Abstract
Background:
Platelet function testing was suggested to help tailor P2Y12-inhibitor therapy; however, the lack of proper standardization is still a limitation.
Methods:
In a prospective study, we enrolled clopidogrel-treated and P2Y12-inhibitor naive patients to investigate the influence of (1) time from blood collection, (2) stability of the stored Adenosine diphosphate (ADP) reagent, and (3) the use of enoxaparin on results of the Multiplate assay. Measurements were performed from samples kept for 0, 30, 60, 120, and 240 minutes at room temperature before processing. To determine the impact of the reagent stability, freshly thawed ADP was compared with ADP kept for 3 to 5 or 8 to 13 days at 2°C to 8°C. Finally, samples containing enoxaparin at therapeutic or prophylactic doses were compared with enoxaparin-free blood.
Results:
A total of 180 measurements were performed. ADP-stimulated platelet reactivity values decreased significantly over time (67 ± 40 U to 68 ± 37 U to 58 ± 37 U to 45 ± 33 U to 35 ± 33 U; P < .0001). Consequently, a dramatic reduction was observed in the proportion of patients with high platelet reactivity (P < .0001). A significant drop in platelet reactivity was observed with ADP stored for 8 to 13 days as compared to freshly thawed ADP (P = .011). Enoxaparin triggered a slight, concentration-dependent increase in platelet reactivity (P < .05).
Conclusion:
Test conditions may have profound impacts on the obtained results with the Multiplate assay. Our findings highlight the large influence of the time from sample collection until testing, suggesting that measurements should be performed within an hour of blood collection.
Introduction
Percutaneous coronary intervention (PCI) with stent implantation is a default treatment option for patients with ischemic heart disease. All patients undergoing PCI need to be treated with combined antiplatelet therapy for a certain time frame, featuring aspirin, and a P2Y12 inhibitor. 1 Among P2Y12 inhibitors, clopidogrel is still the most widely used agent; however, its platelet inhibitory effect shows high variability in the population due to the unpredictable bioavailability of the drug. 2 To overcome this limitation, novel ADP-receptor antagonists, such as Prasugrel and ticagrelor, have been developed, which have more predictable and more potent antiplatelet effect compared to clopidogrel. However, the administration of such potent platelet inhibitors was associated with a significant increase in the rate of bleeding events. 3,4 Therefore, balancing between thrombotic and bleeding risk after stent implantation is challenging in the daily practice. Platelet function testing (PFT) might be a helpful tool to obtain information regarding the expected rate of bleeding and thrombosis since high platelet reactivity (HPR) is a predictor of adverse thrombotic events and early stent thrombosis after PCI, 5,6 whereas low platelet reactivity (LPR) is associated with higher risk of bleeding. 5,7
However, routine use of PFT is not only supported by current guidelines in part because of negative outcome studies on platelet function–guided therapy but also due to potential methodological issues influencing the results of platelet reactivity testing. 8 -10 To increase reliability and reproducibility, devices with a certain level of standardization were developed such as the Multiplate Analyzer (Roche Diagnostics, Rotkreuz, Switzerland) that was shown to render more reproducible results compared to conventional light transmission assay. 11 However, there are still important methodical aspects of the testing process that are not well standardized in current protocols or user manuals that may result in false results and misclassification of patients. Therefore, we sought to investigate the potential impacts of some important test conditions on ADP-dependent platelet reactivity values obtained by the Multiplate analyzer.
Methods
Patient Population
In a single-center, prospective study, we aimed to enroll clopidogrel-treated (>5 days on 75 mg clopidogrel) and clopidogrel-naive patients to test the impact of various methodical issues on ADP-dependent platelet reactivity values obtained with the Multiplate analyzer. All patients were treated with 100 mg of aspirin. Detailed clinical features of the investigated cohort are shown in Table 1.
Baseline Clinical Characteristics of the Patient Group.

Abbreviations: CABG, coronary artery bypass grafting; DIET, dietary; INS, on insulin treatment; MI, myocardial infarction; OAD, on oral antidiabetic treatment; PAD, peripheral arterial disease; PCI, percutaneous coronary intervention; SD, standard deviation.
Studied Methodology Issues
In this study, 3 different methodical issues were evaluated: (1) time from blood collection until testing, (2) stability of the thawed ADP reagent, and (3) impact of enoxaparin on platelet reactivity values.
Blood samples were drawn by direct venipuncture using a 19-gauge needle into three 9-mL plastic tubes, containing 1/10 volume recombinant hirudin. To investigate the influence of the time elapsed until testing, 1 hirudin tube was kept at room temperature and sampled immediately (0 minutes), 30 minutes, 60 minutes, 120 minutes, and 240 minutes after blood collection. According to the recommendation of the manufacturer, Multiplate testing should be performed within 3 hours of blood collection. 12,13
To investigate the stability of the ADP reagent, blood samples were tested with freshly thawed ADP, with ADP kept for 3 to 5 days and 8 to 13 days after thawing at temperatures of 2°C to 8°C in a refrigerator. All “freshly thawed” ADP reagents were frozen before use, for no more than 20 days. The reagent kept for 8 to 13 days served as a “negative” control for the other 2 measurements because according to the recommendation of the manufacturer, thawed ADP can be used for up to 7 days if kept on 2°C to 8°C. 12,14
Finally, there is no specific recommendation regarding the presence or absence of enoxaparin in the tested samples. However, there may be many clinical scenarios when patients on enoxaparin may undergo PFT. Therefore, to determine the potential influence of enoxaparin on the results of PFT, 2 different enoxaparin concentrations were established. Due to the fact that enoxaparin blood concentrations may be subject of high interpatient variability when administered in subcutaneous or intravenous route, we aimed at creating a standardized environment within the standard hirudin tubes by adding enoxaparin directly into the tube 10 minutes before assessments. In detail, 1:100 mixture of enoxaparin and saline was added to the hirudin blood tube to achieve therapeutic (0.6 IU/mL anti-Xa activity) or prophylactic (0.3 IU/mL anti-Xa activity) concentrations. An independent anti-Xa activity test (Technochrom anti-Xa; Technoclone GmbH, Vienna, Austria) was used to confirm the appropriate enoxaparin concentrations in the blood collection tube. A schematic description of the protocol is depicted in Figure 1.

Time points of platelet function testing during the study.
Platelet Function Testing
Mutliplate electrode aggregometry is a point-of-care assay based on impedance aggregometry and utilizes the analysis of hirudin-anticoagulated whole blood diluted with 0.9% NaCl solution. No platelet-rich plasma preparation or platelet count adjustment is needed when performing the measurement. Platelets activated by added ADP adhere and aggregate on the surface of bare metal electrodes, demonstrating the setting of stent thrombosis. The changes in electric impedance between the 2 electrodes are transformed into arbitrary aggregation units (U) and plotted over time drawing an aggregation curve. The area under the aggregation curve (AUC) value represents the efficacy of platelet inhibition. This assay is a computer-based system, possesses standardized measurement methods 13 and operates with automatic pipette and fix concentrations of ADP. 2,14 HPR was defined as an ADP-dependent platelet reactivity value greater than 46 U according to the recent consensus document. 2 Multiplate analyzer has been validated in several prior clinical studies to predict clinical outcomes. 15,16
Statistical Analysis
Continuous variables are presented as means (standard deviation). Categorical variables are expressed as frequencies and percentages. Presence or absence of the normal distribution of continuous variables were tested with the Kolmogorov-Smirnov test. In case of values of normal distribution, groups were compared with repeated measures of analysis of variance tests. In case of values of non-Gaussian distribution, the nonparametric Wilcoxon matched pairs signed rank test was performed. Differences between the rates of HPR were determined using contingency tables in χ2 test. A P value < .05 was considered statistically significant in all analysis. Statistical analyses were performed with GraphPad Instat version 3.00 for Windows (GraphPad Software, San Diego, California; www.graphpad.com).
Results
From January 2015, 20 patients were enrolled in the study: 11 clopidogrel-treated and 9 clopidogrel-naïve patients. Baseline characteristics of the studied patients are depicted in Table 1. Nine separate assessments were performed in all patients, resulting in a total number of 180 measurements in the study cohort.
Regarding the time elapsed from blood collection, our results showed that platelet reactivity significantly decreased over time: after an initial, slight increase at 30 minutes, a significant drop was observed from 60 until 240 minutes (67 ± 40 U to 68 ± 37 U to 58 ± 37 U to 45 ± 33 U to 35 ± 33 U; P for trend <.0001; Figure 2A and C). Consequently, the rate of HPR decreased significantly from 70% observed at 30 minutes to 20% at 4 hours (P <.0001; Figure 2B).
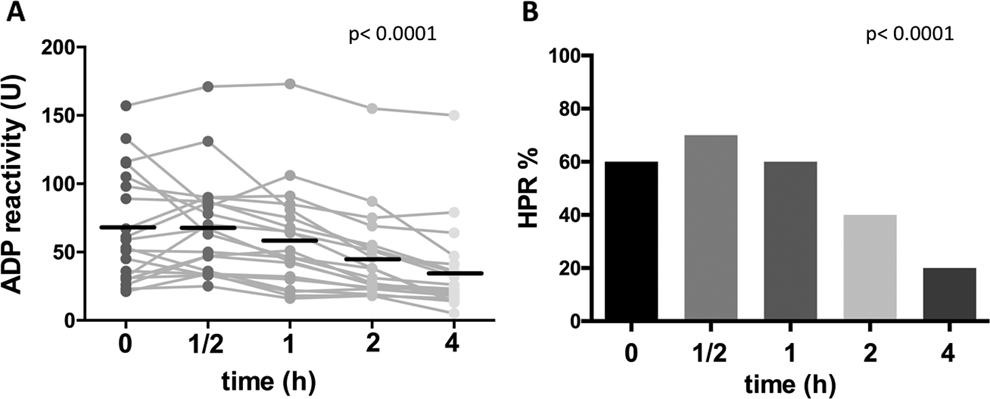
Impact of time from blood collection until assessment on the level of ADP-dependent platelet reactivity values (A) and on the rate of high platelet reactivity (HPR, B). Results show Multiplate ADP-test values in U. HPR was defined as a value above 46 U.
When clopidogrel-treated and naive patients were analyzed separately, similar results were observed with a slight, but more pronounced peak at 30 minutes in the clopidogrel-treated patients (Figures 3 and 4).
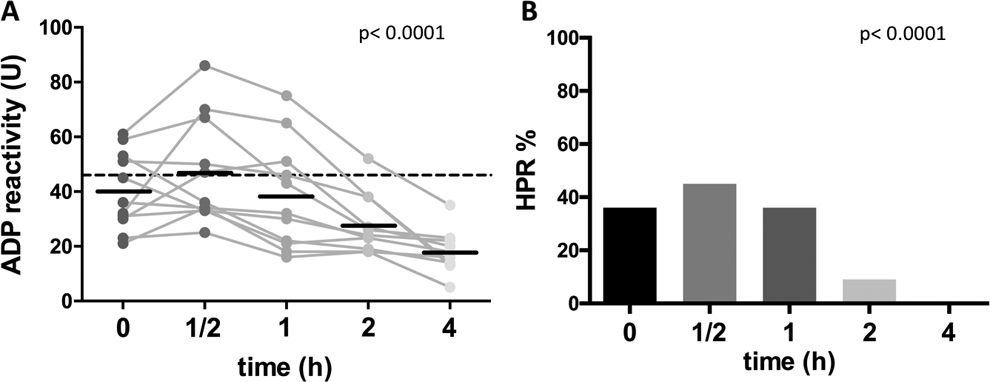
Impact of time from blood collection until assessment on the level of ADP-dependent platelet reactivity values (A) and on the rate of high platelet reactivity (HPR, B) in clopidogrel-treated patients. Results show Multiplate ADP-test values in U. HPR was defined as a value above 46 U.

Impact of time from blood collection until assessment on the level of ADP-dependent platelet reactivity values (A) and on the rate of high platelet reactivity (HPR, B) in clopidogrel-naive patients. Results show multiplate ADP-test values in U. HPR was defined as a value above 46 U.
Regarding the stability of the ADP reagent, a statistically significant, yet slight decrease was observed in platelet reactivity values using ADP stored for 8 to 13 days compared to freshly thawed reagents (P = .011); however, there was no statistically significant difference between the freshly thawed ADP, and the 1 kept for 3 to 5 days at 2°C to 8°C (P = .97; Figure 5). Similarly, no decrease was observed regarding the rate of HPR between the 3 groups (P = .23).
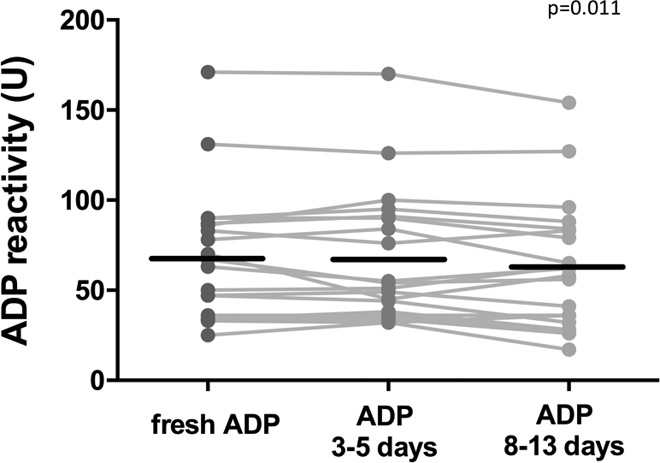
Impact of ADP stability on the level of ADP-dependent platelet reactivity values. Results show multiplate ADP-test values in U.
In case of enoxaparin usage, a significant, concentration-dependent, but slight increase was observed regarding platelet reactivity when therapeutic and prophylactic concentrations were added to hirudin tubes (P = .0024; Figure 6A). The increase in the rate of HPR was not significant (P = .33; Figure 6B).

Impact of 2 enoxaparin concentrations on the level of ADP-dependent platelet reactivity values (A) and on the rate of high platelet reactivity (HPR, B). Results show multiplate ADP-test values in U. HPR was defined as a value above 46 U.
Discussion
Multiplate is a relatively well-standardized platelet function assay based on impedance principles that was validated to predict stent thrombosis, bleeding, and mortality in a large sample of coronary patients treated with clopidogrel. 15,16 In a recent meta-analysis, the cutoff of 46 U and 19 U were shown to be able to separate patients with high platelet reactivity (HPR) and LPR that were associated with an increased risk of stent thrombosis and bleeding, respectively. 7 Therefore, a recent consensus document recommended these sharp cut points to define HPR and LPR. 2 However, there are many aspects of the platelet function assessment that are not strictly regulated and may interfere with the obtained results.
In case of the historical gold standard, light transmission aggregometry (LTA), unstandardized methodical issues including centrifuging time and speed, agonist concentration, and the preferred estimate to express results (maximal or final aggregation) were major drawbacks. 17 This may have been an important reason why the assessed platelet function phenotype was less reproducible with LTA. 11 In this regard, the Multiplate analyzer is a more standardized assay working from diluted whole blood with a fixed dose of ADP and using a uniform estimate to express results. However, there are still some aspects of the testing process that are not well defined and regulated including time from blood collection until testing, optimal reagent handling, and possible interference of antiplatelet and anticoagulant agents. As most of the technical problems might lead to false low levels of platelet reactivity during testing, they might result in misclassifications of patients according to platelet reactivity. Also, unstandardized methodical issues might have had an impact on previous clinical trial results and may be—in part—the reason behind the lack of general recommendation of PFT by clinical practice guidelines. 8,10
Due to the above-mentioned knowledge gaps, we aimed to investigate the impact of 3 important methodical aspects of PFT. Our main findings can be summarized as follows:
First, time from blood collection until assessment is a profound determinant of platelet reactivity both on and off clopidogrel. Thirty minutes after blood collection, platelet reactivity decreases dramatically leading to a large decrease in the proportion of patients with HPR. As a result, compared to the 70% HPR rate at 30 minutes, only 20% of patients were classified as HPR at 4 hours. This means that 71% of the HPR patients would be misclassified from the same blood samples, if processed 210 minutes later. These changes are surprisingly large as the manufacturer recommends that samples may be processed until 3 hours of collection with the Multiplate assay. 12,13 Similarly, laboratory guidelines such as the guideline by the British Committee for Standards in Haematology also recons that samples should be tested between 30 minutes and no more than 4 hours from blood collection. 18
In contrast to these recommendations, our results suggest that all samples need to be processed within an hour and preferably within 30 minutes of blood collection. Some manufacturers as well as the above-cited committee recommend an initial waiting period of 20 to 30 minutes after sample collection in patients taking P2Y12 inhibitors. In the subgroup of clopidogrel-treated patients, we also detected a small increase in platelet reactivity 30 minutes after collection (Figure 3). Therefore, it may be appropriate to follow these recommendations and prevent immediate testing. However, it should be highlighted that the magnitude of increase in case of immediate testing was far smaller than the decrease after delayed testing, performed more than 1 hour after collection. Therefore, our results strongly emphasize that most of the attention should be focused to prevent late testing, that is, more than 1 hour after blood collection. Our results are similar to a recent study by Stohlawetz et al. Although the authors used only 2 time points for analysis (1 and 3 hours after blood collection), they also demonstrated a decline in platelet function 3 hours after blood collection. 19
Second, we aimed to investigate the impact of reagent storage on platelet reactivity values. As a result, we observed a small, yet significant decrease in platelet reactivity when the thawed ADP was stored for more than a week at 2°C to 8°C. Our results are consistent with the recommendations of the manufacturer, allowing the storage of thawed ADP at 2°C to 8°C for a week. 12,14
Third, we analyzed the effect of enoxaparin on platelet function results. The impact of unfractionated heparin (UFH) on platelet function was earlier demonstrated: UFH enhances platelet activation and aggregation in vivo. 20 -22 The role of enoxaparin is much more controversial in the literature: it was described to have only minor pro-activation properties or no effect on platelets 21,22 or to decrease platelet reactivity compared to UFH. 23,24 Enoxaparin is widely used in patients with ACS for parenteral anticoagulation before and during PCI, as well as after PCI in patients requiring chronic oral anticoagulation before the therapeutic INR level is reached. 1,25 In the setting of long-term bed rest after ACS, a prophylactic dose of enoxaparin may be also used to prevent venous thromboembolism. Therefore, our aim was to investigate prophylactic and therapeutic concentrations of enoxaparin on results of PFT. Our outputs showed that similarly to UFH, enoxaparin enhances platelet aggregation in a slight, dose-dependent manner (Figure 6A). Despite statistical significance, the clinical relevance of this phenomenon may be questionable due to small absolute differences and lack of significant increase in the rate of HPR. Compared to enoxaparin-free samples, prophylactic and therapeutic concentrations did not modify the rates of HPR (Figure 6B). However, our results show that due to their impact on the test results, enoxaparin usage should also be standardized together with other test conditions.
Limitations
Our study has some obvious limitations including the relatively low sample size, lack of Prasugrel- or ticagrelor-treated patients, and no other platelet function devices. However, due to the several time points and intrasubject comparisons, the obtained P values strongly reject chanceful findings. Although ticagrelor and Prasugrel were not tested in this experiment, the less potent P2Y12 inhibitor, clopidogrel, may be more subject to a false-negative result for HPR, given the rate of HPR is very low with Prasugrel and ticagrelor but may be as high as 30% to 40% with clopidogrel.
Conclusion
Even in a relatively well-standardized platelet function assay, test conditions may have a large impact on the obtained results. Time delay from blood collection seems to have a dramatic influence on platelet reactivity values, suggesting that platelet function measurements should be performed preferably within an hour of blood collection with the Multiplate assay. Further clinical studies with platelet function assays should include these strict recommendations to prevent misclassification of patients based on PFT.
Footnotes
Authors’ contribution
Döme A Dézsi contributed to conception and design, acquisition, analysis and interpretation, drafted the manuscript, and gave final approval. Béla Mekely critically revised the manuscript and gave final approval. Judit Skopál contributed to analysis and interpretation. Eszter Barabás and Katalin Várnai contributed to acquisition and analysis. József Faluközy critically revised the manuscript. Gábor Veress critically revised the manuscript and gave final approval. Nasri Alotti gave final approval. Dániel Aradi contributed to conception and design, acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; and gave final approval. Döme A. Dézsi and Dániel Aradi agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Dániel Aradi: Consulting for Verum Diagnostica GmbH, Lecture fees from AstraZeneca, DSI/Lilly, Verum Diagnostica, Roche Diagnostics.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
