Abstract
The obesity pandemic is accompanied by increased risk of developing metabolic syndrome (MetS) and related conditions: non-alcoholic fatty liver disease (NAFLD)/non-alcoholic steatohepatitis (NASH), type 2 diabetes mellitus (T2DM) and cardiovascular (CV) disease (CVD). Lifestyle, as well as an imbalance of energy intake/expenditure, genetic predisposition, and epigenetics could lead to a dysmetabolic milieu, which is the cornerstone for the development of cardiometabolic complications. Glucagon-like peptide-1 (GLP-1) receptor agonists (RAs) and dual glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 RAs promote positive effects on most components of the “cardiometabolic continuum” and consequently help reduce the need for polypharmacy. In this review, we highlight the main pathophysiological mechanisms and risk factors (RFs), that could be controlled by GLP-1 and dual GIP/GLP-1 RAs independently or through synergism or differences in their mode of action. We also address the evidence on the use of GLP-1 and dual GIP/GLP-1 RAs in the treatment of obesity, MetS and its related conditions (prediabetes, T2DM and NAFLD/NASH). In conclusion, GLP-1 RAs have already been established for the treatment of T2DM, obesity and cardioprotection in T2DM patients, while dual GIP/GLP-1 RAs appear to have the potential to possibly surpass them for the same indications. However, their use in the prevention of T2DM and the treatment of complex cardiometabolic metabolic diseases, such as NAFLD/NASH or other metabolic disorders, would benefit from more evidence and a thorough clinical patient-centered approach. There is a need to identify those patients in whom the metabolic component predominates, and whether the benefits outweigh any potential harm.
Keywords
Introduction
The pandemic proportions of obesity, defined as “abnormal or excessive fat accumulation that presents a health risk”, have received major attention. 1 Obese patients are at increased risk of developing metabolic syndrome (MetS) and its related conditions: non-alcoholic fatty liver disease (NAFLD)/non-alcoholic steatohepatitis (NASH), type 2 diabetes mellitus (T2DM) and cardiovascular (CV) disease (CVD). 2
MetS includes a cluster of several CV risk factors (RFs) that may lead to CVD and NAFLD-related complications. 3 The overlap between CV and NAFLD-related RFs (e.g., obesity, dyslipidaemia, hypertension, and T2DM), is driving the increase in the worldwide prevalence of NAFLD. 4 The prevalence of NAFLD is >90% in patients undergoing bariatric surgery (severely obese), >76% of T2DM patients, and about 70% among CVD patients, compared with ∼25% for the global general adult population. 5 -7
Genetic predisposition, epigenetics, lifestyle, as well as an imbalance of energy intake/expenditure could lead to a dysmetabolic milieu, which is the cornerstone for the development of both, NAFLD/NASH and CVD. 8 -11 In fact, the considerable impact of dysmetabolism on NAFLD incidence and severity has been widely recognized and led to the proposal of a nomenclature change from NAFLD to Metabolic Associated Fatty Liver Disease (MAFLD). 12,13 Given the similar pathophysiology, there is emerging evidence that medications that can reduce CV morbidity and mortality (as well as reducing RFs for CVD) could also be used for the treatment of NAFLD/NASH. 14 Furthermore, treatment of NAFLD/NASH must extend beyond the liver disease to include CVD risk reduction to reduce CVD mortality. 14
Following the US Food and Drug Administration’s (FDA) requirement, since 2008 every new treatment option for T2DM 15 is subject to major CV outcome trials (CVOTs). 15 Glucagon-like peptide 1 receptor agonists (GLP-1 RAs) represent significant progress in the treatment of T2DM patients because they affect a broad array of CVD RFs through body weight (BW) reduction, 16 lipid level improvement, and positive effects on blood pressure (BP). 17 Due to their CVD risk reduction properties, 18 -23 GLP-1 RAs should be considered in patients with T2DM and possibly NAFLD. 14
Glucose-dependent insulinotropic polypeptide (GIP), the dominant incretin hormone in healthy individuals, also enhances meal-stimulated insulin secretion in a glucose-dependent manner; however, in patients with T2DM insulin secretion after GIP stimulation is diminished. 24 Although GIP acts synergistically with GLP-1 RAs in promoting BW reduction by signaling satiety through receptors in the hypothalamus, there are also differences between the action of GIP and GLP-1 RAs. 25,26 GIP showed no effect on glucagon response to hyperglycemia, while it strongly potentiated insulin secretion. 26 In contrast, during hypoglycemic and fasting conditions, GIP increased glucagon levels without a significant effect on insulin secretion. 26 In addition, dual GIP and GLP-1 RAs showed robust effects on glycemic control and meaningful BW loss, indicating their potential use in the treatment of T2DM. 27 This has also raised the question of the possibility of their use in the treatment of other dysmetabolic conditions such as NAFLD/NASH.
This review will address the evidence on the use of GLP-1 and dual GIP/GLP-1 RAs for the treatment of obesity/MetS, prediabetes, NAFLD, T2DM and CV complications. We also highlight the main pathophysiological mechanisms and RFs that could be controlled by GLP-1 and dual GIP/GLP-1 RAs.
Literature Search
A PubMed electronic database search was conducted, without any limitation by publication date. A focus was placed on clinical studies, especially those published in the last 2 decades. A combination of Medical Subject Heading (MeSH) terms and non-MeSH terms was used, including glucagon-like peptide-1 receptor agonists, glucose-dependent insulinotropic polypeptide agonists, obesity, nonalcoholic fatty liver disease, nonalcoholic steatohepatitis, metabolic syndrome, prediabetes, type 2 diabetes mellitus, pathophysiology, and cardiovascular disease. Reference lists of selected publications were also searched. Since this was a narrative review, some articles, not retrieved during this search strategy, were added at the discretion of the authors if they were considered useful. All selected articles were in English and were peer-reviewed.
Pathophysiology of GLP-1 Receptor (GLP-1R) Agonism in Cardiometabolic RFs
GLP-1 binds to G-protein receptors on target tissues: pancreatic α- and β-cells, central and peripheral nervous system, skeletal muscle, heart, endothelium, kidneys, lungs, digestive system, adipose tissue (AT), lymphocytes, and eyes. 28 Food is the strongest stimulus for GLP-1 secretion. GLP-1 exerts insulinotropic effects through GLP-1R which are dominantly expressed in β pancreatic cells. 29 However, the expression of GLP-1Rs in other organs is responsible for the extrapancreatic actions of GLP-1 and its metabolic effects. 29,30 When released into the blood, GLP-1 exerts several physiological effects (Figure 1): in the pancreas it increases the mass of β-cells and decreases apoptosis, stimulates glucose-dependent insulin secretion, reduces glucose-dependent glucagon secretion and slows gastric emptying, postprandial hyperlipidemia, intestinal motility, and gastric acid secretion. 30 -35

The pleiotropic effects of GLP-1 and GIP. GIP indicates glucose-dependent insulinotropic polypeptide; GLP-1, glucagon-like peptide 1.
The effect of GLP-1 RAs on BW is independent of and distinct from its effect on plasma glucose levels. 36 The mechanisms for reducing BW are inhibition of gastric emptying and induction of satiety. 37 GLP-1 reduces appetite, and food intake, and increases satiety via GLP-1Rs in the hypothalamus. 24,37,38 The action of GLP-1 RA affects meal onset and energy intake, not energy expenditure. 39 The sites of GLP-1R expression within the central nervous system and consequently representative targets for GLP-1 action are neurons in the area postrema, hypothalamus, hindbrain, hippocampus, thalamus, lateral parabrachial nucleus, nucleus accumbens, vagal afferent neurons and nucleus tractus solitaries. 40
It is not completely clear whether BP and lipid profile improvement is a direct consequence of GLP-1 RA treatment, or a result of changes in body composition, BW loss, and/or better glucose regulation. 16 The pathophysiological mechanisms responsible for the antihypertensive effect of GLP-1 RAs have not been fully clarified. Nevertheless, treatment with GLP-1 RAs affects cardiac performance and peripheral vascular resistance, which may partly explain their effect on BP. 17 In addition, GLP-1R in the kidney can induce natriuresis and diuresis in the renal proximal tubule, through inhibition of NHE3 (Na+/H+ exchanger isoform 3). 41 GLP-1R-induced natriuresis and diuresis are also accompanied by increases in glomerular filtration rate and renal plasma flow. 41
Apart from the BW loss and positive effect on glucose regulation, GLP-1 can also influence triglyceride (TG) and total cholesterol (TC) levels. 42 GLP-1R signaling modulates key enzymes of lipid metabolism in the liver and reverses cholesterol transport, reduces hepatic TG content (HTGC) and very low very-low-density lipoprotein (VLDL)-TG production rate from the liver, and impairs hepatocyte de novo lipogenesis and β-oxidation. 42 Short- and long-term treatment with GLP-1 RAs reduced fasting and postprandial lipid levels in T2DM patients as well as in healthy populations. 42 There is also evidence that some GLP-1 RAs can lower the concentration of distinct atherogenic lipoproteins in plasma 43 ; it is of clinical significance that GLP-1 RAs with proven cardiovascular benefit, such as liraglutide, are able to reduce the levels of small, dense LDL, 44,45 since these particles are associated with the formation and progression of atherosclerosis. 46
In the liver, AT, and skeletal muscle cells, GLP-1 increases glucose utilization by stimulating glycogen and lipid synthesis independent of insulin. 47 To date, it is still controversial whether the liver expresses GLP-1Rs, so, the effects of GLP-1 on the liver may be mediated indirectly. Indeed GLP-1 and GLP-1 RAs showed beneficial effects on the liver. 48 Some beneficial liver effects include postprandial GLP-1-mediated insulin secretion and inhibition of hepatic gluconeogenesis indirectly via the gut-pancreas-liver axis. 48 In addition, exendin-4 (a peptide with 39 amino acid residues isolated from the saliva of the Gila monster) or GLP-1 can activate key signaling molecules downstream of insulin receptor substrate-2 (a key substrate for insulin signaling in β-cells), 48 which consequently leads to insulin-like effects.
Numerous CV trials have reported improvements in CV outcomes in patients treated with GLP-1 RAs. 49,50 GLP-1 has pleiotropic CV effects, including anti-inflammatory effects, cardioprotection in ischemia, and decreased platelet aggregation. 38 In addition, activation of GLP-1R resulted in various anti-atherosclerotic effects, e.g., reversing endothelial dysfunction (stimulates nitric oxide [NO] production and endothelial NO synthase [eNOS] activation, eliciting vasorelaxation in arterial endothelium), improving vascular smooth muscle cell dysfunction, reducing macrophage inflammation and foam cell formation, inhibiting NLRP3 (Nucleotide-Binding Domain, Leucine-Rich-Containing Family, Pyrin Domain-Containing-3) inflammasome and improving cardiomyocyte/cardiac fibroblast dysfunction. 51,52 They also influence hypertension through renal and non-renal mechanisms. 30,53 Based on the above mentioned cardiometabolic effects of GLP-1 RAs, these drugs are likely to be increasingly used to treat cardiometabolic disorders, other than DM.
Pathophysiology of GIP Receptor (GIPR) Agonism on Cardiometabolic RFs
GIP exerts important biological effects in many tissues, some of which remain to be explored (Figure 1). In addition to the pancreas, the human GIPR is widely distributed in AT, intestine, bone, and trachea, and is also detected in smaller amounts in the brain, heart, spleen, thymus, blood cells, lungs, and kidneys. 54 In the pancreas, it mediates its insulinotropic effects like GLP-1, but unlike GLP-1, GIP acts as a bifunctional stabilizer of blood glucose and stimulates glucagon secretion at lower glucose concentrations. 55,56 Some effects of GIP differ from GLP-1: under conditions of insulin resistance (IR); it promotes lipid deposition in mice subcutaneous fat cells, it does not affect gastric emptying, and its effect on blood glucose is impaired in chronic hyperglycemia. 24,57 -59
The reports on the role of GIP and GIPR in regulating BW make it an interesting target for addressing obesity. Obesity leads to GIP resistance in AT and a decrease in GIPR, whereas BW loss leads to an increase in GIPR and improves GIP activity. 60 -62 In mice models, GIPR deficiency protects against diet-induced neuronal leptin resistance, a key problem in obesity. 63 A centrally administered antibody that neutralizes GIPR was effective against obesity in obese mice, therefore antagonism of GIP could be a therapeutic approach to prevent obesity. 63 However, the metabolic benefits of GIP antagonism have not yet been tested in humans. 64 Paradoxically, studies have shown that GIPR agonism and antagonism have similar effects on BW and glucose tolerance. 65 The effect of GIP on the hypothalamus and food intake is not clear, but in recent years hybrid molecules have been developed that promote greater BW loss than GLP-1 RAs alone by activating GIPR. 66,67 However, GIP is another incretin that may enhance the metabolic effects of GLP-1 through complementary or synergistic actions. Surprisingly, when GIP was combined with GLP-1 and glucagon, glycemic control in obese mice improved, as did BW loss and reversal of NASH. 67,68
The effect of GIP on arterial hypertension is still under investigation. Patients with peripheral arterial disease have an increased GIP concentration, which correlates with peripheral arterial disease independently of diabetes mellitus (DM), CVD, age, sex, body mass index (BMI), hypertension, and smoking. 69 These observations identified GIP as a counterregulatory vasoprotective peptide. 69
GIP plays an important role in human lipid metabolism. 70,71 Fat is the most potent nutrient that stimulates the release of GIP in humans. 72,73 Results have shown that GIP plays a physiological role in promoting lipid deposition in AT in response to overeating by increasing lipoprotein lipase activity. 74,75 On the other hand, GIP has been shown to reverse the accumulation of TG in AT, liver, and muscle. 76 Interestingly, GIP is a mediator of the increased TG storage in AT that leads to BW gain and obesity. 24,51
The presence of GIPR in the liver is controversial. It is also known that GIP mediates the development of fatty liver in response to the consumption of high glycemic index foods. 77 Studies in animal models suggest that GIP promotes the deposition of lipids in the liver, and GIP release/signaling mediates the development of fatty liver in response to high intake of carbohydrates, particularly sucrose. 78
The role of GIP in CVD is still unclear. 79,80 In vitro, GIP can exhibit both pro-atherogenic (e.g., endothelin-1 production in vascular endothelial cells) and anti-atherogenic properties (enhancement of nitric oxide [NO] production, inhibition of cell proliferation in vascular smooth muscle cells, and suppression of inflammatory processes in adipocytes, macrophages, and monocytes). 81 -83 In animal studies, GIP also increases hormone-sensitive lipase and cardiac TG. 79 Human studies showed that GIP increases heart rate, 84 -87 NO levels, 88,89 interleukin (IL)-6, IL-1β, monocyte chemoattractant protein 1, 90 vascular adhesion molecule-1 in blood and epithelium, 91 and stimulates osteopontin expression in the vasculature via endothelin-1. 92 It could lead to the progression of atherosclerosis in obese humans but prevent plaque formation in mice. 93,94
In addition, in vivo, pharmacological concentrations of GIP are likely to be protective against atherosclerosis in both, DM and non-DM patients. 81 However, the question arises whether GIPR agonism or antagonism has CV benefits in humans. There is a need for further clarification.
Effects of GLP-1 RAs and GIPR agonists on dysmetabolic milieu/diseases are shown in Figure 2.

Effects of GLP-1 RAs and GIPR agonists on dysmetabolic milieu/diseases. CNS indicates central nervous system; CVD, cardiovascular disease; GIP, glucose-dependent insulinotropic polypeptide; GLP-1 RAs, glucagon-like peptide 1 receptor agonists; IR, insulin resistance; MetS, metabolic syndrome; NAFLD, non-alcoholic fatty liver disease; NASH, non-alcoholic steatohepatitis; SAT, subcutaneous adipose tissue; T2DM, type 2 diabetes mellitus.
GLP-1 RAs and Dual GIP/GLP-1 RAs in the Treatment of Non-Diabetic Obesity and Pre-Diabetes—Current Evidence
Obesity is phenotypically diverse, with some proportion of obese individuals who are metabolically healthy, without BP- or lipid-lowering medication, and with no abnormalities in insulin action. 95 However, in most individuals, obesity is closely associated with IR, the key pathophysiological driver of disturbed glucose-metabolism and a CV RF. 96 BW loss has benefits regardless of whether the pathophysiological origin of T2DM is β-cell dysfunction or IR. 97 While insulin-resistant individuals benefit most from BW loss, in patients with a primary defect in β-cell function, BW reduction might reduce β-cell gluco-lipotoxicity and minimize pharmacotherapy requirements. 97
Moderate weight loss of 5% to 10% is encouraged for glycemic and cardiometabolic RF control. 98 However, sustained loss of at least 15% BW has a major effect on the progression of T2DM, inducing remission in a large proportion of patients and markedly improving metabolic status. 99
Achieving the desired BW loss is extremely challenging and pharmacotherapy can be used in this setting. Treatment of obesity based on GLP-1 RAs proved to decrease morbidity and mortality in the adult population. 100 Two agents of the GLP-1 RA class are approved by one or more regulatory authorities worldwide for chronic BW management (liraglutide 3.0 mg subcutaneously (SC) and semaglutide 2.4 mg SC), in individuals without DM. 97 The remaining GLP-1 RA are recognized to promote BW loss while primarily prescribed for their glucose-lowering actions. Only exenatide, liraglutide, and semaglutide have been studied for their role in prediabetes progression in patients with obesity. 101,102 Obesity may also complicate type 1 DM (T1DM) and “obese-induced insulin resistance” in T1DM children could be the main “driver” for NAFLD development. 103
Exenatide and liraglutide have been studied in these patients who also have suboptimal glycemic control or obesity; consistent BW loss was demonstrated. 104 Dual agonists combining GLP1 with GIP are in the pipeline for obesity management and their potential role in T2DM. 105 Due to impressive BW loss in nondiabetic obese, who received tirzepatide, this agent will presumably affect T2DM risk and reversion of prediabetes. 106 The dual GIP/GLP-1 RAs, directly and indirectly, improve white AT function and ectopic lipid accumulation and subsequently whole-body insulin sensitivity. 107
Exenatide
Exenatide, the first approved GLP-1 RA, was developed for the treatment of T2DM. In this category of patients, in addition to its glucose-lowering effects, exenatide has shown significant improvement in many cardiometabolic parameters, including endothelial function and subclinical atherosclerosis. 108 Exenatide was also studied in nondiabetic patients with obesity and patients with prediabetes (with impaired fasting glucose [IFG] or impaired glucose tolerance [IGT]) for its effects on BW and glucose tolerance. Exenatide-treated (10 µg SC with a 4-week 5 µg dose-initiation period) participants lost 5.1 ± 0.5 kg from baseline versus 1.6 ± 0.5 kg with placebo (P < .001), and the placebo-adjusted difference in percent BW reduction was −3.3% ± 0.5% (P < .001). Consequently, after 24 weeks, 77% of patients with prediabetes returned to normoglycemia, compared with 56% of patients treated with placebo. 101 Another study targeted BW loss in nondiabetic women with short exenatide intervention and demonstrated that a subset of women had a robust response to exenatide treatment (30% of subjects who lost ≥5% BW), while the other 30% even gained BW. 109
Liraglutide
Liraglutide was the first GLP-1 RA approved by the FDA and the European Medicines Agency (EMA) specifically for BW loss (ranging from 5%-10% reduction of initial BW). In a network meta-analysis, liraglutide 3.0 mg SC was associated with significant excess BW loss versus placebo at 1 year −5.3 kg (95% confidence interval [CI]: −6.06 to −4.52 kg). 110
The SCALE (Effect of Liraglutide on Body Weight in Non-diabetic Obese Subjects or Overweight Subjects With Co-morbidities) trial aimed to evaluate the efficacy and safety of 3.0 mg of liraglutide administered once-daily SC in overweight or obese adults who did not have DM at baseline. 111 Patients (n = 3731; 56-week duration) treated with liraglutide lost 8.4 ± 7.3 kg, while those in the placebo group lost 2.8 ± 6.5 kg of BW (95% CI: −6.0 to −5.1 kg; P < .001). 111 In addition, fewer patients in the placebo group achieved at least 5% of BW loss from the baseline, compared with patients treated with liraglutide (27.1% vs 63.2%, P < .001). Besides the BW loss of 8.0% ± 6.7%, patients in the liraglutide group had improvements in beta-cell function and insulin sensitivity. Furthermore, T2DM developed in more patients in the placebo group than in the liraglutide group during the course of treatment. 111 One of 4 trials in the SCALE program targeted individuals (n = 2254) with prediabetes with the primary outcome: duration to the onset of DM. Over 160 weeks of treatment with liraglutide showed greater BW loss compared with placebo (−6.1% vs −1.9%, P < .0001), and the time to onset of DM among all randomized individuals was 2.7 times longer with liraglutide than with placebo (95% CI: 1.9-3.9, P < .0001), resulting in a hazard ratio (HR) of 0.21 (95% CI: 0.13-0.34). 112 Only 3% in the liraglutide-treated group developed DM by the end of the trial, compared with 11% in the placebo group. The main finding was highly prevalent prediabetes to normoglycemia conversion in the liraglutide group; 66% versus 36% in the placebo group (P < .0001). 112
In accordance with this finding, an earlier, but a small, double-blind study of liraglutide 1.8 mg SC versus placebo that included older individuals (n = 68) with overweight/obesity and prediabetes, demonstrated that 75% of individuals treated with liraglutide achieved normal fasting glucose compared with 19% on placebo. 113 Peripheral IR, as opposed to hepatic IR, was directly measured (with the modified version of the insulin suppression test) at baseline and after 14 weeks of liraglutide or placebo demonstrated that BW loss achieved with liraglutide was associated with a significant 29% improvement in IR. 113
Semaglutide
Semaglutide, the most recently approved once-weekly (OW) GLP-1 RA, has been associated with pronounced BW loss in combination with dietary and physical activity counseling. 102,114 -116 The STEP (Semaglutide Treatment Effect in People with Obesity) trial program supported a market authorization approval for semaglutide for obesity treatment. 102,115,116
The STEP-1, a double-blind trial (n = 1961 adults), included either patients with obesity or overweight patients with at least one CV RF, who did not have DM. 102 After randomization (in a 2:1 ratio) enrolled patients were treated with semaglutide (2.4 mg, OW SC) or placebo, plus lifestyle intervention, for 68 weeks. Treatment with semaglutide was associated with sustained, clinically relevant −14.9% mean change in BW from baseline. 102 In addition, treatment with semaglutide showed greater BW loss, from baseline to week 68, compared with patients in the placebo group (−15.3 vs −2.6 kg; 95% CI: −13.7 to −11.7). Weight loss of 15% or more of initial BW was achieved in 50.5% of patients treated with semaglutide and 4.9% of patients treated with placebo (P < .001). 102 In the exploratory endpoints, the authors noted that 84.1% of participants in the semaglutide group who had prediabetes at baseline, as compared with 47.8% of participants in the placebo group with prediabetes reverted to normoglycemia. 102
In the STEP-3 trial (n = 611 patients with obesity/overweight plus one additional CV RF, without DM; randomized double blind), treatment with semaglutide (2.4 mg, OW SC) compared with placebo showed significantly greater estimated mean BW change from baseline to week 68 (−16.0% vs −5.7%, 95% CI: −12.0 to −8.6; P < .001). 116 Weight loss of 15% or more of initial BW was achieved by 55.8% of patients treated with semaglutide and 13.2% of patients treated with placebo (P < .001). 116
In the STEP-4 trial, after a 20-week run-in period (semaglutide, 2.4 mg OW SC), Rubino et al 115 demonstrated that maintaining semaglutide treatment compared with switching to placebo showed continued BW reduction during the next 48 weeks (mean BW change −7.9% in the semaglutide group vs +6.9% with the switch to placebo, P < .001).
Daily SC semaglutide (doses from 0.05 to 0.4 mg) versus liraglutide (3.0 mg) and placebo for the treatment of non-diabetic obesity over 52 weeks led to the following estimated mean percentage BW reductions; −6.0% (0.05 mg), −8.6% (0.1 mg), −11.6% (0.2 mg), −11.2% (0.3 mg), and −13.8% (0.4 mg) for the semaglutide group versus −2.3% BW loss for the placebo group. Mean reductions in BW for 0.2 mg or more of semaglutide were all significantly greater than with liraglutide (−13.8% to −11.2% vs −7.8%). Estimated BW loss of ≥10% occurred in 37% to 65% of participants receiving ≥0.1 mg of semaglutide. 114
The STEP 1, 3, and 4 trials 102,115,116 collectively included a large cohort of adults with prediabetes (n = 1536) and provide robust data on the effects of semaglutide on normoglycemia conversion during the 68 weeks of treatment. Among participants with baseline prediabetes, significant improvements in glucose metabolism and a high likelihood of normoglycemia were proven (STEP 1, 84.1%; STEP 3, 89.5%; STEP 4, 89.8%). 117
Tirzepatide
A novel dual GIP/GLP-1 RA, tirzepatide (39-amino acid synthetic peptide), was developed for the treatment of T2DM, obesity/overweight with weight-related comorbidities, NAFLD/NASH, CV complications in T2DM, and heart failure. Its efficiency and safety led, in May 2022, to the first approval in the USA for improving glucose-regulation in T2DM patients. 118(p202) Furthermore, in October 2022, the FDA granted tirzepatide a Fast Track designation for the treatment of obesity and overweight. 119
The SURMOUNT (A Study of Tirzepatide in Participants With Obesity or Overweight) trials were designed to test the ability of tirzepatide to produce BW loss in people who are overweight/obese but do not have DM. Recently, the SURMOUNT-1 trial (n = 2539 obese/overweight patients without DM, randomized double-blind phase 3 trial; duration 72 weeks) was published and showed that treatment with tirzepatide (5, 10, and 15 mg, OW SC) induced substantial and sustained BW loss (the mean percentage change in BW −15.0%, −19.5%, −20.9%, respectively). This was statistically significant when compared with placebo (mean percentage change in BW −3.1%) (P < .001). 106 The percentage of patients who had BW loss of ≥20% was 50% (95% CI: 46-54) in the 10 mg and 57% (95% CI: 53-61) of patients in the 15 mg groups, compared with 3% in patients treated with placebo (P < .001 for all). 106 Furthermore, most of the prediabetes patients (95.3%) at baseline, treated with tirzepatide, had reverted to normoglycemia, when compared with 61.9% of patients treated with placebo. 106 Tirzepatide improved markers of beta-cell function and insulin sensitivity, and analysis of homeostasis model assessment-estimated (HOMA)-IR suggests that only 20% to 30% of the improvement in insulin sensitization by tirzepatide is due to BW loss, suggesting dual receptor agonism confers distinct mechanisms regarding glycemic control. 120
The antiobesity potential of GLP-1 and dual GIP/GLP-1 RAs in obese nondiabetic patients and individuals with prediabetes is summarized in Table 1.
The Antiobesity Potential of GLP-1 and Dual GIP/GLP-1 RAs in Obese Nondiabetic Patients and Individuals With Prediabetes.

Abbreviations: BW, body weight; GIP, glucose-dependent insulinotropic polypeptide; GLP-1 RAs, glucagon-like peptide 1 receptor agonists; SC, subcutaneously; SCALE trial, Effect of Liraglutide on Body Weight in Non-diabetic Obese Subjects or Overweight Subjects With Co-morbidities trial; STEP trial, Semaglutide Treatment Effect in People with Obesity; SURMOUNT-1, A Study of Tirzepatide in Participants With Obesity or Overweight.
Considerations Regarding Potential Clinical Use of GLP-1 and Dual GIP/GLP-1 RAs for the Treatment of Prediabetes in the Obese: Simplicity, Safety, and Tolerability
Since GLP-1 and GIP signaling is impaired in obesity and T2DM, restoring these mechanisms with incretin-based therapies is a reasonable option. Furthermore, the GLP-1 RA proven improvements in β-cell function in both preclinical studies and short-term clinical studies are promising for individuals with prediabetes. However, since T2DM prevention can be achieved by intensive lifestyle modification alone 122 and since lifestyle modifications provide better long-term effects than medications, 123 tolerability and safety profile of potential pharmacotherapy should be thoroughly considered before clinical use. Several pharmacologic interventions besides GLP-1 RA have been shown to lower the incidence of T2DM in specific populations: metformin, 122 acarbose, 124 rosiglitazone, 125 insulin glargine 126 and testosterone. 127 Up to now, no pharmacologic agent has been approved by the FDA specifically for T2DM prevention. However the use of metformin is encouraged in specific high risk populations by the American Diabetes Association. 98
A Cochrane database systematic review investigating the prevention or delay of T2DM with GLP-1 RA use included 7 trials with 2702 participants at increased risk for developing T2DM. 128 The analysis found limited-quality evidence that at-risk patients taking GLP-1 RAs are less likely to progress to DM (number needed to treat (NNT) = 23). More individuals who received GLP-1 RA had serious adverse events than those who received placebo, 15.1% versus 12.7% (RR 1.18, 95% CI 0.94-1.47); absolute risk increase = 2.4%; number needed to harm (NNH) = 42. No comparisons among the different GLP-1 RA could be made because of the small number of participants for some GLP-1 RA class members. 128 The potential of a DM prevention intervention lies predominantly in the BW loss achieved by individuals at risk of T2DM. 129 It is also likely that some sub-phenotypes of individuals with prediabetes may benefit more of GLP-1 RAs rather than other therapeutic agents, and this highlights the importance of a more tailored approach for screening, prevention and management. 130 Therefore, further long-term comparative studies of interventions with novel potent anti-obesity medications of the GLP-1 RA class are needed to identify any differences in intervention effects. 123
GLP-1 RA for Weight Control in DM Patients
All GLP-1 RA class agents have proven efficacy in both, glucose control and concurrent clinically relevant BW reduction in patients with T2DM. 131 However, patients with T2DM have more difficulty in losing BW compared with individuals without DM, 132 and the mean BW loss difference between GLP-1 RAs and placebo in patients with and without DM was 4% to 6.2% versus 6.1% to 17.4%, respectively. 132
Trujillo et al 133 rated the drug members according to BW lowering potency in the following order: semaglutide > liraglutide > dulaglutide > exenatide extended-release = exenatide (twice daily) = lixisenatide. In addition, a meta-analysis rated SC semaglutide as the most potent in reducing BW, followed by oral semaglutide, exenatide twice daily and liraglutide, in patients with T2DM. 134
Liraglutide
In the SCALE Diabetes, among patients with T2DM who are overweight/obese, liraglutide treatment led to a more significant BW reduction, 6.0% (3.0 mg SC daily) and 4.7% (1.8 mg SC daily) when compared with placebo (estimated treatment difference [ETD]: −4.00% and −2.71%; P < .001 for both). 135 BW reduction 10% or more occurred in 25.2% and 15.9% versus 6.7% of patients treated with liraglutide 3.0 mg and 1.8 mg SC versus placebo (P < .001 and P = .006, respectively). 135 Furthermore, in the SCALE Insulin randomized controlled trial (n = 396 T2DM patients with obesity treated with insulin) liraglutide (3 mg SC daily) as an adjunct to intensive behavioral therapy induced greater BW reduction when compared with placebo (mean BW change: −5.8% vs −1.5%, ETD: −4.3% [95% CI −5.5 to −3.2]; P < .0001). 121
Semaglutide
In the STEP-2 trial (n = 1210 patients with T2DM, phase 3, double-dummy and double-blind superiority study, BMI ≥27 kg/m2) treatment with semaglutide (2.4 mg OW SC), compared with placebo, resulted in 6.2% ETD in mean BW loss (P < .0001). 136 In addition, after 68 weeks of the treatment, 68.8% of patients in the semaglutide (2.4 mg) group achieved BW reduction of ≥5%, compared with 28.5% of patients in the placebo group (odds ratio [OR]: 4.88, P < .0001). 136 It is important to mention that patients treated with insulin were excluded from the STEP-2 trial. 136
In the SUSTAIN-7 trial, semaglutide (1.0 mg OW SC) showed a significantly higher potency for BW reduction compared with dulaglutide (1.5 mg OW SC) in patients with T2DM (6.5 vs 3.0 kg, ETD −3.55 kg; P < .0001). 137
Tirzepatide
Tirzepatide is being investigated in the SURPASS (A Study of Tirzepatide in Participants With Type 2 Diabetes Not Controlled With Diet and Exercise Alone) phase 3 clinical trial program. 138 A 5 mg dose led to 7.6 kg (8.2%) BW loss, a 10 mg dose led to 9.3 kg (9.3%) and a 15 mg dose led to 11.2 kg (11.9%) BW loss over 40 weeks, in the SURPASS-2 study (n = 1879), and the reductions in BW were dose-dependent. 139 Reductions in BW were greater with tirzepatide at all doses (5, 10, or 15 mg, OW SC) than with semaglutide at a dose of 1 mg, with ETD −1.9, −3.6, and −5.5 kg, respectively (P < .001 for all). 139 In addition, approximately 8% of patients treated with semaglutide (1 mg OW SC) achieved a BW loss of ≥15%, compared with 15%, 24%, and 36% of patients treated with tirzepatide at doses of 5, 10, and 15 mg, respectively. 139
The SURMOUNT-2 trial, designed to test the ability of tirzepatide to produce BW reduction in participants with obesity who also have T2DM, is ongoing and is expected to be published in the first half of 2023.
Other Components of MetS: BP and Lipid Levels
The importance of CV RF is best illustrated by the fact that a 10 mmHg decrease in systolic BP reduces CVD risk by 11%, 140 while a 1 mmol/L lowering in low-density lipoprotein cholesterol (LDL-C) reduces the annual rate of CVD by approximately 20% in very diverse groups of patients. 141
Although determining the isolated (independent of glycemic and BW reduction or changes in body composition) effects of GLP-1 RA on lipid levels and BP, requires well-designed studies, expert opinions confirm positive, but moderate effects on lipid levels and BP. 17 According to a 2018 narrative review of head-to-head trials of 6 GLP-1 RA, 142 the greatest statistical difference in BP was 2.7 mmHg (liraglutide resulted in a significantly greater decrease from baseline in systolic BP compared with dulaglutide). 143 Semaglutide 1.0 mg SC once weekly (OW) also significantly decreased systolic BP compared with exenatide 2 mg SC OW with a difference of 2.4 mmHg. 144 For diastolic BP, no significant between-treatment differences were reported in any of the included trials. 142 The most recent GLP-1 RA, oral semaglutide (that was not included in the previously mentioned review) demonstrated reductions of systolic BP (−2 [−5 to 0] mmHg; P = .07) compared with placebo. 145
Several large as well did some smaller dedicated GLP-1 RA clinical trials collected lipid data as secondary outcomes proving favorable effects on lipoprotein metabolism, with modest LDL-C, total cholesterol (TC), and fasting TG lowering. Some have also demonstrated substantial blunting of the postprandial rise in serum TGs 146 Postprandial hypertriglyceridemia is a potential risk predictor of atherosclerotic CVD even in the presence of normal fasting TG levels. 147,148 Its relevance is further enhanced because of the assumption that postprandial rise in serum TGs is a contributor to residual vascular risk despite aggressive LDL-C lowering. 147,148 Notably, in a real-world prospective study involving obese T2DM patients, the reduction in triglyceride levels by liraglutide was the main contributor of the reduction in subclinical atherosclerosis, highlighting the importance of the triglycerides-lowering effect by GLP-1 RAs for vascular risk reduction. 149
One of the first double-blinded, randomized, placebo-controlled, crossover studies examining the lipid-lowering effect of GLP-1 RA was conducted in 35 subjects with IGT or recent-onset T2DM. It demonstrated the effect of a single SC injection of exenatide on postprandial lipids, remnant lipoproteins, and apolipoproteins. Exenatide reduced postprandial lipid and lipoprotein levels without the reduction in BW or fasting glucose and TG levels that usually occur with prolonged therapy. 150 In a recent first randomized trial, that addressed the possible effects of antidiabetic interventional drugs such as GLP1 RAs on endothelial and arterial stiffness indexes as surrogate markers of vascular damage, subjects treated with dulaglutide showed significantly lower values of mean serum TC and LDL-C. 151
Oral semaglutide at the highest dose improves the fasting lipid level profile compared with placebo; it demonstrated a modest, but superior reduction in TC, LDL-C, and most pronounced reduction (OR 0.90 [0.83 to 0.99] in triacylglycerols (oral semaglutide 14 mg). 145 Compared with another GLP-1 RA (liraglutide), oral semaglutide was not significantly different. 145,152 A randomized clinical trial is currently investigating the direct and indirect anti-atherosclerotic mechanisms of oral semaglutide, including its effect on the full spectrum of plasma lipids and lipoproteins. 153
Tirzepatide and any further dual GLP-1/GIP RAs that could be developed seem to be promising options for lipoprotein profile improvement in patients with T2DM. Tirzepatide dose-dependently decreased TG over time compared with placebo and dulaglutide, tirzepatide 5, 10, and 15 mg decreased TG levels by 28.8%, 37.7%, and 41.4%, respectively, compared with placebo. 154 It is speculated that the relative treatment effect would not be different regardless of the participant’s baseline CV risk level, since similar changes in CV surrogate markers (BP and lipoproteins) by tirzepatide were observed in studies, that included participants with either high or moderate CV risk. 155 The SURMOUNT-1 trial demonstrated that in nondiabetic obese treatment with tirzepatide (5, 10, and 15 mg, OW SC) induced substantial placebo subtracted change in systolic and diastolic BP (−6.2 and −4.0 mmHg, accordingly) and −20.3% change in TG level. 106 The reductions in BP and lipid levels of GLP-1 RA and dual incretin agonists cannot be directly translated into CV outcomes and seem to be complementary to other protective pleiotropic actions of this drug class.
GLP-1 RAs in the Treatment of NAFLD/NASH
The association of NAFLD and NASH with obesity and T2DM encouraged the investigation of the effect of GLP-1RAs in animal and human models of NAFLD/NASH. 156,157 The fact that GLP-1RAs lead to improvement in hepatic steatosis through weight loss has been reported. 158,159 However, experimental evidence based on both, animal models and human hepatocytes, have shown that GLP-1RAs may also play a direct role in ameliorating hepatic steatosis (by reducing de novo lipogenesis), mediated by the activation of genes associated with insulin sensitivity and fatty acid β-oxidation and improving several elements of the insulin signaling pathways. 88,158,160,161 In patients with NAFLD/NASH, glucose-induced GLP-1 secretion is deficient, while GIP secretion is preserved. 162
The beneficial effects of GLP-1 RAs on the individual histological scores of NASH are multifactorial, due to the combined indirect effects on IR/hyperglycemia and BW reduction, as well as the result of a direct beneficial effect on the liver (regardless of weight loss and hyperglycemia). 163 These direct effects are fundamental for the establishment of GLP-1 RAs as a treatment option for NAFLD/NASH in patients in whom the fatty liver disease is not mainly driven by obesity. 164
Nevertheless, the presence or absence of GLP-1R in hepatocytes is controversial. 34,160,165,166 GLP-1 increases glucose utilization in the liver, skeletal muscle, and AT by inducing glycogen and lipid synthesis independently of insulin level. 47 On the other hand, GLP-1RAs can stimulate lipolysis, AT browning, and pre-adipocyte differentiation. 34 Liraglutide and exenatide showed antioxidant effects via the c-Jun N-terminal kinase pathway or upregulation of protective antioxidant enzymes. 161 GLP-1RAs attenuate endoplasmic reticulum stress responses by suppressing apoptosis. 167 In addition, GLP-1 RA exenatide attenuated fatty liver by activating sirtuin 1, an upstream regulator of adenosine monophosphate-activated protein kinase in hepatocytes, supporting the rationale for GLP-1-based NAFLD/NASH therapy. 168 Preclinical studies that recruited patients with NASH suggested that GLP-1 RAs might reduce hepatic inflammation through mechanisms that are at least partly independent of weight loss. 156 Mechanistically, as hepatocytes, stellate cells, and Kupffer cells do not express the canonical GLP-1R, the actions of GLP-1 on the liver are primarily indirect.
Exenatide may be an effective treatment for reducing hepatic fat content in patients with T2DM and obesity, but these effects are mainly dependent on BW reduction. 169 In a study (n = 44 patients with T2DM and obesity, HTGC was assessed with MRI and proton MRI spectroscopy, at baseline and 26 weeks after). In patients treated with exenatide, HTGC reduction was significantly related to body weight reduction (r = 0.47, P = .03), but was not associated with an improvement in IR, glycated hemoglobin level or adiponectin. 169
Armstrong et al in a randomized, double-blinded, multicenter, placebo-controlled phase 2 study evaluated SC injections of liraglutide (1.8 mg daily) versus placebo in patients who are overweight and showed clinical evidence of NASH. 170 Treatment with liraglutide led to histological resolution of NASH and was well tolerated and safe. After treatment, 39% of patients who were treated with liraglutide and underwent end-of-treatment liver biopsy had resolution of definite NASH versus 9% of patients treated with placebo (relative risk [RR] 4.3, 95% CI: 1.0-17.7; P = .019). 170 However, the LEAN (Liraglutide Efficacy and Action in NASH) trial suggested that liraglutide should be started early during the course of NAFLD, since in NASH patients with advanced fibrosis, it might not be useful. 170 In women (n = 72) with polycystic ovary syndrome (a well-known IR state), treatment with liraglutide promoted BW loss (−5.2 kg [5.6%]), reduced liver fat content (LFC) (by 44%) and showed reduction of the NAFLD prevalence by two-thirds (P < .01 for all). 171
Katsiki et al in 2017 emphasized the need for investigation of the effects of semaglutide in patients with NAFLD. 172 In a randomized, double-blind, placebo-controlled trial, liver stiffness was evaluated by MRI elastography and LFC by MRI proton density fat fraction [MRI-PDFF]); treatment with semaglutide significantly reduced LFC compared with placebo (P < .0001) but did not change liver stiffness in patients with NAFLD. 173 In patients with T2DM and obesity, semaglutide significantly reduced ALT activity and high-sensitivity C-reactive protein (hsCRP) levels. 174 Summarized data collected from CV and weight outcome trials showed that at the end-of-treatment ALT activity reductions were 6% to 21% (P < .05 for doses ≥0.2 mg/day). 174 The results of the meta-analysis showed that semaglutide, when compared with other GLP-1RAs liraglutide and dulaglutide was superior in decreasing the AST activity. 175 Newsome et al 176 evaluated the effects of semaglutide treatment in the resolution of NASH without worsening of fibrosis (n = 320 patients with biopsy-proven NASH and liver fibrosis of stage F1, F2, or F3). NASH resolution was achieved in 40%, 26% and 59% in the semaglutide 0.1-, 0.2-, and 0.4 mg groups versus 17% in the placebo group (P < .001 for semaglutide 0.4 mg SC vs placebo). 176
Treatment with dulaglutide improved AST, ALT, and gamma-glutamyl transferase (GGT) activity when compared with placebo, in a pattern consistent with LFC reductions. 177 Kuchay et al 178 in their study (n = 64 patients with T2DM and MRI-derived proton density fat fraction-assessed LFC of ≥6.0% at baseline) evaluated the effects of dulaglutide treatment (OW for 24 weeks) on LFC. Treatment with dulaglutide significantly reduced GGT levels (mean between-group difference −13.1 U/l, P = .025) compared with controls, but there were no significant reductions in ALT, AST activity and change in liver stiffness measurement (LSM in kPa) (P = .10, P = .075, and P = .123, respectively). 178 In addition, in patients with NAFLD dulaglutide significantly reduced LFC (control-corrected absolute change in LFC −3.5%, P = .025; relative change of −26.4%, P = .004), corresponding to a 2.6-fold greater reduction compared with controls. 178
Recent meta-analyses 163,179 -181 reported that GLP-1 RAs (mostly semaglutide and liraglutide) are highly promising pharmacological agents in the treatment of NAFLD or NASH. In the meta-analysis by Mantovani et al (n = 2597 individuals, 10 studies, included T2DM patients treated with GLP-1 RAs) treatment with semaglutide or liraglutide, as well as treatment with pioglitazone promoted resolution of NASH without worsening of fibrosis. GLP-1 RA treatment improved some of the NASH histological features, such as steatosis, lobular inflammation or ballooning. 181 In another meta-analysis (n = 936 patients, 11 studies), Mantovani et al 163 reported that treatment with GLP-1 RAs, when compared with reference treatment or placebo, for a median of 26 weeks significantly decreased ALT (pooled weighted mean difference [WMD] −7.21 IU/L, P = .02) and GGT activity (pooled WMD −0.97 IU/L, P < .001), while AST activity did not differ between groups (P = .27). 163 More importantly, there was a significant reduction in the absolute percentage of LFC (using MRI-based techniques) (pooled WMD −3.92%, 95% CI: −6.27% to −1.56%) and greater histological resolution of NASH without worsening of liver fibrosis (pooled random-effects odds ratio [OR] 4.06, 95% CI: 2.52-6.55; for liraglutide and semaglutide only). 163 GLP1-RAs improve hepatic function, histology, and inflammation, by body weight reduction, improving glycemia, and decreasing hepatic fat. In this context, Ghosal et al 179 in their meta-analysis (n = 615 patients, 297 on GLP1-RA and 318 in the control group, 8 randomized controlled trials) evaluated effects of GLP-1 RAs in patients with both, T2DM and NAFLD. Treatment with GLP-1 RAs showed a significant improvement in AST activity (standardized mean difference [SDM]: −0.44, 95% CI: −0.64 to −0.24, P < .01), ALT (SDM −0.56, 95% CI: −0.88 to −0.25, P < .01), and GGT activity (SDM −0.60, 95% CI: −0.86 to −0.34, P < .01). As expected, GLP-1 RAs treatment showed a decrease in BW (SDM −0.66, 95% CI: −0.88 to −0.44, P < .01), glycosylated hemoglobin (SDM −0.40, 95% CI: −0.61 to −0.19, P < .01), as well as the reduction in LFC (SDM −0.43, 95% CI: −0.74 to −0.12, P < .01), compared with standard of care or placebo. This meta-analysis also showed a very significant improvement in liver histology in the group of patients treated with GLP-1 RAs (rate ratio, 6.60, 95% CI: 2.67-16.29, P < .01). 179
GLP-1 RAs demonstrated higher efficacy in LFC lowering when compared with insulin-based therapies or metformin. 180,182 In a meta-analysis (n = 1454 patients, 8 studies), treatment with GLP-1 RAs significantly reduced LFC (SMD −1.05; P < .001) when compared with controls, and when compared with insulin-based therapies and metformin (SMD −0.66, P < .001 and −0.63, P = .02, respectively). 180 In addition, GLP-1RA significantly reduced ALT (SMD −1.69, P < .001), AST (SMD −1.46, P < .001), and GGT (SMD −2.10; P < .001) activity. 180 When compared with insulin-based therapies, treatment with GLP-1 RAs resulted in significantly lower ALT activity (SMD −0.96; P = .02), and when compared with metformin treatment with GLP-1 RAs resulted in significant decreases in GGT and ALT activity (SMD −1.04; P < .001 and SMD −0.66; P = .03, respectively). 180 However, thiazolidinediones were superior when compared with GLP-1 RAs in the improvement in ALT, AST, and GGT activity. 180 When added to metformin, liraglutide reduced intrahepatic lipid content, promoted weight loss and reduced subclinical atherosclerosis (carotid intima-media thickness) in patients with NAFLD and T2DM. 183,184 Furthermore, when added to pioglitazone, liraglutide and semaglutide were considered the most effective intervention for achieving NASH resolution. 185,186
Dual GIP/GLP-1 RAs in the Treatment of NAFLD/NASH
In patients with NASH, ingestion of saturated fatty acids is associated with prolonged GIP response, ß glucometabolic parameters and carotid intima-media thickness-cell dysfunction, and with adipokine imbalance, and liver injury, which favors GIP antagonism in non-diabetic patients with NASH. 187 In addition, a doubling in fasting GIP levels may be associated with improved LDL clearance but with an unhealthy fat distribution independent of insulin levels. 188 These effects of GIP have not encouraged the further use and establishment of GIP monotherapy in metabolic-associated diseases, including NAFLD/NASH.
GIP and GLP-1 appear to activate distinct subsets of hypothalamic neurons coupled to a reduction in food intake, 189 and furthermore, the glucose-lowering effects of simultaneous GLP-1R agonism could rapidly restore GIP sensitivity, thus enabling superior glucose-lowering action when combined. 190 In patients with T2DM, higher tirzepatide doses significantly increased adiponectin levels and decreased NASH-related biomarkers. 191 Hartman et al 192 in their phase 2 trial (n = 316 T2DM patients, 26-weeks duration, treated with dulaglutide (1.5 mg SC), tirzepatide OW [1, 5, 10, or 15 mg SC] or placebo) evaluated the effect of tirzepatide on AST and ALT activity as well asadiponectin levels. Treatment with tirzepatide significantly decreased ALT (all tirzepatide doses, P < .01) and AST (all tirzepatide doses except 10 mg, P < .05) activity at 26 weeks. In addition, at 26 weeks of treatment, adiponectin increased significantly from baseline with tirzepatide 10 and 15 mg (P < .001). 192
In a recent substudy of the SURPASS-3 trial (SURPASS-3 MRI study; n = 296, randomized, open-label, parallel-group, phase 3, T2DM patients, BMI ≥25 kg/m2, evaluated change in LFC measured by MRI-PDFF, 52-week duration), pooled tirzepatide (10 and 15 mg doses, OW SC) promoted a significant reduction in LFC, visceral AT when compared with insulin degludec (−8.09% vs −3.38%), with the ETD versus degludec −4.71% (95% CI: −6.72 to −2.70; P < .0001). 193
The potential of GLP-1 and dual GIP/GLP-1 RAs in the treatment of NAFLD/NASH is summarized in Table 2.
The GLP-1 and Dual GIP/GLP-1 RAs in the Treatment of NAFLD/NASH.
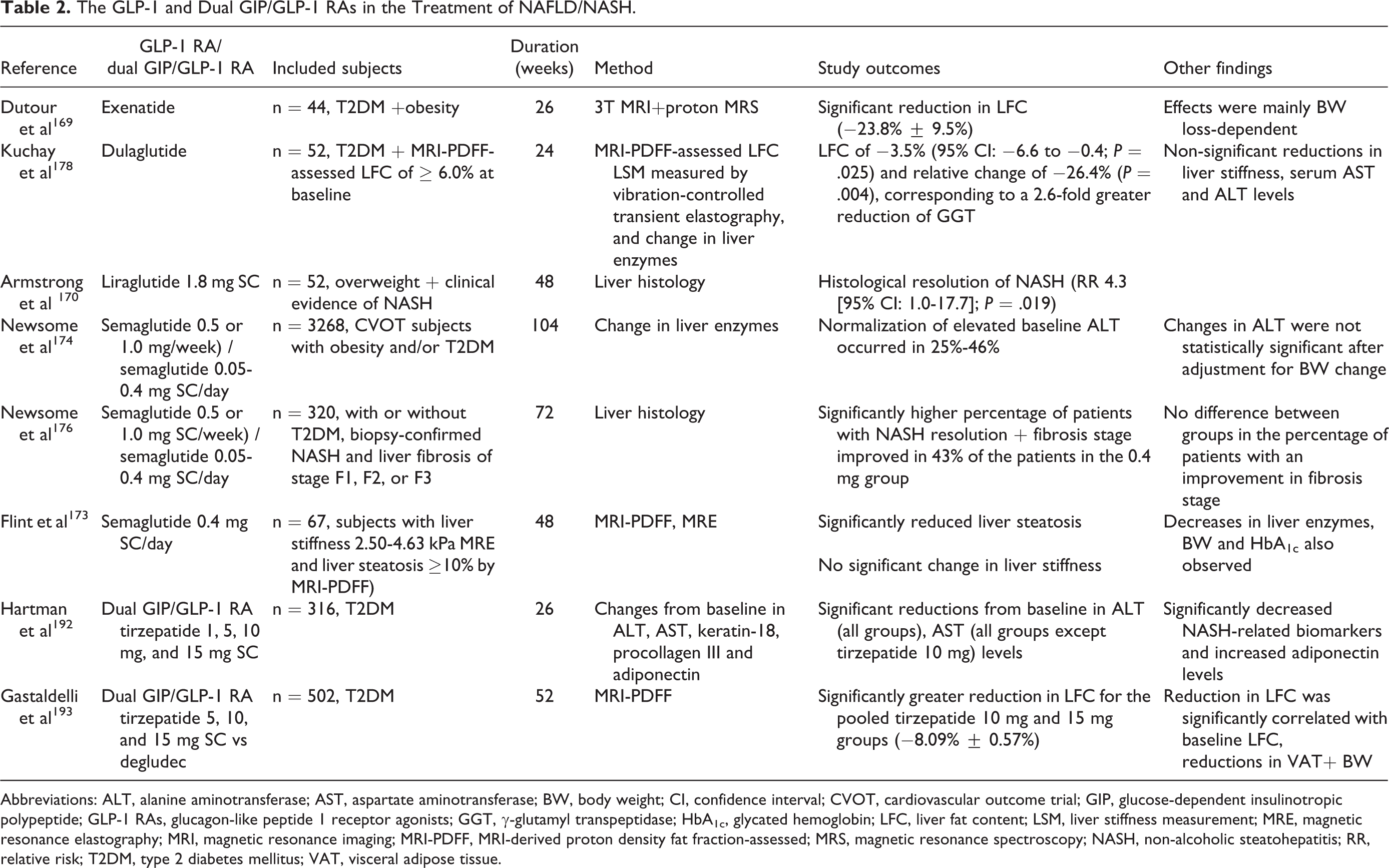
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; BW, body weight; CI, confidence interval; CVOT, cardiovascular outcome trial; GIP, glucose-dependent insulinotropic polypeptide; GLP-1 RAs, glucagon-like peptide 1 receptor agonists; GGT, γ-glutamyl transpeptidase; HbA1c, glycated hemoglobin; LFC, liver fat content; LSM, liver stiffness measurement; MRE, magnetic resonance elastography; MRI, magnetic resonance imaging; MRI-PDFF, MRI-derived proton density fat fraction-assessed; MRS, magnetic resonance spectroscopy; NASH, non-alcoholic steatohepatitis; RR, relative risk; T2DM, type 2 diabetes mellitus; VAT, visceral adipose tissue.
Current Evidence on GLP-1 RAs Use in Patients With DM and CVOTs
The risk of CVD is increased 2-fold in people with DM regardless of other conventional RFs. 194 In the last decade, the CV safety of drugs to lower blood glucose in T2DM has been questioned, such that any drug should preclude increased CV risk. 15 Since then, several CVOTs have been conducted. Either a neutral effect or a reduction in CV events has been reported for the different GLP-1 RAs 18 -21,36,195,196 and, overall, a beneficial reduction in the risk for the distinct cardio-renal endpoints has been obtained by the analysis of available data. 197
Indeed, nine published CVOTs studies examined the effects of GLP-1 RAs compared with placebo on major adverse CV events (MACE) incidence in T2DM patients: (1) Lixisenatide 20 μg/day SC (the Evaluation of Lixisenatide in Acute Coronary Syndrome [ELIXA] trial; n = 6068; duration 2.1 years; 100% prevalence of CVD), 195 (2) Liraglutide 1.8 mg SC/day (Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results [LEADER]; n = 9340; duration 3.8 years, 81% prevalence of CVD), 18 (3) Exenatide ER 2 mg SC /week (Exenatide Study of Cardiovascular Event Lowering Trial [EXSCEL]; n = 14752; duration 3.2 years, 73% prevalence of CVD), 196 (4) Albiglutide 30 to 50 mg SC/week (HARMONY; n = 9463; duration 1.6 years, 100% prevalence of CVD), 36 (5) Dulaglutide 1.5 mg SC/week (Researching Cardiovascular Events With a Weekly Incretin in Diabetes [REWIND] n = 9901; duration 5.4 years, 31% prevalence of CVD), 20 (6) Semaglutide 0.5 to 1 mg SC/week (Semaglutide unabated Sustainability in Treatment of Type 2 Diabetes [SUSTAIN]-6 n = 3297; duration 2.1 years; 83% prevalence of CVD), 19 and (7) Oral semaglutide 14 mg/day (Peptide Innovation for Early Diabetes Treatment [PIONEER-6] n = 3181; duration 1.3 years; 85% prevalence of CVD). 21 Furthermore, [AMPLITUDE-O] (Effect of Efpeglenatide on Cardiovascular Outcomes) n = 4076; duration 1.8 years) included more patients (32%) with renal disease (eGFR 25-60 mL/min) than the other trials and was the first clearly positive CVOT with an exendin-4-based GLP-1 receptor agonist. 198
The FREEDOM (Clinical Impact of ITCA 650 [an injection-free preparation delivering exenatide via a mini osmotic pump placed in the subdermal tissue of the abdominal wall], a Novel Drug-Device GLP-1 Receptor Agonist, in Uncontrolled Type 2 Diabetes and Very High Baseline HbA1c; n = 4,156, duration 16 months) studies evaluated exenatide delivered via an implanted minipump (ITCA 650) in patients with T2DM. ITCA 650 was non-inferior to placebo in CV outcomes, however longer-duration CVOT is needed to more precisely define the CV effects. 199 Trials that reported a reduced HR for the primary composite CV endpoint of first occurrence of nonfatal myocardial infarction or stroke or death from CV causes were LEADER (HR 0.87, 95% CI: 0.78-0.97), SUSTAIN-6 (HR 0.74, 95% CI: 0.58-0.95), HARMONY (HR 0.78, 95% CI: 0.68-0.90), and REWIND (HR 0.88, 95% CI: 0.79-0.99). Lixisenatide (ELIXA), exenatide (EXSCEL), and oral semaglutide (PIONEER) were non-inferior to placebo on the primary composite outcome of MACE (respectively, 1.02, 95% CI: 0.89-1.17; 0-91, 95% CI: 0.83-1.00; 0.79, 95% CI: 0.57-1.11).
CVOT for tirzepatide (SURPASS-CVOT) is ongoing. However, Sattar et al 155 recently published a pre-specified CV meta-analysis of all 7 randomized controlled trials (n = 144 T2DM patients) from the SURPASS trial program. The HRs comparing tirzepatide versus controls were 0.90 (95% CI: 0.50-1.61) for CV death, 0.80 (95% CI: 0.57-1.11) for MACE-4 and 0.80 (95% CI: 0.51-1.25) for all-cause death. 155
A recent meta-analysis of 7 CVOTs 49 showed that GLP-1 RAs reduced MACE by 12% (HR 0.88; P < .001), CV mortality by 12% (HR 0.88; P = .003), fatal or nonfatal stroke by 16% (HR 0.84; P < .001), and fatal or nonfatal myocardial infarction by 9% (HR 0.91; P = .043). Another meta-analysis of the same 7 CVOTs found that the number of treatments required to prevent MACE was 73 (95% CI: 45-212). 200
Expert Opinion
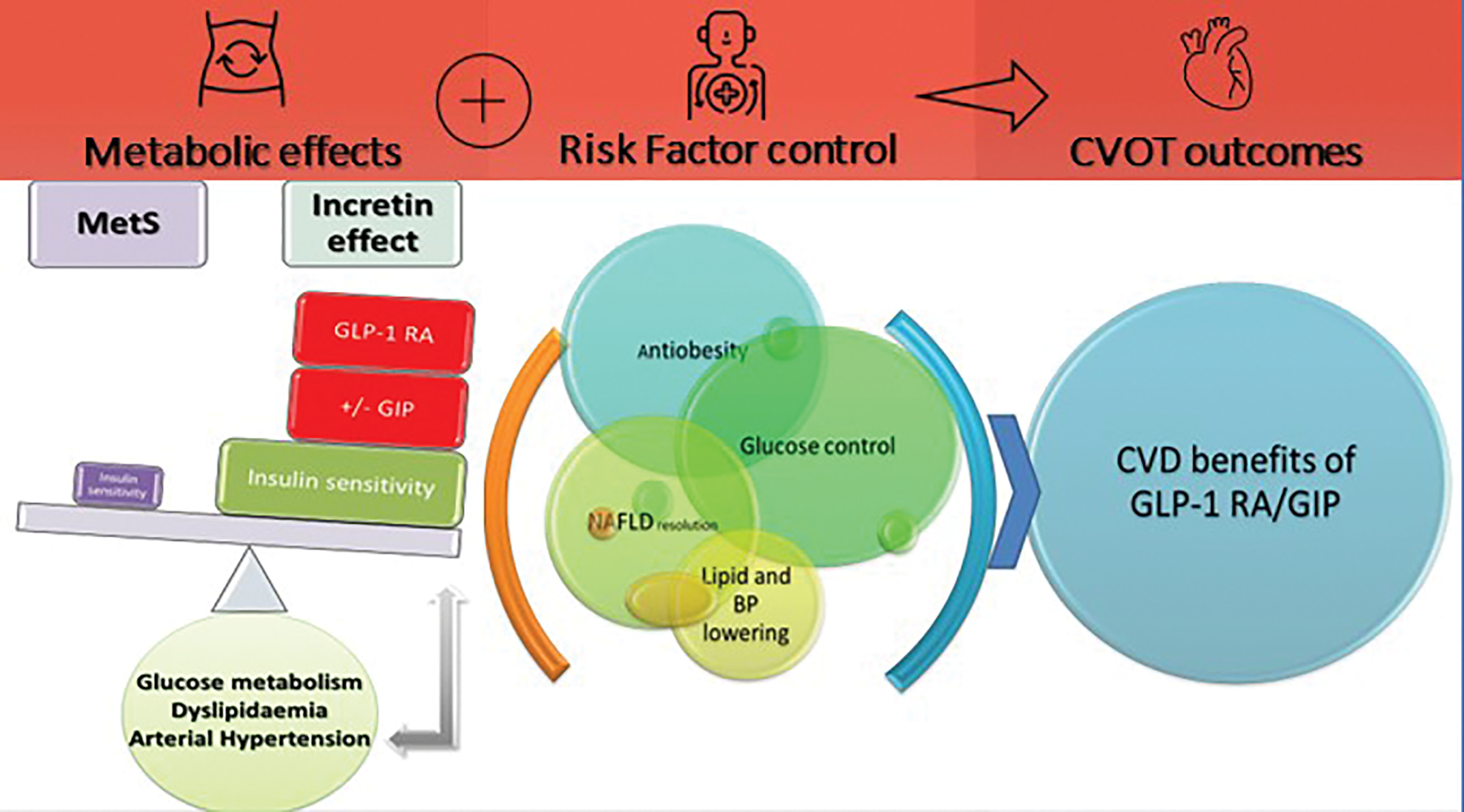
In conclusion, the well-established GLP-1 RAs and novel dual GIP/GLP-1 RAs are drug classes with various effects that cover a wide range of “dysmetabolic milieu” disorders associated with obesity. Given the clear association of obesity with MetS and its components, prediabetes, T2DM, cardiometabolic complications, and NAFLD/NASH, GLP-1 RAs and dual GLP-1/GIP RAs promote crucial positive effects on the most components of the “cardiometabolic continuum” and consequently help reduce the need for polypharmacy.
Clinical evidence from well-designed trials and real-life data indicates that currently, the GLP-1 RAs are the most promising drugs for the treatment of obesity. Moreover, the novel GLP-1/GIP RA tirzepatide shows even greater potential for weight reduction. This finding is in line with the GIPR agonism potential for ameliorating the GLP-1 RA dose-related gastrointestinal side effects and the proposed synergy of the incretins to promote satiety. Their combined weight-related potential is translated into the improvement of components of the MetS (e.g., dyslipidaemia, hypertension, reduction of visceral AT, etc.), which are also CV RFs. The postulated BW-independent effects of GIP RA on lipid metabolism suggest that the dual agonists play an important role in improving the lipoprotein profile.
It is well-known that GLP-1 RAs are the most potent non-insulin drugs for the treatment of T2DM, with currently limited use in terms of the treatment of prediabetes and the prevention of T2DM. With few exceptions, 201 there is a large lack of head-to-head trials with the use of 2 GLP-1 RAs as direct comparators, since in any trial the comparator to the studied agent is the so-called “standard of care”, and this limits the utility of information for clinicians. Although direct comparisons are not possible since differences in study designs exist, considering normoglycemia conversion rates were approximately 66% with liraglutide, 84% with semaglutide, and >95% with tirzepatide. Furthermore, tirzepatide demonstrated restoration of insulin sensitivity irrespective of the BW loss magnitude. It is irrefutable and clear that an adequate dietary regimen is still the cornerstone of T2DM prevention and prediabetes treatment, with possible additional and favorable use of both, GLP-1 and dual GLP-1/GIP RAs after assessing the benefits and risks for each patient individually. The results of the registration studies of GLP-1/GIP RA tirzepatide indicate robust improvements in glycemic control in patients with T2DM, without increased risk of hypoglycemia, with the safety profile which was consistent with GLP-1 RAs. Comparatively, these results are likely to pave the way for tirzepatide in clinical practice guidelines and its incorporation in the treatment algorithm, as one of the initial treatment options.
Understanding and positioning of GLP-1 and GLP-1/GIP RAs in the treatment of NAFLD/NASH is complex. It is important to emphasize that the pathogenesis of NAFLD/NASH can be explained in part by a genetic component, and in part by a metabolic component. The mechanisms underlying these components are fundamentally different. Metabolic components are characterized by the hepatic oversupply of substrates (e.g., sugars, lipids, and amino acids), while the genetic component is characterized by impaired hepatic mitochondrial function, making the liver less able to metabolize these substrates. This leads to the conclusion that the use of GLP-1 and dual GLP-1/GIP RAs requires more evidence and a thorough clinical patient-centered approach, intending to identify those patients in whom the metabolic component predominates. Given the complex etiology of NAFLD, future studies require better design to avoid the various biases that cloud this field and to pave the way for the right use of GLP-1 and GLP-1/GIP RA in the treatment of NAFLD/NASH and other “dysmetabolism” complications.
The place of GLP-1 RAs in CV diabetology is clearly and unequivocally expressed by the guidelines for the treatment of T2DM, as a crucial and most important drug class in patients with atherosclerotic CVD, which was clearly shown through published GLP-1 RAs CVOTs. 202 Moreover, CVOT for tirzepatide (SURPASS-CVOT) is ongoing and its results are expected soon.
Footnotes
Author Contributions
Emir M. Muzurović contributed to the conception and design, wrote the manuscript, contributed to the research data discussion, and reviewed the data and manuscript. Špela Volčanšek contributed to the conception and design, wrote the manuscript, contributed to the research data discussion, and reviewed the data and manuscript. Karin Zibar Tomšić contributed to the conception and design, wrote the manuscript, contributed to the research data discussion, and reviewed the data. Andrej Janež contributed to the conception and design, drafting, and critical review of the manuscript. Dimitri P. Mikhailidis contributed to the conception and design, drafting, and critical review of the manuscript. Manfredi Rizzo contributed to the conception and design, drafting, and critical review of the manuscript. Christos S. Mantzoros contributed to the conception and design, drafting, and critical review of the manuscript. All authors reviewed and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EMM has given talks or attended conferences sponsored by Novo Nordisk, Boehringer Ingelheim, AstraZeneca, Medtronic, Merck Sharp & Dohme, Novartis, Sanofi and Servier. ŠV has given talks or attended conferences sponsored by Eli Lilly, Novo Nordisk and SANOFI. KZT has given talks or attended conferences sponsored by Novo Nordisk, Medtronic, Boehringer Ingelheim and AstraZeneca. AJ receiving lecture fees from AstraZeneca, Boehringer Ingelheim, Eli Lilly, Merck Sharp and Dohme, and Sanofi Pasteur Biologics and advisory board fees and lecture fees from Novo Nordisk. DPM has given talks, acted as a consultant or attended conferences sponsored by Amgen and Novo Nordisk. MR has given lectures, received honoraria and research support, and participated in conferences, advisory boards, and clinical trials sponsored by several pharmaceutical companies including Amgen, Astra Zeneca, Boehringer Ingelheim, Kowa, Eli Lilly, Meda, Mylan, Merck Sharp & Dohme, Novo Nordisk, Novartis, Roche Diagnostics, Sanofi, and Servier. CSM reports grants through his institution from Merck and Boehringer Ingellheim, has been a shareholder of and has received grants through his Institution and personal consulting fees from Coherus Inc. and AltrixBio, he reports personal consulting fees from Novo Nordisk, reports personal consulting fees and support with research reagents from Ansh Inc., collaborative research support from LabCorp Inc., reports personal consulting fees from Genfit, Lumos, Amgen, Corcept, Intercept, and Regeneron, reports support (educational activity meals through his institution or national conferences) from Amarin, Novo Nordisk, Astra Zeneca, Boehringer Ingelheim and travel support and fees from TMIOA, Elsevier, the California Walnut Commission, College Internationale Researche Servier and the Cardio Metabolic Health Conference. None is directly related to the work presented herein.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
