Abstract
Objective:
To evaluate the safety and efficacy of extended-interval dabigatran dosing in older Chinese patients with non-valvular atrial fibrillation.
Methods:
We conducted an observational study on non-valvular atrial fibrillation patients administered dabigatran at different dosing intervals at the Department of Geriatrics, Peking University First Hospital, China. We enrolled 121 consecutive non-valvular atrial fibrillation patients aged ≥60 years on dabigatran therapy (mean age, 79.6 ± 7.4 years); they were administered conventional low-dose dabigatran (110 mg twice daily) or extended-interval dosing with dabigatran (110 mg every 16 h or every 24 h). All patients received follow-up care, and we evaluated the presence of bleeding and thromboembolic events.
Results:
All patients exhibited creatinine clearance greater than 30 mL/min with an average of 56.6 ± 17.3 mL/min. Sixty-two patients received extended-interval dosing with dabigatran at a mean dose of 117.1 ± 18.6 mg daily. Patients on extended-interval dosing were older; they exhibited lower creatinine clearance and bodyweight and higher CHA2DS2-VASc and HAS-BLED scores. The mean follow-up time was 25.8 ± 15.6 months. No significant differences were observed in the trough and peak values of the activated partial thromboplastin time and in thromboembolic or bleeding events between the 2 groups.
Conclusion:
Extended-interval dabigatran dosing in older patients with non-valvular atrial fibrillation and lower creatinine clearance can maintain activated partial thromboplastin time trough and peak values comparable to the conventional low dose. Physician-prescribed practices regarding dabigatran dosing intervals do not lead to worse outcomes in the above-mentioned population.
Introduction
Dabigatran has been proven to be non-inferior to warfarin in the prevention of stroke and embolism in patients with non-valvular atrial fibrillation (NVAF). 1 Additionally, low-dose dabigatran (110 mg twice daily) was associated with a reduced risk of ischemic stroke, systemic embolism, death, and intracranial bleeding compared with warfarin in Asians. 2 -5 Dabigatran is recommended for use at fixed doses without routine laboratory monitoring; however, the plasma concentrations of dabigatran demonstrated more than 5-fold variation between individual patients at the same dose in a pharmacokinetic analysis of RE-LY trial, 6 which indicates that an individual patient might benefit from tailoring the dose of dabigatran to optimize risk of ischemic and bleeding events, particularly the elderly or patients with impaired renal function. Balancing bleeding and stroke risk is a huge challenge when considering anticoagulant therapy in NVAF patients with bleeding or at a high risk of bleeding. Older age is usually associated with several comorbidities, decreased creatinine clearance, increased risk of bleeding, and atrial fibrillation-related ischemic stroke, that is, high risk of bleeding and thrombosis. 7,8 Off-label dosing of dabigatran occurs frequently, 9,10 with under-dosing, especially in older patients. No dosage forms of dabigatran are available with a dose lower than 110 mg in China. Extending the interval of administration is often used to reduce the dosage in patients with a high risk of bleeding in clinical practice. However, no studies have reported the efficacy and safety of extended interval administration before, and the consequences of extended-interval dabigatran dosing remain uncertain. Therefore, this study sought to observe the efficacy and safety of extended-interval dabigatran dosing in older Chinese patients with NVAF and to identify factors associated with extended-interval dosing.
Methods
Design and Study Population
This is an observational study of NVAF patients on different dabigatran dosing intervals in a large university teaching hospital in China. The study protocol was approved by the Ethics Committee of Peking University First Hospital (2019 RESEARCH No. 129). Each participant provided informed consent to be included in the study.
Consecutive oral anticoagulant treatment-naive NVAF patients aged 60 years or older who were newly prescribed dabigatran were enrolled from October 2013 to September 2017 at the Department of Geriatrics, Peking University First Hospital, a 1500-bed tertiary care teaching hospital in China. Patients were excluded if they had significant valvular heart disease and previous valvular replacement; could not be followed up or had renal insufficiency with creatinine clearance rate <30 mL/min. Atrial fibrillation was diagnosed by ECG or 24-h Holter with valvular heart disease excluded by echocardiography. Patients treated with dabigatran 110 mg twice daily were defined as the conventional low-dose group, and patients who received dabigatran 110 mg every 16 h or every 24 h were defined as the extended-interval dosing group. Dabigatran dosage was determined by the treating physician; the researchers did not intervene.
Data Collection
The study data were collected from the computerized database of the clinical management system in hospital. As a legal document, medical records have accuracy, validity, and integrity. Hospital admissions data included patient demographics, dates of admission and separation, major diagnoses based on ICD-10 codes, details of the concomitant prescription of anti-platelet agents (aspirin or clopidogrel) and/or strong P-gp inhibitors (such as amiodarone, diltiazem, and clarithromycin), and activated partial thromboplastin time (aPTT). aPTT was tested in our hospital using an ACL-TOP automatic coagulation analyzer and reagents from Beckman Coulter USA. The normal reference interval was 26.9–37.6 seconds. Administration of dabigatran generally prolongs aPTT. aPTT values measured before the first administration of dabigatran were considered the baseline, and values measured before and 3–4 h after the morning dose at steady state were defined as the trough and peak values, respectively. A steady state was defined as 5 days or more on dabigatran therapy. Creatinine clearance (Ccr, mL/min) was calculated using the Cockcroft–Gault equation ([140 − age (years)] × body weight (kg) × (0.85 if female)/[72 × serum creatinine (mg/dL)]). 11
Clinical Outcomes and Follow Up
All patients were monitored for bleeding and thromboembolic events. The definition of bleeding events was based on the criteria of the International Society on Thrombosis and Haemostasis. Major bleeding was defined as a decrease of >2.0 g/dL in hemoglobin, transfusion of >2 units of blood, or symptomatic bleeding in a critical area or organ. Other types of bleeding were defined as minor bleeding. Thromboembolic events included ischemic stroke, systemic embolism, and thromboses in the left atrium. All patients were followed up at outpatient clinics once a month for any bleeding or thromboembolic events. Bleeding or thrombotic events were comprehensively determined by follow-up doctors according to the patients’ symptoms, laboratory tests, and imaging results. Patients with acute thromboembolic or major bleeding events were hospitalized. Minor bleeding was usually treated at outpatient clinics. All events were recorded in case report form. Patients were followed up until discontinuation of dabigatran, patient death, or the end of the study period (November 2018), whichever occurred the earliest.
Statistical Analysis
Statistical analysis was conducted using the SPSS version 14.0 software (SPSS Inc., Chicago, IL, USA). Continuous and categorical variables are presented as the mean ± standard deviation and frequency (percentage), respectively. The incidence rate of bleeding or thromboembolism was calculated using a person-time approach. Pearson χ2 or Fisher exact tests, if appropriate, were used to make comparisons between groups for categorical variables. Independent t-tests were used to compare groups with continuous variables. Binary logistic regression analysis was used to adjust factors for the reduction of dabigatran dosage. Kaplan–Meier survival analyses with the log-rank test were performed for outcome comparisons. All tests were 2-sided, and P values were considered significant if P < .05.
Results
Baseline Characteristics of Study Population
We enrolled a total of 121 consecutive patients with NVAF, comprising 89 males and 32 females. All patients exhibited a CHA2DS2-VASc score ≥2. The mean age was 79.6 ± 7.4 years, and 57.0% of the patients were aged 80 years or older. All patients exhibited a Ccr greater than 30 mL/min, with an average of 56.6 ± 17.3 mL/min. The mean follow-up duration was 25.8 ± 15.6 months.
There were 59 patients on conventional low dose and 62 patients on extended-interval dosing, with 54 patients on 110 mg every 24 h and 8 patients on 110 mg every 16 h. The mean CHA2DS2-VASc scores were 3.9 ± 1.4 in the conventional low-dose group and 4.7 ± 1.4 in the extended-interval dosing group. The mean dose of dabigatran was 117.1 ± 18.6 mg daily in the extended-interval dosing group. As shown in Table 1, patients on extended-interval dosing were older, and they exhibited lower Ccr, lower bodyweight and BMI, and higher CHA2DS2-VASc and HAS-BLED scores than patients who received the conventional low dose.
Baseline Characteristics of 121 Older NVAF Patients on Dabigatran.

* Strong P-gp inhibitors.
Ccr, creatinine clearance rate; BMI, body mass index.
Factors that were statistically different between groups and considered clinically relevant (age, Ccr, BMI, and HAS-BLED scores) were entered into the multivariate logistic regression model, along with the number of samples. The results revealed that age ≥80 years (odds ratio [OR]: 2.963, 95% CI: 1.268–6.927, P = .012) and Ccr < 50 mL/min (OR: 3.370; 95% CI: 1.252–9.070; P = .016) were independently associated with extended-interval dosing.
Monitoring of Activated Partial Thromboplastin Time in Patients on Dabigatran
The aPPT baseline and trough levels were tested and drawn for all patients. A total of 41 patients on extended-interval dosing and 53 patients on conventional low-dose dabigatran tested aPTT peak values. As seen in Table 2, aPTT baseline values were higher in patients on extended-interval dabigatran dosing; however, no significant difference was observed in the aPTT trough and peak values between the 2 groups.
Comparison of aPTT Values Between Patients on Extended-Interval Dosing and Those on Conventional Low Dose of Dabigatran.
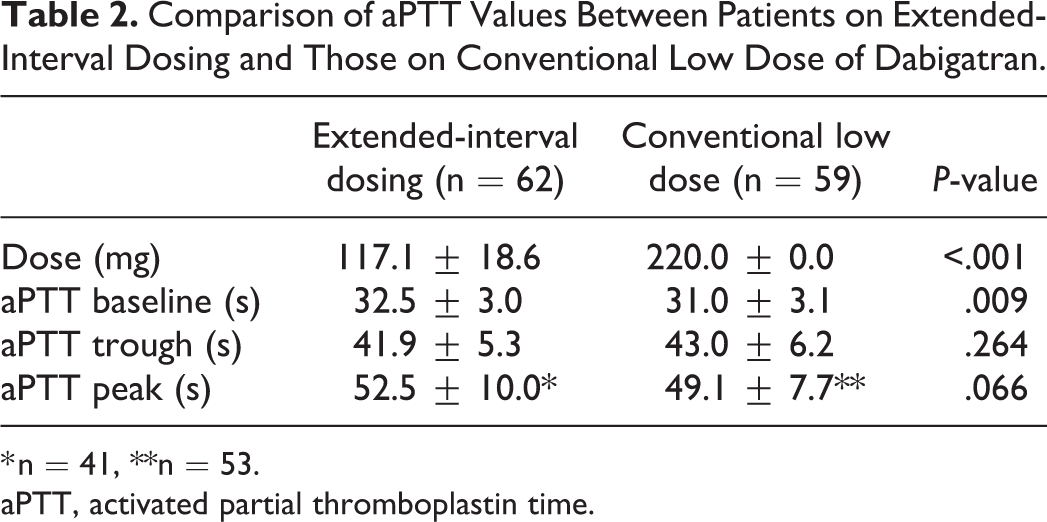
* n = 41, **n = 53.
aPTT, activated partial thromboplastin time.
A total of 23 patients underwent a dose switch between dabigatran 110 mg twice daily and 110 mg every 24 h at the beginning of the dabigatran therapy. The aPTT baseline and trough values at different interval are presented in Figure 1; the aPTT trough value was higher for the dose of 110 mg twice daily than for 110 mg every 24 h (53.6 ± 8.3 s vs. 43.8 ± 4.1 s, P < .001) in the same group of patients.
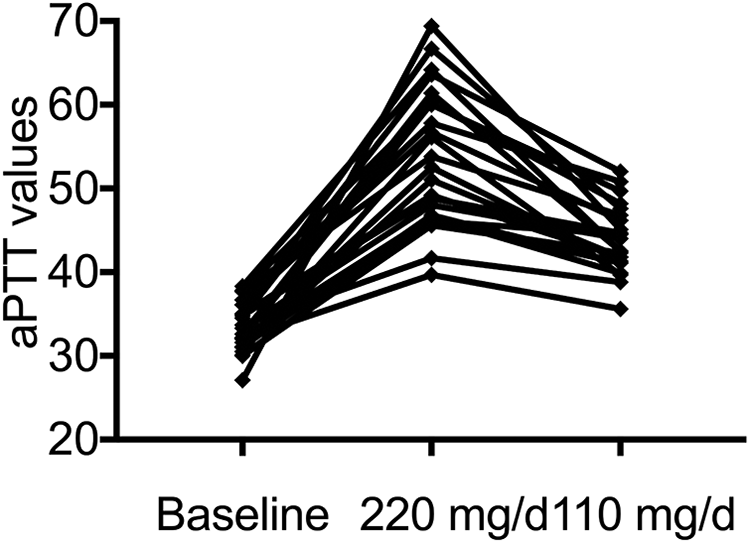
Changes in aPTT with different administration intervals of dabigatran in 23 patients who underwent dose switching.
Efficacy and Safety Outcomes
All patients were followed up at outpatient clinics once a month, with a mean follow-up of 25.8 months. A total of 8 thromboembolic events occurred, with 4 cases in the extended-interval dosing group (2.91% per year) and 4 cases in the conventional low-dose group (3.25% per year). A total of 19 bleeding events occurred, with gastrointestinal bleeding and subcutaneous ecchymosis the most common manifestations; 14 (10.19% per year) occurred in patients on extended-interval dosing and 5 (4.06% per year) at the conventional low dose. There were 3 episodes of major bleeding (2.18% per year) on extended-interval dosing and 1 (0.8% per year) at the conventional low dose.
Although there was a significant difference in the baseline data between the groups, due to the small sample size, we conducted a rough comparison of event rates between the groups. There were no more thrombotic events in the extended-interval dabigatran dosing group. No significant differences were observed with respect to bleeding or major bleeding between the 2 study groups, as assessed through the Kaplan–Meier survival analyses (Figure 2).

Extended-interval dabigatran dosing (Extended-interval) did not increase the risk of thromboembolism compared with the conventional low dose (Conventional) in older NVAF patients (A). There were no significant differences in all bleeding (B) or major bleeding (C) events between the 2 study groups.
The details of the episodes of thromboembolism and major bleeding in 12 patients are summarised in Table 3. All patients were hospitalised and discharged after recovery.
Details of Thromboembolic and Major Bleeding Events in 12 Patients.
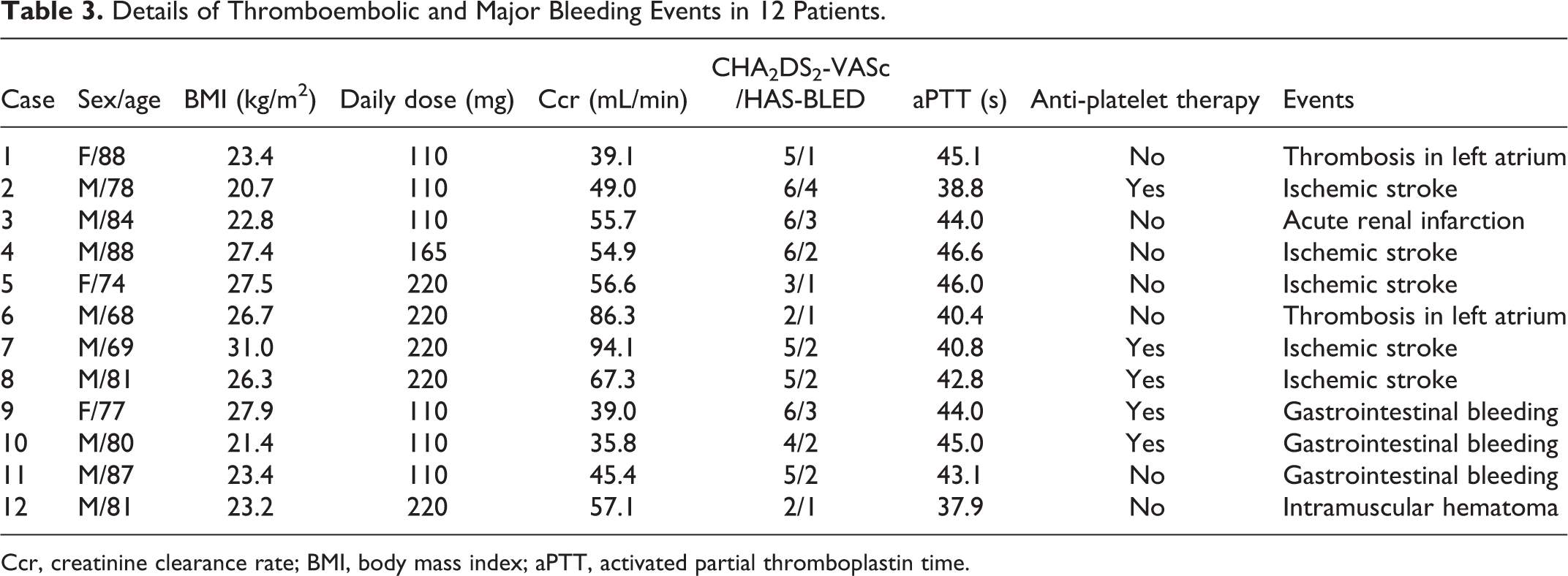
Ccr, creatinine clearance rate; BMI, body mass index; aPTT, activated partial thromboplastin time.
Discussion
The findings of this observational study of NVAF patients treated with dabigatran can be summarized as follows: i) more than half of the NVAF patients at the study center received extended-interval dabigatran dosing; ii) patients on extended-interval dosing were older, and they exhibited lower Ccr, lower body weight and BMI, and higher CHA2DS2-VASc and HAS-BLED scores. Patients of age ≥80 years and with Ccr < 50 mL/min were independently associated with extended-interval dosing; iii) there were no significant differences in the aPTT trough and peak values between patients on extended-interval dosing and those on conventional low dose; iv) there were no more thrombotic events and bleeding events in the extended-interval dabigatran dosing group. Extended-interval dabigatran dosing did not increase the risk of thromboembolism in older patients with lower Ccr.
The results of our study are consistent with the findings of previous studies on off-label dosing of dabigatran. 12,13 In a study by Briasoulis et al., 1013 patients eligible for the standard dose (150 mg twice daily) of dabigatran received a low dose (75 mg twice daily). According to propensity score matching, the use of a low dose was not associated with stroke or major, gastrointestinal, or intracranial bleeding. Similar conclusion was reached for edoxaban in an analysis of data from the randomized, double-blind ENGAGE AF-TIMI 48 trial, 14 in which the authors concluded that tailoring of the dose of edoxaban on the basis of clinical factors preserved the efficacy of edoxaban compared with warfarin and achieves the dual goal of preventing excess drug concentrations and helps to optimize an individual patient’s risk of ischemic and bleeding events. The 2 groups in our study exhibited significant baseline differences, such as average age, average creatinine clearance, and CHA2DS2-VASc and HAS-BLED scores, because of which their major bleeding and thrombotic event outcomes appear incomparable. However, compared with the patients on the conventional low dose, patients on the extended-interval dosing were older; the latter exhibited worse renal function and higher CHA2DS2-VASc and HAS-BLED scores, which indicates a higher risk of thromboembolic and bleeding events. There were no more thrombotic events and bleeding events in the extended-interval dabigatran dosing group, which suggests that extended-interval dosing of dabigatran was safe and effective for the affected populations. This deserves further investigation through larger trials. We suppose that physicians may tailor the doses of dabigatran to the specific patient’s underlying risk of thromboembolism and bleeding. Through such individual adjustment, more patients may be able to tolerate anticoagulation therapy, which would enable optimize an individual patient’s risk of ischemic and bleeding events.
Thrombotic events have been associated with low blood levels for anticoagulants, not necessarily with low doses. 15 The plasma concentrations of dabigatran demonstrated more than 5-fold variation between individual patients at the same dose in the subsequent pharmacokinetic analysis of RE-LY trial. 6 Nearly 80% of ingested dabigatran is metabolized by the kidney. The half-life of dabigatran is extended in chronic kidney disease, leading to potentially higher plasma levels of dabigatran. 16 Patients on extended-interval dabigatran dosing in the current study were older with lower Ccr, which would increase the dabigatran plasma levels. The mean daily dose of dabigatran was 117.1 ± 18.6 mg in the extended-interval dosing group. To the best of our knowledge, this is the lowest daily dose of dabigatran ever reported. Administration of dabigatran twice daily appears better according to its pharmacokinetics. 17 However, there are no dosage forms of dabigatran with a dose lower than 110 mg in China. Therefore, when a dose reduction is needed due to bleeding or a high risk of bleeding, the interval of administration can only be extended. aPTT was evaluated as a surrogate marker for the anticoagulant activity of dabigatran in the current study. Studies 18,19 have shown that aPTT is linearly correlated with plasma dabigatran levels within the therapeutic range; however, it is insensitive with higher levels. Dabigatran was used at a lower dose in the current study, and the likelihood of the blood levels of the drug being considerably higher than the therapeutic range is low. There were no significant differences in the aPTT trough and peak values between the 2 groups, which indicates that extended-interval dabigatran dosing in older NVAF patients with lower Ccr can maintain anticoagulant activity comparable to the conventional low dose.
This study has 2 main limitations. First, this study enrolled a relatively small number of consecutive patients at a single center; therefore, the Kaplan–Meier survival analyses of the outcomes were not stratified, and there were significant differences in the baseline data between the 2 compared groups. Second, the extended-interval dabigatran dosing was off-label according to the current guidelines; however, it was passively reduced due to bleeding or high risk of bleeding. Therefore, these results warrant further investigation through large-scale prospective studies in the future.
Conclusion
The current study provides a real-world picture of dabigatran prescribing practices. In summary, we determined that extended-interval dabigatran dosing in older NVAF patients with lower Ccr can maintain aPTT trough and peak values comparable to the conventional low dose. The prescribing practices of physicians with respect to dabigatran dosing intervals do not lead to worse outcomes in the above-mentioned population.
Footnotes
Acknowledgments
We express our gratitude to all the patients who participated and the staff members of the Department of Geriatrics at Peking University First Hospital for their assistance in the current study.
Author Contributions
Lian-fang Ni and Meilin Liu were involved in study conception and design; Lian-fang Ni, Xia-huan Chen, Yan Fan, Zhi-fang Fu, Dan Sun, and Zhi-jie Zhao contributed to data collection; Lian-fang Ni and Xia-huan Chen helped in data analysis and data interpretation; Lian-fang Ni drafted the manuscript; Meilin Liu was involved in obtaining funding and supervised the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Major Research Program of China [2016YFC1301300].
