Abstract
After thickening of the cardiac chamber walls during embryogenesis, oxygen and nutrients can no longer be adequately supplied to cardiac cells via passive diffusion; therefore, a primitive vascular network develops to supply these vital structures. This plexus further matures into coronary arteries and veins, which ensures continued development of the heart. Various models have been proposed to account for the growth of the coronary arteries. However, lineage-tracing studies in the last decade have identified 3 major sources, namely, the proepicardium, the sinus venosus, and endocardium. Although the exact contribution of each source remains unknown, the emerging model depicts alternative pathways and progenitor cells, which ensure successful coronary angiogenesis. We aim to explore the current trends in coronary artery development, the cellular and molecular signals regulating heart vascularization, and its implications for heart disease and vascular regeneration.
Introduction
Within the last decade, heart diseases have been the leading cause of death around the world. 1 The urgency of understanding coronary development is crucial for millions of patients who undergo expensive and often invasive surgical procedures. Research into coronary angiogenesis and vascularization of the heart may lead to the development of more cost-effective treatments. 2 –10 To this end, the origin of coronary arteries (CAs) has been a topic of great interest, which has led to a greater understanding of anatomic principles and the creation of more accurate and sophisticated tools for mapping cell differentiation (Figure 1).

Anatomy of the coronary arteries.
Models depicting the genesis of CAs have evolved over time. A century ago, CAs were thought to bud off directly from the aortic root. 11 –14 Later, Hutchins et al proposed a mechanical explanation for the development of CA from the right and left aortic sinuses of Valsalva. 15 This mechanical explanation appeared plausible due to the outwardly convex shape of the nonfacing sinus and the 3 pulmonary sinuses. However, studies using immunohistochemistry have challenged this view by demonstrating that coronary vessels develop even before invading the aortic root. 16 Since then, other biological models or pathways have been proposed to account for coronary origin, namely, the proepicardium, the endocardium, and the sinus venosus. 17 –20 As a result, there is a need for a comprehensive review that weighs the current evidence in the literature on coronary embryogenesis and its potential role in vascular repair.
The Developing Heart
The heart develops from an avascular primordial tube. It is one of the first organs to develop during embryogenesis, which maintains its pumping action while undergoing continuous physiological remodeling. 21 In the early stages of development, the heart undergoes structural reorganization into 3 layers of cardiomyocytes: the endocardium (inner layer), myocardium (middle layer), and the epicardium (outer layer). These layers receive nutrients and oxygen through diffusion across the endocardial endothelium. As the myocardial wall thickens, the endocardial surface area also increases by progressive trabeculation. 22
Coronary development involves an intricately timed series of events, namely vasculogenesis, angiogenesis, and/or arteriogenesis (Figure 2). 23 –25 Vasculogenesis is the differentiation of endothelial precursor cells that leads to the creation of new vessels. On the other hand, angiogenesis is the generation of microvessels from preexisting vessels by means of sprouting or nonsprouting. 5,26,27 Once new vessels are formed, further development may occur through intussusception, which increases the surface area of a vascular network via the formation of capillary pillars that bridge adjacent capillaries. 28 A more clinically significant form of collateral growth is arteriogenesis, which occurs following coronary arterial obstruction. Arteriogenesis involves the creation of mature vessels from preexisting interconnecting arterioles. 29,30

Temporal development of coronary arteries. Progenitor cells migrate to form a primitive vascular plexus. Then, the plexus attaches to the aorta, which allows blood flow. The coronary arteries undergo remodeling, which ultimately leads to mature coronary vessels.
As the heart folds, a primordial vascular network replaces the mechanism of passive diffusion that became insufficient in supplying oxygen and nutrients to the walls of the heart as they continued to develop and thicken. This primary vascular plexus subsequently expands into the heart in an epi-to-endocardial direction. Through coronary angiogenesis, capillaries sprout toward the heart base, which eventually connect to the aorta. The endothelial strands can be found at all 3 sinuses, but later decrease in number until only 2 channels persist to form the proximal segments of the left and right CAs. 31,32 These channels acquire a muscular coat via the migration of supporting smooth muscle cells (SMCs) and pericytes from the epicardium. 33 –39
The CAs are anatomically positioned at the thinnest part of the aortic wall. Until now, it has been postulated that postnatal expansion of the myocardial vascular bed only proceeds through angiogenesis. 40 However, recent findings suggest that bone marrow–derived endothelial precursor cells contribute to neovascularization in the heart during the neonatal period, consistent with a vasculogenic paradigm. 41,42 This vascular network remodels and expands to form the mature coronary vascular system that supplies the adult heart. 43
Reevaluating Vascular Origin
The predominant view from the early 1990s through the mid-2000s was that coronary vessels formed through a vasculogenic process from proepicardial-derived mesenchymal cells. 44 –48 The proepicardial organ (PEO) is a transient embryonic outgrowth of the septum transversum that contacts and spreads over the developing heart tube to form the outer layer. Interestingly, the PEO is a molecularly compartmentalized structure that consists of subsets of multipotent cells. 18 Concomitantly, some of these cell subpopulations within the PEO, such as Semaphorin 3D (Sema3c), contribute more toward coronary endothelial cells (ECs) than others, that is, Tbx18. 18 However, precursor cells migrate anteriorly from the posterior side of the heart toward the aortic root. 49,50 Proepicardial organ cells also undergo an epithelial to mesenchymal transition (EMT), highly conserved throughout species. 51 –53
Subsequent studies have identified other extra-cardiac structures as the predominant source of CA ECs. Poelmann et al traced endothelial precursor cells that were derived from the liver primordium. 31 This study demonstrated that precursor cells merged into small vessels that attached to the sinus venosus before connecting to subepicardial cells followed by the aortic root. Red-Horse et al and De Andres et al described CA as being principally derived from differentiated ECs that sprout from the sinus venosus. 17,54
Clonal labeling experiments using the endothelial-specific marker tamoxifen-induced Cre (Cdh5-CreERT2) demonstrated that a majority of clones (74%) were genetically similar to sinus venosus cells and not to PEO cells. Coronary ECs arise from sinus venous precursor ECs through angiogenesis, where they dedifferentiate and proliferate to form the coronary vasculature. 55 Differentiated sinus venous cells maintain elastic potential, which suggest that site-specific cardiac signals trigger their dedifferentiation into the CA, capillaries, and veins. 17
Lineage tracing in mammalian studies all do not mirror findings in avian studies and propose that although the proepicardium gives rise to myocardial stroma and vascular smooth muscle, it is only a minor contributor to coronary ECs. 56 –59 Further investigations using ventricular endocardium-specific Nfatc1 regulatory elements 55 and AplnCreERT219 in mice did not label PEO, epicardium, liver bud, or sinus venosus cells. These fate mapping studies demonstrated that ventricular endocardium-derived cells are not terminally differentiated but contribute to the development of the endothelium of the CA (72%) and their intramyocardial network (81%). 55 Although Nfatc1 expression was not traced to the sinus venous, other markers for ventricular endocardial-derived cells, Nfatc1Cre-mediated β-gal or enhanced green fluorescent protein (EGFP) expression was observed in some sinus venous ECs. This indicated that Nfatc1+ endocardial cells contribute to a subgroup of sinus venous ECs.
Furthermore, Tian et al showed, using genetic lineage tracing, that a substantial portion of postnatal coronary vessels arise de novo rather than expanding from preexisting embryonic vasculature. 60 The study reported that myocardial compaction of coalesce trabeculae leads to regional hypoxia, which stimulates trapped endocardial cells to form a rapid vascular supply. The finding shows an alternative pathway for coronary development, which may have implications for regenerative therapy.
Indeed, CA arise from multiple sources, namely, the sinus venosus, endocardium, and proepicardium, the relative contributions of which changes between species. 61 It is also possible that ventricular endocardium-derived cells have a major contribution to the development of CA endothelium and their intramyocardial network. Angiogenic sprouting from the sinus venosus may generate most subepicardial coronaries and a subset of intramyocardial vessels. Proepicardial organ cells contribute only a fraction of EC cells and seem to play a more important role in the development of the myocardial stroma and vascular smooth muscle. These different sources or alternative pathways ensure successful development of the CA.
Smooth Muscle Cells and Pericytes
Another important source for CA is the smooth muscle layer, which develops during the remodeling of the primitive vascular plexus. In humans, coronary artery smooth muscle cells (CASMCs) envelops the CA, forming several smooth muscle layers, which serve to provide structural support. 62 Clinically, CASMCs have also been implicated in the pathogenesis of cardiovascular diseases. 63 As to their source, CASMCs are regarded as being highly heterogeneous as lineage-tracing studies have identified 8 possible sources including the proepicardium, neural crest, and more recently, endocardial-derived cardiac cushion mesenchyme. 64 –66 However, the proepicardium and neural crest are considered main contributors of CASMCs. 64
Although a large contribution is derived from the proepicardium, epicardial cells undergo EMT to allow migration into deeper layers of the myocardium and differentiate into SMCs or fibroblasts. 67,68 The decision to differentiate into SMCs or fibroblasts occurs prior to migration and EMT. Other studies indicate that epicardial-derived SMCs do not express the same biomarkers as mature SMCs. Volz et al identified pericytes as precursors of SMCs. 69 It is suggested that epicardial cells acquire pericytes during its migration beneath the epicardium. 70 Later, pericytes lining the primitive vascular plexus differentiate into SMCs through Notch signals. 71,72 Similar to epicardial cells, Chen et al demonstrated that endocardial ECs differentiate into mural cells (ie, pericytes and vascular SMCs). 66 Using a murine model, endocardial cells underwent EMT and reverted to a primitive mesenchymal progenitor, which expressed platelet-derived growth factor receptors (PDGFR), PDGFRα and PDGFRβ. The progenitor cells migrated into the myocardium where it differentiated and assembled the wall of the CA. 66
Kumar et al further characterized the origin of mural cells using human embryonic stem cells. 73 The study showed that clonal precursor mesenchymoangioblast, which originates from mesoderm, differentiated into CASMCs and pericytes. These findings further elucidate a lineage tree, which can be used to further explore development and regeneration. Overall, the current literature suggests that the CA has multiple sources or pathways for smooth muscle layer development. Sharma et al reported that having alternative pathways and/or progenitor cells ensures successful development when the original is compromised. 74
Coronary Artery Malformations and Vascular Endothelial Growth Factor–Induced Angiogenesis
Anatomic variations of CAs are rare in the general population and can be classified into anomalies of origin, course, or distribution. 75 Clinically important variations include those with an ectopic origin, an interarterial, or intramural course due to reduced coronary blood flow. 76 –79 The duplication of coronary vessels is usually benign; however, multiple anterior interventricular arteries can create technical difficulties during surgical reconstruction when present alongside other cardiac congenital defects such as tetralogy of Fallot. 80
Congenital coronary artery anomalies (CAAs) have been shown to increase the risk of myocardial infarction or ischemia, congestive heart failure, and sudden cardiac death especially in young athletes. 81 –88 The faulty migration of precursor ECs or the downregulation of vascular endothelial growth factor (VEGF) receptors can explain the occurrence of aberrant coronary vessels. Members of the VEGF family are key regulators of the development of blood and lymphatic vessels. The VEGF family includes 5 growth factors in mammals: VEGF (VEGF-A, VEGF-B, VEGF-C, and VEGF-D) and placenta growth factor (Figure 3). 89,90 Increased expression of VEGF has been demonstrated in epicardial and subepicardial cells at the aortic root, and these molecules are thought to regulate EC penetration of the aorta. 91 Moreover, a direct intravenous injection of VEGF into ECs induces an angiogenic phenotype similar to those in coronary vessels. 92
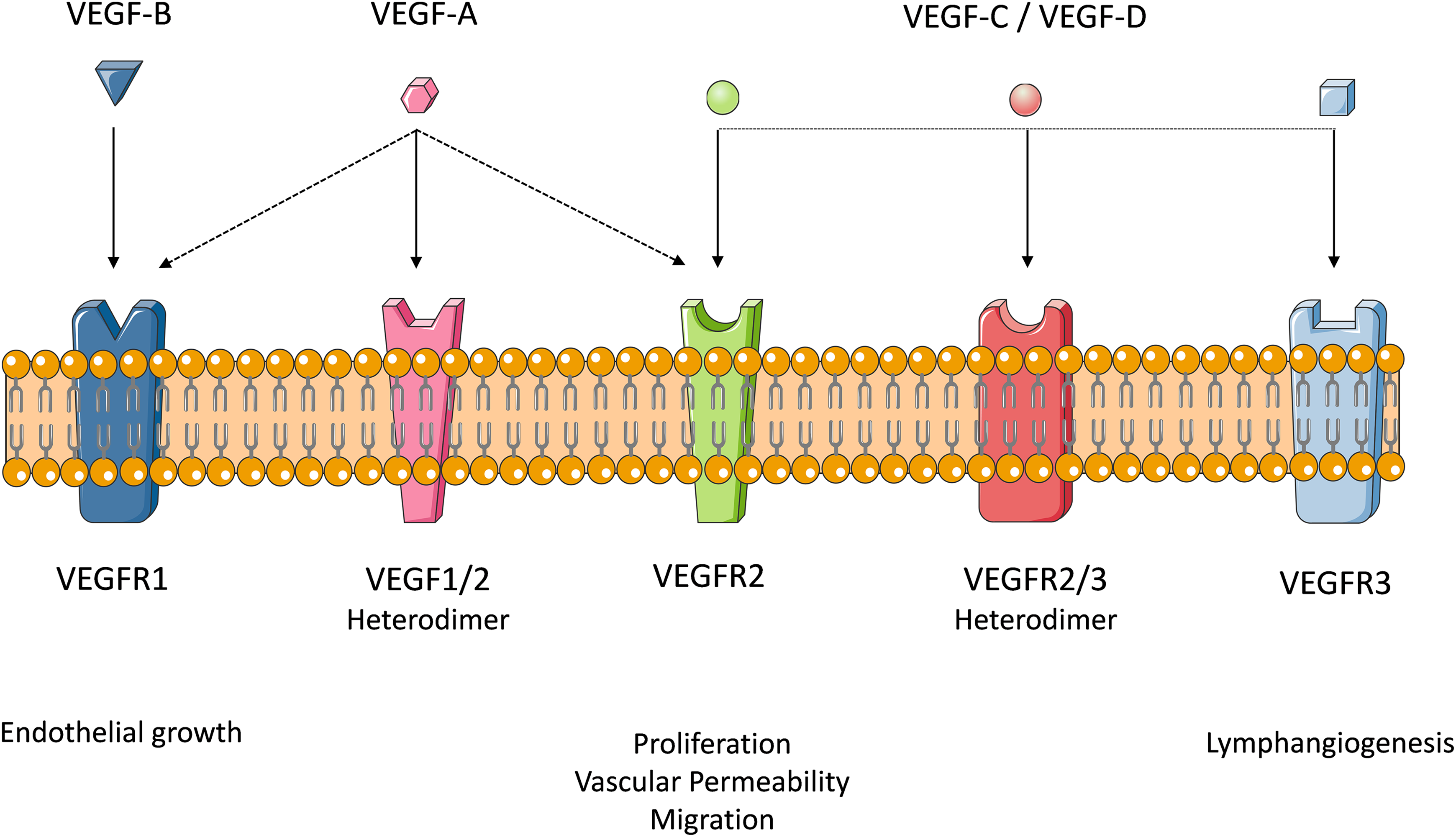
Members of the vascular endothelial growth factor (VEGF) family.
Specifically, VEGF-C, which is expressed around the aorta, guides the growing CA to the aorta, while avoiding connections to the pulmonary artery; specific cardiomyocytes within the aorta facilitate correct positioning of the coronary vessels. 93 The VEGF-C deletion studies show minimal development of coronary vessels or aberrant connections to the aorta. 93 Furthermore, while VEGF-B is spatially and temporally correlated with endothelial growth in the heart, its role in CA development appears minimal. The VEGF-B deficient mice demonstrate no vascular or developmental abnormalities. 94 However, when a myocardial infraction is induced, VEGF-B knockout mice show larger infarct scars but with intact heart function. Therefore, VEGF-B may play a more pathological role rather than developmental. 94
Myocardial VEGF-A is important for the development of the intramyocardial network. 55 Messenger RNA for the tyrosine kinase receptors, VEGF receptor (VEGFR) 2 and VEGFR3, were shown to be highly expressed at the site where the primitive capillary plexus invades the aorta to form the major CA. 95 The VEGFR-2-null embryos did not develop coronary plexuses in the peritruncal/coronary sulcus area or ventricular septum, which resulted in severely diminished or absent CAs in these embryos. 55 Together, the data from studying VEGF-A and VEGFR-2 null hearts indicate a hypoxic-dependent VEGF-A concentration gradient in the myocardium of the early heart. This gradient provides a signal through VEGFR-2, which leads to the migration of angiogenic forerunner cells from the endocardium to the myocardium.
Hypoxia, mechanical forces associated with blood flow, and stretch due to diastolic filling are potent stimulators for vasculogenesis, angiogenesis, and arteriogenesis through increased expression of VEGF, its receptors, and subsequent paracrine signaling. 96,97 To this end, understanding the embryogenesis of coronary vasculature continues to have a clinical impact in detecting CAA and methods for correcting subsequent defects. Wang et al identified a pool of angiogenic precursor cells expressing high levels of VEGFR3 with endocardial origin. 98 These precursor cells, VEGFR3high, are regulated by protein o-fucosyltransferase 1 (POFUT1), a regulator of Notch signaling. The study showed that inactivation of POFUT1 resulted in excessive angiogenic cell proliferation and plexus formation and ultimately to anomalous CA, myocardial infarction, and heart failure. The study provides evidence for genetic screening, which may potentially detect individuals at risk of coronary anomalies and regenerative therapies.
Additional cellular signaling events linked to the proper vasculogenesis and patterning of the CA include connexin43 (Cx43), Perlecan, T-box-containing transcription factor (Tbx-1), Sema3c, and morphogenetic protein (BMP) -binding EC precursor-derived regulator (BMPER). 99 Many of the patterning variations have been reported in mice strains with Cx43 mutations. Although Cx43 is part of the connexin family that forms gap junctions mediating diffusion of ions and signaling molecules, Cx43 is also capable of protein interactions, generating cell polarity during development of CA and maintaining proper cytoarchitecture. 100 The Cx43 knockout mice show phenotypes that include ectopic origin, abnormal course, and intramural tunneling. 101 –103 A single knockout of the Cx43 gene increases the likelihood of CA pattern abnormality, suggesting that vasculogenesis is sensitive to the signaling strength. 103
Another candidate gene with a role in CA development is Tbx-1, located on the human chromosome 22. This gene has also been associated with heart anomalies in DiGeorge syndrome, and knockout of Tbx-1 gene in mice has shown abnormalities of aortic arch arteries. 104 –106 Theveniau-Ruissy et al suggested that Tbx-1 plays a role in the positioning of second heart field cells. 107 Knockout of the gene affects the placement of CA at the base of the great arteries. Moreover, the Sema3c signaling pathway has been identified as a Tbx-1-dependent gene with the role of preventing CA growth in subpulmonary myocardium and establishing vessel free zones. 107 Other studies have highlighted BMPER as a new regulator for CA patterning. Depending on the signaling strength, BMPER can promote or inhibit CA growth. This method had shown to help remodeling of the coronary plexus during CA development and provides a novel approach for studying coronary embryogenesis. 108
Potential Translational Role in Vascular Repair
Research into coronary angiogenesis and vascularization of the heart could potentially lead to the development of more targeted treatments. The presence of multiple pathways leading up to CA development offers the possibility of vascular repair. Moreover, because these cell types are present in adults, it is possible that they may be stimulated to form new blood vessels. Phase 1 clinical trials have demonstrated that the administration of high doses of human recombinant VEGF significantly improved conditions of ischemia. 109 The same study showed insignificant results when low doses of human recombinant VEGF were administered. In another study, a phase 2 clinical trial using catheter-mediated VEGF-A gene delivery using plasmid liposomes or adenoviruses showed improved vascularity in patients experiencing intermittent claudication. However, the study showed no significant difference when compared to placebo. 110 Both studies appear to have promising results, although clinically ineffective. Hsu et al showed that the effectiveness of VEGF could be optimized when administered concurrently with CSF1, a macrophage recruiting cytokine. 111 The study proposes a synergistic strategy in promoting vascularization and tissue survival that is clinically effective compared to VEGF only.
The side-effect profile associated with exposure to angiogenic factors is concerning and represents a barrier for translational scientists. Experimental models show that long-term exposure to VEGF resulted in severe hypotension due to nitric oxide release, arteriolar vasodilation, and edema. 109,112,113 Other areas of concern are plaque growth or destabilization due to the mitogenicity and chemotactic activity toward macrophage and proliferative retinopathy. 114 Additionally, the effect of angiogenic factors on tumor growth and metastasis bears concern in susceptible patients. However, these effects have not been well established and appear to be rare occurrences. Overall, VEGF-induced angiogenesis has been physiologically well tolerated and effective, although concerns over its side effects and route of administration require further investigation to optimize its therapeutic potential. 115
Conclusion
Coronary arteries originate from 3 major sources, namely, the proepicardium, sinus venosus, and endocardium. Evaluating and quantifying the contribution of each proposed cell source in CA development are the next important steps in furthering our understanding of coronary growth patterns. As highlighted in this review, advances made in the development of CA endothelium-specific markers, high-resolution, and real-time imaging have paved the road to a better interpretation of these embryonic events. Understanding the cellular origin of coronary vessels has potential translational biomedical applications especially in cardiac revascularization and reparative treatments, as well as stem cell research for the management of coronary diseases. However, clinical trials are needed to evaluate their long-term safety.
Footnotes
Acknowledgments
The authors would like to acknowledge Mr Sahrin Samad for medical illustration.
Author Contribution
Ramai contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; and gave final approval. Lai contributed to conception and design; contributed to acquisition, analysis, and interpretation; critically revised the manuscript; and gave final approval. Monzidelis critically revised the manuscript and gave final approval. Reddy critically revised the manuscript and gave final approval. All authors agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
