Abstract
Background:
Previous studies have shown that adherence to low-dose aspirin (LDA) is suboptimal. However, these studies were based on an average measure of adherence during follow-up, ignoring its dynamic process over time. We described the trajectories of adherence to LDA treatment among the French population over 3 years of follow-up.
Methods:
We identified a cohort of 11 793 new LDA users, aged ≥50 years in 2010, by using the French national health-care database. Patients included had at least 3 years of history in the database before study entry to exclude prevalent aspirin users and to assess baseline comorbidities. They were followed from the first date of LDA supply (the index date) until the first date among death, exit from the database, or 3 years after the index date. Adherence to LDA was assessed every 3 months by using the proportion of days covered (PDC) and dichotomized with a cutoff of PDC of 0.8. We used group-based trajectory modeling to identify trajectories of LDA adherence. Predictors of LDA adherence trajectory membership were identified by multinomial logistics regression.
Results:
We identified 4 trajectories of adherence among new LDA users: the not-adherents (4737 [40.2%]), the delayed not-adherents (gradual decrease in adherence probability, 1601 [13.6%]), the delayed adherents (gradual increase in adherence probability, 1137 [9.6%]), and the persistent adherents (4318 [36.6%]). The probability of belonging to the not-adherent group was increased with female sex, low socioeconomic status, and polymedication and was reduced with a secondary indication for LDA use, such as diabetes, hypertension, and dementia, at least 4 consultations in the previous year, or 1 hospitalization or a cardiologist consultation in the 3 months before the index date.
Conclusion:
This study provides a dynamic picture of adherence behaviors among new LDA users and underlines the presence of critical trajectories that intervention could target to improve adherence.
Keywords
Introduction
Cardiovascular diseases (CVDs) are the leading cause of death worldwide, accounting for 31% of all global deaths. 1 To prevent cardiovascular morbidity and mortality, low-dose aspirin (LDA) treatment is among the most widely used treatment. 2,3 Its efficacy in the secondary prevention of CVD has been established: the benefits in reducing cardiovascular events outweigh the risk of hemorrhage. 4 However, its efficacy in primary prevention is more controversial, with recent published trials that did not support its use 3,5 and conflicting guidelines between American and European societies. 6 -9 Many American societies recommended LDA in the primary prevention settings, among individuals aged 50 years or more, 6,7,10 -12 but European guidelines were more cautious because of the associated risk of bleeding 13 ; however, in 2014, a position paper of the European Society of Cardiology suggested that aspirin might be considered in the primary prevention of CVD in both sexes at a high risk of major cardiovascular events and no increased risk of bleeding. As for French recommendation, the French National Authority of Health recommended, in a paper in 2012, aspirin in the primary prevention among individuals with a high cardiovascular risk. 14
To achieve optimal efficacy, LDA must be taken daily and maintained indefinitely. 15,16 Still, previous studies have shown that adherence to LDA is suboptimal, 17,18 with the lowest rates among all cardiovascular preventive therapies (65%). 17 Consequences of this poor adherence are decreased treatment effectiveness and increased cardiovascular morbidity and mortality. 19 -21 Poor adherence is complex, given its multifactorial nature, 22 and needs to be well understood and described to be better managed.
With the advent of medico-administrative databases, drug adherence has been extensively described in studies using these databases. However, many of these studies were based on an average measure of adherence during follow-up, 23 -27 ignoring its dynamic process over time. Actually, individuals with similar average measures of adherence may show different profiles of adherence evolution during follow-up. We addressed this limitation by applying another approach, group-based trajectory modeling (GBTM), which identifies the presence of latent groups of individuals sharing similar evolution of an outcome of interest during follow-up. 28 These models were first developed in the context of sociological and behavioral research to identify subgroups of individuals showing different trajectories of outcome. 28 However, several studies have recently applied them in the context of adherence to medications, 29 -32 to clusters individuals with similar trajectories of adherence over time and to explore predictors of each trajectory.
Therefore, the main objective of this study was to describe the trajectories of LDA use among the French population in the primary and secondary prevention settings and over 3 years of follow-up. Secondary objectives were to (1) describe the trajectories of LDA use according to the presence or not of a secondary indication for LDA use and (2) identify predictors of LDA adherence trajectory.
Methods
Data Source
The Système National des Données de Santé (SNDS) is the French national health-care database that contains prospectively recorded data on all beneficiaries’ medical reimbursements covered by the different health insurance schemes: the general scheme covers about 86% of France residents, and 14 other schemes cover the rest. 33 It contains information on beneficiaries’ age, sex, date of death, complementary universal health coverage (CMU-C) status, and all outpatient health-care consumption including all reimbursed prescription drugs coded according to the Anatomical Therapeutic Chemical (ATC) classification system, 34 the date of delivery, quantity, and brand name. It also contains the long-term chronic disease (LTD) status, allowing for full medical reimbursement; the date of the LTD diagnosis; and its nature, coded according to the International Classification of Diseases, Tenth revision (ICD-10). 35 Through the Programme de Médicalisation des Systèmes d’Information, the SNDS also includes medical summaries of all hospitalizations, including the date of stay, medical procedures and expensive drugs during the hospital stay, the primary diagnosis (main reason for admission), related diagnoses (specifies the disease context of the primary diagnosis), and diagnoses related to other comorbidities, all encoded according to the ICD-10.
In this study, we used the Echantillon Généraliste de Bénéficiaires (EGB) database, which is a 1/97th dynamic random sample of the SNDS, containing the same data, and more easily available for researchers. 33
Study Design and Follow-Up
This was a historical cohort study to identify and describe trajectories of LDA use among the French population during 3 years of follow-up. The index date was the first LDA delivery between January 1, 2010, and December 31, 2012. All individuals were then followed from the index date to the earliest of death from any cause, exit from the database, or month 36 after the first LDA delivery.
Study Population
All individuals in the EGB sample covered by the French national general health insurance scheme since January 1, 2007, and who were at least 50 years old on January 1, 2010, were eligible for inclusion. We restricted our study population to the general health insurance scheme because only these data were available since 2007. We required at least 3 years of history in the database between January 1, 2007, and January 1, 2010, to exclude prevalent LDA users defined by at least 1 LDA delivery in this 3-year period and to assess baseline characteristics and comorbidities. We also required at least 3 months of follow-up after the index date to have at least 1 adherence measure during follow-up.
Definition of LDA Adherence
We selected all reimbursed drugs with ATC codes corresponding to an aspirin dose of 50 to 325 mg (the antiplatelet dose). The definition of adherence was based on the proportion of days covered (PDC) according to the Centers for Medicare and Medicaid Services method. 36 This method allows for calculating adherence to a treatment over a given period, taking into account hospitalizations during each assessment period and the overlap between 2 deliveries. After the first aspirin delivery, we calculated a PDC for each 3 calendar months until the end of follow-up by dividing the number of days covered with aspirin treatment delivered over the previous 90 days by 90. However, if any hospitalization occurred during the assessment period, we excluded the total number of hospital days from the PDC calculation (by dividing the number of nonhospital days covered with aspirin treatment delivered over the previous 90 days by the total number of nonhospital days). As recommended, good adherence was defined as PDC ≥0.8 and poor adherence otherwise. 37,38 In a sensitivity analysis, we also considered an alternative cutoff of 0.5 to define good adherence to account for over-the-counter (OTC) use among LDA users. The PDC for individuals who died or exit database was considered missing after the date of exit from the database or death.
Covariates
We considered the following covariates for describing individuals and multivariate adjustment: Cardiovascular risk factors included age, sex, morbid obesity (defined by bariatric surgery or hospitalization related to obesity), heavy alcoholism (defined by alcoholic liver cirrhosis or hospitalization related to alcohol use disorder), smoking-related conditions (defined by chronic obstructive pulmonary disease or hospitalization related to smoking-related disorder), diabetes, hypertension, and dyslipidemia.
Indications advocating secondary prevention treatment with LDA were defined at baseline in the 3-year period before the index date and included coronary heart disease (coronary artery disease, unstable angina, myocardial infarction, history of coronary artery bypass grafting, or percutaneous coronary intervention), stroke or transient ischemic attack, peripheral artery disease (atherosclerosis or obstructive arteriopathy of lower limbs), atrial fibrillation, or bioprosthetic or mechanical valvular replacement surgery.
Other comorbidities of interest were psychiatric disorder, dementia, end-stage chronic renal failure, cancer, and major hemorrhagic events.
Low socioeconomic status, defined by the CMU-C ( a free complementary health insurance for individuals of low socioeconomic status). All these previous covariates were defined at baseline in the 3-year period before the index date.
Health-seeking behavior was addressed by the proxy number of physician visits and polymedication by the number of distinct ATC classes delivered per year (assessed during the year before the index date). We also considered the presence of at least 1 hospitalization or a cardiologist consultation during the 3 months before the index date.
Co-treatments were other antithrombotics including nonaspirin antiplatelets and anticoagulants, each defined by reimbursement for one of these therapies during each assessment period.
The occurrence of acute coronary events, acute stroke, transient ischemic attack, or major hemorrhagic events was described during follow-up.
Previously developed algorithms were used to define each indication or comorbidity and combined information from drug reimbursements, medical procedures Classification Commune des Actes Médicaux (CCAM), and hospital and LTD diagnosis. 39,40 Supplemental codes of identification for comorbidities are presented in the Supplemental Material.
Statistical Analysis
Group-based trajectory models
Group-based trajectory model is a semi-parametric mixture model that captures the heterogeneity in a population by clustering individuals following distinct trajectories of adherence into different groups. 28 We applied this model using proc traj, an SAS macro for GBTM, 41,42 which allows comparing different models with several prespecified number of groups (1-5 groups). Each model predicted the probability of belonging to each group, then assigned the individual to the group for which the participant had the highest probability of belonging. The selection of the optimal model was based on the lowest value of the Bayesian Information Criterion, a minimum number of individuals allocated to each trajectory of 5%, a minimum average probability of being assigned to a group of 70%, a minimum entropy of 0.7 (a discrimination measure to aid in determining how well individuals are classified into their groups; the nearest this measure is to 1, the better the individuals are classified), 43 and the clinical relevance of the model (more details available in Supplemental Material).
Predictors of adherence group
Once the optimal model was selected, we described baseline characteristics between the different groups with median (interquartile range [IQR]) for continuous variables and frequency (%) for categorical variables by trajectory. We then identified baseline predictors of belonging to each trajectory by multinomial (logit) regression analysis, considering the high adherence group as the reference. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated. Polymedication was classified into 3 classes (≤10, 11-20, and ≥21) and the number of consultations per year in 4 classes (≤3, 4-6, 7-12, >13). Predictive accuracy of the model was tested with C-statistics.
Secondary analysis
For secondary analyses, first the same trajectory modeling analysis was repeated to identify the trajectory of adherence for individuals with and without a secondary prevention indication for LDA at baseline. Second, we described the presence of co-treatment with antithrombotics (anticoagulants or antiplatelets) as well as the occurrence of thromboembolic and major hemorrhagic events during follow-up by each adherence group.
Finally, in a sensitivity analysis, we considered first an alternative cutoff of 0.5 for PDC calculation and second, included only individuals without any missing PDC values during follow-up (N = 10 416; ie, death or exit from the database before month 36 and hospitalization period longer than 3 months) to test for the impact of missing data on the results. All analyses were performed with SAS Enterprise Guide V.7.1. P value <.05 was considered statistically significant.
Results
Study Population
We included 5853 men and 5940 women with a first LDA delivery between January 1, 2010, and December 31, 2012 (Figure 1). The median (IQR) age at inclusion was 69.0 (61.0-79.0) years (Table 1), and the median follow-up was 36 months. Only one-third of the study population had a secondary prevention indication for LDA at baseline (coronary heart disease [17.8%] and/or stroke or transient ischemic attack [6.9%] and/or peripheral artery disease [13.5%] and/or atrial fibrillation [9.1%] and/or bioprosthetic or mechanical valvular replacement surgery [0.7%]). During follow-up, we observed 1293 (11%) deaths, and 81 (0.7%) exit from the database (Table 2).

Flow of the population in the study.
Characteristics of the Study Population by Adherence Trajectory.

Abbreviations: ATC class, Anatomical Therapeutic Chemical class; LDA, low-dose aspirin; IQR, interquartile range.
aDefined by complementary universal health coverage.
bIncludes coronary artery disease, unstable angina, myocardial infarction, undergoing coronary artery bypass grafting, or percutaneous coronary intervention.
Occurrence of Events During Follow-Up by Adherence Group.

Abbreviation: LDA, low-dose aspirin.
aOnly major hemorrhagic events that required hospitalization could be identified in the database.
Identification and Characteristics of Trajectories
We identified 4 trajectories of adherence among new LDA users (Figure 2): the not-adherent group (40.2% of the study population), the delayed not-adherent group (who showed a high probability of being adherent at the beginning of follow-up but gradually decreasing probabilities during follow-up; 13.6% of the study population), the delayed adherent group (who showed a low probability of being adherent during the first 12 months of follow-up and then their probability of adherence increased gradually thereafter; 9.6% of the study population), and the persistent adherent group (36.6% of the study population). Characteristics of these 4 groups are given in Table 1. During follow-up, the delayed not-adherent group showed increased frequency of major hemorrhagic events (Table 2) and a gradual increase in antithrombotic treatment during follow-up (Figure S1), mainly due to a gradual increase in anticoagulant treatment (Figure S2). Individuals in the delayed adherent group showed increasing frequency of an event that required a secondary prevention indication for LDA during follow-up (Table 2). The persistent adherents had the highest mortality rate (Table 2) and a gradual decrease in nonaspirin antithrombotic treatment during follow-up (Figure S1), mainly due to a gradual increase in nonaspirin antiplatelet treatment (Figure S3).

Trajectories of adherence to low-dose aspirin (LDA) treatment over 36 months of follow-up in the study population. The 95% confidence intervals are represented by the gray band around each gray line.
Identification of Trajectories by Presence or Not of a Secondary Prevention Indication for LDA Use at Baseline
We found 4 similar trajectories of adherence among the 4079 and 7714 individuals with a baseline secondary prevention indication for LDA use or not, respectively (Figure 3A and B). The proportion of persistent adherents was higher for those with than without a baseline secondary indication for LDA (46.5% vs 31.4%). Moreover, about half of not-adherents individuals and delayed not-adherents individuals had at least 1 reimbursement for a nonaspirin antithrombotic treatment during each assessment period (Figure S4). For individuals without a secondary indication for LDA, 45.1% were not adherent to LDA (Figure 3B).
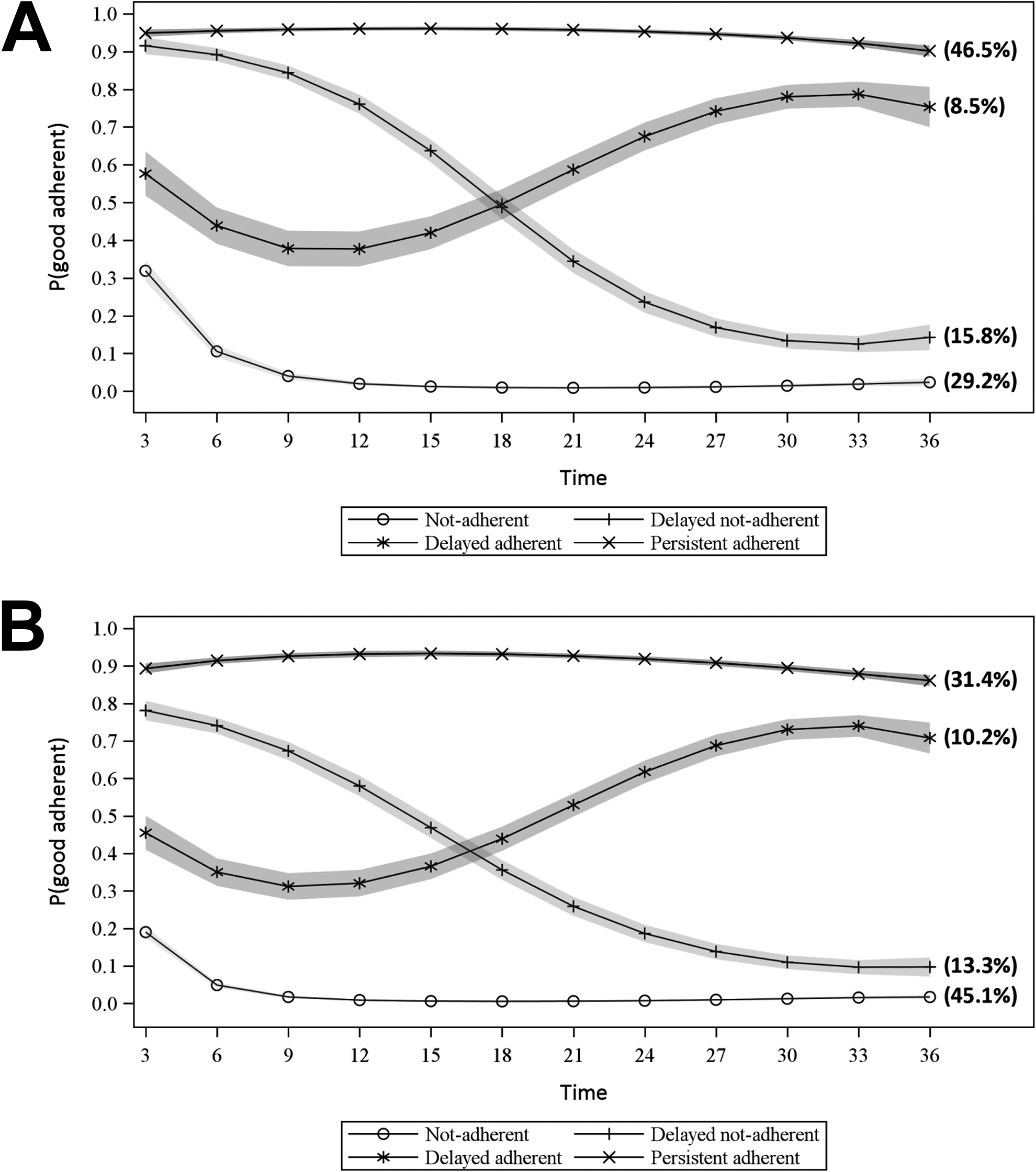
(A) Trajectories of adherence to low-dose aspirin (LDA) treatment over 36 months of follow-up among those with a secondary prevention indication for low-dose aspirin (LDA). (B) Trajectories of adherence to low-dose aspirin (LDA) treatment over 36 months of follow-up among those without a secondary prevention indication for low-dose aspirin (LDA).
Predictors of Adherence
On multinomial logistic regression, the absence of a secondary indication for LDA use at baseline was the main predictor of being in the not-adherent, delayed not-adherent, or delayed adherent group versus persistent adherent group (OR: 0.40 [95% CI: 0.36-0.45], 0.53 [95% CI: 0.45-0.63], and 0.74 [95% CI: 0.64-0.85], respectively; Table 3). Moreover, the probability of being not-adherent was increased with female sex, low socioeconomic status, and polymedication the year before the index event and was decreased with diabetes, hypertension, dementia, ≥4 physician consultations in the previous year, and at least 1 hospitalization or 1 cardiologist consultation in the previous 3 months (Table 3); C-statistics from logistic regression models are shown in Table S1.
Multivariate Analysis of Predictors of Adherence Trajectories During Follow-Up.a

Abbreviations: ATC class, anatomical therapeutic chemical class; CI, confidence interval; LDA, low-dose aspirin; OR, odds ratio.
aOnly variables significant at P < .05 were included in the multivariate analysis.
bDefined as complementary universal health coverage.
cNumber of distinct ATC classes delivered per year was classified into 3 classes: ≤10, 11-20, and ≥21.
dNumber of consultations per year was classified into 4 classes: ≤3, 4-6, 7-12, and ≥13.
The boldface correspond to OR significantly different from 1.
Sensitivity Analysis
In a sensitivity analysis, we also considered a 0.5 cutoff of PDC for good adherence (Figure S5). As expected, the proportion of individuals was greater in the persistent adherent group than other groups (44.3% vs 35.4% in the main model), and 2 decreasing adherence groups were identified: early decreasing adherence (10.2%) and delayed decreasing adherence (8.3%). When we excluded all individuals with missing PDC data during follow-up, results were similar to those observed in the first model, so missing data had a low impact on the results.
Discussion
Group-based trajectory modeling underlined the presence of 4 distinct profiles of adherence among the French population. Approximately 80% of the population had stable adherence trajectories and were divided between 40% of not-adherents and 37% of persistent adherents throughout the follow-up. Two smaller groups changed their adherence behaviors during follow-up: the delayed not-adherent and the delayed adherent groups, which accounted for 14% and 10% of the study population, respectively.
First, we found a very low rate of persistent adherents during follow-up, as compared with other LDA adherence studies (65%-92.5%). 17,23 -27 However, comparison between our results and other conventional adherence studies is complicated because most previous studies combined poor adherence and good adherence periods into one average measure. Group-based trajectory modeling is advantageous over these conventional methods because it underlines all the clinically relevant periods of adherence that individuals would experience during follow-up. As follows, GBTM underlined the presence of a delayed not-adherent group that showed a gradual decrease in the probability of adherence during follow-up. Some explanations for this apparent decrease in adherence could be physician’s decision to interrupt the treatment (reevaluation of the benefit–risk balance, respecting recommendation in the context of primary prevention…), a switch to another antiplatelet or anticoagulant treatment among some individuals, and the occurrence of major hemorrhagic events, which is a contraindication to maintain the treatment; these possibilities were underlined by the gradual increase in nonaspirin antithrombotic treatment among 25% of this group (Figure S1), and the highest rates of hemorrhagic events were observed in this group (4.3%) versus other groups. Indeed, this decrease in adherence behavior has been identified in many adherence trajectory studies independent of the drug under study, 29,30,44 -46 which suggests that a decrease in adherence could also be due in part to a personal attitude regarding the treatment rather than the treatment itself. Conversely, 9.6% of the study population showed a gradual increase in probability of being adherent after 15 months of intermediate probability of adherence. Other studies showed comparable rate: 11.4% of new statin users 29 and 10% of new antiplatelet users. 30 This gradual increase may be related to a gradual occurrence of events that advocated secondary prevention treatment with aspirin and thus also increased the motivation and awareness among individuals. Accordingly, in 14%, the highest rate among all groups, a condition developed that required secondary prevention treatment during follow-up.
Finally, the not-adherent group accounted for the largest proportion of the study population (40.2%), which underlines a serious problem of adherence to LDA, which, apart from the occurrence of a contraindication or a switch to another antithrombotic therapy, should be taken lifelong. 15,16 Previous studies evaluating trajectories of adherence among new statin and antidiabetic users showed a comparable rate of not-adherents, 29,46 whereas a study of adherence to antihypertensive drugs showed a very low proportion of not-adherents (7%). 31 However, this latter study was not restricted to new users, among whom discontinuation is frequent in the first year. 31 A possible explanation for this behavior is the low proportion of individuals with a secondary prevention indication for LDA (24%), with the uncertain effectiveness of LDA in primary prevention. 4
Indeed, when we searched for predictors of low adherence to LDA, the absence of a secondary indication for LDA use was the main predictor of being in the not-adherent or delayed not-adherent group versus persistent adherent group. The probability of being in the not-adherent group was also increased with female sex, low socioeconomic status, or polymedication, which agreed with other studies, 17,20,37,38 and was decreased with other covariates that are associated with an increased rate of CVDs. Finally, dementia was also associated with good adherence, which suggests that these individuals are in an advanced stage and might be relying on caregivers to administer medications and thus show good adherence behavior. 47
To our knowledge, this is the first study to evaluate dynamic trajectories of adherence to LDA use among new users, regardless of the indication for use. However, some limitations to this study should be noted. First, we chose a cutoff of 80% because it is the most adequate cutoff for cardiovascular medications 38 ; however, for LDA use, this cutoff may be lower, especially because of the possible OTC purchase of this medication. A cutoff of 50% did not greatly change our results, especially because LDA users are more likely to refill their monthly prescriptions not in OTC to be reimbursed and also because LDA is frequently associated with other cardiovascular treatments that could not be purchased as OTC. Moreover, a validation study of a prescription database showed that unrecorded OTC use had a small impact on misclassification of LDA use. 48 Second, filling a prescription does not guarantee that the patient actually takes the drug nor the date of the actual consumption; however, a systematic review showed a good association between refill records and adherence. 49 Third, we could not test for primary nonadherence (patients who did not fill their first prescription for LDA), given that no prescription data are available in the French databases. Fourth, we could not investigate all potential LDA adherence predictors, given the lack of information in administrative databases on potential predictors such as patient–physician relationships, social behaviors, lifestyle, and education. This was underlined by the poor predictive accuracy of the model (C-statistics = 0.6). However, some predictors were identified in this study. Finally, GBTM ignores the intracorrelation between repeated adherence measures in the same individual, which could overestimate the number of identified groups. However, the objective of our study was only exploratory, with no attempt to classify each individual into 1 class, and GBTM was found to be preferable to some other methods, analyzing developmental trajectories 50 and summarizing longitudinal adherence with visual patterns more accurately than the conventional approaches. 29
Conclusion
This study provides a better understanding of adherence behaviors among new LDA users in France over 3 years and underlines the presence of critical trajectories and time periods when adherence behaviors worsened and that intervention could target to increase adherence. Only one-third of the study population showed persistent adherence behaviors, and the situation that most conditioned adherence profiles was the presence of a secondary prevention indication for LDA treatment, which may go in line with the uncertain efficacy of aspirin in the primary prevention and the contradictory guidelines.
Supplemental Material
Supplemental_Material - Trajectories of Adherence to Low-Dose Aspirin Treatment Among the French Population
Supplemental_Material for Trajectories of Adherence to Low-Dose Aspirin Treatment Among the French Population by Aya Ajrouche, Candice Estellat, Yann De Rycke and Florence Tubach in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
This manuscript is an original work that has not been published and is not under consideration for publication elsewhere.
Author Contributions
All authors designed the research and conducted the research. AA and YDR did the statistical analysis. AA wrote the first draft of the manuscrit. All authors contributed to the data interpretation, revised each draft for important intellectual content, and read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a research grant from the French ministry of health [Grant number PHRC-K 14-158]; by an academic grant from Paris Diderot University-Sorbonne Paris Cité [No grant number is applicable] to A.A.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
