Abstract
Introduction:
Myocardial infarction is one of the leading causes of morbidity and mortality worldwide. Cellular interactions of red blood cells (RBCs) and platelets with endothelial cells and cardiomyocytes play a crucial role in cardiac ischemia/reperfusion (I/R) injury. However, addressing the specific impact of such cell-to-cell interactions in commonly employed in vivo models of cardiac I/R injury is challenging due to overlap of neuronal, hormonal, and immunological pathways. This study aimed to refine a Langendorff-based ex vivo transfer model to evaluate the impact of specific blood components on cardiac I/R injury.
Material and methods:
Murine whole blood, defined murine blood components (RBCs, platelet-rich plasma [PRP], and platelet-poor plasma [PPP], respectively) as well as human RBCs were loaded to the coronary system of isolated murine hearts in a Langendorff system before initiating global ischemia for 40 minutes. Following 60 minutes of reperfusion with Krebs Henseleit Buffer, left ventricular function and coronary flow were assessed. Infarct size was determined by specific histological staining following 120 minutes of reperfusion.
Results:
Loading of murine whole blood to the coronary system of isolated murine hearts at the beginning of 40 minutes of global ischemia improved left ventricular function after 60 minutes of reperfusion and reduced the infarct size in comparison to buffer-treated controls. Similarly, isolated murine RBCs, PRP, and PPP mediated a protective effect in the cardiac I/R model. Furthermore, human RBCs showed a comparable protective capacity as murine RBCs.
Conclusion:
This Langendorff-based transfer model of cardiac I/R injury is a feasible, time-, and cost-effective model to evaluate the impact of blood components on myocardial infarction. The presented method facilitates loading of blood components of genetically modified mice to murine hearts of a different mouse strain, thus complementing time- and cost-intensive chimeric models and contributing to the development of novel targeted therapies.
Introduction
Myocardial infarction (MI) is one of the leading causes of morbidity and mortality, especially in the Western world. 1 The pathophysiological causation of MI is a sudden occlusion of a coronary artery leading to ischemic injury of the affected myocardium. Treatment of choice is the catheter-based revascularization of the culprit lesion. 2 However, it is known that not only the ischemic insult but also reperfusion of the ischemic area causes myocardial damage. 3 Recent studies suggest that cell-to-cell interactions between endothelial cells and cardiomyocytes with red blood cells (RBCs) and platelets might play an important role in the pathogenesis of cardiac ischemia/reperfusion (I/R) injury. 4,5 However, common in vivo models of MI fail to address unequivocally these effects due to interference of neuronal, hormonal, and immunological pathways.
Since the development of the ex vivo-perfused mammalian heart preparation by Oscar Langendorff in the end of the 19th century, 6 the Langendorff preparation has proven invaluable for cardiovascular research. Traditionally, for the Langendorff preparation, mammalian hearts are perfused ex vivo with gassed Krebs Henseleit Buffer (KHB) containing essential electrolytes as well as glucose in a pressure- or flow-constant manner under continuous registration of physiological parameters, such as coronary flow, heart rate, and left ventricular function. 7 This method allows for investigating cardiac physiology and the impact of pathophysiological and pharmacological stressors on the heart function. Thus, it is not surprising that the isolated perfused heart model fundamentally contributed to the current understanding of heart physiology. However, the traditional buffer-perfused working heart model has several limitations, as it cannot address fundamental interactions of blood cells and blood-borne molecules with cardiomyocytes and endothelial cells. These cell-to-cell interactions appear very important, especially in pathophysiological conditions such as myocardial I/R injury. Thus, several attempts were made to establish whole blood–perfused isolated heart preparations 8,9 or preparations with continuous perfusion with RBC- or platelet-augmented crystalloid buffers. 5,9 -11 These attempts bear a high methodical complexity and pose several limitations. For all models with continuous perfusion of RBCs, the occurring hemolysis constitutes a major problem. 12 For ex vivo perfusion of hearts with whole blood or blood cell-augmented crystalloid buffers, larger amounts of blood are needed thus limiting experiments to larger animal species such as rats or rabbits. Accordingly, these efforts could not utilize material from mice bearing the advantages of the availability of a wide array of gene-modified strains and less cost intensity. Alternatively, isolated RBCs from other species (bovine or human) have to be used. 12 Furthermore, in many attempts, the blood is diluted to concentrations below the physiological range most likely influencing the results. In a novel approach, Yang and colleagues loaded mitochondrial DNA fragments to the coronary system during the period of global ischemia leading to a significant increase in the infarct size. 13 In a similar attempt, Yang et al loaded murine blood to the coronary circulation of isolated murine hearts in the Langendorff apparatus at the beginning of a global ischemia. 14,15 Afterward, the hearts were reperfused with KHB. This experimental model avoided the problem of hemolysis caused by continuous perfusion of blood and minimized the required amount of blood to less than 1 mL. The studies also demonstrated the feasibility to investigate human RBCs in rodent hearts rendering this model more relevant for investigations of human disease. However, this promising method requires further characterization in detail: Yang et al did not in detail investigate the presence of the blood components in the coronary system throughout the ischemic period or how fast the blood components were washed out following reperfusion. 14,15 The lack of these data impede the comparability of the data with the results of other studies using different models of cardiac I/R injury. Furthermore, the experiments of Yang et al were restricted to whole blood, RBCs, and plasma. 14,15
Here, we aimed to further characterize and refine this approach to establish a Langendorff-based ex vivo transfer model allowing to evaluate the impact of specific blood components on cardiac I/R injury.
Materials and Methods
Volunteer Recruitment
The study was approved by the institutional ethic board of the Heinrich-Heine-University Düsseldorf (No. 5903R) and adhered to the principles of the Declaration of Helsinki. Written informed consent was obtained from all volunteers. Six (3 men and 3 women) healthy volunteers aged 23 to 30 years with no medical history and no medication except oral contraceptives in women were recruited between August and October 2017.
Isolated Mouse Heart Perfusion
All experiments were conducted in accordance with the European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes (Council of Europe Treaty Series No. 123) and 2010/63/EU. C57BL/6J mice were purchased from Janvier Labs (Saint-Berthevin, France). Animal care was in accordance with the institutional guidelines with access to food and water ad libitum.
For characterization of the impact of blood and distinct blood components on the cardiac I/R injury, a modified protocol for isolated mouse heart perfusion was used as shown in Figure 1. 14 Briefly, 9- to 15-week-old male C57BL/6J mice were anesthetized with intraperitoneal (IP) injection of 50 mg/kg ketamine (Pfizer Pharma PFE GmbH, Berlin, Germany) and 40 mg/kg xylacin (Bayer, Leverkusen, Germany). Following injection (IP) of heparin (1000 IU/mouse; Rotexmedica, Trittau, Germany ), hearts were excised rapidly and placed in ice-cold KHB. The ascending aorta was cannulated, and hearts were mounted into a Langendorff-apparatus (Hugo Sachs, March-Hugstetten, Germany) and perfused at a constant pressure of 100 mm Hg with modified gassed (5% CO2 in O2) KHB containing 118 mM NaCl, 4.7 mM KCl, 0.8 mM MgSO4, 25 mM NaHCO3, 1.2 mM KH2PO4, 5 mM glucose, 110 mM Na-pyruvate, and 2.5 mM CaCl2 at 37°C. A water-filled balloon connected to a pressure transducer was inserted through the mitral valve into the left ventricle (LV) for recording of isovolumetric LV developed pressure, its positive and negative first derivate (+dP/dt and −dP/dt), and the LV end-diastolic pressure (LVEDP). Coronary flow was continuously registered using an ultrasound flow probe (Type MC1PRB-HSE for HSE-TTFM; Hugo Sachs, March-Hugstetten, Germany) positioned in the Langendorff system upstream of the cannulated heart (Figure 1). At the beginning of the experiment, the LVEDP was set to 5 to 15 mm Hg. Hearts were paced at a constant rate of 600 beats/min. After a stabilization period of 20 minutes, hearts were subjected to 20 seconds of global zero flow ischemia to test the coronary reserve. Following the transient ischemia, hearts were allowed to recover for at least 5 minutes. Hearts were excluded from the analysis when they met one of the following exclusion criteria after the 5-minute recovery phase: (1) coronary flow >4 mL/min, (2) LV developed pressure <50 mm Hg, or (3) a coronary flow reserve revealed by a transient ischemia <70% of the baseline flow. Zero flow global ischemia was applied by interruption of the coronary flow for 30 to 45 minutes as indicated. In the first minute of the global ischemia, 400 µL of blood or defined blood components were loaded to the coronary system of the murine hearts via a side arm using a syringe driver set to 0.4 mL/min. Control hearts were loaded with 400 µL KHB. Despite the presence of blood products in the coronary system during ischemia, no-flow ischemia resulted in cessation of LV contractile activity as reported before. 16 After the defined period of ischemia, flow was reestablished. Several minutes upon reperfusion, LV contractile activity restarted. Hearts were reperfused with KHB for 120 minutes (Figure 1). During perfusion, the temperature of the hearts was kept at 37°C using prewarmed KHB and by perfusion of the walls of the organ bath chamber with warm water (38°C). The buffer was also prewarmed at a temperature of 38°C in a water bath. After passing the Langendorff apparatus, the KHB had a temperature of 37°C when it perfused the hearts. During ischemia, the hearts were immersed in an organ bath at 37°C to prevent dropping of the temperature in the absence of prewarmed KHB perfusion.

Experimental design. A, Schematic illustration of the experimental setup. B, Setup of the Langendorff apparatus. Left ventricular function was monitored by a water-filled balloon inserted into the left ventricle connected to a pressure sensor. Hearts were paced at a frequency of 600 bpm. The 2-way tap served to switch off the perfusion for global ischemia. Blood or blood components were loaded to the coronary system via the side arm. C, Timeline of the experiment. After 20 minutes of stabilization, hearts were subjected to 20 seconds of ischemia to test the coronary reserve. After a 5-minute recovery period, baseline values were recorded. At the beginning of the 40-minute global ischemia, blood or blood components were loaded to the coronary system. After 120 minutes of reperfusion, hearts were taken out of the Langendorff system and subjected to TTC staining. LVEDP indicates left ventricular end diastolic pressure; LVP, left ventricular pressure; LD, Langendorff; KHB, Krebs Henseleit Buffer.
Infarct Staining
After the reperfusion period of 120 minutes, hearts were tightly wrapped into plastic wrap and were frozen at −20°C for 1 hour. Hearts were cut into 6 approximately 1-mm-thick slices and stained in a 2,3,5-triphenyltetrazolium chloride (Sigma Aldrich, St. Louis, Missouri) solution (1% [w/v])dissolved in phosphate buffer (80% [v/v] 0.1 M Na2HPO4 and 20% [V/V] 0.1 M NaH2PO4, pH adjusted to 7.4) for 4 minutes at 37°C. Slices were photographed under a stereomicroscope. Viable (red) and necrotic (white) areas were analyzed by computer-assisted planimetry (Diskus software; Hilgers Technisches Büro, Königswinter, Germany). Infarct size was calculated as percentage of the left and right ventricular volume (% LV + RV).
Histology
For histological analyses, 2 hearts were taken out of the Langendorff system at each of the following time points of the Langendorff protocol with loading of whole blood or RBC-enriched KHB: (1) after stabilization of the heart in the Langendorff system, (2) directly after RBC loading at the beginning of global ischemia; (3) after global ischemia before reperfusion, and (4) after 60 minutes of reperfusion. Hearts were fixed in 4% paraformaldehyde and processed for routine paraffin histology. Sections were cut using a microtome (Leica, Wetzlar, Germany) and hematoxylin/eosin-staining was performed.
Blood Sampling and Isolation of Blood Components
Murine blood was collected from anesthetized (50 mg/kg ketamine and 40 mg/kg xylacin) 11- to 15-week-old male C57BL/6J mice via puncture of the heart using heparinized 1-mL syringes. Human blood samples were obtained from the cubital vein using heparinized 10-mL syringes. Blood samples were transferred to heparinized tubes.
Before loading of murine whole blood to the coronary system of isolated murine hearts, blood samples were prewarmed at 37°C for 30 minutes. For isolation of RBCs, blood samples were immediately centrifuged at 830g for 10 minutes at 4°C. Plasma and the buffy coat were discarded. The RBCs were washed twice with KHB and stored at 4°C up to 24 hours before use. Before loading to the coronary system of isolated murine hearts, RBCs were diluted with KHB resulting in a hematocrit (HCT) of 20% to 50% as indicated and prewarmed at 37°C for 30 minutes.
For isolation of platelet-poor plasma (PPP), blood samples were immediately centrifuged at 830g for 10 minutes at 4°C. Platelet-poor plasma was transferred to new tubes, and 3 parts of PPP were diluted with 2 parts of KHB to obtain physiologic concentrations of all plasma components and prewarmed at 37°C for 30 minutes before loading to the coronary system of isolated murine hearts.
For isolation of platelet-rich plasma (PRP), blood samples were centrifuged at 210g for 10 minutes at 18°C. The PRP was transferred to a new tube and diluted (6:4) with KHB to obtain physiologic platelet concentrations. Samples were incubated for 30 minutes at room temperature before loading to the coronary system of isolated murine hearts.
Blood Count Analyses and Hemoglobin Measurements
Blood counts (n = 7) were performed on murine blood before loading to the Langendorff perfused hearts and on the effluent of the hearts collected while loading of the blood to the hearts at the beginning of global ischemia and during the first minute of reperfusion using VetAbc Animal Blood Counter (SCIL animal care company, Viernheim, Germany) For blood count analyses, the effluent was collected. For detection of hemoglobin (Hb) concentrations in the effluent at different points (during the 1st, 5th, 10th, 20th, 30th, 40th, 50th, and 60th minute of reperfusion) of the protocol, 1 to 2 mL of the effluent from the murine hearts in the Langendorff system were collected and stored at −80°C. Hemoglobin concentrations were detected using a DetectX Hemoglobin Colorimetric Detection Kit from Arbor Assays (Ann Arbor, Michigan) following the manufacturer’s instructions.
Blood Gas Analyses and Calculation of O2 Content and O2 Extraction
Blood gas analyses were performed on KHB and in KHB-resuspended washed RBCs with a HCT of 40% using an ABL800 FLEX Radiometer (Copenhagen, Denmark) following the manufacturer’s instruction. O2 content was calculated using the following formula: cO2 = sO2 × Hb [g/dL] × 1.34 [mL/g] + paO2 [mm Hg] × 0.0031 [1/mm Hg × mL/dL]. O2 extraction was calculated using the following formula: before/after heart perfusion DO2 = CbeforeO2 − CafterO2 = (SbeforeO2 × Hb [g/dL] × 1.34 [mL/g] + pbeforeO2 [mm Hg] × 0.0031 [1/mm Hg × mL/dL]) − (SafterO2 × Hb [g/dL] × 1.34 [mL/g] + pafterO2 [mm Hg] × 0.0031 [1/mm Hg × mL/dL]).
Statistical Analyses
LV-developed pressure, +dP/dt, −dP/dt, and coronary flow registered after 60 minutes of reperfusion are expressed in percentage of the preischemic baseline values. The LVEDP registered after 60 minutes of reperfusion is expressed as absolute value. Data are presented as mean ± standard deviation. Multiple comparisons were analyzed by 1-way analysis of variance (ANOVA). When a significant difference was detected, 1-way ANOVA was followed by Tukey or multiple comparison post hoc tests. For comparison of 2 groups, an unpaired Student t test was used. P < .05 was set as a threshold of significance. Data were analyzed with GraphPad Prism version 6.01 (GraphPad Software, San Diego, California).
Results
For characterization of the effects of defined blood components on the cardiac I/R injury, a modified Langendorff preparation was used and further refined. The experimental setup is shown in Figure 1. First, we evaluated the appropriate time of global ischemia in KHB-perfused mouse hearts in the Langendorff preparation by varying the time of global ischemia between 30 and 45 minutes. A global ischemia time of 40 minutes was determined as optimal with a resulting infarct size of approximately 50% of the LV and right ventricular volume following 120 minutes of reperfusion (Figure 2; Supplemental Table 1).
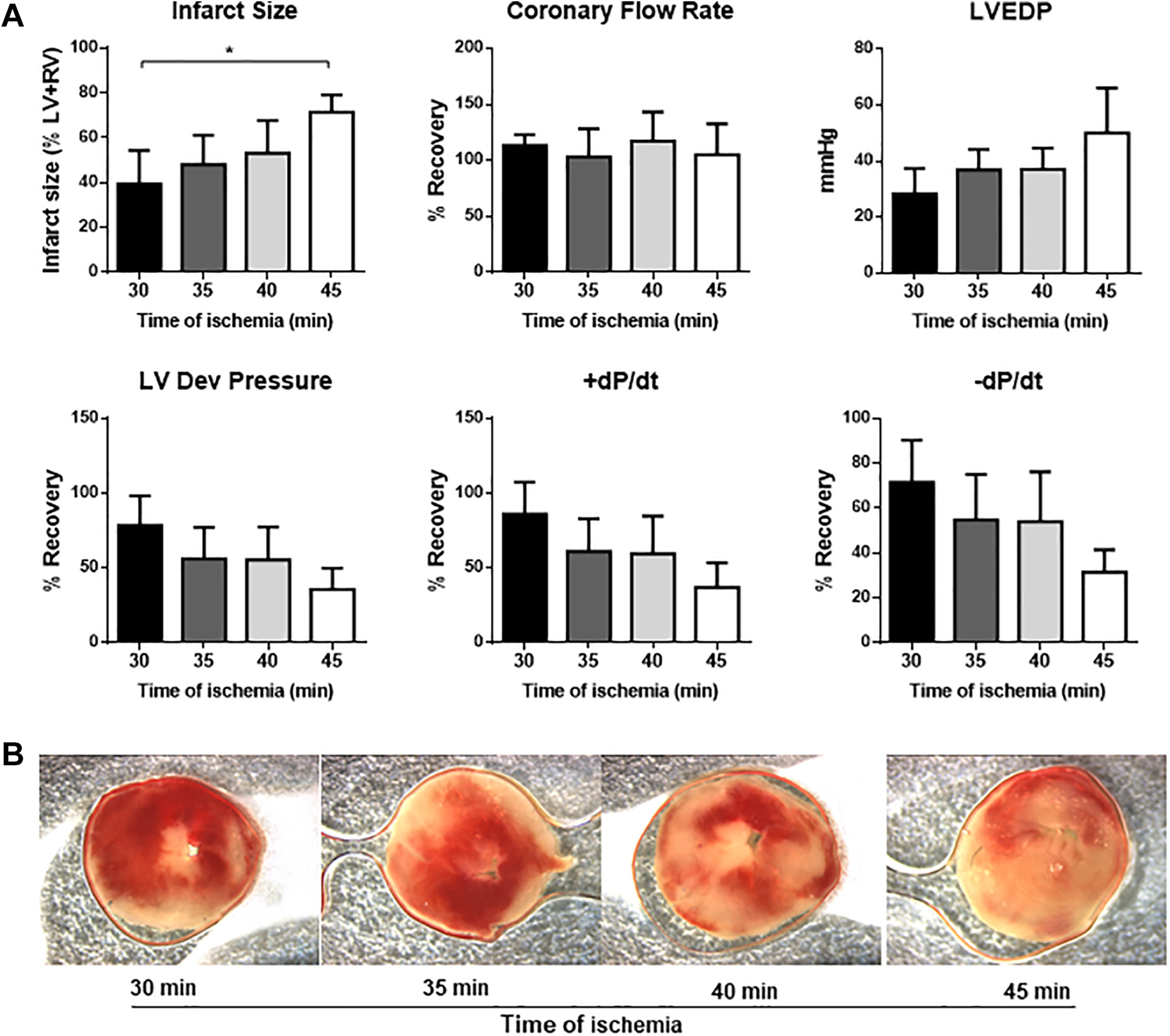
Evaluation of the optimal duration of global ischemia. A global ischemia duration of 40 minutes of buffer-treated hearts results in an infarct size of approximately 50% and a recovery of left ventricular function of 50% compared to baseline values. Thus, this condition is optimal to test the effects of distinct blood components on the cardiac I/R injury (n = 3-4 per group). A, Impact of the duration of global ischemia on infarct size, coronary flow, and left ventricular systolic and diastolic function. B, Representative pictures of TTC-stained midventricular sections of murine hearts subjected to global ischemia for 30 to 45 minutes in the Langendorff apparatus. LV indicates left ventricle; RV, right ventricle; LV dev pressure, left ventricular developed pressure; LVEDP, left ventricular end diastolic pressure; +dP/dt, −dP/dt, first positive and negative derivate of LV pressure; TTC, 2,3,5-triphenyltetrazolium chloride. Data are shown as mean and SD. Significant differences between groups are shown (1-way analysis of variance [ANOVA]). *P < .05.
Next, murine whole blood was loaded to the coronary system via a side arm before a 40-minute period of global ischemia. Histological analyses and measurement of Hb concentrations in the effluent confirmed that no blood cells were present in the hearts before loading of blood (Figure 3). Analyses of the effluent collected during the blood loading procedure and at the beginning of reperfusion revealed slightly lower blood cell counts than the whole blood before loading most likely due to dilution with KHB in the Langendorff system. Blood counts and histologic analyses confirmed that the loaded blood remained stable within the hearts throughout the 40 minutes of global ischemia (Figure 3). Following 60 minutes of reperfusion with KHB, no blood cells were detectable in the coronary system (Figure 3). Measurements of Hb in the effluent of the hearts collected before loading of blood and during the 60 minutes of reperfusion confirmed that the RBCs were washed out rapidly upon reperfusion with KHB (Figure 3).
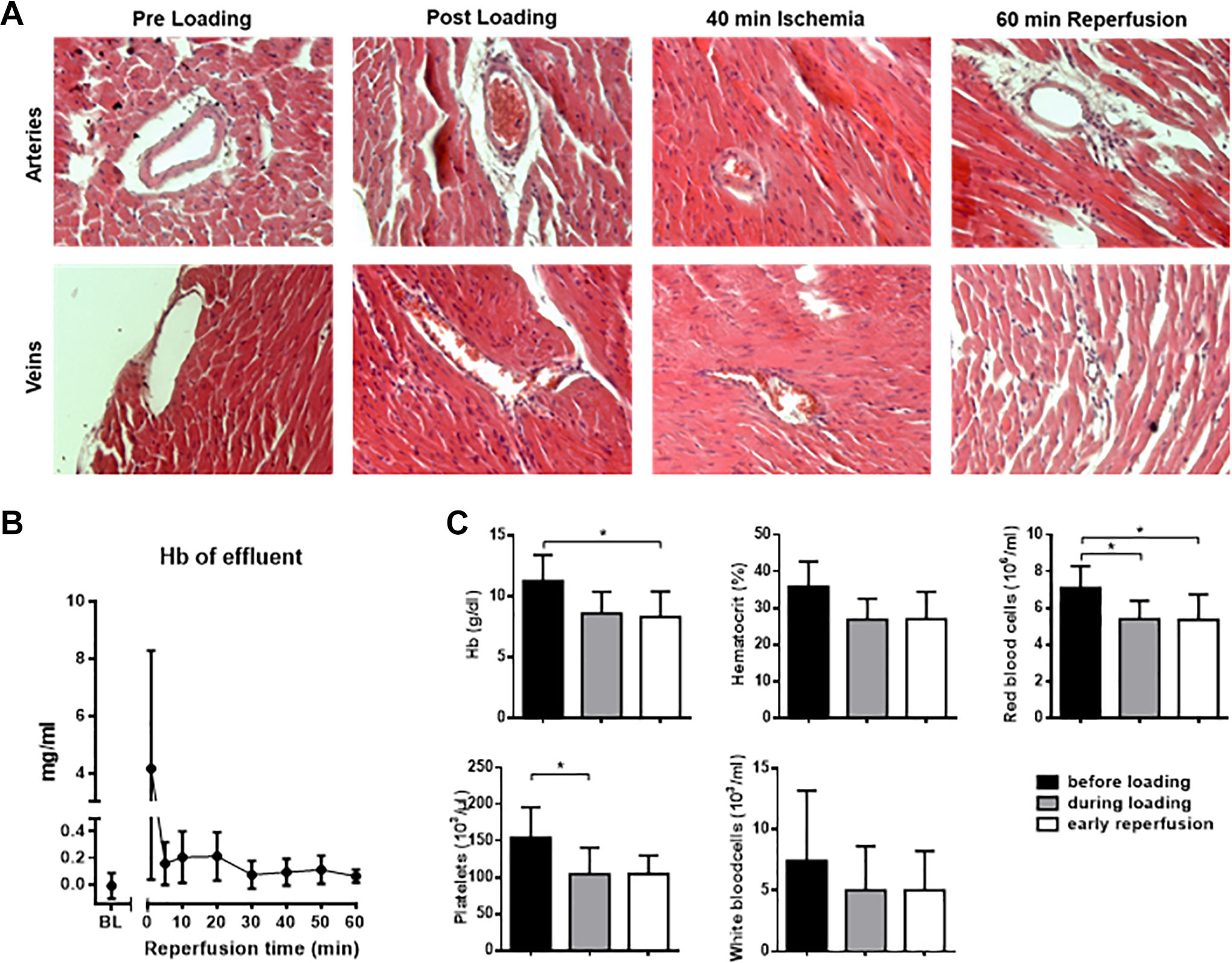
Loading of murine blood to murine Langendorff perfused hearts. A, Histological analysis of hematoxylin and eosin (HE)-stained sections of murine hearts taken out from the Langendorff system at different time points of the experiment (before loading of blood to the coronary system, directly after loading of blood to the coronary system at the beginning of global ischemia, after 40 minutes of global ischemia, and after 60 minutes of reperfusion with KHB [time point zero]; n = 2). Representative pictures are shown. B, Hemoglobin analyses of the effluent before blood loading and upon reperfusion (n = 8). C, Blood counts (n = 7) were performed on murine blood before loading to the Langendorff perfused hearts and on the effluent of the hearts collected while loading of the blood to the hearts at the beginning of global ischemia and during the first minute of reperfusion. Data are shown as mean and standard deviation (SD). *P < .05. Hb indicates hemoglobin; KHB, Krebs Henseleit Buffer.
After confirmation that loading of murine blood to the coronary system of murine hearts at the beginning of global ischemia in the Langendorff system was feasible, we evaluated the effects of murine whole blood, specific blood components on LV function and infarct size. Indeed, loading explanted hearts during ischemia with murine whole blood significantly improved LV function after 60 minutes of recovery compared to KHB-treated controls (Figure 4; Supplemental Table 2). Defined murine blood components (RBCs, PRP, and PPP) improved LV recovery as effective as murine whole blood. Furthermore, loading of murine RBCs, PRP, or PPP at the beginning of global ischemia significantly reduced the infarct size compared to buffer-treated controls (Figure 4). In a cross-species transfer approach, we loaded human RBCs to murine hearts in the Langendorff system at the beginning of global ischemia. Interestingly, human RBCs showed a cardioprotective effect comparable to that of murine RBCs in the Langendorff system (Figure 5; Supplemental Table 3).
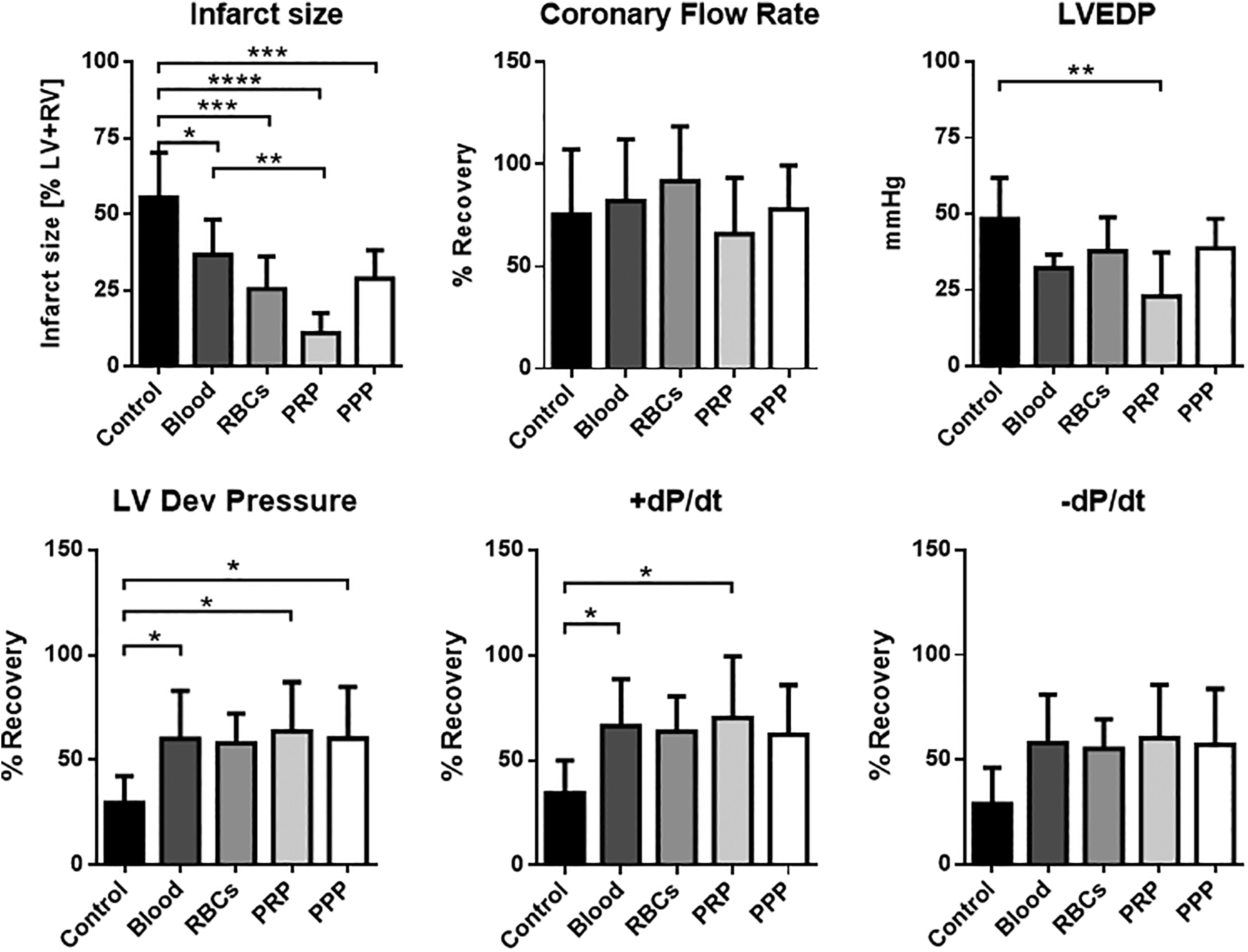
Cardioprotective effects of murine blood and blood components on ischemia–reperfusion (I/R) injury of in the Langendorff system. Langendorff-perfused mouse hearts were loaded with murine whole blood or with defined murine blood components at the beginning of 40 minutes of global ischemia. After 60 minutes of reperfusion with Krebs-Henseleit buffer, left ventricular function and the coronary flowrate were assessed. Infarct size was determined by histological analyses (TTC-Staining) after 120 minutes of reperfusion (n = 5-6 per group). Data are shown as mean and standard deviation (SD). Significances between groups are shown (1-way analysis of variance [ANOVA]). *P < .05, **P < .01, ***P < .001. LV indicates left ventricle; RV, right ventricle; LV dev pressure, left ventricular developed pressure; LVEDP, left ventricular end diastolic pressure; +dP/dt, −dP/dt, first positive and negative derivate of LV pressure; RBCs, red blood cells; PRP, platelet-rich plasma; PPP, platelet-poor plasma; TTC, 2,3,5-triphenyltetrazolium chloride.

Human and murine RBCs have a comparable protective effect on the ischemia–reperfusion (I/R) injury in isolated perfused murine hearts. Loading of human RBCs (n = 6) to Langendorff perfused murine hearts at the beginning of 40 minutes of global ischemia are equal potent in restoring the left ventricular function after 60 minutes of reperfusion and reducing the infarct size after 120 minutes of reperfusion as murine RBCs (n = 7). Control hearts (n = 6) were treated with buffer only. Data are shown as mean and standard deviation (SD). Significances between groups are shown (1-way analysis of variance [ANOVA]). *P < .05, **P < .01, ***P < .001. LVm left ventricle; RVm right ventricle; LV dev pressurem left ventricular developed pressure; LVEDP, left ventricular end diastolic pressure; +dP/dt, −dP/dtm first positive and negative derivate of LV pressure; mRBCs, murine red blood cells; hRBCs, human red blood cells.
To examine whether the abovementioned cardioprotective effects of RBCs were dose dependent, we loaded washed human RBCs diluted in KHB to different concentrations ranging from an HCT of 20% to 50% to the coronary system of Langendorff-perfused hearts at the beginning of global ischemia. All concentrations applied markedly reduced infarct size following 120 minutes of reperfusion. However, only RBC suspensions with almost physiologic HCTs of 30% or 40% could improve LV function after 60 minutes of reperfusion (Figure 6; Supplemental Table 4).

Dose-dependency of the protective effect of human RBCs on I/R injury in murine hearts. Isolated perfused murine hearts were perfused with human RBCs diluted with different concentrations of KHB resulting in a hematocrit of 20% to 50% at the beginning of 40 minutes of global ischemia (n = 4-7 per group). Control hearts were treated with KHB only. Data are shown as mean and standard deviation (SD). Significances between groups are shown (1-way analysis of variance [ANOVA]). *P < 0.05, **P < .01, ***P < .001. Ctrl indicates control; LV, left ventricle; RV, right ventricle; LV dev pressure, left ventricular developed pressure; LVEDP, left ventricular end diastolic pressure; +dP/dt, −dP/dt, first positive and negative derivate of LV pressure; RBCs, red blood cells; HCT, hematocrit; KHB, Krebs-Henseleit-Buffer.
To evaluate whether the observed RBC-mediated cardioprotective effect relied on the RBC-dependent increased oxygen supply, blood gas analyses on KHB as well as on RBC suspended in KHB were performed before and after heart perfusion. Blood gas analyses revealed an 8-fold higher oxygen supply delivered by KHB-containing RBCs with an HCT of 40% compared to KHB alone (Supplemental Figure 1).
Discussion
The concise intention of this study was to characterize a Langendorff-based assay to allow the specification of the influence of isolated blood components on myocardial I/R injury. Here, we show that 0.4 mL of blood or isolated blood components injected to the coronary system of murine hearts in the Langendorff preparation at the beginning of a 40-minute global ischemia are sufficient to fill the coronary system until the beginning of reperfusion. The fact that our histological analyses reveal that most but not all coronary vessels are filled with blood after loading blood preparations can most likely be explained with the coronary reserve. Our study shows that isolated murine RBCs, PRP, and PPP protect against the I/R injury in isolated murine hearts confirming previous observations. 4,5 Indeed, platelets may play an important role in cardiac I/R injury by releasing of numerous cardioprotective factors 17 that activate the Reperfusion Injury Salvage Kinase (RISK) and survivor activating factor enhancement (SAFE) pathways 17,18 or induce PAK1/AKT/NOS3 signaling. 19 One of these cardioprotective factors is the platelet-derived shingosine-1 phosphate (S1P) that can mediate both pro- and antiaggregatory effects via G-protein-coupled receptors on platelets. 17 Furthermore, S1P shows a cardioprotective effect by S1P1, S1P2, and S1P3 receptors in cardiomyocytes, leading to activation of the RISK and SAFE pathways. 19 Previous studies have shown that the effects of platelets on cardiac I/R injury depend on the presence and extent of platelet activation. 16 Mirabet and colleagues showed that activated platelets have a detrimental effect on LV function after myocardial I/R injury, whereas nonactivated platelets did not influence LV function after I/R injury. 16 In contrast, our results indicate that even PRP has a protective effect on the LV function after I/R injury. The discrepancy between our and the previous study might derive from the fact that we used PRP, whereas Mirabet and colleagues tested isolated platelets. The additional centrifugation necessary for platelet isolation might lead to a higher extent of platelet activation. Furthermore, Mirabet and colleagues used a syringe containing acid citrate dextrose A as anticoagulant, whereas we used heparinized syringes for blood collection. The different anticoagulants might have effects on platelet activation. Interestingly, there is no additive effect of the observed cardioprotective potential of isolated blood components when murine whole blood is loaded to the coronary system. This phenomenon might be explained by interference with leukocyte-mediated inflammatory effects. Previous studies have shown that leukocytes play an important role in the pathogenesis of MI and that high leukocyte counts are independently associated with high thrombus burden and detrimental outcome after MI. 20 Thus, the presence of leukocytes in murine whole blood might attenuate the cardioprotective effects mediated by leukocyte-free blood preparations. In previous studies on the effect of blood or RBCs on Langendorff-perfused hearts, diluted blood or RBC-enriched buffer was used with blood cell concentrations far below the physiologic range. 21 However, our study reveals that although RBCs in low concentrations can reduce the infarct size, only more physiologic concentrations confer improved LV function upon reperfusion. Thus, our results highlight the importance of physiologic blood cell concentrations for proper characterization of blood cell-mediated processes in cardiac (patho)physiology.
Our model appears much easier to handle and more cost effective than previously described attempts of blood-perfused Langendorff preparations. 8,10,21,22 Furthermore, our method paradigmatically reduces the problem of hemolysis which is one major issue in Langendorff preparations with continuous perfusion of whole blood or RBC-augmented crystalloid buffers. 12 The small amounts of blood or blood components required for the experimental setup described here facilitates the utilization of murine blood, while previously described models relied on blood of rabbits, rats, humans, or cows due to the minimally required blood volume. 5,10,12,21 Thus, our experimental setup opens the opportunity to take advantage of numerous genetically modified mouse models for further characterization of specific blood cell-mediated pathways on the cardiac I/R injury. Loading of blood components obtained from genetically modified mice to murine hearts of a different mouse strain can replace chimeric models or tissue-specific, gene-targeted models. These time- and cost-intensive models are traditionally used to examine blood cell-specific signaling pathways on the one hand and vascular cell- and cardiomyocyte-related responses on the other hand in the setting of myocardial I/R injury. 23,24 The use of blood from genetically modified mouse models to an isolated wild-type heart has in fact been demonstrated in a study in which RBCs from mice with gene-targeted deletion of endothelial nitric oxide synthase were given to isolated hearts from wild-type mice. 14 Using this approach, it was convincingly demonstrated that the cardioprotective effect of arginase inhibition was critically dependent on endothelial nitric oxide synthase in RBCs. Furthermore, it was recently demonstrated that RBCs from a disease model of type 2 diabetes (db/db) attenuated postischemic cardiac function and increased infarct size in wild-type hearts. 15 This observation extends the importance of this model to investigate the role of blood components in cardiovascular disease.
In addition, our study shows that RBCs from young and healthy human volunteers injected to the coronary system of Langendorff-perfused murine hearts exert a cardioprotective effect comparable to the influence of murine RBCs. Hence, this experimental setup can be employed as an ex vivo assay to identify RBC dysfunction in various human diseases. This was recently demonstrated by Yang et al 15 who administered RBCs from patients with type 2 diabetes to nondiabetic rat hearts. It was found that these human RBCs markedly attenuated postischemic cardiac function in comparison with RBCs from healthy controls, emphasizing the utility of this model for investigations of human disease. Furthermore, RBCs could be treated pharmaceutically ex vivo before loading to the Langendorff system as demonstrated in previous studies. 14,15 Thus, this experimental setup may help to identify new treatment strategies for various forms of RBC dysfunction.
Certain important limitations of our study should be mentioned. In the model described here, Langendorff-perfused hearts are only exposed to the blood components during the period of global ischemia. Thus, interactions of blood components with endothelial cells and cardiomyocytes before onset of ischemia or during reperfusion cannot be addressed. Furthermore, the presented method is an ex vivo model, and any extrapolation to the in vivo situation should be drawn with caution. The results of our study cannot be transferred to patients with acute MI. However, this attempt opens the opportunity to identify the effect of pharmacological modification of isolated blood components to the myocardial I/R injury or to use blood components obtained from genetically modified mice to test the influence of such modifications on the myocardial injury. Thus, this model may also help to generate hypotheses to be tested in further studies with a more clinical focus.
Conclusion
In conclusion, this Langendorff-based assay is a feasible, time-, and cost-efficient model to evaluate the impact of blood components on myocardial I/R injury. The presented method facilitates loading of blood components of genetically modified mice to murine hearts of a different mouse strain. Accordingly, this model can replace or complement traditional chimeric models and can contribute to the development of novel targeted therapies.
Supplemental Material
Supplemental Material, Supplemental_Figure_1 - A Model of Blood Component–Heart Interaction in Cardiac Ischemia–Reperfusion Injury using a Langendorff-Based Ex Vivo Assay
Supplemental Material, Supplemental_Figure_1 for A Model of Blood Component–Heart Interaction in Cardiac Ischemia–Reperfusion Injury using a Langendorff-Based Ex Vivo Assay by Johanna M. Muessig, Sema Kaya, Luise Moellhoff, Johanna Noelle, Leonie Hidalgo Pareja, Maryna Masyuk, Norbert Gerdes, John Pernow, Malte Kelm and Christian Jung in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Supplemental_Table_1_revised - A Model of Blood Component–Heart Interaction in Cardiac Ischemia–Reperfusion Injury using a Langendorff-Based Ex Vivo Assay
Supplemental Material, Supplemental_Table_1_revised for A Model of Blood Component–Heart Interaction in Cardiac Ischemia–Reperfusion Injury using a Langendorff-Based Ex Vivo Assay by Johanna M. Muessig, Sema Kaya, Luise Moellhoff, Johanna Noelle, Leonie Hidalgo Pareja, Maryna Masyuk, Norbert Gerdes, John Pernow, Malte Kelm and Christian Jung in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Supplemental_Table_2_revised - A Model of Blood Component–Heart Interaction in Cardiac Ischemia–Reperfusion Injury using a Langendorff-Based Ex Vivo Assay
Supplemental Material, Supplemental_Table_2_revised for A Model of Blood Component–Heart Interaction in Cardiac Ischemia–Reperfusion Injury using a Langendorff-Based Ex Vivo Assay by Johanna M. Muessig, Sema Kaya, Luise Moellhoff, Johanna Noelle, Leonie Hidalgo Pareja, Maryna Masyuk, Norbert Gerdes, John Pernow, Malte Kelm and Christian Jung in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Supplemental_Table_3_revised - A Model of Blood Component–Heart Interaction in Cardiac Ischemia–Reperfusion Injury using a Langendorff-Based Ex Vivo Assay
Supplemental Material, Supplemental_Table_3_revised for A Model of Blood Component–Heart Interaction in Cardiac Ischemia–Reperfusion Injury using a Langendorff-Based Ex Vivo Assay by Johanna M. Muessig, Sema Kaya, Luise Moellhoff, Johanna Noelle, Leonie Hidalgo Pareja, Maryna Masyuk, Norbert Gerdes, John Pernow, Malte Kelm and Christian Jung in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Supplemental_Table_4_revised - A Model of Blood Component–Heart Interaction in Cardiac Ischemia–Reperfusion Injury using a Langendorff-Based Ex Vivo Assay
Supplemental Material, Supplemental_Table_4_revised for A Model of Blood Component–Heart Interaction in Cardiac Ischemia–Reperfusion Injury using a Langendorff-Based Ex Vivo Assay by Johanna M. Muessig, Sema Kaya, Luise Moellhoff, Johanna Noelle, Leonie Hidalgo Pareja, Maryna Masyuk, Norbert Gerdes, John Pernow, Malte Kelm and Christian Jung in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
The work reported was performed at the University Hospital Düsseldorf, Heinrich-Heine-University, Medical Faculty, Division of Cardiology, Pulmonology, and Vascular Medicine, Düsseldorf, Germany.
Acknowledgments
Author Contribution
Muessig, J, Jung, S, and Kelm, M contributed to conception and design. Masyuk, M, Gerdes, N, and Pernow, J contributed to conception. Muessig, J, Kaya, S, Moellhoff, L, Noelle, J, and Hidalgo Pareja, L contributed to acquisition and analysis. All authors except Muessig critically revised manuscript and gave final approval. All authors agrees to be accountable for all aspects of work ensuring itegrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the German Research Foundation (DFG SFB1116-B06 and -09), the Forschungskommission of the Faculty of Medicine of the Heinrich-Heine-University Düsseldorf, Germany and the Swedish Research Council (2016-01284). No financial support by a sponsor can be stated with an influence on study design, collection, analysis and interpretation of data and furthermore on writing of the manuscript and the decision to submit the manuscript for publication.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
