Abstract
Background:
Testosterone replacement therapies may increase blood pressure (BP) with chronic use but the mechanism is not clear. TLANDO™ is a new oral testosterone undecanoate (TU) under development for the treatment of male hypogonadism.
Methods:
We studied the effects of the TU at 225 mg twice daily on ambulatory BP (ABP) and heart rate, in 138 men with hypogonadism (mean age, 54 years, 79% white, 48% with hypertension). Ambulatory BP and heart rate and hematologic assessments were obtained at baseline and following 4-months of therapy.
Results:
Changes from baseline in ambulatory 24-hour, awake, and sleep systolic BP (SBP) of 3.8 (P < 0.001), 5.2 (P < 0.001), and 4.3 mmHg (P = 0.004) were observed post-treatment, respectively. Lesser changes in the diastolic BP (DBP) were observed (1.2 (P = 0.009), 1.7 (P = 0.004), and 1.7 mmHg (P = 0.011) for 24-hour, awake, and sleep, respectively). Hematocrit and hemoglobin were increased by 3.2% and 0.9 g/dL (P < 0.001), respectively. In those men in the top quartile of changes in hematocrit (range of 6% to 14%), the largest increases in ambulatory SBP (mean, 8.3 mmHg) were observed, whereas the changes in ambulatory SBP in the lower 3 quartiles were smaller (mean, 1.9, 3.3, and 2.1 mmHg in 1st, 2nd and 3 rd quartiles, respectively).
Conclusion:
These data demonstrate that small increases in ABP occurred following 4 months of the oral TU. For those men whose hematocrit rose by >6%, BP increases were of greater clinical relevance. Hence, hematocrit may aid in predicting the development of BP increases on testosterone therapy.
ClinicalTrials.gov identifier:
NCT 03868059.
Introduction
Testosterone replacement therapy for male hypogonadism or testosterone deficiency is administered by intramuscular and subcutaneous injections, transdermal gels and lotions, dermal patches, intranasal gels, and more recently by oral formulations. 1 Oral formulations of testosterone undecanoate have been developed to provide average serum testosterone levels in the eugonadal range (typically 300-1,000 ng/dL) and avoid peak concentrations that are 1.5 times the upper limit of normal. Undecanoic fatty acid esterification of testosterone to create the testosterone-ester allows for absorption via the intestinal lymphatic system and bypasses first pass metabolism. 2 Testosterone is then liberated from testosterone undecanoate by endogenous non-specific esterases in the systemic circulation avoiding the hepatotoxicity observed with the orally active 17-alpha-alkyl androgen: e.g. methyltestosterone. An oral testosterone undecanoate therapy (TLANDO™) that does not require dose titration has been developed to restore testosterone to eugonadal levels in hypogonadal males and has met regulatory criteria for approval. 3
During recent clinical trials with both injectable and oral testosterone formulations, small increases in both clinic and ambulatory blood pressure (BP) have been observed and the mechanism of these increases is unclear although increases in red blood cell mass with resultant increases in hemoglobin and hematocrit may play a role. 4 In the present study, we evaluated this new oral testosterone undecanoate at 225 mg twice daily on clinic and ambulatory BP monitoring performed at baseline and the end of nearly 4 months of treatment along with clinical and laboratory parameters to determine its effects on BP and the potential predictors of changes from baseline in BP.
Methods
Study Design
The study was an open-label, multicenter, single arm study with a 2-month screening period, a baseline visit to assess BP and heart rate via ambulatory BP monitoring (ABPM) prior to administration of study medication, and a visit about 3.6 months after initiating the fixed-dose oral testosterone undecanoate (TLANDO™) at 225 mg approximately 12 hours apart to re-assess BP and heart rate by ABPM (Supplementary Figure 1). The dose of 225 mg twice daily is the approved dose for administration of this oral testosterone undecanoate formulation based on clinical development studies. During the screening period, 2 consecutive blood samples were obtained between 6 and 10 AM, on 2 separate days, following an appropriate washout of current testosterone replacement therapy if required (typically 2 up to 12 weeks depending on the type of testosterone therapy used). Those men meeting the study inclusion criteria were confined for approximately 26 hours for study procedures, including seated clinic and ambulatory BP measurements. There were no restrictions on physical activity during the ABPM recordings. Subjects with valid ABPM data were enrolled in the study and provided study drug for 60 days; all returned to clinic for a resupply of study drug for days 60-120. Study subjects were re-confined to the study site on day 107 of the study, and clinical and ambulatory BPs were performed with no restrictions on activity. The trial was registered with ClinicalTrials.gov (identifier: NCT 03868059).
Study Participants
All participants were men between 18 and 80 years of age, inclusive, with documented onset of hypogonadism (congenital or acquired) prior to the age of 65. The total serum testosterone level was required to be below 300 ng/dL on 2 consecutive blood samples obtained between 6 and 10 am on 2 separate days at approximately the same time, either in individuals naïve to androgen replacement or following washout of previous androgen therapy. Exclusion criteria included any unstable medical condition including uncontrolled hypertension, defined as a repeated observation of a systolic BP ≥ 160 mmHg systolic or diastolic BP ≥ 100 mmHg during the screening period, or a change in any antihypertensive therapy in the preceding 6 weeks prior to study enrollment. In addition, laboratory exclusion criteria included any of the following: hemoglobin <11.5 g/dL or >16.5 g/dL; hematocrit <35% or 54%; serum transaminases > 2.5 times the upper limits of normal; serum creatinine >2.0 mg/dL; prostatic specific antigen (PSA) >4 ng/ml; or prolactin levels >17.7 ng/ml.
The trial was conducted in accordance with Good Clinical Practice requirements, as described in the current revision of International Conference on Harmonization guidelines of Technical Requirements of Pharmaceuticals for Human Use and the Declaration of Helsinki. Before any study procedures could occur, a written informed consent was obtained from each study participant.
Safety and Laboratory Assessments
At each clinic visit following the screening visit, all study participants were queried about clinical adverse events. Safety assessments also included physical examination, vital signs, 12-lead electrocardiography (at screening and at 107 days post-initiation of study medication), and clinical laboratory tests.
Serum testosterone and testosterone undecanoate (TU) were analyzed by both PPD Bioanalytical Laboratory (Richmond, VA, US) and ARUP (Salt Lake City, UT) using their respectively validated LC-MS/MS methods (LCMSC 260.10, entitled “Quantitation of Testosterone and Dihydrotestosterone in Human Serum via HPLC with MS/MS Detection”) in this study. The quality control (QC) samples were prepared in bulk, divided into aliquots, and frozen at nominal −20°C prior to analysis. Five QC levels were used, with a minimum of 3 QC levels prepared in natural unmodified human serum. The same serum sample set (or sub-aliquots thereof), along with a set of “blinded” QCs were prepared by PPD and sent to ARUPS Labs to be similarly analyzed using their T/DHT LC-MS/MS assay. The method was validated for linearity, precision, accuracy, recovery, and specificity. For the hematocrit and hemoglobin assays, whole blood samples were filled into 3.0 mL K2-EDTA tube, then gently inverted 10 times. The samples were shipped at ambient temperature to the lab. Hemoglobin and hematocrit were analyzed as part of an automated blood count using the Coulter LH 750/780 Hematology Analyzer.
Blood Pressure Monitoring
The BP was monitored manually in the clinic at the baseline and post-treatment study visits (the first was in the untreated state while the second visit was following approximately 4 months of testosterone undecanoate). The clinic measurements were made in the seated position in triplicate after 10 minutes of rest and using appropriate sized cuff and bladder with a digital recorder. Study participants were then fitted with an ambulatory BP monitor that was initiated to measure the BP at 15-minute intervals between 7 am and 11 pm and at 20-minute intervals between 11 pm and 7 am (Ambulo™ 2400, Tiba Medical, Inc., Portland, OR, USA). This device was validated in an independent study in 2010 5 and passed 2 well-known standards (ISO and BHS) with A grades. Differences between the device and manual standards for SBP/DBP averaged 1.8/0.7 mmHg. The ABPM data were evaluated both manually and programmatically by standardized, computerized methods, for validity including a minimum of one valid reading per hour and at least 22 of 24 hours of valid data over the monitoring period.
Statistical Analyses
The primary endpoint in this study was the change from baseline to end of study of the weighted average 24-hour systolic BP. Other endpoints included changes from baseline in the clinic systolic and diastolic BPs and pulse rate and changes from baseline in the awake (daytime) and sleep (nighttime) systolic BPs, the 24-hour, awake, and sleep diastolic BPs and the 24-hour, awake and sleep heart rates. Additionally, the BP and heart rate changes were evaluated in subgroups of study participants with and without a history of hypertension and with and without a history of diabetes mellitus. The clinical analyses were performed in those with complete data at baseline and end of study (defined as the full analysis set (FAS), n = 126) and the ambulatory BP analyses were performed in those with complete ambulatory BP data (defined as the 24-hour ABP FAS, n = 118). Finally, the incidence of adverse events was tabulated in all participants who received at least one dose of study drug (defined as the safety set, n = 138).
A linear regression model was used with baseline BP as a covariate to analyze the primary and secondary endpoints. The change of 24-hour BP from baseline was calculated using the time-weighted average of area under the curve of BP obtained over 24 hours divided by the time duration. Changes in hourly average BPs were calculated by taking the difference between the corresponding hourly BP at the end of treatment visit and the baseline visit for a given post-dosing hour. Post-hoc regression analyses were also performed to assess relationships among changes from baseline in ambulatory systolic and diastolic BPs with changes in body weight, heart rate, and hematocrit and hemoglobin. Furthermore, categorical changes in ambulatory BP according to the quartiles of changes in hematocrit were also analyzed.
Results
Subject Disposition and Baseline Characteristics
A total of 144 study participants were enrolled and 138 received at least 1 dose of study drug. One hundred twenty-six (91.3%) of the participants treated with the study drug completed the trial and had valid baseline and end of treatment ABPM studies. Among all the participants (N = 144), the primary reasons for early termination were withdrawal by the subject (8.0%), adverse events (2.2%), lost to follow-up (2.2%) and a family death (0.7%). The demographic and baseline characteristics of the study participants are shown in Table 1. The mean age at baseline was 53.8 years (13% older than 65 years), 79% were white and 18% were black. The mean body mass index (BMI) was 33.1 kg/m2. Nearly half of the study participants had a history of hypertension and about one-fourth had a history of type 2 diabetes mellitus. The mean serum testosterone level at study entry was 204 ± 11 ng/dL. The average baseline clinic and 24-hour BPs were comparable (Table 1).
Characteristics of the Patient Population at Baseline.

* Awake time: 7 AM to 10:59 PM.
** Sleep time: 11 PM to 6:59 AM.
Blood Pressure and Heart Rate
Changes from baseline in the 24-hour systolic BP on oral testosterone undecanoate 225 mg twice daily were small but increased significantly (3.8 mmHg, 95% CI, 1.7-6.0 mmHg, P < 0.001). Mean 24-hour systolic BP values at baseline were 127.2 ± 15.8 mmHg and at the end-of-study 131.0 ± 15.6 mmHg. Lesser effects were seen for the ambulatory diastolic BP and were only significantly greater for the awake, daytime period (Table 2). Mean 24-hour diastolic BP values at baseline were 79.3 ± 5.9 mmHg and at the end of study were 80.5 ± 6.2 mmHg. The nocturnal decline in systolic BP was unchanged by oral testosterone undecanoate therapy (6.2% at baseline versus 6.8% at the end of the study, P = 0.15). Small increases in the 24-hour ambulatory heart rate were also observed (2 beats/minute, P < 0.001, see Table 2) and primarily driven by increases during the daytime (awake) period.
Changes From Baseline in Clinic and Ambulatory Blood Pressure and Heart Rate Following 4 Months of Oral Testosterone Undecanoate (225 mg BID).

* P-values based on weighted averages derived by comparing the area under the curve of BP values over time using paired t tests.
** Awake (Daytime): 7 AM to 10:59 PM.
*** Sleep (Nighttime): 11 PM to 6:59 AM.
Outlier analyses showed that 12/118 study participants (10.1%) had a change from baseline in 24-hour systolic BP > 20 mmHg. Only 1/118 (0.8%) of the study participants had a change from baseline in 24-hour diastolic BP > 15 mmHg. For the 25 study participants who had a baseline systolic BP > 140 mmHg, 32% had a post-treatment systolic BP
Ambulatory systolic and diastolic BPs over 24 hours at baseline and at the end of treatment are shown in Figure 1. The systolic BP over time was modestly higher during the 24 hours following approximately 4 months of treatment with oral testosterone undecanoate. The 24-hour diastolic BPs over time were similar to the systolic BP profiles (Figure 1).

Hourly changes in ABP at baseline and end of study (about 4 months of oral testosterone undecanoate therapy). (A) Systolic BP; (B) diastolic BP.
In the clinic, seated BPs increased significantly by 4.8/1.7 mmHg (P < 0.001 for systolic BP; P = 0.017 for diastolic BP) following 107 days of treatment with testosterone undecanoate 225 mg twice daily (Table 2). The clinic pulse rate increased by 2.3 beats/minute (P = 0.007).
Blood Pressure Changes in Pre-Specified Subgroups
Changes from baseline in 24-hour BP and heart rate in study participants with and without hypertension and with and without type 2 diabetes are shown in Table 3. There were comparable numbers of patients with and without hypertension (57 and 61, respectively) with ABPM data at both baseline and end of treatment. Changes from baseline in 24-hour systolic and diastolic BPs were nominally greater in patients with hypertension at baseline versus those without hypertension but the differences were not statistically significant. Changes in ambulatory heart rate were smaller in patients with hypertension versus those without hypertension (Table 3).
Changes From Baseline in 24-Hour Ambulatory BPs and Heart Rate in Study Participants With and Without Hypertension and With and Without Type 2 Diabetes following Treatment With Oral Testosterone Undecanoate, 24-Hour ABP FAS (n = 118).

* All values are mean ± standard error.
There were fewer patients with type 2 diabetes than without type 2 diabetes (34 and 84, respectively) who had ABPM data at both baseline and end of treatment. Changes from baseline in 24-hour systolic and diastolic BPs were nominally but not statistically greater in patients with diabetes versus those without diabetes, whereas changes in ambulatory heart rate were nominally smaller in patients with diabetes versus those without diabetes.
Blood Pressure Findings Associated With Body Weight, Heart Rate, and Age
The change from baseline in the body weight was not significant following about 4 months of oral testosterone undecanoate (baseline 105.2 ± 1.8 kg and change from baseline 0.5 ± 0.3 kg, P = 0.84). There was no relationship between the change in body weight and change in 24-hour ambulatory systolic BP (r2 = 0.073, P = 0.43). While ambulatory heart rate increased modestly over 24-hours (Table 2), there was no relationship between the change from baseline in 24-hour systolic BP and 24-hour heart rate (r2 = 0.077, P = 0.41).
Changes from baseline in the ambulatory BP parameters over 24-hour, daytime and nighttime periods were evaluated by median age cut-offs (54 years) and showed no impact of age on the changes in ambulatory BP. Changes in 24-hour BP for study participants < 54 years were 3.6/0.9 mmHg and for those ≥ 54 years, changes from baseline were 4.0/1.5 mmHg (p values between age groups, 0.87 and 0.55, respectively for SBP and DBP).
Blood Pressure Findings Associated With Hematologic Results
The hematocrit increased from 44.2% ± 3.2% to 47.4% ± 4.5% (mean change of 3.2% ± 3.6%, P < 0.001) following treatment with oral testosterone undecanoate. Similarly, the hemoglobin rose from 14.6 ± 1.1 g/dL to 15.4 ± 1.5 g/dL (mean change of 0.87 ± 1.05 g/dL, P < 0.001). There were 6 study participants (4.3%) with hematocrit values greater than the upper limit of the lab normal range (54%) at the final study visit.
Changes from baseline in the 24-hour BP and heart rate according to changes in hematocrit during the study are shown in Table 4. The quartiles of change in hematocrit ranged from means of −1.5% (1st quartile) to 7.9% (4th quartile). The changes from baseline in ambulatory systolic and diastolic BP were greater in the 4th quartile of hematocrit change compared to the lower 3 quartiles. Correlational analyses of change in ambulatory BP and change in hematocrit showed positive relationships (supplementary Figure 2). For changes from baseline in 24-hour systolic BP, the correlation coefficient with change in hematocrit was 0.16, P = 0.075. For the changes in ambulatory diastolic BP, the correlation coefficient with hematocrit changes was 0.20, P = 0.03. Changes in ambulatory heart rate were small and comparable in the quartiles of change in hematocrit (Table 4).
Changes From Baseline in 24-Hour Ambulatory BPs and Heart Rate According to Quartiles of Changes in Hematocrit Following Treatment With Oral Testosterone Undecanoate, 24-Hour ABP FAS (n = 118).
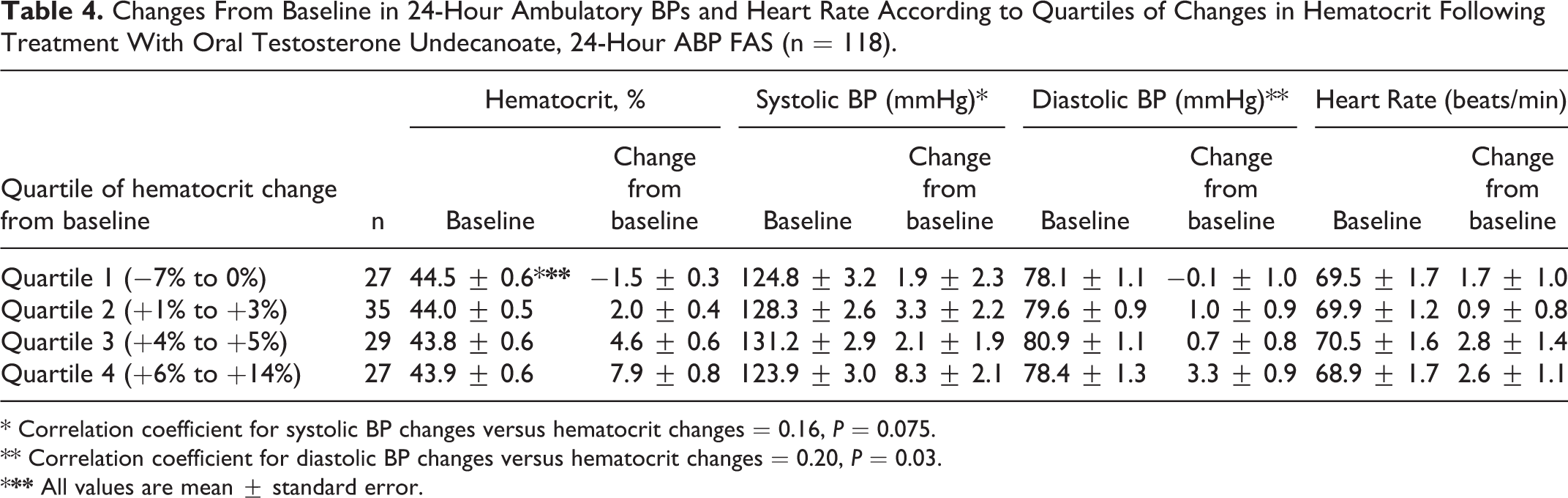
* Correlation coefficient for systolic BP changes versus hematocrit changes = 0.16, P = 0.075.
** Correlation coefficient for diastolic BP changes versus hematocrit changes = 0.20, P = 0.03.
*
Changes from baseline in the 24-hour BP and heart rate according to changes in hemoglobin during the study are shown in supplementary Table 1. The quartiles of change in hemoglobin ranged from an average of −0.5 g/dL (1st quartile) to 2.2 g/dL (4th quartile). Similar to the data for the hematocrit, the changes from baseline in ambulatory systolic and diastolic BPs were greater in the 4th quartile of changes in hemoglobin compared to the lower 3 quartiles. Correlational analyses of change in ambulatory BP and change in hemoglobin also showed positive relationships (supplementary Figure 3). The correlation coefficient for changes in 24-hour systolic BP and changes in hemoglobin was 0.18, P = 0.056. The correlation coefficient for changes in 24-hour diastolic BP and hemoglobin changes was 0.21, P = 0.023. Changes in ambulatory heart rate were small and comparable in the quartiles of change in hemoglobin (Supplementary Table 1).
Adverse Events
There was no deaths and no treatment-related serious adverse events. Fourteen (10.1%) subjects experienced at least 1 treatment-related adverse event that was characterized as mild to moderate. The most frequently reported treatment-related adverse events (>1%) were increased hematocrit (2.9%) and hypertension, polycythemia, and gynecomastia (1.4% each). There were no changes from baseline in kidney or liver function tests during the study period. Of 59 subjects who were taking antihypertensive medication at baseline, 3 study participants had a change in the class of therapy during the study. During the study, 2 subjects (1.4%) were started on a new antihypertensive medication or required increases to their antihypertensive medication regimen.
Discussion
The results of this BP safety study demonstrated that oral testosterone undecanoate was associated with small, but significant increases in clinic and ambulatory awake systolic BP following nearly 4 months of replacement therapy in hypogonadal men. The increases were larger for the systolic BP than for the diastolic BP, both when measured in the clinical setting as well as by the use of 24-hour ambulatory BP monitoring. There were also small increases in the clinic and daytime (awake) heart rates on oral testosterone undecanoate. The increases in ambulatory systolic BP were not correlated with the increase in the ambulatory heart rate discounting the possibility of an adrenergic etiology of the increases in ambulatory BP. There were also no changes in body weight in the men treated with oral testosterone undecanoate nor any relationship between changes in body weight and ambulatory BP that might have supported an increase in plasma volume as one possible mechanism for the increase in BP.
Change in the 24-hour ambulatory BP was the primary outcome measure in this study rather than changes in the clinic BP. In recent years, the US Food and Drug Administration has advocated for use of ABPM in drug safety research 6,7 since these devices have the potential to detect smaller changes in BP with improved reproducibility and minimal-to-no placebo effects. In fact, regulators often do not require a placebo treatment arm for studies such as ours; the goal of which was to provide evidence of a modest-to-moderate BP effect and for which a placebo would be unlikely to obscure the accuracy of the result. Our study also employed frequent readings over the 24-hour period which improves the precision of the studies and allows for considerable power to exclude 3 to 4 mmHg shifts in BP without a placebo arm. 6 -8 It is noteworthy that there were similar changes in clinic BP and changes in awake ambulatory BP in this study. This phenomenon is not uncommon when both the clinical readings and the ambulatory BP measurements are made in confined patients in clinical research centers. 6
We observed small, but significant, increases in the mean levels of hemoglobin and hematocrit following nearly 4 months of oral testosterone undecanoate replacement therapy. Testosterone has a well-known erythrogenic effect that increases red blood cell production and induces increases in hemoglobin and hematocrit, 4,9 particularly after a few months of replacement therapy. 10 In a recent review comparing the effects of chronic replacement of testosterone products (gels, injections, pellets) on serum hormones and hematological parameters, injectable testosterone increased the hematocrit and hemoglobin more substantially (mean increase of hematocrit: 6.5% and hemoglobin: 1.1 g/dL) compared to gels (2.6% and 0.8 g/dL) and pellets (2.6% and 0.8 g/dL). In addition, the proportion of men developing erythrocytosis (defined as hematocrit ≥ the upper limit of the lab normal range) following the treatment were substantially higher with testosterone replacement by injection (66.7%) compared to gels (12.8%) or pellets (35.1%). 4 There are also mechanistic data that have shown increases in erythropoietin, suppression of hepcidin, and increased expression of ferroportin and the transferring receptor following testosterone therapy. 11 In our study, following 4 months of the oral testosterone undecanoate TLANDO™ 225 mg twice daily, only 4.3% of the study participants developed a hematocrit above the laboratory reference range.
The results of our study showed a positive relationship between increases in hematocrit or hemoglobin and increases in ambulatory BP. To our knowledge, this relationship has either not been studied or not been observed previously. For those patients whose hematocrit rose 6 to 14% in the top quartile of changes, the average increase in 24-hour BPs (systolic/diastolic) was 8.3/3.3 mmHg, while for those whose hematocrit did not change in the lowest quartile, changes from baseline in 24-hour BPs were just 1.9/−0.1 mmHg. This new information should be helpful for clinical practice since monitoring of the hemoglobin and hematocrit is a routine practice when initiating testosterone replacement therapy in men with hypogonadism. In those men who have robust erythogenic effects, clinicians would be advised to be more vigilant about monitoring BP.
Prospective cohort studies with large populations have shown that increases in systolic BP of 4 to 5 mmHg are related to adverse cardiovascular events, particularly heart failure and stroke. 12 However, the clinical importance of a 4 mmHg increase in systolic BP in hypogonadal men is less clear since men with testosterone deficiencies have increases in cardiovascular risk 13 and there is evidence that normalizing testosterone levels in hypogonadal men is associated with lower rates of cardiovascular morbidity. 14 Furthermore, in an observational study with a median follow-up of 7 years by Traish and coworkers, 15 hypogonadal men receiving parenteral testosterone undecanoate had lower cardiovascular events than men not receiving testosterone replacement therapy and their clinical BP values fell over time. Nevertheless, clinical assessment of BP is important for hypogonadal men who require testosterone replacement. We also noted that patients with a history of hypertension had numerically larger increases in ambulatory BP than those individuals without a medical history of hypertension. This finding is consistent with the effects of another oral testosterone undecanoate when compared to a testosterone gel 16 and in those patients receiving parenteral administration of testosterone enanthate. 17 Our findings and those of other studies suggest that small increases in clinic or ambulatory BP are likely to be a class effect of testosterone replacement therapies in hypogonadal men. Hence, careful BP assessments in the clinic would be particularly relevant for hypogonadal patients with hypertension when initiating testosterone replacement therapy.
Our study does have some limitations. This trial was conducted without a placebo or active comparator arm as noted above. While desirable to have a comparator arm, placebo has little to no impact on ambulatory BP values within weeks to months of therapy. 7,18 Hence, the results of the present study are unlikely to be affected by the lack of a comparator treatment arm and can be compared to other recent studies 16,17 in which ambulatory BP monitoring was conducted with other testosterone replacement therapies. Unlike other oral testosterone undecanoate formulations, TLANDOTM does not require dose titration and uses a fixed dose of 225 mg BID, hence assessment of a range of doses is not recommended. In this study, the measured mean level of testosterone undecanoate at the end of treatment was in the therapeutic range (107.8 ± 25.7 ng/mL), and similar to those measured from a previous study that evaluated the efficacy and pharmacokinetics of oral testosterone undecanoate. 3
In conclusion, this new oral formulation of testosterone undecanoate increased clinic and 24-hour ambulatory BP by approximately 5/4 mmHg. There were also small increases in ambulatory daytime heart rate but these were not related to the BP changes. As would be expected, a small proportion of men experienced erythrogenic effects after nearly 4 months of treatment, however only 4.3% had hematocrit levels >54% (the upper range for the laboratory). We did observe that more substantial increases in ambulatory BP occurred in those men who developed the largest increases in hematocrit, a novel finding of this study, and one which may improve clinical monitoring after initiating testosterone replacement therapy in hypogonadal men.
Supplemental Material
Supplemental Material, sj-jpg-1-cpt-10.1177_10742484211027394 - Effects of a Novel Oral Testosterone Undecanoate on Ambulatory Blood Pressure in Hypogonadal Men
Supplemental Material, sj-jpg-1-cpt-10.1177_10742484211027394 for Effects of a Novel Oral Testosterone Undecanoate on Ambulatory Blood Pressure in Hypogonadal Men by William B. White, Adrian Dobs, Culley Carson, Anthony DelConte, Mohit Khera, Martin Miner, Muhammad Shahid, Kilyoung Kim and Nachiappan Chidambaram in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, sj-jpg-2-cpt-10.1177_10742484211027394 - Effects of a Novel Oral Testosterone Undecanoate on Ambulatory Blood Pressure in Hypogonadal Men
Supplemental Material, sj-jpg-2-cpt-10.1177_10742484211027394 for Effects of a Novel Oral Testosterone Undecanoate on Ambulatory Blood Pressure in Hypogonadal Men by William B. White, Adrian Dobs, Culley Carson, Anthony DelConte, Mohit Khera, Martin Miner, Muhammad Shahid, Kilyoung Kim and Nachiappan Chidambaram in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, sj-jpg-3-cpt-10.1177_10742484211027394 - Effects of a Novel Oral Testosterone Undecanoate on Ambulatory Blood Pressure in Hypogonadal Men
Supplemental Material, sj-jpg-3-cpt-10.1177_10742484211027394 for Effects of a Novel Oral Testosterone Undecanoate on Ambulatory Blood Pressure in Hypogonadal Men by William B. White, Adrian Dobs, Culley Carson, Anthony DelConte, Mohit Khera, Martin Miner, Muhammad Shahid, Kilyoung Kim and Nachiappan Chidambaram in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
Dr. White drafted the manuscript and had full access to the data; Dr. Kim performed statistical analyses and provided critical review of the manuscript. Drs. Dobs, Culley, Khera, Miner, DelConte, and Shahid provided critical review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
