Abstract
Introduction:
Ticagrelor might reduce infarct size by exerting a more potent antiplatelet effect or by promoting a potential conditioning stimulus in ST-elevation myocardial infarction (STEMI) patients. Pre-infarction angina (PIA) is an effective preconditioning stimulus that reduces ischemia-reperfusion injury. Because little is known on the interaction of PIA in STEMI-patients loaded with ticagrelor, we sought to determine if patients loaded with ticagrelor had improved clinical outcomes as compared to clopidogrel and to study if it is modulated by the presence of PIA.
Methods:
From 1272 STEMI patients submitted to primary percutaneous coronary intervention and treated with clopidogrel or ticagrelor from January 2008 to December 2018, 826 were analyzed after propensity score matching. Infarct size was estimated using peak creatine kinase (CK) and troponin T (TnT), and clinical impact was evaluated through cumulative major cardiac and cerebrovascular events (MACCE) at 1-year follow-up. Matched patients and their interaction with PIA were analyzed.
Results:
Patients loaded with ticagrelor had lower peak CK [1405.50 U/L (730.25-2491.00), P < .001] and TnT [3.58 ng/mL (1.73-6.59), P < .001)], regardless of PIA. The presence of PIA was associated with lower CK (P = .030), but not TnT (P = .097). There was no interaction between ticagrelor loading and PIA (P = .788 for TnT and P = .555 for CK). There was no difference in MACCE incidence between clopidogrel or ticagrelor loading (P = .129). Cumulative survival was also similar between clopidogrel or ticagrelor, regardless of PIA (P = .103).
Conclusion:
Ticagrelor reduced infarct sizes independently and without a synergic effect with PIA. Despite reducing infarct size, clinical outcomes were similar across both groups.
Introduction
In patients presenting with ST-elevation myocardial infarction (STEMI), the best evidence-based approach in reducing infarct size and mortality is timely primary percutaneous coronary intervention (PPCI). 1 However, despite rescuing the ischemic cardiomyocytes from further infarction, reperfusion is also associated with an additional component of irreversible myocardial damage called the ischemia-reperfusion (IR) injury phenomenon. 2 -6 While several cardioprotective strategies have consistently shown considerable promise in reducing the IR injury in preclinical studies, their translation to clinical practice has been unsatisfactory. 7 -10
Potent oral antiplatelet agents have been a mainstay of current PPCI to limit clot organization and to prevent stent thrombosis. Ticagrelor, a P2Y12 inhibitor, has been used for this purpose in patients presenting with acute coronary syndrome (ACS). 1 It has been suggested that ticagrelor has pleiotropic effects, exerting a potential conditioning stimulus through the inhibition of the Equilibrative Nucleoside Transporter 1 (ENT-1), expressed on the surface of erythrocytes, which leads to reduced adenosine reuptake. 11 -13 Adenosine would then act on cardiomyocytes, activating multiple pathways, including the Reperfusion Injury Salvage Kinase (RISK) and Survival Activating Factor Enhancement (SAFE) signaling pathways. 2,3 These are thought to promote the survival of the cardiomyocyte by inhibiting the mitochondrial permeability transition pore (MPTP) and stimulating the expression of cardioprotective genes. 2,3 However, there are inconsistent data regarding the potential cardioprotective effect of ticagrelor, with conflicting results. 14 -20
On the other hand, and despite not being entirely consensual, the presence of pre-infarction angina (PIA), which is believed to be a very potent conditioning stimulus protecting the cardiomyocyte from the IR injury, 3 has been most consistently shown to limit infarct size, reduce mortality, and improve left ventricle ejection fraction (LVEF) in patients presenting with STEMI. 2,21 -28 Therefore, it is possible that PIA may render additional cardioprotective agents irrelevant.
While several studies have researched the individual cardioprotective effect of ticagrelor and PIA, to the best of our knowledge, the potential synergism between PIA and ticagrelor has not yet been studied. Given the unmet need in identifying a strategy to limit the IR phenomenon, and postulating that ticagrelor loading in an already preconditioned patient might hamper any conditioning maneuver at reperfusion, our aim was to determine if ticagrelor loading in STEMI patients with or without preexisting PIA is related to improved outcomes.
Materials and Methods
Study Population and Definitions
We carried out a retrospective, observational, cohort study which included every consecutive patient with an admission for a STEMI who underwent PPCI, with a clearly identifiable culprit lesion, between January 1st 2008 and December 31st 2018, at a tertiary hospital. All patients were loaded with aspirin (500 mg) and either clopidogrel (600 mg) or ticagrelor (180 mg) before PPCI (given in ambulance or in the emergency room upon arrival). Only naïve patients to clopidogrel or ticagrelor were included in the analysis. The myocardial necrosis biomarkers were serially sampled by local protocol upon admission, then at 6 h, 12 h, 24 h and 48 h. We excluded patients who were not treated with ticagrelor or clopidogrel at admission and those that switched the P2Y12 inhibitor during hospital stay or at follow-up. We also excluded patients who presented with a culprit lesion at left main coronary artery or at a previous coronary bypass graft, and also those who came intubated and hence a loading dose was not given before catheterization. For patients with multiple STEMI-related admissions, we only considered the first admission during the study period, with the remaining admissions being considered on follow-up. The diagnosis of STEMI was established according to the criteria defined by the European Society of Cardiology. 1 The clinical, demographic, laboratory and procedural data for these patients were retrospectively obtained by electronic medical chart review and included in an anonymized database. PPCI was performed mostly by radial approach. As per local protocol, the only anticoagulant agent given was unfractionated heparin (70 Units/Kg) by a peripheral vein to all patients undergoing PPCI. IIb/IIIa inhibitors were administered at the discretion of the interventional cardiologist. After completion of PPCI, all patients were admitted to the coronary care unit where evidence-based care was rendered as per current guidelines.
PIA was diagnosed at admission when a patient presented arm, jaw, or chest pain, at rest or during exercise, lasting less than 20 minutes, in the week before the STEMI diagnosis; this was systematically inquired to every patient, according to a prespecified checklist protocol at admission. Total ischemic time is defined as the time between symptom onset and the passage of coronary guidewire with restoration of blood flow.
We calculated the Thrombolysis in Myocardial Infarction (TIMI) score, which gives an estimate of mortality in patients with STEMI, 29 and the SYNTAX score, which estimates the complexity of multivessel coronary artery disease. 30 We stratified SYNTAX into 3 groups in order to have approximately a third of patients in each group: ≤16.00, 16.01-22.00 and ≥22.01. The estimated glomerular filtration rate (eGFR) was calculated according to the Cockcroft-Gault formula.
LVEF was calculated using 2D-echocardiography and stratified in 4 groups: good LVEF was considered for values equal to or higher than 55%; mild impairment was considered if LVEF was between 45% and 55%; moderate impairment if LVEF ranged from 35% and 45%; and severe impairment if LVEF lower than 35%.
The 1-year clinical follow-up data, regarding the occurrence of major cardiac and cerebrovascular events (MACCE) after the index STEMI, were obtained from electronic records by chart review. This study was approved by the ethics committee of Centro Hospitalar Universitário do Porto (2021.291[237-DEFI/245-CE]). Informed consents were waived due to the anonymization and retrospective nature of the study.
Definition of Clinical Outcomes
Infarct size was estimated using peak creatine kinase (CK) and peak troponin T (TnT) as surrogates from the samples drawn at 6, 12, 24 and 48 h. TnT concentration was measured by Elecsys Modular Analytics e170 (Roche Diagnostics, Mannheim, Germany); CK activity was measured on a Roche Cobas 8000 analyzer (c701 module; Roche Diagnostics). Cumulative MACCE at 1 year follow-up is a composite of death of any cause, stroke with imaging evidence, recurrent myocardial infarction (MI), and target lesion revascularization (TLR) (defined as angina or restenosis with ischemia, requiring reintervention, of a previous culprit lesion).
Statistical Analysis
Continuous variables were expressed as mean ± standard deviation (SD) when they followed a normal distribution or as median and interquartile range, between quartiles 25 (Q1) and 75 (Q3), when non-normally distributed. Categorical variables were represented as absolute values and percentages [n (%)]. The normality of distribution was assessed by Shapiro-Wilk and Kolmogorov-Smirnov tests.
To compare continuous variables between groups, we used an independent Student t test when variables followed a normal distribution or Mann-Whitney U test for non-normally distributed variables. To compare categorical values, we used χ 2 or Fisher exact tests, when appropriate.
The logarithmic transformation was applied to peak CK, peak TnT, total ischemic time and SYNTAX score in order to allow for parametric testing and adequate propensity score (PS) matching.
As the standard of antiplatelet therapy changed during the study period, with ticagrelor being favored over clopidogrel, we attempted to reduce the treatment assignment bias by using PS to match each patient treated with ticagrelor to a comparable patient treated with clopidogrel. To estimate the PS, we used 7 baseline characteristics, namely sex (male/female), age (years), smoking habits (yes/no), diabetes mellitus (yes/no), dyslipidemia (yes/no), hypertension (yes/no), and eGFR (mL/min). We also used 6 variables related to procedural data, particularly TIMI and SYNTAX risk scores, anterior acute myocardial infarction (yes/no), LVEF at discharge (good/mild/moderate/severe), Killip class (≤2/≥3) and total ischemic time (minutes). The match tolerance was set at 0.01. We then performed all comparative analyses after PS matching.
To analyze the interaction between PIA and the use of ticagrelor or clopidogrel, a 2-way factorial ANOVA was computed.
We estimated cumulative survival at follow-up with a Kaplan-Meier analysis, using the log-rank test for comparisons.
For all tests, a two-sided P value less than .05 was considered statistically significant. We used SPSS version 27.0 (SPSS Inc., Chicago, IL, USA) for all comparative statistics and propensity score matching.
Results
Study Population
A total of 1272 STEMI patients were treated by PPCI between January 1st 2008 and December 31st 2018. Of these, 1214 patients (95%) fulfilled the entry criteria and comprised our study sample. Tables S1 and S2 show the demographic and procedural characteristics and in-hospital outcomes, respectively, in accordance with ticagrelor vs clopidogrel loading, prior to PS matching. Patients who received ticagrelor were younger, had better renal function and were pre-treated more often with calcium channel blockers. Both groups presented similar procedural characteristics, except for door-to-balloon time, IIb/IIIa inhibitors and thrombectomy usage, and length of stay, which were lower on patients treated with ticagrelor. Furthermore, patients treated with clopidogrel had lower radial access and coronary disease complexity, as measured by the SYNTAX score. At discharge, patients treated with ticagrelor were less prescribed with β-blockers and angiotensin-converting enzyme inhibitors. LVEF was not different at discharge from hospital.
By applying the PS matching as explained in the previous section, 413 patients loaded with ticagrelor were matched with 413 patients loaded with clopidogrel (Figure 1). Table 1 shows the baseline and procedural characteristics, and Table 2 shows the in-hospital outcomes after PS matching. In patients treated with clopidogrel, although more patients [151 (36.7%)] presented PIA at admission, this difference was not statistically significant. The remaining baseline characteristics were identical between the studied groups. Regarding procedural characteristics, total ischemic time was comparable, whereas door-to-balloon time, IIb/IIIa inhibitors infusion, and length of stay were lower in patients loaded with ticagrelor. Moreover, radial access and coronary artery disease complexity (SYNTAX score) were lower in patients treated with clopidogrel. No follow-up data was available in 3 patients (2%).

Flowchart of the study, illustrating the distribution according to propensity score matching. PPCI indicates primary percutaneous coronary intervention; STEMI, ST-elevation myocardial infarction.
Baseline and Procedural Characteristics After Propensity Score Matching.
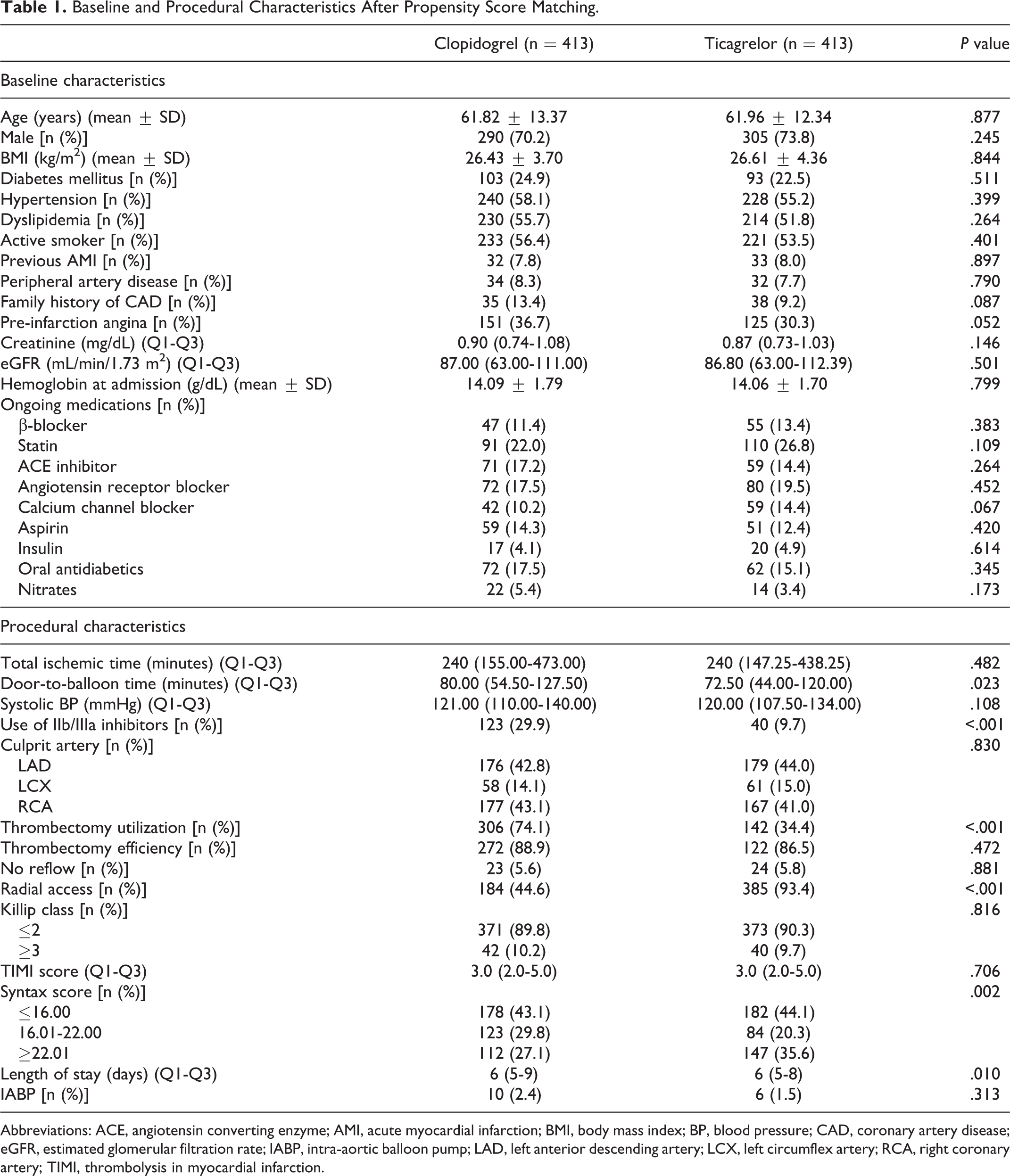
Abbreviations: ACE, angiotensin converting enzyme; AMI, acute myocardial infarction; BMI, body mass index; BP, blood pressure; CAD, coronary artery disease; eGFR, estimated glomerular filtration rate; IABP, intra-aortic balloon pump; LAD, left anterior descending artery; LCX, left circumflex artery; RCA, right coronary artery; TIMI, thrombolysis in myocardial infarction.
In-Hospital Outcomes After Propensity Score Matching.

Abbreviations: ACE, angiotensin converting enzyme; ICD, implantable cardioverter defibrillator; LVEF, left ventricle ejection fraction.
Outcomes
Patients treated with ticagrelor had a lower peak CK and TnT [1405.50 U/L (730.25-2491.00) for CK, P < .001, and 3.58 ng/mL (1.73-6.59) for TnT, P < .001)], regardless of PIA (P < .001 for CK and TnT without PIA; P = .009 for CK and P = .006 for TnT in patients presenting with PIA) (Table 3, Figure 2). The presence of PIA was associated with lower CK values (P = .030), but not TnT (P = .097). No interaction was found between ticagrelor vs clopidogrel loading and PIA (P = .788 for TnT and P = .555 for CK, Figures S1 and S2).
Main Outcomes.

Abbreviations: CK, creatine kinase; MACCE, major adverse cardiac and cerebrovascular events; MI, myocardial infarction; PIA, pre-infarction angina; TLR, target leasion revascularization.
a Compared by Student t test after logarithmic transformation.

Distribution of peak CK and peak troponin T according to PIA status. CK indicates creatinine kinase; PIA, pre infarction angina. +Compared by Student t test after logarithmic transformation.
Regarding MACCE at 1-year follow-up (Table 3, Figure 3), while cumulative MACCE tended to be higher in patients treated with clopidogrel (13.3% vs 8.0% in patients loaded with ticagrelor), there was no statistically significant difference between clopidogrel or ticagrelor loading (P = .129). Cumulative survival was also similar between clopidogrel or ticagrelor, regardless of PIA (P = .103, Figure 4).

Proportion of cumulative major cardiac and cerebrovascular events (MACCE) according to clopidogrel or ticagrelor loading.

Kaplan-Meier curves for cumulative survival. P value represents the difference between groups. PIA indicates pre infarction angina.
Discussion
In this study, we showed that ticagrelor led to lower peak CK and TnT when compared to clopidogrel, and this effect is independent of PIA, which reduces peak CK, but not peak TnT. At discharge, there was no difference in LVEF between both groups. Also, there was no difference in MACCE at 1-year follow-up.
It has been consistently shown that the larger the infarct size, the higher the incidence of death, heart failure (HF) and an overall worse prognosis. 31 Our results show that, despite reducing peak CK and TnT, ticagrelor had no clinical impact in terms of MACCE reduction, despite tending to be lower in ticagrelor-loaded patients. The impact of ticagrelor as a cardioprotective strategy on MACCE or other hard endpoints has been controversial. The PLATO trial showed that ticagrelor reduced death from vascular causes, stroke, and MI in ACS patients. 32 Also, in a network meta-analysis comparing the efficacy and safety of P2Y12 inhibitors in ACS, Navarese et al showed that ticagrelor reduced mortality when compared to clopidogrel and prasugrel. 18 However, in a STEMI population, Hjortbak et al demonstrated that, as compared to clopidogrel, ticagrelor led to lower hospitalization for HF at 1-year follow-up, while failing to show a difference in MACCE incidence. 20 It is worth noting that many studies showing a reduction in mortality enrolled ACS patients, whether it referred to STEMI or not. 19,32,33 STEMI-patients present with complete coronary occlusion, 34 thus being more prone to IR injury than non-STEMI patients and the confounding factor induced by non-STEMI patients could explain the disparities between studies. Although the concentration of myocardial necrosis biomarkers has been related to final infarct size, as evaluated by cardiac magnetic resonance (CMR), 35 factors like the timing of blood sample withdrawal and the washout from the necrotic myocardium underperforms contemporary cardiac imaging modalities, which can evaluate both the area at risk and better estimate final infarct size. 31
While ticagrelor is related to smaller infarct sizes when compared to clopidogrel, 36,37 it is important to consider that clopidogrel is a prodrug, requiring a two-step metabolic activation process that leads to a delay between when the patient is given the drug and when an adequate plasma level is reached. 38,39 This could explain why clopidogrel loading did not result in a further reduction in CK and TnT levels, as the timing of administration might not have been sufficient to allow for proper metabolism and adequate therapeutic plasma levels. This aspect is also infrequently taken into consideration in most studies comparing ticagrelor to clopidogrel. However, while this might interfere with platelet reactivity, in itself suggested to be potentially and partially cardioprotective, 20,40 the potency of P2Y12 inhibition does not linearly translate to smaller infarct sizes. 20 In a study comparing ticagrelor to cangrelor, Ubaid et al demonstrated that, while cangrelor led to more potent P2Y12 inhibition when compared to ticagrelor, it did not lead to further cardioprotection. 41 This adds further support to the thesis that the pleiotropic effects attributed to ticagrelor, which are not a class effect, are the main driver behind its cardioprotective properties. 20 Another important aspect to consider in our study is that ticagrelor-loaded patients presented with an overall better baseline health status, which might have influenced the results, despite the lack of difference in MACCE incidence.
Our results show that PIA reduces peak CK, but not TnT, suggesting that its cardioprotective effect occurs in the earliest MI stages. 42 In our study, patients with PIA and loaded with ticagrelor had similar enzymatic activity to patients treated with ticagrelor who did not present PIA and the lack of interaction between each other suggests that there was no synergistic effect. It is not clear how PIA exerts cardioprotection, but it is thought to involve the activation of signaling pathways, among which the RISK and SAFE pathways, which ultimately target the MPTP. 3,43,44 As previously described, it is believed that ticagrelor exerts its cardioprotective effect through these pathways by enhancing local concentration of adenosine. In a preclinical trial in rabbits, Yang et al showed that when cangrelor, a P2Y12 inhibitor, was combined with ischemic preconditioning, there was no additional cardioprotection when compared to cangrelor alone, suggesting they acted through a similar mechanism. 45 Therefore, this could explain the lack of additive effect in our study, 46 as the cardioprotection provided by PIA might be superimposed by ticagrelor loading. However, there is still an advantage in ticagrelor loading, and this benefit, even if slight, is better than no benefit at all, especially among patients without PIA.
It is also possible that the current medical therapy for STEMI associated with PPCI is optimized in such a way that cardioprotection is already at its possible maximum. 47 This would possibly explain, in part, why most clinical trials testing other cardioprotective strategies show disappointing results. 48,49 In some clinical trials aimed to evaluate cardioprotective strategies, this may very well be due to a lower than anticipated event rate in an era where state-of-the-art, timely reperfusion, has already proved to be so effective that a small but nevertheless significant infarct size reduction might not have clinical impact. 47 In our series, nearly 90% of patients presented with Killip class 1-2 and a follow-up mortality rate of around 5%. Hence, a clinically meaningful benefit derived from the differences in biomarkers release would be hard to demonstrate. Finally, it is reasonable to consider that the observed benefits of ticagrelor might be more a reflection of a better STEMI-care in later years where ticagrelor replaced clopidogrel in most STEMIs than an advantage of the drug itself.
Because cardioprotection strategies aiming to mitigate the IR injury phenomenon have consistently been shown to reduce infarct sizes in a preclinical setting, 2,3,46,50 and even in some clinical trials, 51 -53 a possible future direction would be to study cardioprotection maneuvers or drugs that signal different cardioprotective mechanisms in a population which benefits the most from cardioprotection on top of current STEMI-care (e.g., patients presenting with large anterior infarcts and heart failure). 47
By sharing some of signaling cardioprotective pathways, our data suggests that ticagrelor loading might overtake the preconditioning effect afforded by PIA and might as well limit any postconditioning maneuver, which should be taken in consideration in future trials. Having those pathways already saturated after ticagrelor loading is a possible explanation. Of course, we cannot neglect the fact that its more potent antiplatelet effect might also have a role in limiting microvascular embolization and hence infarct size. 54 In fact, to take a step forward in current STEMI-care, the microvascular compartment cannot be overlooked anymore despite optimal epicardial flow restoration by PPCI. 55 Coronary microembolization is a complex and multifactorial phenomenon caused by either device manipulation during PCI or spontaneous embolization of atherothrombotic plaque debris and clots. This leads to physical obstruction that causes micro-infarcts and to the release of soluble substances causing endothelial dysfunction, vasoconstriction and platelet aggregation. Leucocyte recruiting enhances the inflammatory response, culminating in cardiomyocyte death and contractile dysfunction. 54 Interestingly, although platelets are thought to play a key role in attaching and facilitating leucocyte infiltration into tissues, animal experiments have shown that preventing the P-selectin-dependent platelet-leucocyte complex does not improve myocardial salvage nor leucocyte transmigration. 56 Therefore, the relative potency of an antiplatelet agent is not a guarantee for a better microvascular reperfusion.
At discharge, there was no difference in LVEF between groups. This could be partly explained by our stratification in 4 groups, according to a range of LVEF values. Regarding HF, by only evaluating LVEF, we are only identifying patients with HF with reduced ejection fraction, as we did not take HF signs and symptoms into consideration for this study. Another important aspect is that we may have overlooked an impact in cardiac remodeling that is not possible to identify from the evaluation of LVEF alone. The HEALING-AMI trial showed that, despite absolute LVEF being similar across the study sample, ticagrelor was associated with positive left ventricle remodeling. 17 This could explain the lower hospitalization for HF, as described in the referred studies above, and be the main endpoint to evaluate in further studies of cardioprotection.
Limitations
There are some limitations to our study. Firstly, this is a retrospective analysis, which has its inherent limitations, and despite using PS to reduce the treatment assignment bias, it is possible that other confounders were still present. We did not evaluate hospitalization due to HF, which is an important clinical endpoint regarding the prognosis, mortality, and disease severity. We also did not take the value of NT-proBNP into consideration, as well as the variation in left ventricle diastolic or systolic volumes at 1 year follow-up, which could be better indicators of adverse cardiac remodeling.
Despite the correlation between peak CK and TnT with imaging and clinical outcomes, 57 we did not use cardiac magnetic resonance or scintigraphy to evaluate infarct size, which are more rigorous in detecting true differences in infarct size and cardioprotective effects, as well as detecting the salvaged myocardia per the area at risk. 58 We acknowledge that this may influence the validity of our results.
The interval between prodromal angina and its impact in microvascular obstruction in previous reports is wide, ranging from 48 h 59 up to 3 months 60 before STEMI. Because it is arguable that those patients who have PIA more closely to the index event may benefit the most from the preconditioning stimulus, narrowing the definition of PIA to 24 to 72 h before STEMI could have had enhanced the effect.
We also did not consider whether hypertensive patients presented with left ventricle hypertrophy, which has been suggested to interfere with cardioprotection. 61 Regarding clopidogrel loading, we did not perform genetic testing or platelet function assays. This could partly explain the lower efficiency of clopidogrel when compared to ticagrelor, as it has been shown that certain CYP isoenzymes result in a lower metabolism, leading to lower plasma clopidogrel concentrations. 38,39
Conclusions
In this propensity score-matched study of STEMI patients, we demonstrated that ticagrelor reduced infarct size. This effect is independent of PIA, and there was no synergic effect between P2Y12 inhibitor loading and PIA. The reduction of infarct size did not translate into better clinical outcomes, as the incidence of cumulative MACCE was not significantly different between groups. Since ticagrelor may significantly modulate infarct size as compared to clopidogrel, this should be taken into account in upcoming trials aiming to study cardioprotection in STEMI-patients.
Supplemental Material
Supplemental Material, sj-docx-1-cpt-10.1177_10742484231169644 - Ticagrelor Loading on ST-Elevation Myocardial Infarction: Interaction With Prodromal Angina on Infarct Size and Clinical Events
Supplemental Material, sj-docx-1-cpt-10.1177_10742484231169644 for Ticagrelor Loading on ST-Elevation Myocardial Infarction: Interaction With Prodromal Angina on Infarct Size and Clinical Events by João Pedro Faria, Pedro Oliveira, André Alexandre, David Sá Couto, Ricardo Costa, Andreia Campinas, André Frias, Bruno Brochado, Raquel Santos, João Silveira, Severo Torres and André Luz in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, sj-jpg-1-cpt-10.1177_10742484231169644 - Ticagrelor Loading on ST-Elevation Myocardial Infarction: Interaction With Prodromal Angina on Infarct Size and Clinical Events
Supplemental Material, sj-jpg-1-cpt-10.1177_10742484231169644 for Ticagrelor Loading on ST-Elevation Myocardial Infarction: Interaction With Prodromal Angina on Infarct Size and Clinical Events by João Pedro Faria, Pedro Oliveira, André Alexandre, David Sá Couto, Ricardo Costa, Andreia Campinas, André Frias, Bruno Brochado, Raquel Santos, João Silveira, Severo Torres and André Luz in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, sj-jpg-2-cpt-10.1177_10742484231169644 - Ticagrelor Loading on ST-Elevation Myocardial Infarction: Interaction With Prodromal Angina on Infarct Size and Clinical Events
Supplemental Material, sj-jpg-2-cpt-10.1177_10742484231169644 for Ticagrelor Loading on ST-Elevation Myocardial Infarction: Interaction With Prodromal Angina on Infarct Size and Clinical Events by João Pedro Faria, Pedro Oliveira, André Alexandre, David Sá Couto, Ricardo Costa, Andreia Campinas, André Frias, Bruno Brochado, Raquel Santos, João Silveira, Severo Torres and André Luz in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
The datasets generated during and/or analyzed during the current study are not publicly available, but are available from the corresponding author on reasonable request. Informed consents were waived by the Ethics Committee of Centro Hospitalar Universitário do Porto due to the anonymization and retrospective nature of the study. This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Centro Hospitalar Universitário do Porto (2021.291[237-DEFI/245-CE], March 2022).
Author Contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by João Pedro Faria, André Luz, André Alexandre, David Sá Couto, Ricardo Costa, Andreia Campinas and André Frias. Data analysis was performed by João Pedro Faria, André Luz, Pedro Oliveira, Bruno Brochado, Raquel Santos, João Silveira and Severo Torres. The first draft of the manuscript was written by João Pedro Faria and André Luz and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The UMIB is funded by the Foundation of Science and Technology (FCT) Portugal (UIDB/00215/2020; UIDP/00215/2020; LA/P/0064/2020).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
