Abstract
Background:
We investigated whether the cardioprotective, volatile gas anesthetic agent, isoflurane, could improve survival and organ function from hemorrhagic shock in an experimental rat model, compared to standard nonvolatile anesthetic agent ketamine/xylazine.
Methods:
Sprague Dawley rats (both genders) were randomized to receive either intraperitoneal ketamine/xylazine (K/X, 90 and 10 mg/kg; n = 12) or isoflurane (5% isoflurane induction and 2% maintenance in room air; n = 12) for anesthesia. Blood was withdrawn to maintain mean arterial blood pressure at 30 mm Hg for 1 hour, followed by 30 minutes of resuscitation with shed blood. Rats were allowed to recover and survive for 6 weeks.
Results:
During the shock phase, the total withdrawn blood volume (expressed as % of estimated total blood volume) to maintain a level of hypotension of 30 mm Hg was significantly higher in the isoflurane group (51.0% ± 1.5%) than in the K/X group (45.3% ± 1.8%; P = .023). Recovery of blood pressure during the resuscitation phase was significantly improved in the isoflurane group compared to the K/X group. The survival rate at 6 weeks was 1 (8.3%) of 12 in rats receiving K/X and 10 (83.3%) of 12 in rats receiving isoflurane (P < .001). Histology performed at 6 weeks demonstrated brain infarction in the 1 surviving rat receiving K/X; no brain infarction occurred in the 10 surviving rats that received isoflurane. No infarction was detected in heart, lung, liver, or kidneys among the surviving rats.
Conclusions:
Isoflurane improved blood pressure response to resuscitation and resulted in significantly higher long-term survival rate.
Introduction
Hemorrhagic shock remains a leading cause of mortality and morbidity on the battlefield as well as in the civilian world following traumatic blood loss. 1 Therapies have focused largely on the nature of the fluid that is used for resuscitation including the composition of blood elements. There has been less focus on developing therapies aimed at directly protecting the vital organs themselves from periods of ischemia/hypoxia due to low blood pressure. Recent studies suggest that agents and therapeutic maneuvers that protect the heart during myocardial infarction may be applied to the situation of hemorrhagic shock. For example, our group and others have made the observation that remote ischemic conditioning, a therapy shown to reduce myocardial infarct size 2,3 in both experimental and clinical studies, can improve survival in experimental models of hemorrhagic shock. 4,5 This therapy involves brief repetitive 5 minutes inflations and deflations of a blood pressure cuff placed around a limb to induce transient limb ischemia that then protects remote organs from longer periods of ischemia that would occur with myocardial infarction, stroke, or hemorrhagic shock. As a potential therapy against hemorrhagic shock in the battlefield, remote ischemic conditioning makes sense as it could be applied prophylactically before a soldier went into a high-risk combat situation. That is, it could serve as a “vaccination” against the deleterious effects of blood loss.
There are other potential cardioprotective therapies that could be applied to the treatment of hemorrhagic shock. One potential therapy would be the anesthetic agent isoflurane. Isoflurane has been shown to mimic ischemic preconditioning in experimental models and reduce the size of experimentally induced myocardial infarction. 6 Some studies have suggested that isoflurane mimics the benefits of ischemic preconditioning on myocardial infarct size by activation of the KATP channel. 7,8 However, isoflurane’s potential as a therapy for hemorrhagic shock has not been systematically investigated. Our present study was to determine the effects of the cardioprotective agent, isoflurane, on long-term survival in an experimental model of hemorrhagic shock.
Methods
All procedures were approved by Huntington Medical Research Institute’s Animal Care and Use Committee and were performed in accordance with the Guidelines for the Care and Use of Laboratory Animals (NIH publication No. 85-23, National Academy Press, Washington DC, revised 2011). Huntington Medical Research Institutes is an AAALAC-accredited facility.
Surgical Procedures for Fixed-Pressure Hemorrhagic Shock Model
Sprague Dawley rats of either sex were randomly anesthetized with either intraperitoneal ketamine/xylazine (K/X, 90 and 10mg/kg, respectively; n = 12) or with isoflurane (5% isoflurane induction and 2% maintenance in room air; n = 12). The rats were intubated and ventilated with room air. The neck and groin were shaved and cleaned. Cut downs were performed to expose the left carotid artery, left jugular vein, and the left femoral artery. Polyethylene catheters were inserted into the left carotid artery for blood withdrawal, inserted into the left jugular vein for shed blood reinfusion, and inserted into the left femoral artery for blood pressure monitoring. Body temperature was continuously monitored via a rectal temperature probe and was maintained at approximately 37°C, with an adjustable heating pad placed under the rats. Prior to blood withdrawal to induce hemorrhagic shock, rats received 500 U/kg of intravenous heparin, so that blood that was withdrawn and would then be returned to the rats as part of resuscitation, would not thrombose. The predetermined hypotensive level and duration were 30 mm Hg (mean blood pressure) and 60 minutes, respectively. For each rat, blood was withdrawn during an interval of 10 to 12 minutes to reach the target blood pressure of 30 mm Hg and shed blood was stored in a heparinized 10-mL syringe. At the end of 60 minutes of shock duration, shed blood was reinfused over 30 minutes to resuscitate. Rats remained anesthetized for 1 hour during which hemodynamics were monitored (femoral artery catheter) and then were allowed to survive for 6 weeks. There were 2 applications of K/X during the surgical procedures, one was at the time of induction of anesthesia; a second one was at 10 minutes before blood withdrawal. Postoperative analgesia (buprenorphine, 0.01 mg/kg body weight, subcutaneous) was maintained for 2 days.
Blood Gas Analyses
Arterial blood samples (0.3 mL) were collected from the carotid arterial catheter at 1 hour after initiation of resuscitation for blood gas analyses, including blood pH, arterial partial pressure of carbon dioxide, arterial partial pressure of oxygen, Na, K, Cl, iCa, glucose, and lactate concentration which were measured with a blood-gas analyzer. In addition, at 1 hour after initiation of resuscitation, blood was collected for cell counts, platelet count, and other electrolytes and chemistries.
Echocardiographic Assessment
Echocardiogram was performed at baseline, 55 minutes into the shock phase, 1 hour after blood reinfusion and at 6 weeks after recovery in those rats that survived using a 15-MHz linear array transducer of a Philips ultrasound system. Left ventricular (LV) diastolic and systolic internal dimensions (LVIDd and LVIDs, mm), and LV fractional shortening (LVFS, %) were measured based on 2-dimensional parasternal short-axis views and 2-dimensional targeted M-mode tracings.
Histopathologic Analysis
As part of the terminal study, rats were euthanized at 6 weeks under deep anesthesia using ketamine/xylazine and their vital organs were stained with triphenyltetrazolium chloride for assessment of gross organ infarctions. Heart, brain, lungs, liver, and kidney were collected and fixed in 10% neutral buffered formalin and embedded in paraffin. Slides (5-µm tissue sections) were stained for hematoxylin and eosin (H&E) staining to assess tissue structure, and picrosirius red staining to assess fibrosis.
Statistical Analyses
All data are reported as mean ± standard error of mean. Student t tests were used for echocardiographic results and blood parameters. A linear mixed models analysis for repeated measures was used to analyze hemodynamic variables, testing for a group × time interaction. Differences between specific time points of hemodynamic variables are post hoc contrasts from the same mixed models. Survival analysis was performed by log-rank testing. Values were considered significant at P < .05.
Results
During the shock phase, the total withdrawn blood volume (expressed as % of the estimated total blood volume) to maintain mean blood pressure at 30 mm Hg was significantly higher in the isoflurane group (51.0% ± 1.5%) compared to the K/X group (45.3% ± 1.8%; P = .023). At 6 weeks, 1 (8.3%) of 12 rats survived in the K/X group versus 10 (83.3%; P < .001 by Fisher exact test) of 12 in the isoflurane group (Figure 1, left panel). All deaths occurred within 24 hours of shock. The rats were found dead in the cage in the morning on the day after surgery; therefore, the cause of death (24 hours) was not investigated. Rats that survived 24 hours survived for 6 weeks. Kaplan–Meier curves showing survival are shown in Figure 1 (right panel); there was a significant group difference favoring improved survival with isoflurane anesthesia compared to K/X anesthesia (by log-rank statistics: χ2 = 16.0, P < .0001).

There are significantly higher survival rate at 6 weeks after hemorrhagic shock in the isoflurane group compared to the K/X group. A, The survival rate at 6 weeks in the isoflurane group was 10/12 (83.3%), which was significantly higher than 1/12 in the K/X group (8.3%; P < .001); B, Kaplan–Meier curve showed that all deaths occurred within 24 hours of hemorrhagic shock; rats surviving past 24 hours survived for 6 weeks. Overall survival was improved in the isoflurane versus the K/X group (P < .0001).
Blood pressure and heart rate data during the acute phase of the study are shown in Figure 2. Rats were anesthetized with K/X showed significant higher blood pressure at baseline in Figure 2 because this baseline blood pressure was measured at 5 minutes after K/X application and K/X induces transient (∼10 minutes after injection) increases in blood pressure followed by a reduction in blood pressure. 9 Recovery of blood pressure (systolic, diastolic, and mean) during the resuscitation phase was significantly improved in the isoflurane group compared to the K/X group. Heart rate was significantly higher in the isoflurane group compared to the K/X group during the acute procedures.

Blood pressure and heart rate measurements begin about 5 minutes prior to blood-shedding. A, The time course of systolic arterial pressure is significantly different for the 2 groups (group × time interaction term, F(30,657) = 3.54, P < .0001); B, The time course of diastolic arterial pressure is significantly different for the 2 groups (group × time interaction term, F(30,657) = 3.32, P < .0001); C, The time course of mean arterial pressure is significantly different for the 2 groups (group × time interaction term, F(30,657) = 3.62, P < .0001); D, The time course of pulse pressure is significantly different for the 2 groups (group × time interaction term, F(30,657) = 2.74, P < .0001); blood pressure decreased gradually after initiation of blood withdrawing and maintained at 30 mm Hg during 60 minutes of shock phase. Then the blood pressure rapidly increased after initiation of shed blood reinfusion in both groups but significantly higher in the isoflurane group. E, The time course of heart rate is not significantly different for the 2 groups (group × time interaction term, F(30,657) = 0.75, P = .8), but there is a significant main effect for group (F(1,22) = 123.55, P < .0001). Absolute heart rate remained significantly higher in the isoflurane group compared to the K/X group during the entire time course, including baseline, and the shock and resuscitation phase (all P < .0001). *Indicates the time points, at which there is a significant difference between the 2 groups.
Echocardiographic data are shown in Table 1. There were no differences in baseline echocardiographic measurements. During shock, diastolic and systolic diameters were smaller in the K/X group. Left ventricular fractional shortening was higher during the shock phase in the K/X group compared to the Isoflurane group. At 1 hour after reinfusion of blood, diastolic diameter in the K/X group remained smaller than in the Isoflurane group, but the systolic diameter and LVFS did not differ between the 2 groups at that time. At 6 weeks after hemorrhagic shock, the LVFS was 44.4% ± 2.4% (n = 10) in the isoflurane group; it was comparable with the LVFS at the baseline before hemorrhagic shock by paired t test (48.7% ± 2.0%, n = 10; P = .13).
Echocardiographic Results.

Abbreviations: ID, internal dimension; LVFS, left ventricular fractional shortening.
At 1 hour after resuscitation with shed blood, K/X was associated with lower pH, lower level of standard bicarbonate concentration and oxygen saturation (SO2%), and more negative base excess and elevated anion gap compared to the isoflurane group (Table 2). In addition, K/X was associated with elevated potassium, sodium, and chloride levels compared to isoflurane (Table 2). Platelet counts were preserved and there was less elevation of white blood cell counts in the isoflurane compared to the K/X groups. There was a nonsignificant trend toward a higher bicarbonate level in the isoflurane group compared to the K/X group. There were no differences in the PO2, PCO2, hematocrit, hemoglobin, or glucose levels between the 2 types of anesthesia.
Comparison of Blood Parameters Between Isoflurane and K/X Groups.a
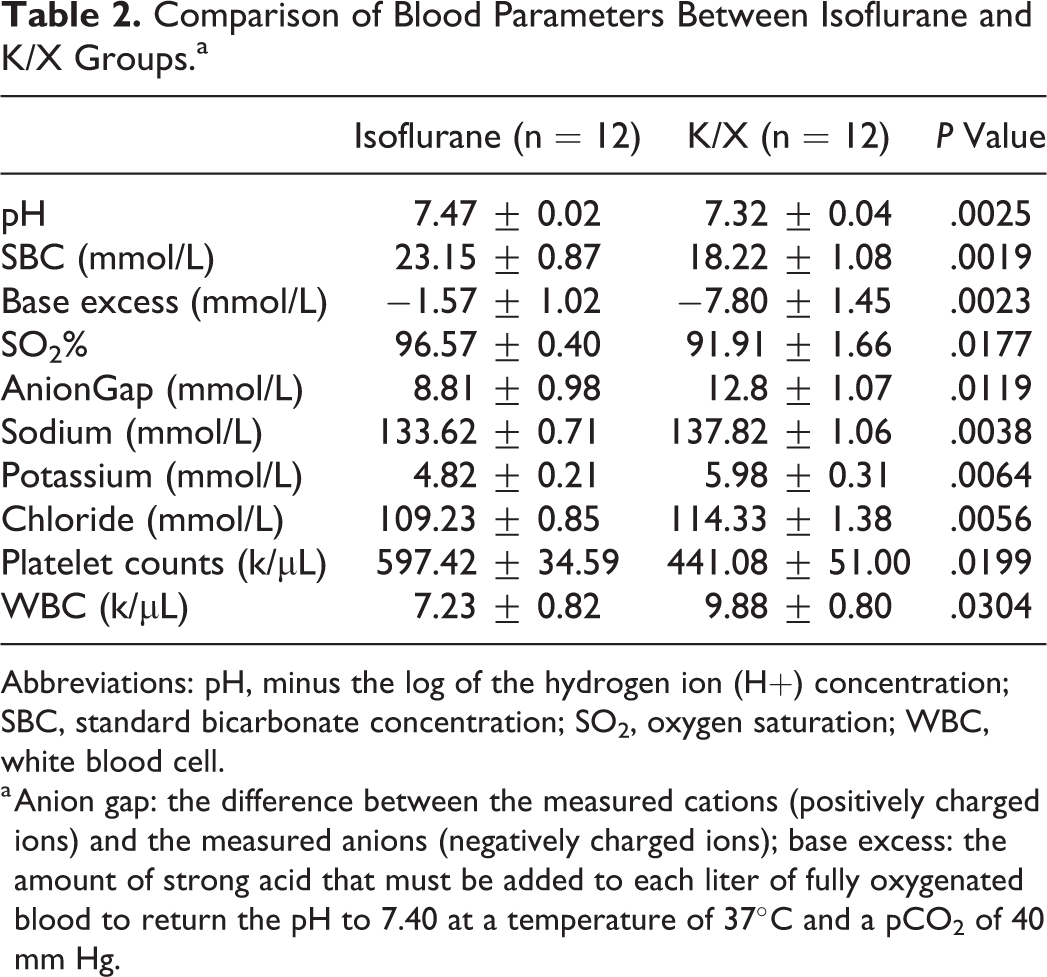
Abbreviations: pH, minus the log of the hydrogen ion (H+) concentration; SBC, standard bicarbonate concentration; SO2, oxygen saturation; WBC, white blood cell.
a Anion gap: the difference between the measured cations (positively charged ions) and the measured anions (negatively charged ions); base excess: the amount of strong acid that must be added to each liter of fully oxygenated blood to return the pH to 7.40 at a temperature of 37°C and a pCO2 of 40 mm Hg.
Analysis of organs from the survivors, stained using triphenyltetrazolium chloride, showed no gross infarcts in the hearts, kidneys, or livers. There was evidence of a small brain infarction in one of the surviving rats in the K/X group. There was no evidence of infarction in the brains of the isoflurane-treated rats (Figure 3). Of interest, there were no gross neurologic abnormalities in any of the rats that survived for 6 weeks. There was no inflammation, fibrosis, or necrosis in H&E-stained heart, kidney, or liver sections. Hematoxylin and eosin–stained lung sections showed lung lesions, including thickened alveolar walls, lost alveoli morphology, erythrocyte leakage, and mononuclear cell infiltration in K/X and isoflurane group. There was no difference in lung lesions between these 2 groups (Figure 4).

At 6 weeks, all of the surviving rats were neurologically intact. Pathology demonstrated brain infarction in the 1 surviving rat that received ketamine/xylazine (panels A and C); no brain infarction was observed in the 10 surviving rats that received isoflurane (panels B and D). The top panels are unstained and the bottom panel shows the same sections stained with triphenyltetrazolium chloride. A and C, brain from the ketamine/xylazine group; B and D, brain from the isoflurane group.
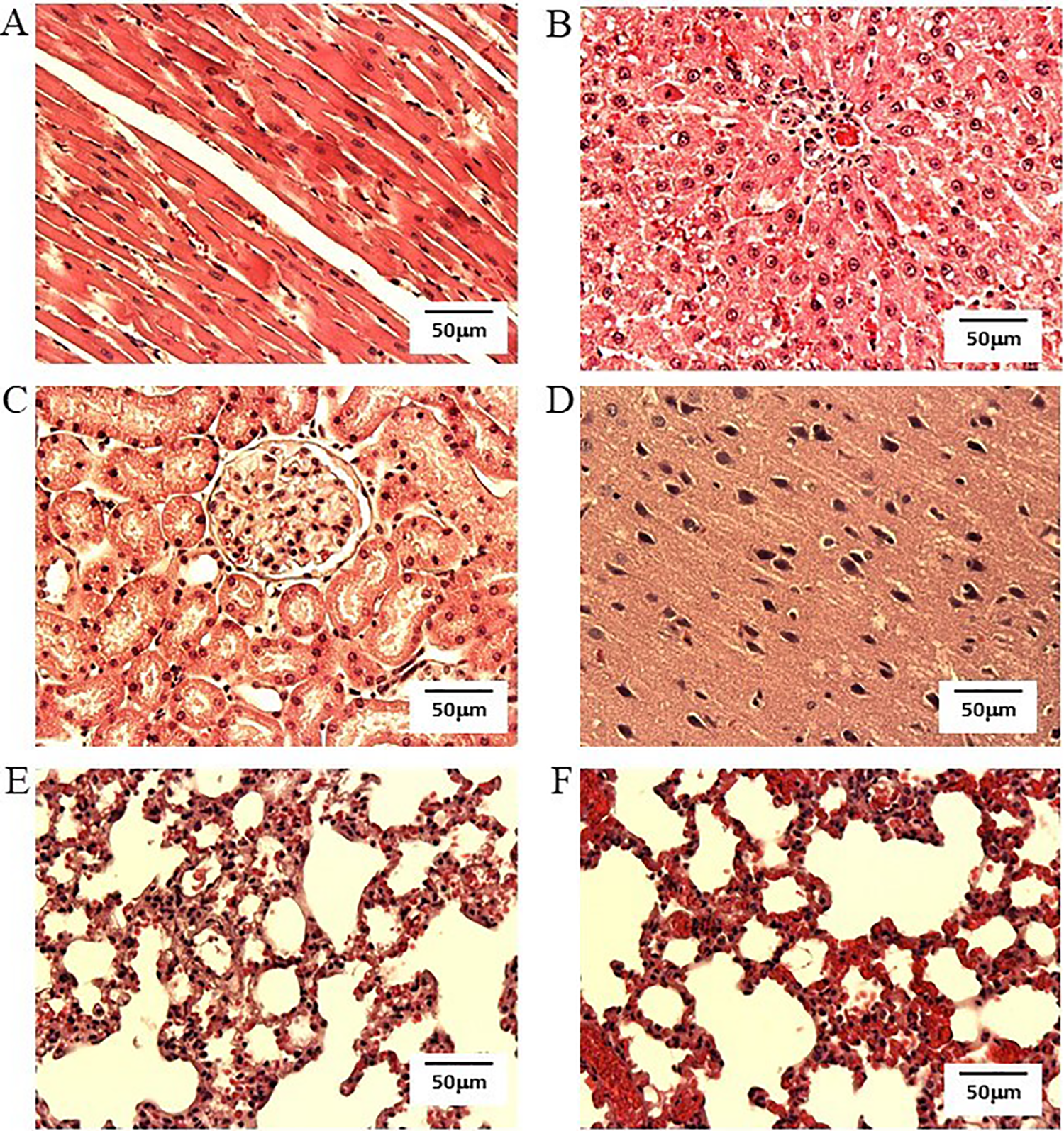
Images from hematoxylin and eosin–stained sections of heart, liver, kidney, brain, and lung (magnification ×20). A, Representative image of heart in isoflurane group. B, Representative image of liver in isoflurane group. C, Representative image of kidney in isoflurane group. D, Representative image of brain in isoflurane group. E, Representative image of lung in isoflurane group. F, Representative image of lung in K/X group. The histology of heart, liver, and kidney appeared normal in the isoflurane and K/X group. The histology of brains was normal in the isoflurane group. Lung lesions were observed in both groups (Scale bar = 50 µm).
Discussion
The main findings of the present study were that isoflurane significantly improved long-term survival in an experimental hemorrhagic shock model; improved the recovery of blood pressure; preserved LV diastolic dimension during the early resuscitation phase; and improved blood pH, electrolytes, SO2 levels, compared to ketamine/xylazine anesthesia. Of note, during shock, diastolic and systolic LV diameters were smaller in the K/X group, which may have represented less intravascular volume, perhaps due to more vascular leak with this anesthetic cocktail. Note that this occurred despite the fact that more blood volume had to be withdrawn from the isoflurane group in order to maintain a blood pressure of 30 mm Hg during the shock phase. Left ventricular fractional shortening was higher during the shock phase in the K/X group compared to the isoflurane group, possibly representing a hyper-contractile state to maintain cardiac output as a compensation for the lower volumes of blood during shock. At 1 hour after reinfusion of blood, diastolic diameter in the K/X group remained smaller than in the isoflurane group, but the systolic diameter and LVFS did not differ between the 2 groups at this time.
Few previous studies have examined the protective effects of isoflurane on survival after hemorrhagic shock and none that we could find that examined the long-term outcome. One study by Zhang et al 10 studied rats subjected to a hemorrhagic shock protocol and observed that intralipid emulsified isoflurane (a preparation that can be administered intravenously) improved survival compared to no treatment at 7 days (32.5% control group vs 55.0%; P < .05); however, inhaled isoflurane alone did not improve survival (37.5%) compared to controls. The rats in the control group were anesthetized intraperitoneally with 2% sodium pentobarbital. The emulsified isoflurane reduced malondialdehyde formation and increased superoxide dismutase activity of the mitochondria of both liver and lung. Emulsified isoflurane also improved liver enzymes and reduced protein and white blood cells in the bronchoalveolar lavage fluid and reduced liver and lung apoptosis. Isoflurane alone did increase liver mitochondrial superoxide dismutase levels and reduced other evidence of liver damage. Why emulsified isoflurane was effective in improving survival in this study, but inhaled was not, is not clear. This study does differ from ours in that we observed a protective effect of inhaled isoflurane on survival and we examined survival out to 6 weeks.
Platelets may play an essential role in trauma/hemorrhagic shock–induced acute lung injury. In a study by Harr et al, 11 rats were anesthetized by either pentobarbital or inhaled isoflurane. Then the rats were subjected to trauma (laparotomy) plus hemorrhagic shock (mean arterial pressure, 30 mm Hg × 45 minutes). Post-traumatic acute lung injury was determined by protein and pulmonary immunofluorescence bronchoalveolar lavage fluid. Pretreatment with isoflurane decreased trauma/hemorrhagic shock–mediated acute lung injury through platelet inhibition, in part, through inhibition of the platelet adenosine diphosphate pathway.
Bahrami et al 12 studied the short-term effects of 3 anesthetic agents on hemodynamics and organ injury in a rat model of hemorrhagic shock. Under normal conditions, ketamine–diazepam anesthesia resulted in a higher systemic vascular resistance than isoflurane. Isoflurane was associated with a higher cardiac output than ketamine–diazepam or ketamine–xylazine under normal conditions. After resuscitation from hemorrhagic shock, cardiac index was higher and systemic vascular resistance was lower in the isoflurane group compared to the ketamine–diazepam group. Although shed volume was greater in the isoflurane group, these rats did not decompensate within the same time frame as the ketamine–diazepam or ketamine–xylazine groups. Biological markers of tissue damage were less with isoflurane compared to ketamine–diazepam. Damage assessed by histology to the adrenal glands, kidney and liver were more pronounced in rats anesthetized with ketamine–diazepam and ketamine–xylazine compared to the isoflurane group. The authors concluded that the effect of different anesthetic agents should be taken into account when studying models of hemorrhagic/traumatic stress. Our current study extends findings in these earlier studies by confirming a protective effect of isoflurane but showing that in a standardized model of pressure-controlled hemorrhagic shock, isoflurane improves long-term survival at 6 weeks and improves aspects of blood pressure recovery, and early blood gas values with improvement in SO2, less acidosis, improved electrolytes, and certain hematologic improvements.
There are some important potential clinical implications of the current research. In the battlefield, anesthesia with ketamine is common 13 ; however, our study questions whether this therapy is the best for preserving tissue from ischemia/reperfusion injury. Hemorrhagic shock and resuscitation are, in essence, a form of total body ischemia/reperfusion injury. Using agents that will reduce such injury may preserve organs and enhance survival. Although gas anesthesia with isoflurane may not be as convenient, the present study suggests that its known cardioprotective properties extend to the entire body in the setting of total body ischemia/reperfusion injury as occurred in hemorrhagic shock and might be a better choice. There are efforts to miniaturize gas anesthesia delivery that could make this form of therapy easier to use in the field. 14
One limitation of the study is that the mechanism of isoflurane’s protective effect was not determined in the present study, but it is likely that the mechanism is similar to isoflurane’s cardioprotective effect and therefore could involve the KATP channel opening property. In experimental models, isoflurane has cardioprotective effects and can reduce myocardial infarct size when applied in a preconditioning fashion 7,15,16 and in a postconditioning fashion. 8,17 Isoflurane was shown to reduce myocardial infarct size when started at 1 hour into a 6-hour permanent coronary artery occlusion model. 18 In addition, isoflurane was shown to reduce damage in an experimental model of Takotsubo cardiomyopathy. 19 This anesthetic agent reduced damage to isolated cells that were deprived of oxygen and glucose. 20 Several studies showed that isoflurane treatment improved the recovery of cardiac function in models of stunned myocardium and preserved adenosine triphosphate levels in stunning models. 19,21 -23 While the mechanism(s) by which isoflurane has these protective effects is still being studied, several likely candidates include activation of the KATP channel 7 which may include the KATP channel of the mitochondria. 8 Administering KATP channel blocking agents inhibited the beneficial effects observed with isoflurane in many of these studies. Opening of the KATP channel has also been suggested as a mechanism by which ischemic preconditioning shows benefits. The concept is that when the KATP channel opens, potassium exits the cell, bathing the external membrane of the cell, which then induces a local cardioplegic effect; much like surgeons would use potassium to quiet the heart during cardiopulmonary bypass procedures. Another limitation of this study is that peripheral resistance was not measured; this valuable marker should be studied in future work.
In conclusion, in this experimental model of hemorrhagic shock, isoflurane demonstrated protective effects including a profound survival advantage over ketamine and xylazine anesthesia. Clinical studies examining the efficacy of isoflurane in the situation of hemorrhagic shock (soldiers in battle, car accidents, gunshot wounds, and blood loss due to other trauma) should be considered in the future.
Footnotes
Authors’ Note
Opinions, interpretations, conclusion, and recommendations are those of the authors and are not necessarily endorsed by the Department of Defense.
Author Contributions
The authors made substantial contributions to the conception or design of the work (R.A.K.) or to the acquisition, analysis, or interpretation of data for the work (W.D., J.S., J.C., R.A.K.); participated in critically revising the manuscript (W.D., J.S., J.C., R.A.K.); approved the final version to be published (W.D., J.S., J.C., R.A.K.); and agreed to be accountable for all aspects of the work (W.D., J.S., J.C., R.A.K.).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Dr Robert A. Kloner is the principal investigator of this work, and the project was supported by the Office of the Assistant Secretary of Defense for Health Affairs, through the Peer-Reviewed Medical Research Program under Award No.W81XWH-16-1-0606.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Office of the Assistant Secretary of Defense for Health Affairs, through the Peer-Reviewed Medical Research Program under Award No.W81XWH-16-1-0606.
