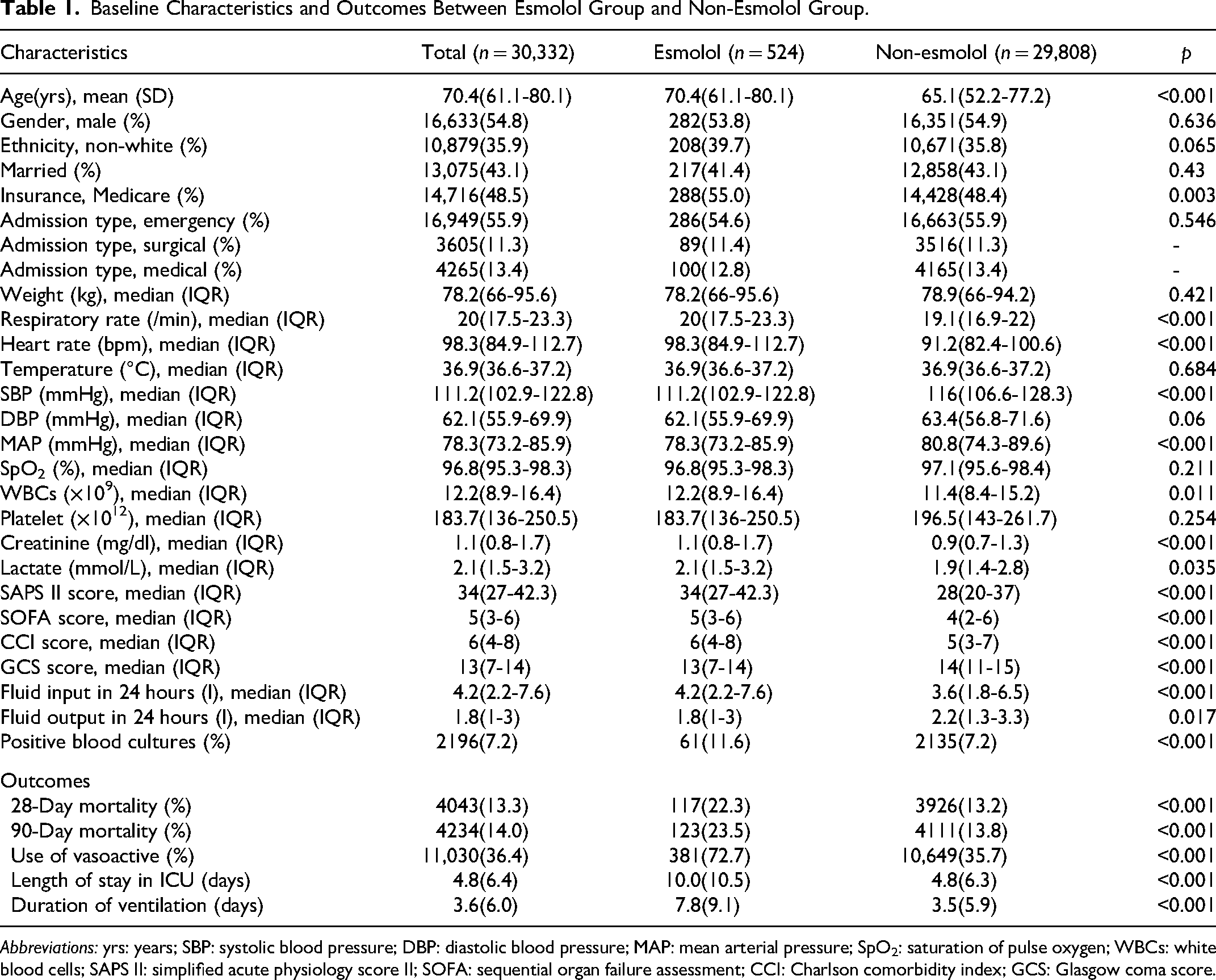
Abbreviations: yrs: years; SBP: systolic blood pressure; DBP: diastolic blood pressure; MAP: mean arterial pressure; SpO2: saturation of pulse oxygen; WBCs: white blood cells; SAPS II: simplified acute physiology score II; SOFA: sequential organ failure assessment; CCI: Charlson comorbidity index; GCS: Glasgow coma score.
We conducted subgroup analyses stratified by gender, sepsis, AKI (acute kidney injury), and surgery to assess the consistency of treatment effect among the subgroups. Furthermore, two sensitivity analyses were conducted to examine the robustness of our results: (a) excluding patients with delayed use of esmolol after ICU admission (the timing of esmolol use larger than the median of the LOS ICU); (b) restricting patients whose LOS ICU larger than 2 days. All analyses were performed with the statistical software R version 4.0.2 (R Foundation for Statistical Computing, Vienna, Austria) and SAS (9.4). The threshold of p < 0.05 (two-sided) was considered statistically significant.
Results
Patient Characteristics
Flowchart of eligible patients is shown in Figure 1. A total of 69,619 patients were screened and finally, there were 30,332 patients with a heart rate over 100 bpm during the ICU stay. Among them, 524 (1.7%) patients were administered with esmolol (esmolol group) and 29,808 (98.3%) patients were not used esmolol (non-esmolol group). Overall, the mean age was 70.4 years and over 50% of patients were admitted from the department of emergency. The baseline heart rate was 98.3 bpm and baseline MAP was 78.3 mmHg. Patients in the esmolol group had older age, higher respiratory rate, and baseline heart rate, lower systolic blood pressure (SBP) and MAP, higher white blood cells (WBCs), creatine levels, lactate levels than the non-esmolol group. In addition, simplified acute physiology score (SAPS II), sequential organ failure assessment (SOFA), Charlson comorbidity index (CCI), and Glasgow coma scale (GCS) were all significantly different between the esmolol group and the non-esmolol group (Table 1). Specifically, potential medications that would affect heart rate and blood pressure, such as lidocaine, amiodarone, verapamil, diltiazem, nitroprusside, norepinephrine, nitroglycerin, dopamine, dobutamine, atropine, phenylephrine and relative drugs of sedation were also listed in Table 2.

Flowchart of eligible patients.
Medication and Therapeutic Measure Between Esmolol Group and Non-Esmolol Group.

Abbreviations: AKI: acute kidney injury.
Esmolol Treatment was not Significantly Associated With Reduced 28-Day and 90-Day Mortality
In univariate analysis, esmolol treatment was significantly associated with increased 28-day and 90-day mortality (all p < 0.001) (Table 1). When adjusting for all the confounders listed in Table 1 (except for the outcomes), there was no significant difference in 28-day mortality between the esmolol group and non-esmolol group before (HR = 0.90, 95% CI = 0.73-1.12, p = 0.343) and after PSM (HR = 0.84, 95% CI = 0.65-1.08, p = 0.167). Similar results were shown in 90-day mortality before (HR = 0.93, 95% CI = 0.75-1.14, p = 0.484) and after PSM (HR = 0.85, 95% CI = 0.67-1.09, p = 0.193) between the two groups (Table 3, Figure 2). Furthermore, our subgroup analyses also indicated that there was no significant difference in 28-day and 90-day mortality between the esmolol group and the non-esmolol group (all p > 0.05) (Table 4). We also performed two sensitivity analyses to further confirm the relationship between the use of esmolol and 28-day and 90-day mortality. Model 1 excluded those who started using esmolol after 2.66 days (median of the length of stay in ICU) in the ICU admission and Model 2 restricted those who stayed in ICU for more than 2 days. After adjustments, the use of esmolol showed no significant association with both 28-day and 90-day mortality before and after PSM (all p > 0.05) (Table 5). Therefore, esmolol treatment was not significantly associated with a reduced risk of death.

Mortality rates and vasopressor use between esmolol group and non-esmolol group. Adjusted for all confounding factors listed in Table 1. Hazard ratio for vasopressor use was replaced by odds ratio (OR) calculated with the multivariable logistic model.
Changes in Heart Rate and Blood Pressure Within the First 7 Days When Heart Rate Over 100 bpm Occurred.
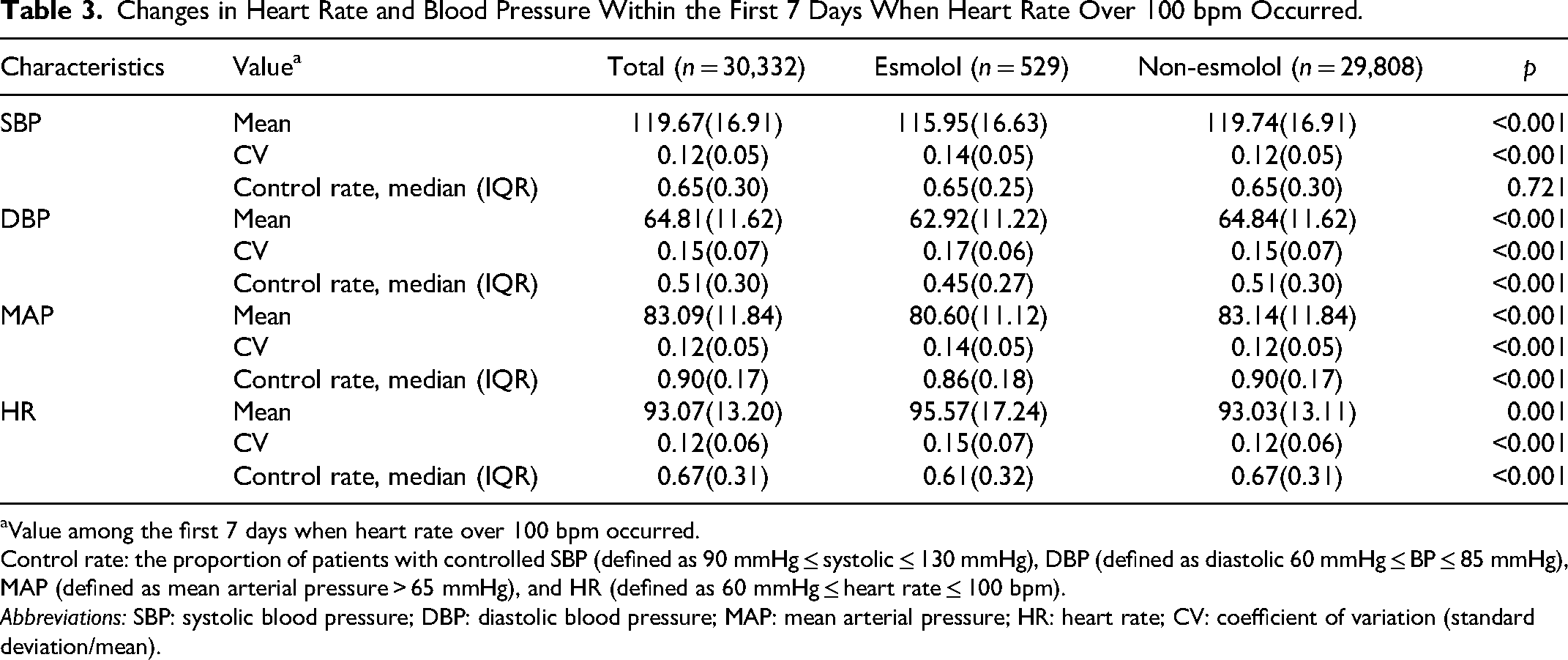
Value among the first 7 days when heart rate over 100 bpm occurred.
Control rate: the proportion of patients with controlled SBP (defined as 90 mmHg ≤ systolic ≤ 130 mmHg), DBP (defined as diastolic 60 mmHg ≤ BP ≤ 85 mmHg), MAP (defined as mean arterial pressure > 65 mmHg), and HR (defined as 60 mmHg ≤ heart rate ≤ 100 bpm).
Abbreviations: SBP: systolic blood pressure; DBP: diastolic blood pressure; MAP: mean arterial pressure; HR: heart rate; CV: coefficient of variation (standard deviation/mean).
Subgroup Analysis in Sepsis, AKI and Ventilation Subgroups.
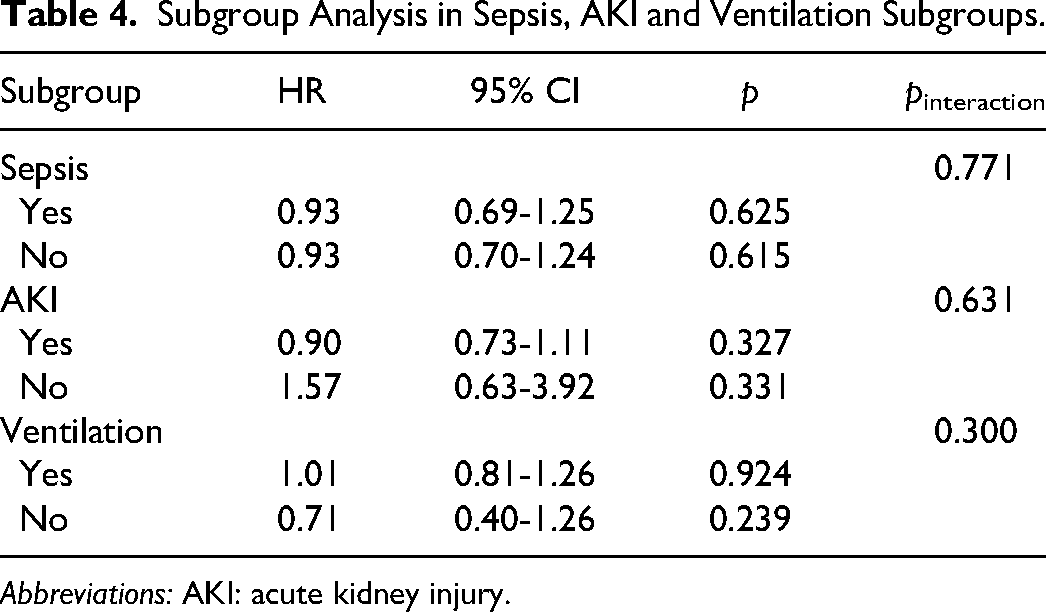
Abbreviations: AKI: acute kidney injury.
Sensitivity Analysis.
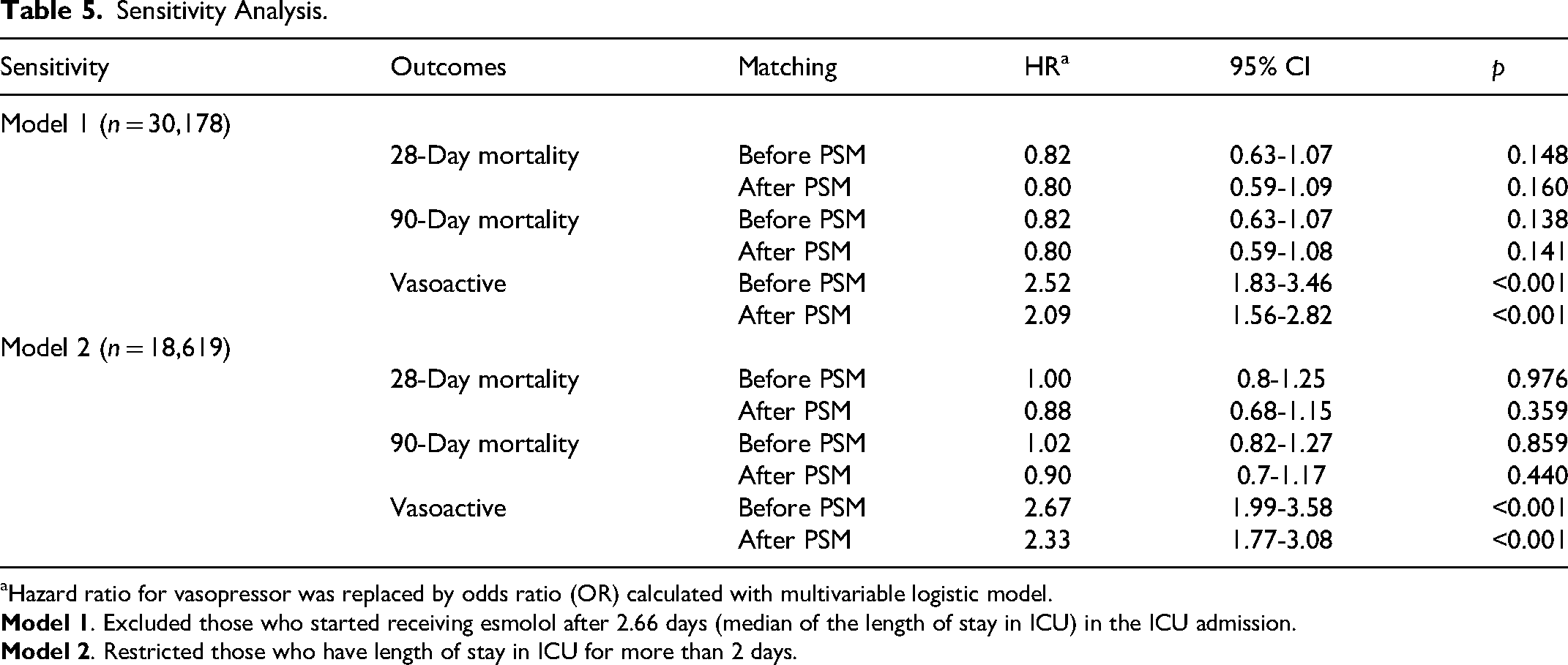
Hazard ratio for vasopressor was replaced by odds ratio (OR) calculated with multivariable logistic model.
The Association of Esmolol Treatment and Hemodynamic Parameters/Vasopressor Utilization Rate
We then focused mainly on the data including control rate, and coefficient of variation in the first 7 days when a heart rate over 100 bpm occurred, to determine changes in heart rate and blood pressure between the esmolol group and non-esmolol group. Patients in the esmolol group had significantly lower mean SBP, DBP, MAP, and higher mean HR when compared with those in the non-esmolol group. We concluded that esmolol treatment statistically reduced DBP, MAP, and heart rate (all p < 0.001) but did not significantly lower SBP (p = 0.721) based on the analysis of the control rate (Table 2).
Moreover, we also explored whether esmolol administration increased vasopressor utilization rate. Vasopressor drugs used in this study included norepinephrine, epinephrine, phenylephrine, vasopressin, dopamine, and dobutamine. After adjusting for confounders, esmolol administration was statistically associated with increased vasopressor utilization rate before (odds ratio [OR] = 3.26, 95% CI = 2.49-4.29, p < 0.001) and after PSM (OR = 2.33, 95% CI = 1.82-2.99, p < 0.001) (Table 2, Figure 2). In addition, the two sensitivity analyses also demonstrated that esmolol treatment significantly increased the rate of vasopressor use (all p < 0.001) (Table 5).
The Association of Esmolol Treatment and Lactate Levels, Daily Urine Output, and Fluid Therapy for Each Point of Time
Patients in the esmolol group showed no significant difference in lactate levels at the time point of 24 hours, 48 hours, and 72 hours when compared with those in the non-esmolol group (p = 0.737, p = 0.580, and p = 0.125) (Figure 3A). Similar results in daily urine output at the time point of 24 hours, 48 hours, and 72 hours (p = 0.051, p = 0.162, and p = 0.825) (Figure 3B). In terms of fluid therapy, no significant difference was found between the two groups at 48 hours and 72 hours (3868.02 ml vs 4026.77 ml, p = 0.259), while patients in the esmolol group had statistically higher positive fluid balance than that in the non-esmolol group at the time of 24 hours (p < 0.05) (Figure 3C).
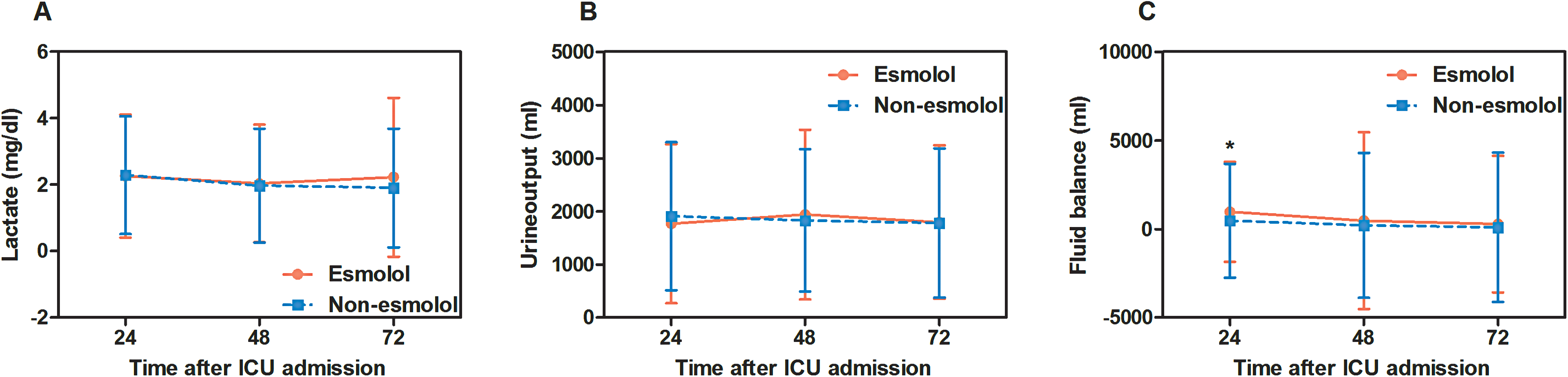
The association of esmolol treatment and lactate levels, daily urine output, and fluid therapy. Adjusted for all confounding factors in Table 1 except for the stratified variables (subgroups). Hazard ratio for vasopressor was replaced by odds ratio (OR) calculated with multivariable logistic model.
Discussion
We evaluated the association between the use of esmolol and clinical outcomes in this retrospective study. A large number of patients with a heart rate over 100 bpm were included and 1.7% of patients received esmolol treatment to control their heart rate. Inconsistent with previously published papers, 8 our findings indicated that no significant difference was shown between esmolol treatment and 28-day and 90-day mortality after adjusting for confounders or PSM. This result was consistent with the results in our subgroup analyses and sensitivity analyses. In addition, esmolol statistically reduced DBP, MAP, and HR and was significantly associated with increased vasopressor utilization rate and higher fluid input at the timepoint of 24 hours.
Cardiac rhythm disturbances, especially tachyarrhythmias, are frequently happened in critically ill patients. 12 Patients with atrial fibrillation, the most common tachyarrhythmia, have been reported to suffer from longer ICU length of stay and their ICU and hospital mortality are twice as high as those without atrial fibrillation. 13 Esmolol, a cardio-selective β-blocker, has been proved to be effective to control tachyarrhythmia including supraventricular tachyarrhythmia and tachycardia. 14 In our study, we disclosed that esmolol treatment significantly reduced heart rate, which was consistent with previous studies.8,15 Moreover, in some conditions, intravenous esmolol can be used to lower blood pressure.16,17 Since the use of sedation drugs may influence patients’ conditions that would indirectly interfere with heart rate, we regarded sedation as one of the confounders; and the underlying medicines that might affect heart rate including lidocaine, amiodarone, verapamil, diltiazem, nitroprusside, norepinephrine, nitroglycerin, dopamine, dobutamine, atropine, and phenylephrine were also regarded as confounders in our statistical analysis. Our study showed that esmolol might decrease both DBP and MAP, but no significant difference was found in SBP. In an early study for healthy volunteers, the authors reported that esmolol significantly reduced SBP. 18 This effect of esmolol may attribute to different populations with different levels of sympathetic activity, various sedatives and vasoactive drugs, and different cardiovascular statuses. 14 With the effect of lower blood pressure, patients in the esmolol group had a higher rate of vasopressor utilization rate than that in the non-esmolol group.
More importantly, although esmolol statistically reduced heart rate, no significant difference was found between the use of esmolol and mortality rate in critically ill patients with a heart rate of over 100 bpm in the present study. The core reasons might be that esmolol also reduced blood pressure, which required more frequent vasopressor utilization and more positive fluid balance for patients. At the same time, no significant difference was shown in lactate levels and urine output between the esmolol group and the non-esmolol group after adjusting for all confounding factor. We speculated that lower heart rate with more frequent vasopressor utilization. The effect of insufficient tissue perfusion, 19 and more positive fluid balance, which partly recovered tissue perfusion, 20 had an offsetting effect to the influence for clinical outcomes. A systematic review included 88 randomized controlled trials illustrated that perioperative application of β-blockers like esmolol significantly reduced cardiac burden and lowered the occurrence of arrhythmias in cardiac surgery. However, the influence of β-blockers on mortality remained unclear. 21 In a recently published randomized controlled trial, Zangrillo et al declared that esmolol did not significantly lower mortality in patients with cardiac surgery. 22 However, for those with septic shock, esmolol reduced heart rate to targeted limits more effectively and significantly improved 28-day mortality. 23 In our study, we focused on critically ill patients including all kinds of diseases and populations in ICU. To the best of our knowledge, our study first reported that esmolol treatment was not associated with 28-day and 90-day mortality with over 500 patients who received esmolol treatment were included. With a large sample size and multiple population with critical status, we believed that esmolol was associated with reduced heart rate to normal range without affecting 28-day and 90-day mortality. Specifically, due to the various reasons of increasing heart rate of critically ill patients, such as fever, volume depletion, hypotension and shock, sepsis, anemia, hypoxia, pulmonary embolism, acute coronary ischemia, and myocardial infarction, pain, anxiety, and sleep deprivation, 24 combined with our present study revealing that esmolol showed no significant association with reduced mortality, we should firstly deal with the root cause of increased heart rate, rather than giving priority to using esmolol. The esmolol cohort was associated with increased vasopressor utilization and more positive fluid balance, which indicated that a direct relationship to the lower blood pressure may be premature. However, ultimately, randomized clinical trials will be needed to further demonstrate the specific relationship between esmolol treatment and clinical outcomes in critically ill patients.
Of note, our study is a retrospective study, though a large sample size was enrolled, we still cannot draw causal conclusions about esmolol treatment and clinical outcomes owning to the potential confounding bias and missing values. The esmolol cohort was older, had a statistically significant higher SAPS II, SOFA and CCI score and lower GCS. These unbalances may have affected the cohort acuity. In addition, we have tried to adjust for confounders to obtain valid results and further confirm our results by subgroup analyses and sensitivity analyses. However, the heart rate can be influenced by various medical treatments including sedation and analgesia and some external factors, which may affect the analysis.
Conclusion
Esmolol treatment was associated with reduced heart rate and lowered DBP and MAP, which may increase vasopressor use and fluid balance at the timepoint of 24 hours in critically ill patients during ICU stay. However, after adjusting for confounders, esmolol treatment was not associated with 28-day and 90-day mortality.
Footnotes
Author Contributions
Liang Qihong: methodology, investigation, software, data curation, formal analysis, visualization, writing the original draft.
Li Lulan: conceptualization, investigation, methodology, investigation, formal analysis, visualization, writing the original draft.
Chen Kerong: methodology, investigation, validation, resources, visualization, writing the original draft.
An Sheng: methodology, validation, resources, visualization, writing the original draft.
Deng Zhiya: conceptualization, investigation, resources, visualization, writing the original draft.
Li Jiaxin: investigation, validation, resources, visualization, writing the original draft.
Zhou Shiyu: methodology, investigation, software, data curation, formal analysis, visualization, writing the original draft.
Chen Zhongqing: investigation, validation, resources, visualization, writing the original draft.
Zeng Zhenhua: supervision, project administration, writing, reviewing and editing.
An Shengli: supervision, project administration, writing, reviewing and editing.
Data Availability Statement
The data in this study were extracted from MIMIC-IV, a large critical care database which was also named Multiparameter Intelligent Motoring in Intensive Care Database IV (https://mimic.mit.edu/). One of the authors (Shiyu Zhou) has obtained access (Certification number 35931520) and was responsible for the data extraction. The code for data extraction can be found on GitHub (![]() ).
).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Clinical Research Program of NanFang Hospital, Southern Medical University [grant number: 2018CR047] and Guangdong Basic and Applied Research Foundation [grant number: 2022A1515012152].
