Abstract
Objective:
Chemotherapy-induced cardiotoxicity is a major and leading cause of death in breast cancer survivors. It can present decades after chemotherapy and can manifest in different ways; some chemotherapeutic agents have a powerful dose-dependent relationship with cardiotoxicity. The aim of this study was to investigate the effect of rosuvastatin on preventing chemotherapy-induced cardiotoxicity in patients with breast cancer.
Methods:
Our study was a randomized, single-blind, placebo-controlled trial that involved 89 women with newly diagnosed breast cancer who were scheduled to receive chemotherapy. Patients were randomly assigned to receive rosuvastatin or a placebo in a 1:1 ratio for 6 months. Echocardiography, using 2-dimensional (2D) Doppler, tissue Doppler, and speckle-tracking methods, was used to determine the absolute changes in the left ventricular systolic ejection fraction (LVEF), left ventricular end-diastolic volume (LVEDV), left ventricular end-systolic volume (LVESV), left atrial (LA) diameter, transmitral Doppler early diastolic velocity (E wave), tissue Doppler early diastolic (e′) and peak systolic (s′) mitral annular velocities, E/e′ ratio, and global longitudinal systolic strain.
Results:
The LVEF was significantly reduced in the placebo group at the end of the study when compared with the baseline value. However, there was no significant difference in the LVEF in the intervention group (intergroup P = .012). Furthermore, compared with the intervention group at the end of the study, there was a significant increase in the 4- and 2-chamber LVESV, LA diameter, and E/e′ ratio in the placebo group (intergroup P = .019, P = .024, P < .001, and P = .021, respectively) and a significant decrease in the e′ and s′ velocities in the placebo group (intergroup P < .001 and P < .006, respectively).
Conclusions:
The present study showed that the prophylactic use of rosuvastatin may prevent the development of chemotherapy-induced cardiotoxicity.
Introduction
Breast cancer is the most common cancer and the second most common cause of cancer death in women worldwide. 1,2 The overall survival of patients with breast cancer has improved dramatically because of the increased availability of new and effective chemotherapy drugs. However, these agents may have short- and long-term side effects that can be fatal. One of the most important and life-threatening side effects of chemotherapeutic agents is drug-induced cardiotoxicity. 2 The most serious cardiovascular side effects from chemotherapeutic agents are left ventricular (LV) systolic dysfunction and heart failure (HF). 3 For example, anthracyclines are one of the most commonly used drugs in the treatment of breast cancer and are an essential component of several different adjutant and palliative regimens. However, the clinical utility of anthracyclines is limited by their cumulative dose-related cardiotoxicity, which can result in HF and a reduced health-related quality of life. 2 The most probable mechanism of anthracycline-induced cardiotoxicity is by the promotion of oxidative stress in nontargeted tissues, which inhibits nucleic acid and protein synthesis and induces reactive oxygen species-dependent apoptosis. 4
In addition to anthracyclines, many other chemotherapeutic agents for breast cancer, such as taxanes, alkylating agents (cyclophosphamide), and trastuzumab (Herceptin), have significant adverse effects on the cardiovascular system. 5 These agents can induce several deleterious processes, including the production of reactive oxygen species, calcium overload, cardiac mitochondrial dysfunction, and the release of some chemokines that can contribute to cardiomyocyte death. 6
Cardiotoxicity can be acute, occurring during or within days of the drug infusion, or can occur years after chemotherapy. 7 It usually begins with myocyte injury, progresses to silent LV systolic dysfunction, and eventually becomes symptomatic and irreversible. Therefore, early detection is important both in the prevention and in the treatment of irreversible myocardial damage. 8 Biomarkers, such as atrial and brain (B-type) natriuretic peptides, cardiac troponin, neutrophil myeloperoxidase, and high-sensitivity C-reactive protein, are useful for predicting chemotherapy-induced cardiotoxicity. 9 There are strategies that can reduce the risk of chemotherapy-induced cardiotoxicity, including dose restrictions of chemotherapeutic agents, the development of liposomal formulations, and the use of iron chelators, such as dexrazoxane. In spite of these strategies, chemotherapy-induced cardiotoxicity continues to occur and is responsible for considerable morbidity and mortality among breast cancer survivors. 10,11
Several agents such as angiotensin-converting enzyme (ACE) inhibitors and beta blockers have also had some beneficial effects in preventing cardiotoxicity during chemotherapy. 6 Statins, which lower cholesterol and exert some antioxidant properties, have been shown to reduce morbidity and mortality in patients with atherosclerotic diseases. 12 Some of the cardiac adverse effects of chemotherapy drugs are triggered by cardiac oxidative stress and inflammation. 13 Previous studies in animals have revealed that treatment with statins may decrease the risk of chemotherapy-induced cardiotoxicity without compromising treatment efficacy. 4 Furthermore, recent human studies have shown that treatment with atorvastatin may provide protection against anthracycline-induced cardiomyopathy and the onset of HF. 14 One study showed that rosuvastatin provided a greater reduction in low-density lipoprotein cholesterol (LDL-chol) with no increased risk of adverse effects when compared with atorvastatin at a 2 times higher dose. 15 To the best of our knowledge, no human study has evaluated the effect of rosuvastatin in the prevention of chemotherapy-induced cardiomyopathy. The aim of our study was to investigate the effect of rosuvastatin on preventing chemotherapy-induced cardiotoxicity in women with breast cancer.
Methodology
Our study was a prospective, randomized, single-blind, placebo-controlled trial of women with newly diagnosed breast cancer who were undergoing chemotherapy between 2016 and 2018 in the Toba Oncology Department, Mazandaran University of Medical Sciences. The study followed the guidelines of the Helsinki Declaration and was approved by the ethics committee of the university. Written informed consent was obtained after the objectives of the study had been explained to the participants. Women who were scheduled to receive anthracycline chemotherapy were enrolled in the study. Patients were assigned to receive rosuvastatin or a placebo in a 1:1 ratio during and after their chemotherapy (for the follow-up period). The echocardiographer was blind to the study group. However, the physician assessing the treatment was not blind to the study group.
Eligibility Criteria
Eligible women were 25 to 77 years old, had been newly diagnosed with breast cancer, had preserved LV systolic function in which the left ventricular ejection fraction (LVEF) ≥ 55%, and had normal liver, renal, and hematological functions. Patients were excluded from the study for the following criteria: previous malignancy, pregnancy, male patients with breast cancer, prior chemotherapy or radiation therapy, prior use of 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase inhibitors, significant valvular heart disease, documented coronary artery disease, history of congestive heart failure (CHF) or cardiomyopathy, chemotherapy was contraindicated, and abnormal liver function tests.
Research Design
The intervention group received 20 mg of oral rosuvastatin 24 hours prior to the first cycle of chemotherapy and once daily during the follow-up period. Transaminase levels were measured before chemotherapy was initiated, 3 months after chemotherapy began, and after the completion of the study. Aspartate transaminase (AST)/alanine transaminase (ALT) levels were monitored and if the levels increased more than 3 times the upper limit, the rosuvastatin would have been stopped. 16 All patients underwent 6 cycles of chemotherapy with doxorubicin (60 mg/m2) and cyclophosphamide (600 mg/m2) every 3 weeks. At the same time, some patients received taxanes and Herceptin as an adjuvant therapy. Radiotherapy was performed after the completion of the study. The remaining eligible patients were treated with Herceptin following the end of chemotherapy.
Primary and Secondary Endpoints of the Study
The primary endpoints were changes in the LVEF and the global longitudinal strain (GLS) after completion of chemotherapy when compared with the baseline values. The secondary endpoints included cardiovascular mortality and hospitalization for HF. Furthermore, changes in the left ventricular end-diastolic volume (LVEDV), left ventricular end-systolic volume (LVESV), diastolic function, and left atrial (LA) diameter were determined after the end of chemotherapy. Diabetes mellitus (DM) was identified based on the guidelines of the American Diabetes Association or the need for insulin or oral hypoglycemic agents. 17 Hypercholesterolemia was defined as a total cholesterol (chol) level ≥ 200 mg/dL or the need for chol-lowering agents. 18
Echocardiography
All patients underwent transthoracic echocardiography 24 hours prior to the initiation of chemotherapy; the echocardiographic examinations were repeated after 6 months using a Vivid S6 Ultrasound System (GE Healthcare, Wauwatosa, WI, USA) with a 1.5- to 3.6-MHz transducer. All images were stored on a hard disk and were interpreted by a single expert echocardiographer who was blind to the patients’ data. The grayscale movies were acquired and stored in the following apical views: 4-chamber, 2-chamber, and 3-chamber views. The peak longitudinal strain values were measured from the basal, mid-, and apical segments of the septal, lateral, inferior, anterior, inferobasal, and anteroseptal walls. The 2-chamber and 4-chamber LVEDV, LVESV, and apical 2-chamber, 4-chamber, and 3-chamber GLS were calculated from the stored movies using a software dedicated for a semiautomated analysis in which the endocardial and epicardial borders were traced. The peak GLS was determined as the average of all 18 myocardial segments. The LVEF was calculated by subtracting LVESV from LVEDV and dividing by LVEDV using Simpson biplane method from the apical 4- and 2-chamber views (Figures 1 and 2). The transmitral Doppler early diastolic velocity (E wave), tissue Doppler septal and lateral early diastolic (e′) and peak systolic (s′) mitral annular velocities, the average of these velocities at the septal and lateral mitral annulus, and the E/e′ ratio were manually determined. The LA anterior–posterior diameter was measured in the left parasternal long-axis view perpendicular to the LA walls at end-systole. The reproducibility of the LVEF and GLS measurements were assessed by repeating the measurements in 10 patients within 24 hours; the intraobserver correlation coefficients were 0.90 and 0.93, respectively.
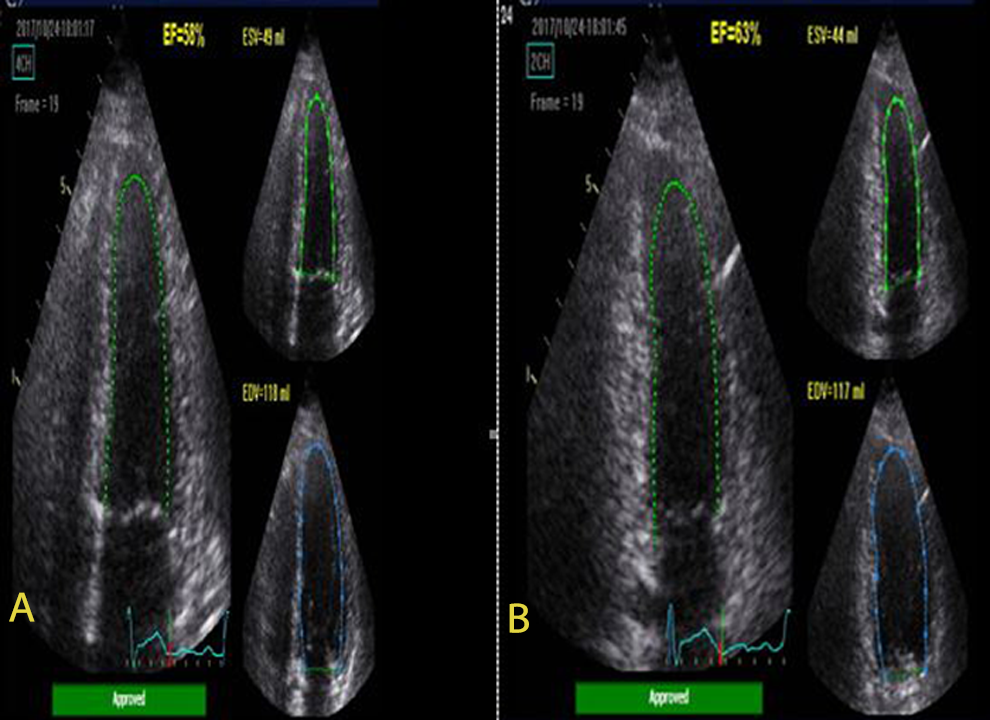
Echocardiographic determination of 4 (A) and 2 chambers (B) LVESV and LVEDV by semiautomated endocardial border detection. LVESV indicates left ventricular end-systolic volume; LVEDV, left ventricular end-diastolic volume.

The longitudinal strain bull’s-eye plot with the standard 2D speckle-tracking echocardiography obtained from the apical 3, 4, and 2 chamber views.
Statistical Analysis
We displayed the continuous variables as the mean (standard deviation) and the categorical variables in terms of frequency and percentage. The normality was determined for all the echocardiographic variables individually using the Kolmogorov–Smirnov test, which showed that the variables were normally distributed. For the individual groups, paired sample t tests were used to compare the means of the variables before and after chemotherapy. An independent t test was used to compare the mean values of the dependent continuous variables between the groups. The categorical variables were compared using the χ2 test and Fisher exact test. A P value ≤ .05 was considered to be statistically significant. In addition, we used Hotelling T 2 test to express the difference between the response variables as a vector. All the statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS)/Predictive Analytics Software (PASW) version 18 (SPSS Inc., Chicago, Illinois). The sample size was obtained from a previous study 13 and was determined using the following statistical equation:
Results
Study Population
Our study included 89 women who had been newly diagnosed with breast cancer and who were scheduled to receive anthracycline chemotherapy between 2016 and 2018. All patients underwent transthoracic echocardiography. However, 6 patients did not receive antracycline chemotherapy due to their age and comorbid cardiac diseases and, thus, were excluded from the study. The remaining 83 patients were randomly assigned to the intervention group or the placebo group in a 1:1 ratio. Because of a lack of follow-up, a total of 6 patients (3 from each group) were excluded from the study (Figure 3). All patients received 6 cycles of anthracycline and cyclophosphamide, which were administrated every 3 weeks. The mean age of the patients was 49.26 (11.20) years. None of the patients underwent radiotherapy during the follow-up period of 6 months. However, 4 patients in the placebo group and 6 patients in the intervention group received Herceptin during the follow-up period. Radiotherapy was considered for suitable patients after completion of the study. Following the end of the study, patients who required Herceptin received it. Patients in the intervention group received 20 mg of rosuvastatin taken orally 24 hours prior to the first cycle of chemotherapy and received 20 mg once daily during the follow-up period of 6 months. The baseline characteristics, total amounts of chemotherapy drugs received, and the mean lipid profile and levels of fasting blood glucose (FBS) and liver enzymes in both study groups at the end of the study are presented in Table 1. Seven patients in the placebo group and 5 patients in the intervention group had DM. In addition, 8 patients in the placebo group and 10 patients in the intervention group had hyperlipidemia, which was not statistically significant: P = .562 and P = .547, respectively. There was no significant difference between the 2 groups in terms of the average total dose of antracycline and cyclophosphamide.

Flowchart of patients entered into the study.
Baseline Characteristics, Total Amounts of the Chemotherapy Drugs and Mean Levels of Lipid Profile, Fasting Blood Glucose and Liver Enzymes at the End of the Study in the 2 Study Groups.
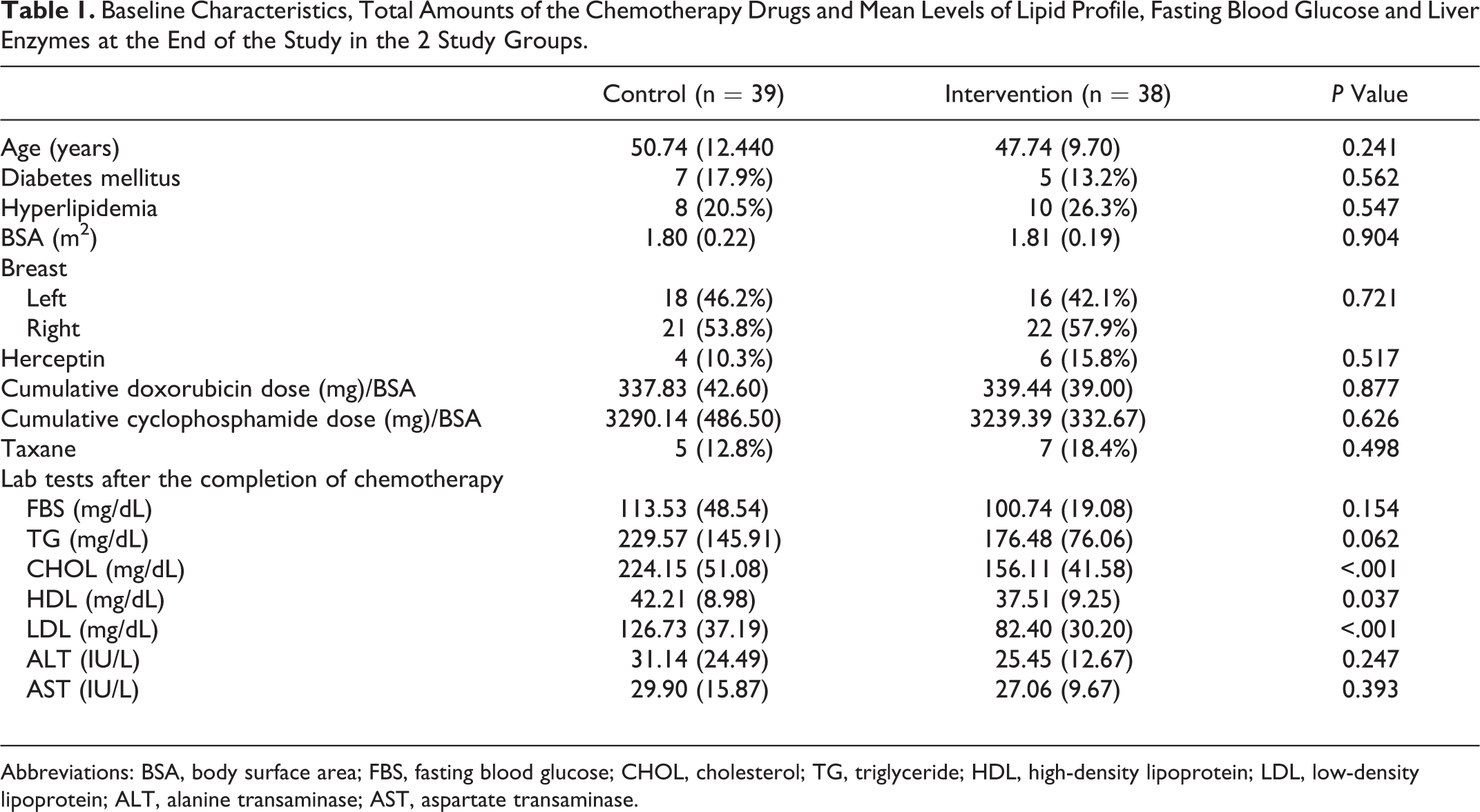
Abbreviations: BSA, body surface area; FBS, fasting blood glucose; CHOL, cholesterol; TG, triglyceride; HDL, high-density lipoprotein; LDL, low-density lipoprotein; ALT, alanine transaminase; AST, aspartate transaminase.
Patient Enrollment and Assignment
Two of the patients in the intervention group did not take rosuvastatin regularly according to our scheduled plan; all other patients took it regularly as scheduled. None of the patients reported any side effects related to rosuvastatin. The plasma levels of triglycerides (TG), chol, high-density lipoprotein (HDL), low-density lipoprotein (LDL), ALT, AST, and FBS were measured at the end of the study. The plasma levels of chol, HDL, and LDL were significantly lower in the intervention group when compared with the control group: P > .001, P > .037, and P > .001, respectively. However, there was no significant difference in the FBS, ALT, and AST levels between the 2 groups: P = .154, P = .247, and P = .393 (Table 1). None of the patients died during the follow-up period, although 1 patient in the placebo group was hospitalized with HF during the follow-up. She also experienced pulmonary metastases and died shortly after the study was completed. The patient’s LVEF was 56% at baseline, which decreased to 33% at the end of the study.
Echocardiography
There was no significant difference in baseline LVEF between the 2 groups (P = .963). At the end of the study, an LVEF ≤ 45% was seen in 6 patients in the placebo group and 4 patients in the intervention group. Compared with the intervention group, the LVEF was significantly reduced in the placebo group at the end of the study (intergroup P = .012; Figure 4). Furthermore, there was a significant increase in the 4- and 2-chamber LVESV, LA diameter, and E/e′ ratio: intergroup P = .019, P = .024, P < .001, and P = .021, respectively. There was also a significant decrease in the e′ and s′ velocities at the end of the study in the placebo group when compared with the intervention group: intergroup P < .001 and P < .006, respectively (Figure 5). However, the differences in the 4- and 2-chamber LVEDV and the 4-, 3-, and 2-chamber GLS changes between the 2 groups were not statistically different: intergroup P = .678, P = .491, P = .995, P = .071, P = .513, and P = .225, respectively (Table 2). Hotelling T 2 test was conducted to express the difference between the response variables as a vector between the 2 groups in which taking or not taking rosuvastatin was the independent variable and the changes in echocardiographic variables before and after chemotherapy were the dependent (response) variables (Table 3). Because the Box test of equality of covariance matrices was significant, Pillai’s statistic trace was computed as 0.563 (statistic = 8.88, η2 = 0.563, and P < .001), which 56.3% of the changes in response variables was explained.

Boxplot of distribution change in 2-chamber left ventricular ejection fraction among the 2 study groups including 39 patients in control and 38 patients in intervention group at the end of the study. The median is represented by the central line inside the box. Upper and lower quartiles correspond with the ends of the boxes. Whiskers represent the extreme values (intergroup P = .012).

Box plot of distribution change in transmitral Doppler early diastolic velocity/mitral annular early diastolic velocity (E/e′) ratio among the 2 study groups including 39 patients in control and 38 patients in intervention group at the end of the study. The median is represented by the central line inside the box. Upper and lower quartiles correspond with the ends of the boxes. Whiskers represent the extreme values (intergroup P = .021).
Echocardiographic Variables of Left Ventricular Systolic and Diastolic Function Before and After Chemotherapy in the 2 Study Groups.

Abbreviations: LVEF, left ventricular ejection fraction; GLS, global longitudinal strain; LVEDV, left ventricular end-diastolic volume; LVESV, left ventricular end-systolic volume; LA, left atrium; E/e′, transmitral Doppler early diastolic velocity/mitral annular early diastolic velocity; s, mitral annular peak systolic velocity.
Comparison Between Changes in Echocardiographic Variables Before and After Chemotherapy in the 2 Study Groups (Pillai's statistic trace).a

Abbreviations: Dif, difference; LVEF, left ventricular ejection fraction; LVESV, left ventricular end-systolic volume; LA, left atrium; E/e′: transmitral Doppler early diastolic velocity/mitral annular early diastolic velocity; s′ mitral annular peak systolic velocity.
a Pillai’s statistic trace (because the Box Test of Equality of Covariance Matrices was significant) was computed as 0.563 (statistic = 8.88, η2 = 0.563 and P value < .001) with 56.3% of variance explanation.
Discussion
Our study showed that the prophylactic use of rosuvastatin in patients with breast cancer may prevent chemotherapy-induced cardiotoxicity. At the end of our study, patients who received rosuvastatin did not show any significant changes in the mean LVEF and other measures of LV systolic function, such as GLS and s′ velocity, LA diameter, and variables of LV diastolic function and filling pressures compared with the baseline values. However, these variables were adversely altered in the placebo group at the end of the study.
Recent advances in the treatment of breast cancer have resulted in improved survival. However, chemotherapy-induced cardiotoxicity is an important side effect of several chemotherapeutic agents, particularly anthracyclines and Herceptin, which is responsible for an increase in mortality. Cardiotoxicity is diagnosed when there is a reduction in LVEF from ≥5% to <55% with associated signs or symptoms of HF or a reduction in LVEF from ≥10% to <55% without accompanying signs or symptoms. 19 Previous studies have shown that adjuvant doxorubicin in a cumulative dose as low as 240 mg/m2 plus cyclophosphamide chemotherapy may lead to frequent, acute decreases in LVEF. 20 The use of iron chelators, such as dexrazoxane, may protect against the cardiotoxicity of doxorubicin, although it may also decrease the drug’s efficacy and increase the risk of secondary tumors. Hence, there is a need to develop other cardioprotective strategies to limit myocardial injury following chemotherapy.
Statins have many pleiotropic properties that are independent of their anti-inflammatory and cholesterol-lowering effects. Their antioxidative effects are exerted via signal transducers on mitogen-activated kinases and transcription factors that are involved in the recognition of DNA damage. In addition to their cardioprotective effects, statins can sensitize different tumor cells to chemotherapy agents. 21 In 2011, Acar et al conducted a study on 40 patients who were undergoing anthracycline chemotherapy. 13 Patients were randomly assigned to a statin or control group. The statin group received 40 mg of atorvastatin daily for 6 months. The statin group showed no difference in the mean LVEF and LV end-systolic and end-diastolic diameters compared with the baseline values. However, the control group showed significant adverse changes in these variables at the end of chemotherapy. 13 Acar et al examined a heterogeneous group of patients undergoing anthracycline chemotherapy; however, the tissue Doppler mitral annular velocities and LV deformation variables were not measured. In our study, the intervention group received 20 mg of rosuvastatin daily for 6 months. We used rosuvastatin due to our personal experience and the fact that it is more effective with no increased risk of adverse effects compared with a higher dose of atorvastatin. 16 In 2012, Seicean et al evaluated the effects of statin therapy on new onset HF in patients with breast cancer undergoing anthracycline chemotherapy. 14 In the study, 67 patients received statin therapy and 134 patients in the control group received no statin therapy. The researchers found that statin use was associated with a lower risk of HF. However, the study was not randomized and patients receiving the continuous use of any type of statins were assigned to the statin group. Furthermore, the researchers only measured the baseline LVEF and did not assess any other echocardiographic variables. 14 In 2015, Stone et al investigated the effect of statin therapy on LVEF in a heterogeneous group of cancer patients who were considered for anthracycline-based chemotherapy. 22 Patients were divided into 2 groups according to their statin status: 14 patients were taking statins for the primary and secondary prevention of cardiovascular diseases and 37 patients were not taking statins. After 6 months, the patients who did not receive statin therapy showed a greater decline in LVEF (6.5% ± 1.5%) compared with the statin group (1.1% ± 2.6%; P = .03). 22 However, this study was not randomized and included a small number of patients who were receiving different statins at different dosages. In an animal study, Kim et al evaluated the effect of rosuvastatin and carvedilol on 61 rats that received doxorubicin for 4 weeks. 4 Rosuvastatin therapy alleviated myocardial injury and LV systolic/diastolic dysfunction, although carvedilol did not show any effect. 4 To the best of our knowledge, no human study has evaluated the effect of rosuvastatin on chemotherapy-induced cardiotoxicity in patients with breast cancer.
LVEF is a widely used echocardiographic parameter for assessing cardiac function during chemotherapy. A decrease in LVEF at baseline or after chemotherapy is a strong predictor of a poor outcome. 23 In our study, the intervention group did not show any significant change in LVEF at the end of study; however, the LVEF was significantly decreased in the placebo group (intergroup P = .012). The changes of the peak systolic deformation indices occur earlier than the decrease in LVEF. 24 GLS is another echocardiographic parameter that may detect early myocardial injury; when comparing the GLS during chemotherapy with baseline values, a relative percentage reduction ≥ 15% is most likely to be abnormal. 25 In our study, there was no significant difference in GLS between the 2 groups after chemotherapy. However, the use of strain imaging and effect of this modality on patient management remains controversial. In our study, patients in the placebo group had a greater increase in LVESV at the end of the study compared with the intervention group. Previous studies have shown that patients with high LVESV had increased rates of death and hospitalization for HF. 26 Patients who developed chemotherapy-induced cardiotoxicity shortly after beginning chemotherapy showed a decrease in the tissue Doppler-derived mitral annular early diastolic and systolic velocities, which remained for several years after treatment. 23 In addition, a decrease in the E/A ratio (the ratio of the transmitral Doppler early to late diastolic velocities) has been associated with a decrease in GLS. 23 These findings are consistent with our study, which showed that the placebo group had a significant decrease in the tissue Doppler e′ and s′ velocities at the end of study when compared with the intervention group: intergroup P < .001 and P < .006, respectively. Some patients may experience diastolic abnormalities after chemotherapy despite a preserved LVEF. The E/e′ ratio can be used to estimate the LV filling pressures in these patients. 27 At the end of our study, the placebo group showed a significant decrease in LV diastolic function, including an increase in the E/e′ ratio and LA diameter when compared with the intervention group: intergroup P = .021 and P < .001, respectively. The average decrease of LVEF in the placebo group was only 5%. However, we tried to assess whether the rosuvastatin could modulate the adverse changes of all echocardiographic variables after chemotherapy due to the correlation between the different echocardiographic variables. The echocardiographic components including the LVEF, LVESV, tissue Doppler e′ and s′ velocities, LA diameter, and filling pressures were compared by Hoteling T 2 test. Pillai’s trace showed statistically significant difference between the 2 groups. The values of the main effects of intervention were significant (P < .05). The partial η2 statistic reported the “56.3%” of the variation of response variables.
Study Limitations
One limitation of our study was the small sample size. Another limitation was the duration of the research, which began 24 hours before the first cycle of chemotherapy and lasted for 6 months. However, chemotherapy-induced cardiomyopathy may develop years after chemotherapy has concluded. In addition, the present study involved research at a single center.
Conclusion
The present study showed that the prophylactic use of rosuvastatin may prevent the development of chemotherapy-induced cardiotoxicity in patients with breast cancer.
Footnotes
Acknowledgments
This research was the postgraduate thesis of Dr. Jamil Smailian and was approved by cardiovascular research center of Mazandaran University of Medical Sciences. The authors thank all patients and staff members who participated in this study. We registered the research to IRCT. The registration code number was IRCT2016121213090N3.
Author Contributions
Maryam Nabati contributed to conception or design; contributed to acquisition, analysis, or interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Ghasem Janbabai and Jamil Esmailian contributed to conception and acquisition, drafted the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Jamshid Yazdani contributed to design and analysis, drafted manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Rosuvastatin and placebo were provided by Fanda Pharmaceutical Company and we would like to thank them for their support.

