Abstract
In previous studies, it was demonstrated that lipid core nanoparticles (LDE) resemble the low-density lipoprotein structure and carrying the antiproliferative agent paclitaxel (PTX) strongly reduced atherosclerosis lesions induced in rabbits by cholesterol feeding. Currently, the aim was to verify whether combining LDE-PTX treatment with methotrexate (MTX) associated with LDE (LDE-MTX) could accelerate the atherosclerosis regression attained with single LDE-PTX treatment, after withdrawing the cholesterol feeding. Thirty-eight rabbits were fed 1% cholesterol chow for 8 weeks. Six of these rabbits were then euthanized for analyses of the aorta (controls). In the remaining rabbits, cholesterol feeding was withdrawn, and those 32 animals were allocated to 3 groups submitted to different 8-week intravenous treatments, all once/week: LDE-PTX (n = 10; 4 mg/kg), LDE-PTX + LDE-MTX (n = 11; 4 mg/kg), and LDE-alone (n = 11). Rabbits were then euthanized and aortas were excised for morphometric, immunohistochemical, and gene expression analyses. After cholesterol feeding withdrawal, in comparison with LDE-alone group, both LDE-PTX and LDE-PTX + LDE-MTX treatments had the ability to increase the regression of plaque areas: −49% in LDE-PTX and −59% for LDE-PTX + LDE-MTX. However, only LDE-PTX + LDE-MTX treatment elicited reduction in the intima area, estimated in −57%. Macrophage presence in aortic lesions was reduced 48% by LDE-PTX and 43% by LDE-PTX + LDE-MTX treatment. Matrix metalloproteinase 9 was reduced by either LDE-PTX (74%) or LDE-PTX + LDE-MTX (78%). Tumor necrosis factor α gene expression was reduced 65% by LDE-PTX and 79% by LDE-PTX + LDE-MTX. In conclusion, treatment with LDE-PTX indeed accelerated plaque reduction after cholesterol feeding; LDE-PTX + LDE-MTX further increased this effect, without any observed toxicity. These results pave the way for the use of combined chemotherapy to achieve stronger effects on aggravated, highly inflamed atherosclerotic lesions.
Introduction
Atherosclerosis is the major cause of cardiovascular diseases. Coronary heart disease, which is essentially related to the atherosclerotic lesions, is one of the chief causes of death in the world. 1,2 From the early stages of atherogenesis, wherein leukocytes are recruited to the arterial wall to repair endothelial lesions, an inflammatory and a proliferative process resulting in invasion of the intima by smooth muscle cells (SMCs) drives the formation of fatty streaks. Fatty streaks may either regress or develop and grow, forming the atheroma plaques in the intima arterial layer. Inflammation is also involved in the concurrent local lipid deposition, as lipoproteins that penetrate the arterial wall are oxidized and trapped by resident macrophages. 3 –6 To date, the medical, noninvasive treatment of atherosclerotic cardiovascular diseases is restricted to cholesterol-lowering and anticoagulant medications. However, research has been increasingly growing aiming to identify specific therapeutic targets and drugs to inhibit the vascular inflammatory process; some of these drugs are object of ongoing clinical trials.
Our laboratory has pioneered the use of anticancer chemotherapeutic agents for atherosclerosis treatment. 7 –9 Anticancer drugs possess the most powerful antiproliferative action in the Pharmacopea and suppressor actions on the immune system that are major side effects of those drugs. As described in previous studies, those agents can be associated with lipid core nanoparticles (LDEs) that resemble the lipid structure of low-density lipoprotein (LDL). The LDE is made without apolipoproteins (apo), but when injected in the bloodstream, LDE acquires apo E from the native lipoproteins. Apolipoprotein E, a 36-kDa protein that is recognized by LDL receptors, bridges the binding of the LDE nanoparticles to the LDL receptors. Thereby, LDE can be taken up by the cells via the LDL receptor-mediated endocytic pathway. 8,10 Low-density lipoprotein receptors are upregulated in proliferating tissues demanding larger amounts of cholesterol and other lipids for membrane synthesis to assist their high mitosis rates. This phenomenon affords the concentration of LDE and drugs incorporated to the nanoparticle structure in neoplastic, inflamed, and other rapidly proliferating tissues. 11,12 Association to LDE of anticancer agents such as paclitaxel (PTX), 13 –15 carmustine, 16 –18 or etoposide 19 –21 resulted in remarkable reduction of their toxicities, as tested in animals, including nonhuman primates, 22 and in clinical trials enrolling patients with advanced hematological or solid cancers. The treatment of rabbits with atherosclerosis induced by cholesterol feeding with LDE-PTX, 23 LDE-etoposide, 24 LDE-methotrexate (MTX), 25 and LDE-carmustine 26 resulted in pronounced decrease, above 60%, of the atheroma lesion areas. By reducing the toxicity to a degree of comfortable tolerability and safety, use of LDE as vehicle makes room for the incorporation of anticancer chemotherapy as a therapeutic tool to achieve strong and fast antiatherosclerotic effects in patients under high risk of undergoing the life-threatening complications of the atherosclerotic cardiovascular diseases.
Combined chemotherapy is a standard scheme for the treatment of malignant neoplasia, attempting to achieve additive or synergic effects of drugs with different mechanisms of pharmacological action. With similar rationale, this study was aimed to investigate in cholesterol-fed rabbits the antiatherosclerotic effects of combining LDE-PTX with LDE-MTX. The experimental protocol used here was different from that used in our previous studies, with longer atherosclerosis induction period and with cholesterol feeding withdrawal before the installment of the LDE drug treatments, purposely to observe effects on more advanced atheromatous plaques.
Methods
Animals and Diets
Thirty-eight male New Zealand white rabbits weighing 3.0 to 3.5 kg were obtained from the central animal settlement of the Medical School of the University of São Paulo. Animals were housed in individual cages, under a 12-hour light–dark cycle in a temperature-controlled room (20°C to 22°C) during the experimental period. Initially, there were 49 animals, but 11 rabbits died because of complications in the atherosclerosis induction process.
The experimental procedures in the study were in accordance with guidelines of the Brazilian College of Experimental Animals. The experimental protocol was approved by the Ethics Committee in Animal Experimentation of the Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo Heart Institute under the research protocol number: 254/10.
Experimental Protocol and Treatment
After a 1-week adaptation period, all animals underwent an atherosclerosis induction model, which consisted of consumption of 1% cholesterol diet for 8 weeks. The amount of chow was restricted to 150 g/d. The food intake and body weight were evaluated daily and weekly, respectively. After the induction period, the atherogenic diet was substituted by a standard chow and animals were allocated to 3 groups, and they received the treatment described below (from 9th to 16th week): LDE-alone group: animals received intravenous injections of LDE only once a week for 8 weeks (n = 11); LDE-PTX group: animals received intravenous injections of LDE-PTX (4 mg/kg) for once a week for 8 weeks (n = 10); LDE-PTX + LDE-MTX group: animals received intravenous injections of LDE-PTX + LDE-MTX (4 mg/kg of both drugs) once a week for 8 weeks (n = 11).
To confirm the presence of atherosclerotic lesions on the aorta, 6 rabbits were euthanized after the 8-week atherosclerosis induction period. These rabbits were included in the control group. After the end of experimental period, all the rabbits were euthanized using a lethal dose of 200 mg/kg of sodium pentobarbital intravenously.
Preparation of LDE-PTX and LDE-MTX
To increase lipophilicity and optimize the formation and stability of the association with LDE, derivatives of PTX (paclitaxel oleate) and MTX (didodecyl methotrexate) were synthetized for the manufacture of the LDE-PTX and of the LDE-MTX preparations, as previously described. 27 Both formulations, LDE-PTX and LDE-MTX, were prepared from a lipid mixture composed of 100 mg cholesteryl oleate, 200 mg egg phosphatidylcholine (Lipoid, Ludwigshafen, Germany), 10 mg triglycerides, and 12 mg cholesterol. To this mixture, 60 mg PTX or MTX were added. The aqueous phase (100 mg of polysorbate 80 and 10 mL of Tris–HCl buffer pH 8.05) was kept at room temperature. Emulsification of the compounds was obtained by high-pressure homogenization using an Emulsiflex C5 homogenizer (Avestin, Ottawa, Ontario, Canada). After homogenization at constant temperature, the nanoemulsion was centrifuged at 3000 rpm for 15 minutes to separate the emulsified from the unbound drug. The nanoemulsion was sterilized by passage through 0.22 μm pore filter (Millipore, Billerica, Massachusetts) and was kept at 4°C until it was used. For dose administration, quantification of PTX or MTX associated with LDE was performed by high-performance liquid chromatography. As measured by laser light scattering (ZetaPALS Zeta Potential Analyzer; Brookhaven Instruments, Holtsville, New York), both formulations had nanoparticle diameter 45 to 60 nm.
Serum Biochemistry and Blood Cell Count
Blood samples were collected from all rabbits before and after the 8-week atherogenic diet as well as after the regression phase. Blood was drawn from the marginal ear vein after 12 hours of overnight fasting for determination of serum levels of total cholesterol, high-density lipoprotein cholesterol (HDL-c), triglycerides, urea, glucose, creatinine, alanine aminotransferase (ALT), aspartate aminotransferase (AST) by commercial kits (Labtest, Lagoa Santa, MG, Brazil), and blood cell count (erythrocytes and leukocytes).
Analysis of the Atherosclerotic Lesion Area in the Aorta
Aortas were excised from the aortic arch to the abdominal aorta opened longitudinally, washed with saline, and placed in 10% buffered formalin at pH 7.0. After fixation, the lipid deposits in the aorta were stained by Scarlat R (Sudan IV; Sigma-Aldrich, Saint Louis, Missouri), and the aortas were photographed to perform the measurements.
Total area and lesion area were measured using Leica Q500 iW (Leica Imaging Systems, Cambridge, United Kingdom). The extent of atherosclerotic lesions was expressed as percentage of aorta covered by lesions.
Histological Analysis
For general architecture of plaques, histological analyses and immunohistochemistry were performed on representative 4-µm segments of aortic arch tissue samples. Samples were stained with hematoxylin–eosin to measure the total and intima layer area. To assess the cellular composition of the lesion, monoclonal antibodies antimuscle α-actin, clone 1A4 for vascular SMCs, and anti-rabbit macrophage clone RAM11 for macrophages (Dako, Glostrup, Denmark) were used . To assess the presence of metalloproteinase 9, the anti-rabbit matrix metalloproteinase (MMP) 9 clone 56-2A4 antibody was used (Abcam, Cambridge, Massachusetts).
The images obtained by microscopy were analyzed using Leica Q500 iW (Leica Imaging Systems, Cambridge, United Kingdom). Total and intima layer area were measured, and the percentage of lesion was calculated by the ratio intima area/total area.
Gene Expression Analysis
Aorta samples were homogenized in 1 mL of Trizol reagent according to the manufacturer’s instructions (Invitrogen, Carlsbad, California). Evaluation of RNA purification and quantification were performed by measuring absorbance at 260 and 280 nm in Nanodrop 2000 spectrophotometer instrument (Thermo Fisher Scientific, Waltham, Massachusetts). Ratios of 260/280 nm measures ranging from 1.8 to 2.0 were considered satisfactory for purity standards. Denaturing agarose gel electrophoresis of RNA was used to assess the quality of the samples. The first strand of complementary DNA (cDNA) was synthesized from 0.5 µg of total RNA, using the high capacity kit (Applied Biosystems, Foster City, California) according to the manufacturer’s specifications. The resulting cDNA was diluted with Tris-ethylenediaminetetraacetic acid buffer and was stored at −20°C for subsequent analyses.
Messenger RNA (mRNA) levels of interleukin (IL) 10, IL18, IL1b, tumor necrosis factor (TNF), vascular cell adhesion molecule 1 (VCAM1), C-C motif chemokine ligand 2 (CCL2 or monocyte chemoattractant protein-1 [MCP-1]), MMP9, and MMP12 were determined by TaqMan quantitative real-time polimerase chain reaction (qRT-PCR) analysis using StepOnePlus Real-Time PCR System (Applied Biosystems). Data were analyzed using the comparative Ct method. 28 In accordance with the method, the mRNA amounts of the target genes were normalized to the endogenous housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH #Oc03823402_g1; Applied Biosystems]. The assay identification of the target genes used are described as follows: IL10 #Oc03396940_m1, IL18 #Oc03399565_m1, IL1b #Oc03823250_s1, TNF # Oc03397715_m1, VCAM1 #Oc03397346_m1, CCL2 # Oc03823583_s1, MMP9 #Oc03397520_m1, and MMP12 #Oc03398610_m1; all assays purchased from Applied Biosystems. The equation 2− ΔΔ Ct was applied to calculate the gene relative expression comparing each sample of all studied groups to the mean of control group where ΔCt = Ct target gene − Ct GAPDH and ΔΔCt = ΔCt sample − mean ΔCt control group. Target and reference genes were amplified in duplicate for each sample in a volume of 12 µL containing 1.2 µL template cDNA diluted 1:20, 0.6 µL of primers and probes mixture (20× Taqman Gene Expression Assays), 6 µL of TaqMan Universal PCR Master Mix (Applied Biosystems) and RNAse-free water to complete a reaction final volume. The cycling conditions were performed as follows: 2 minutes at 50°C, 10 minutes at 95°C, and 40 cycles of 15 seconds at 95°C followed by 1 minute at 60°C.
Statistical Analysis
Normality tests were performed previously to determine the Gaussian distribution. For Gaussian distribution, data were compared using the 1-way analysis of variance (ANOVA) test, followed by Tukey post hoc test. For those data with non-Gaussian distribution, they were compared using the Kruskal-Wallis test, followed by Dunn test. In all analysis, parameters were considered significantly different when P < .05 (2-tailed P).
Results
Effects of Cholesterol Feeding (8th Week) and of the Treatments (16th Week) on Weight and on Hematological, Renal, and Hepatic Parameters
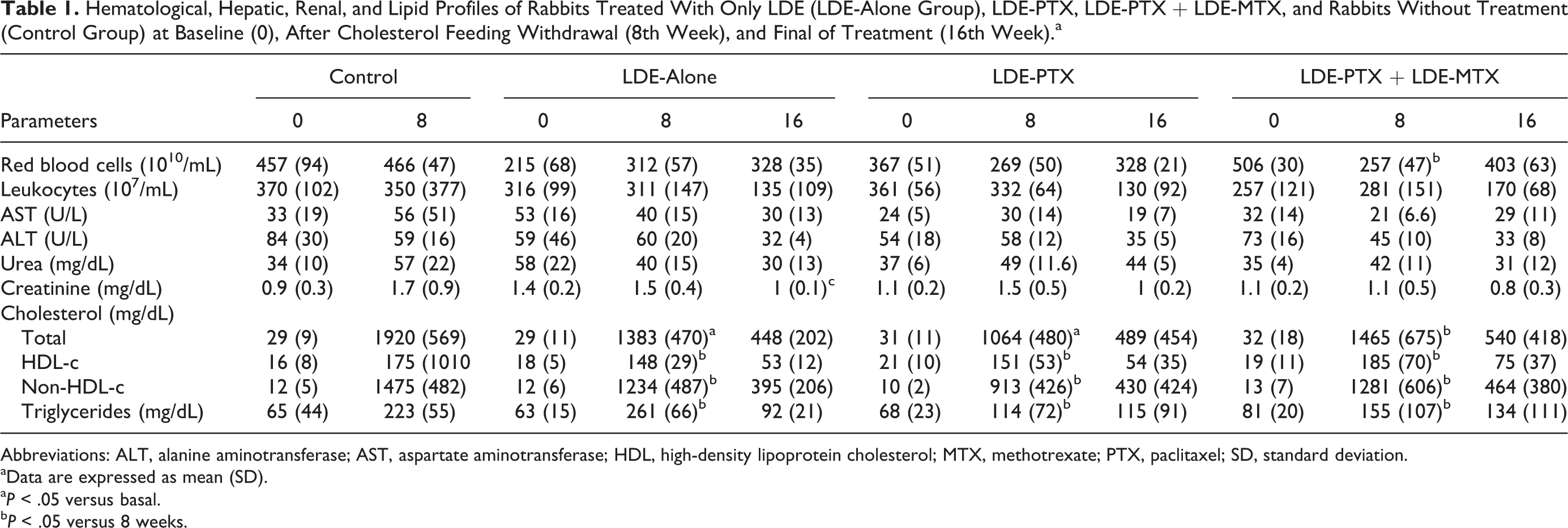
The weight of the animals did not significantly change from baseline (3.4 kg ± 0.12) to the 8th week (3.4 kg ± 0.18) and to the 16th week (3.6 kg ± 0.09). The LDE-PTX + LDE-MTX group showed a 50% reduction in red blood cell (RBC) count at the end of the 8th week, but by the end of the 16th week, the values were recovered back to the baseline level. In the remaining groups, the RBC count was unchanged in either the 8th or the 16th weeks. Leukocyte counts were unchanged after either 8th or 16th weeks in all groups (Table 1). Renal or hepatic toxicity was not observed as evaluated by creatinine, urea ALT, and AST serum concentrations (Table 1).
Hematological, Hepatic, Renal, and Lipid Profiles of Rabbits Treated With Only LDE (LDE-Alone Group), LDE-PTX, LDE-PTX + LDE-MTX, and Rabbits Without Treatment (Control Group) at Baseline (0), After Cholesterol Feeding Withdrawal (8th Week), and Final of Treatment (16th Week).a
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; HDL, high-density lipoprotein cholesterol; MTX, methotrexate; PTX, paclitaxel; SD, standard deviation.
aData are expressed as mean (SD).
a P < .05 versus basal.
b P < .05 versus 8 weeks.
Comparison Between Controls and LDE-Alone Group
As shown in Table 1, the 1% cholesterol chow intake resulted in about 60-fold increase in the values of total cholesterol, and HDL-c increased by 11-fold, as measured 8 weeks after the beginning of cholesterol feeding (controls). The withdrawal of the cholesterol intake during the ensuing 8 weeks (LDE-alone group) considerably decreased the plasma total cholesterol, but by the time they were euthanized, the total cholesterol values were still 15-fold higher than the basal, precholesterol feeding values. Similarly, HDL-c values were diminished 8 weeks after cholesterol feeding stopped, but they were still several-fold higher than the basal values.
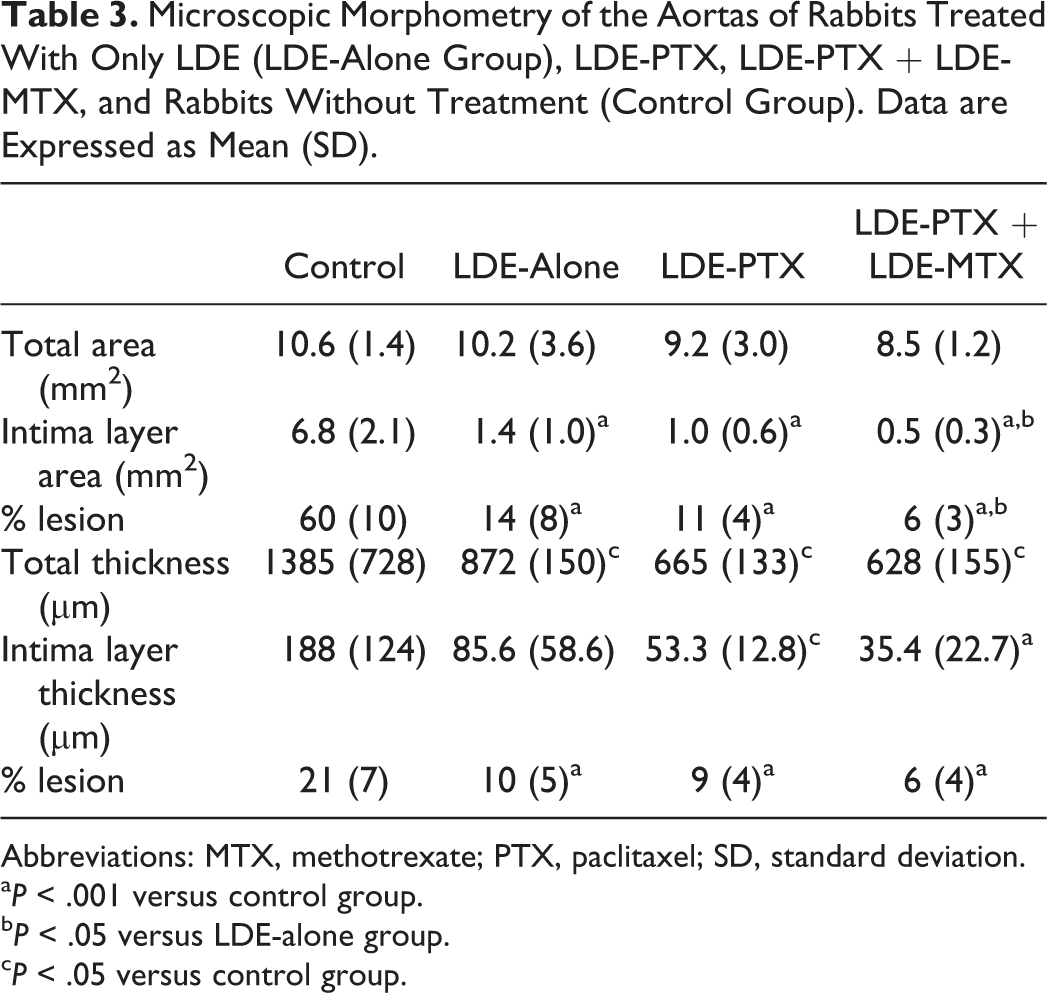
In respect to the atheroma plaque area (Table 2 and Figure 1), the cholesterol feeding withdrawal elicited 29% reduction of the plaque area by macroscopic analysis. This reduction, however, was not statistically different as compared with the group euthanized immediately after the 8-week period of cholesterol feeding. By microscopic analysis (Table 3), the width of intima layer was markedly 77% reduced by the sole withdrawal of cholesterol feeding (P < .001).
Macroscopic Morphometry of the Aortas of Rabbits Treated With Only LDE (LDE-Alone Group), LDE-PTX, LDE-PTX + LDE-MTX, and Rabbits Without Treatment (Control Group). Data are Expressed as Mean (SD).

Abbreviations: MTX, methotrexate; PTX, paclitaxel; SD, standard deviation.
a P < .001 versus control group.
b P < .05 versus LDE-alone group.
c P < .01 versus LDE-alone group.

Atherosclerotic plaques in the aorta. Representative photographs showing atherosclerotic lesions (stained by Sudan IV) in the aorta of rabbits. (A) Control group; (B) LDE-alone group; (C) LDE-PTX group, and (D) LDE-PTX + LDE-MTX group. MTX indicates methotrexate; PTX, paclitaxel.
Microscopic Morphometry of the Aortas of Rabbits Treated With Only LDE (LDE-Alone Group), LDE-PTX, LDE-PTX + LDE-MTX, and Rabbits Without Treatment (Control Group). Data are Expressed as Mean (SD).
Abbreviations: MTX, methotrexate; PTX, paclitaxel; SD, standard deviation.
a P < .001 versus control group.
b P < .05 versus LDE-alone group.
c P < .05 versus control group.
Effects of LDE-PTX and LDE-PTX + LDE-MTX on Serum Lipids and Atheroma Plaques
The different LDE drug treatments administered to the rabbits promptly after cholesterol feeding withdrawal did not affect the plasma lipids, as can be inferred from the comparison between the final lipid values of the groups treated with LDE-PTX or LDE-PTX + LDE-MTX and the LDE-alone group values (Table 1). The results of LDE-PTX and LDE-PTX + LDE-MTX treatments are compared with those obtained in the group of rabbits fed cholesterol during 8 weeks when the cholesterol-rich diet was withdrawn, and the animals were treated with LDE only for additional 8 weeks when they were euthanized (LDE-alone group).
In respect to the macroscopic atheroma plaque area (Table 2 and Figure 1), as compared to the LDE-alone group, LDE-PTX treatment resulted in 49% reduction in the plaque area (P < .05). The LDE-PTX + LDE-MTX resulted in 59% reduction in plaque area, which was significantly greater than that achieved by LDE-PTX (P < .01).
By microscopic morphometric analysis performed in aortic arch sections (Table 3), the area of the intima layer was reduced 57% by LDE-PTX + MTX, as compared to the LDE-alone group (P < .05). In contrast, treatment with LDE-PTX was not efficient to reduce the plaque area compared to LDE-alone group.
Effects of LDE-PTX and LDE-PTX + LDE-MTX on Macrophages, SMCs, and MMP9
Table 4 shows immunochemistry data obtained from the percentage of lesion calculated by the ratio intima area/total area of the LDE groups. The presence of macrophages decreased 48% in the LDE-PTX group (P < .05) and 43% in the LDE-PTX + LDE-MTX group (P < .05) in comparison with LDE-alone group. The presence of MMP9 was markedly 74% reduced by LDE-PTX and 78% by LDE-PTX + LDE-MTX treatments (P < .05). In contrast, the presence of SMCs in the intima was not changed by both treatments. Figure 2 shows photomicrographs illustrating macrophages, SMCs, and MMP9 in aortic slices stained by immunohistochemistry.
Evaluation of Presence of Macrophages, SMCs, and MMP-9 by Immunohistochemistry According to % of Stained Area at Atherosclerotic Plaque in Rabbits Treated With Only LDE (LDE-Alone Group), LDE-PTX, LDE-PTX + LDE-MTX in the Final Protocol (16th Week). Data are Expressed as Mean (SD).

Abbreviations: MMP metalloproteinase, MTX, methotrexate; PTX, paclitaxel, SD, standard deviation; SMC, smooth muscle cells.
a P < .05 versus LDE-alone group.
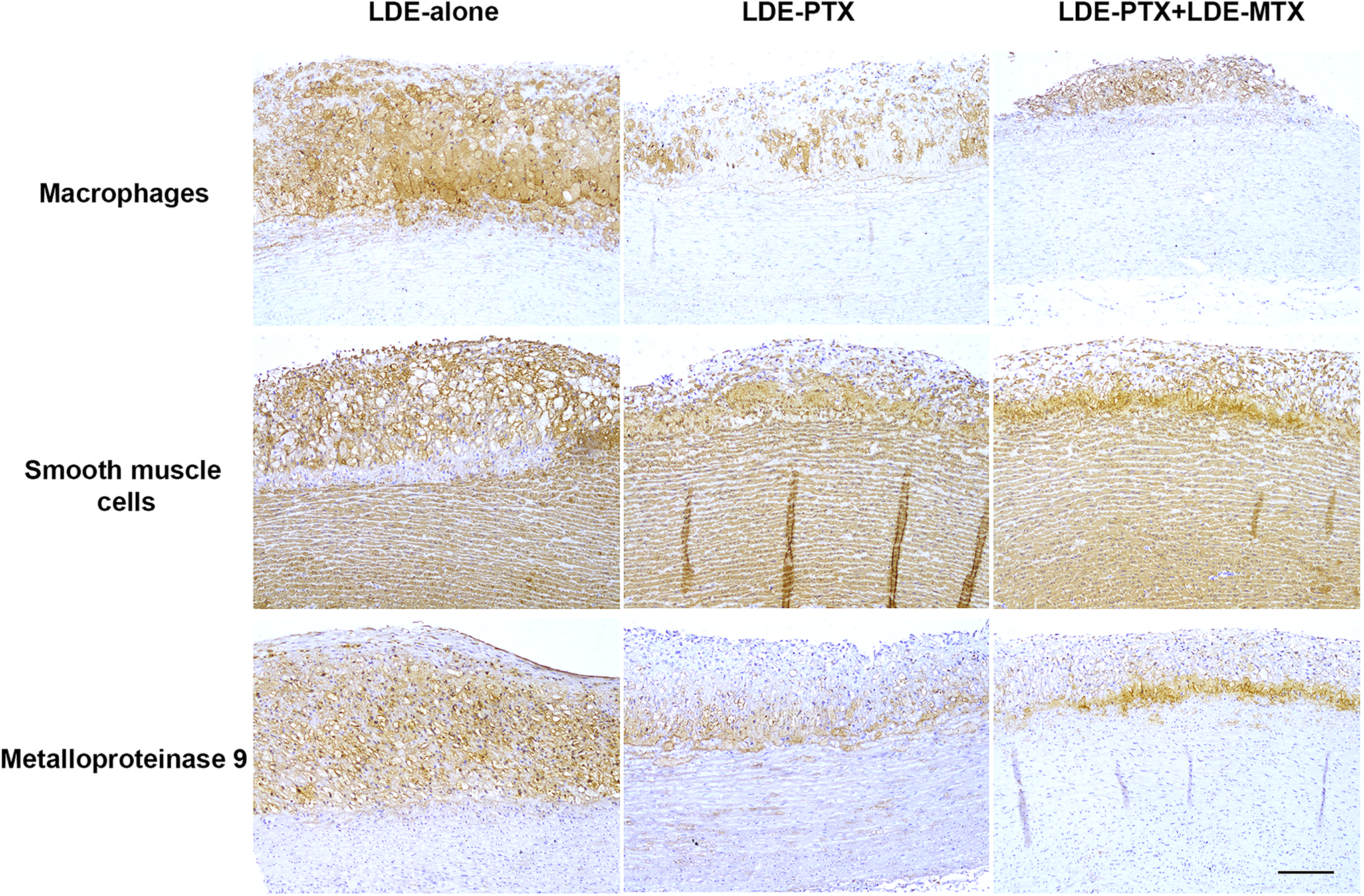
Representative photomicrographs of immunohistochemistry of aortic arch tissue for macrophages, smooth muscle cells, and metalloproteinase 9 under 100× magnification. Bar in photomicrographs stands for 100 µm. PTX indicates paclitaxel; MTX, methotrexate.
Effects of LDE-PTX and LDE-PTX + LDE-MTX on Expression of Inflammatory Genes
The gene expression analysis of IL-10, IL-1β, IL-18, TNF-α, VCAM, MCP-1, MMP-9, MMP-12 is shown in Figure 3. Comparing to the LDE-group, LDE-PTX and LDE-PTX + LDE-MTX treatment groups were ineffective in increasing IL-10 expression, an anti-inflammatory cytokine. Regarding pro-inflammatory markers, both treatments markedly decreased TNF-α expression. The LDE-PTX elicited a 65% of reduction (P = .006), and the effect of LDE-PTX + LDE-MTX was even greater at the order of 79% of reduction (P = .001) upon TNF-α expression. The gene expression of IL-18, IL-1β, VCAM, MCP-1, MMP-9, and MMP-12 was not reduced by either LDE-PTX or LDE-PTX + LDE-MTX treatments.
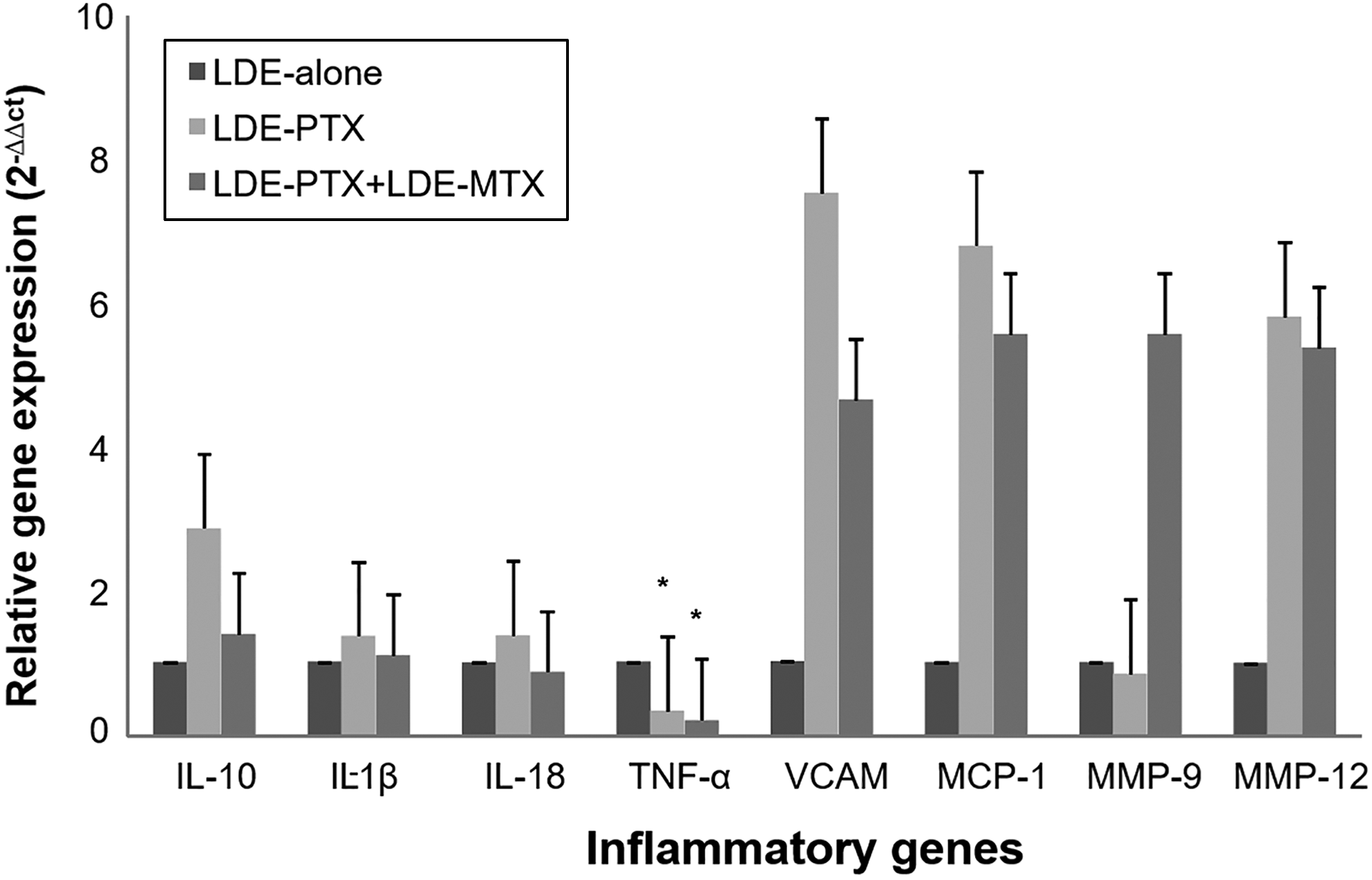
Gene expression of the inflammatory markers IL-10, IL-1β, IL-18, TNF-α, VCAM, MCP-1, MMP-9, MMP-12. Data are expressed as mean ± SE. *P < .01 versus LDE-alone. IL indicates interleukin; MCP, monocyte chemoattractant protein; MMP, matrix metalloproteinase. MTX, methotrexate; PTX, paclitaxel; TNF, tumor necrosis factor; VCAM, vascular cell adhesion molecule.
Discussion
In the previous experiments in which LDE carrying PTX, etoposide, MTX, or carmustine was tested, the classical atherosclerosis model of the high cholesterol-fed rabbits was also employed. 23 –26 In those previous experiments, rabbits were fed cholesterol for 4 weeks, and the LDE-drug treatments were introduced after the completion of the fourth week and lasted until the end of the 8-week cholesterol feeding when they were euthanized for analysis. In the current protocol, the period of cholesterol feeding previous to the commencement of the LDE-drug treatments was twice as long, that is, 8 weeks, and when LDE drugs were introduced, cholesterol feeding was interrupted. With this alternative strategy, we intended to test the effects of treatments on more advanced and presumptively more complicated lesions and also to exclude the effects of cholesterol feeding simultaneous to the ongoing LDE drug treatment.
In the LDE-alone group, the fact that there was a 77% reduction of the width of the intima layer 2 months after the suspension of cholesterol feeding does not imply that the nanoparticle per se has any capability to promote atherosclerosis reduction. It is well known that, in rabbits 29,30 or monkeys 31 with atherosclerosis induced by cholesterol-rich diet, the cholesterol feeding withdrawal results in pronounced lesion regression, in parallel with the reduction in plasma cholesterol.
The treatment of the rabbits with LDE-PTX had the ability to accelerate the reduction of the plaque area at the order of 50% compared with the LDE-alone group. However, the intima width was not changed by LDE-PTX. In our previous study, under the shorter (4 week) period of treatment with LDE-PTX, while the animals were still on continuous cholesterol feeding, LDE-PTX reduced the plaque area by 60% and also the intima width by 75%. 23 Comparing the 2 different protocols, in our previous study, LDE-PTX was probably acting not only to promote plaque regression but also to prevent the plaque development. In the current experimental protocol, only the regression component is at play, since by the time the treatment was introduced, the atherosclerosis causative factor, that is, cholesterol feeding, was interrupted.
Interestingly, whereas in the continuous cholesterol-feeding protocol both the macrophage and the SMC invasions of the intima were diminished by the LDE-PTX treatment, here only the macrophage intimal invasion was significantly accelerated by the treatment.
The most important outcome of the study was the finding that by adding LDE-MTX to LDE-PTX, the effect on regression of the lesion area was increased by 17% in comparison with LDE-PTX monotherapy. The reduction in the intimal width was 63% greater than that achieved by LDE-PTX. The mechanisms of action of the 2 agents are distinct. The antiproliferative action of PTX, the prototype of the taxane class of chemotherapeutic agents, is achieved by inhibiting the disassembly of microtubules during the mitotic spindle. 32,33 On the other hand, MTX, an antimetabolite compound, as a folate analog, competitively interferes with the tetrahydrofolate metabolism, inhibiting the key enzyme dihydrofolate reductase that is crucial to DNA and RNA synthesis. 34 –36 Whether the improvements achieved with the combination scheme resulted from addictive or synergic effects is difficult to establish. It is noteworthy that while LDE-PTX treatment reduced the lesion area, but did not change the width of the intima, LDE-PTX + LDE-MTX had the ability to produce both effects.
Regarding the presence of macrophages in the intima layer, LDE-PTX + LDE-MTX seemed not to have added up to the effects of LDE-PTX since the count of those cells in the intima was not different between the 2 treatments. The effect of LDE-PTX on the intimal content of SMCs was null in comparison with the LDE-alone treatment, but LDE-PTX + LDE-MTX was also ineffective. Then, it can be hypothesized that the effectiveness of the 2 treatments in accelerating the postcholesterol feeding atherosclerosis regression was rather due to the macrophage reduction effect and not as inhibitors of the migration of SMCs to the intima. In our previous studies, in which treatments with LDE-PTX 23 or with LDE-MTX 25 were performed without interrupting cholesterol feeding, the former treatment resulted in marked reduction of both macrophage and SMC migration, whereas the latter elicited macrophage reduction only, without effects on SMC migration to the intima. In this setting, the fact that the treatment being installed during or after cholesterol feeding determined differences in therapeutic response. These differences are conceivably related to the presence or not of continuous aggression to the endothelium by the massive amounts of proatherogenic lipoproteins consequent to the high cholesterol feeding. The presence of MMP-9, an atherogenesis-related factor secreted by macrophages, 37 was also equally reduced by LDE-PTX and LDE-PTX + LDE-MTX. On the other hand, the gene expression of pro-inflammatory TNF-α in the aorta was decreased by LDE-PTX monotherapy but still more deeply inhibited by LDE-PTX + LDE-MTX. Tumor necrosis factor α is secreted by several cell types, including macrophages and endothelial cells. 38 The fact that LDE-PTX + LDE-MTX had a more potent inhibitory effect on this cytokine has implications for the manifestations of atherosclerosis, since high TNF-α levels, as also postulated for MMP-9, are predictors of plaque instability. 39 –43
Recently, Maranhão et al reviewed the toxicity of several nanoparticle systems tested to date at the clinical setting, 9 showing that the ability of the LDE system to almost completely abolish the toxicity of several antineoplastic drugs is indeed unique. In regard to the LDE-PTX preparation, the absence of toxicities at high dose of PTX was extensively observed in studies with experimental animals and patients with advanced cancers, and recently, a safety study was performed that confirmed the viability of the use in patients with cardiovascular diseases. 44 The lack of observable toxicity shown here with the combination LDE-PTX and LDE-MTX and the clear-cut superiority of antiatherosclerotic action of the combined chemotherapy agents may pave the ground for this novel therapeutic approach for cardiovascular diseases.
In conclusion, our current results confirmed by an alternative protocol in which the treatment commences after cholesterol withdrawal that LDE-PTX was effective in accelerating the regression of the atherosclerotic lesions. More importantly, it was also shown that the combined chemotherapy with PTX and MTX can be a more potent antiatherosclerosis approach to be tested in future clinical trials. This treatment could be destined to achieve rapid atherosclerosis regression and control of the manifestations of the disease in severe cases, wherein a more energetic treatment is required.
Footnotes
Authors’ Note
Dr Maranhão has a Research Career Awards from the National Council for Scientific and Technological Development (CNPq, Brasília, Brazil). This study was performed at the Heart Institute (InCor) of the Medical School Hospital, University of São Paulo, São Paulo, Brazil.
Author Contributions
Gomes, F contributed to conception and design contributed to acquisition, analysis, and interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy. Maranhão, R contributed to conception and design contributed to analysis and interpretation drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy. Tavares, E contributed to acquisition and analysis contributed to acquisition and analysis critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy. Carvalho, P contributed to acquisition and analysis drafted manuscript critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy. Higuchi, M contributed to acquisition and analysis critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy. Mattos, F contributed to interpretation critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy. Pitta, F contributed to interpretation critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy. Hatab, S contributed to interpretation critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy. Kalil-Filho, R contributed to interpretation critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the State of São Paulo Research Support Foundation (FAPESP, São Paulo, Brazil) and from the Research Support in Cardiology of Brazilian Society of Cardiology (SBC, São Paulo, Brazil).
