Abstract
Extracorporeal cardiac shock wave therapy (CSWT) has been used to treat patients with severe coronary heart disease and cardiac failure with good results; however, the safety of this treatment is still controversial. Its safety in clinical setting and on microstructures has been confirmed, but the influence of shock wave on the ultrastructures of myocardial cells is not clear. In this study, 12 Sprague-Dawley rats were randomly divided into control (NC) and CSWT therapy (NC+SW) groups. The heart rate, blood pressure, serum troponin I (TNI), and cardiac ultrasound were evaluated, and the myocardial inflammatory responses and fibrosis changes were compared. The samples were observed by transmission electron microscopy to evaluate the changes in myocardial tissue ultrastructure. The CSWT had no significant influence on rat hemodynamics indices and serum TNI, did not affect left ventricular function, and did not cause myocardial inflammatory response and fibrosis changes. The scores of myocardial ultrastructure damage in the NC and NC+SW groups were 1.39 ± 0.982 and 2.42 ± 1.009, respectively (P = .103). The CSWT did not cause significant additional damage to myocardial ultrastructures. The safety of CWST has been preliminarily proved at the clinical, microstructure, and ultrastructure levels, but its long-term safety needs further exploration.
Keywords
Introduction
Cardiac shock wave therapy (CSWT) is a new type of treatment that came to the forefront ∼30 years ago in clinics and that has been used in patients with severe coronary heart disease and cardiac failure with positive results. The important mechanics of the treatment is that CSWT increases local blood flow in myocardial tissue and promotes angiogenesis. First, extracorporeal shock waves promote the release of nitric oxide and increase blood flow, which is the short-term effect. It can also upregulate the expression of vascular endothelial growth factor (VEGF) and promote angiogenesis, which are long-term effects; however, shock waves are nonselective for tissues and cells. They can directly destroy the cytomembranes, cytoskeletons, and microvessels in tissues and cells. It is not known whether shock waves have a negative effect on myocardial cells during ischemia or whether they can irreversibly damage myocardial cells and further influence cardiac malfunction. Although therapeutic, it is unclear whether the mechanical shear force and void effect of the shock waves can lead to additional tissue injury. These issues are cause for concern in clinical treatments.
In 2010, Jargin 1 expressed doubts about the safety and effectiveness of CSWT from a pathological aspect. He believed that extracorporeal shock waves were nonselective and that when the strength of the shock wave was appropriate, it could be therapeutic, but when it was not, it could damage the microstructures of tissues, such as the cytomembranes, cytoskeletons, capillaries, and myocardial cells, to increase injury to ischemic myocardial cells and subsequent apoptosis and necrosis. In addition, whether the void effect and mechanical shear force caused by the shock waves could lead to change on the hemodynamics, damage vascular endothelium, and aggravate vasculopathy were unclear. The increase in neovascularization and upregulation of VEGF expression observed during this study might be the only nonspecific damage–repair process resulting from CSWT. As for the nonrenewable myocardial tissues, CSWT could aggravate fibrosis, increase scar repair, and accelerate myocardial reconstitution. In 2012, Meglio et al 2 used Fischer 344 rats to evaluate CSWT safety. The results showed no increasing tendency in myocardial fibrosis and oxidation which preliminarily proved the safety of shock waves. In 2016, He et al 3 proved that CSWT did not increase apoptosis in rat myocardial tissue. Other clinical trials on CSWT and animal studies reported that shock waves did not cause arrhythmia and that there were no significant changes in serum troponin I (TNI) and brain natriuretic peptide before and after therapy. 4 –8
The safety of CSWT has been confirmed in the research; however, the effects from its nonselectiveness of tissues and cells continue to create concern. Although the results of the studies on protein, RNA, and DNA are positive, 4 the assessment of tissue ultrastructure morphology after CSWT therapy is limited. We used transmission electron microscopy (TEM) to evaluate the possible damage to myocardium ultrastructures from CSWT treatment, specifically the influence on important organelles, such as the mitochondrion, sarcoplasmic reticulum, and cell nucleus. 9
Materials and Methods
Animals
All animal experiments conformed to the protocols approved by Animal Use and Care Committee of Beijing Hospital and the Guide for Care and Use of Laboratory Animals (NIH publication #85-23, revised 1996). During the study, the treatment of the animals was in keeping with the relevant regulations for laboratory animal management. Twelve 2-month-old male Sprague-Dawley (SD) rats (Specific Pathogen Free [SPF] grade) weighing 220∼240 g each were provided by Vital River Laboratories (Beijing, China; license number: SCXK [Jing] 2012-0001). The SD rats were randomly divided into 2 groups as follows: normal control group (NC group, N = 6) and normal control with shock wave group (NC+SW group, N = 6).
The Masson Trichrome Coloration Kit and Sirius Red Staining Kit were purchased from Solarbio Science & Technology Co, Ltd. (Beijing, China). Phenobarbital was purchased from Sigma Aldrich (St. Louis, Missouri). Anhydrous magnesium sulfate, absolute ethyl alcohol, xylene, citric acid, trisodium citrate, sodium chloride, potassium chloride, disodium hydrogen phosphate, and monopotassium phosphate were purchased from Beijing Chemical Reagent Company (Beijing, China).
The equipment used were the CH-8274 animal extracorporeal CSWT apparatus (Storz Medical, Tägerwilen, Switzerland); SC2000 Ultrasound System (Siemens, München, Germany) with probe (10V4, 10 Hz); an FX-2111 single-channel electrocardiograph (Fukuda, Beijing); the BP-2010 caudal artery blood pressure analysis system (Softron, Beijing, China); the RM2236 paraffin-slicing machine (Leica Biosystems, Vista, California); slide warmers (Tianli, Tianjin, China); the EM UC 7 ultramicrotome (Leica Biosystems); the JEOL TEM-1010 transmission electron microscope (Tokyo, Japan); and the Quemesa digital imaging system (EMSIS ASIA, Singapore).
Preparation of Compound Anesthetic
To prepare the anesthetic, 1.772 g pentobarbital sodium, 4.24 g magnesium sulfate, and 8.5 g chloral hydrate were dissolved in 28.5 mL anhydrous alcohol and 50 mL 1,2-propylene glycol and diluted with water to 200 mL. The solution was then filtered using a 0.45-μm membrane and stored in 4°C refrigerator.
Extracorporeal CSWT
At days 1, 3, and 5 after random grouping, the rats were intraperitoneally injected with the anesthetic at 0.1 mL/30 g body weight and fixed on the surgery table in the supine position. The target area for CSWT was the inclined left ventricle in the middle and lower sternum with significant cardiac impulse (left ventricular anterior wall).
The extracorporeal shock wave probe was placed in the precordium in close contact with the skin and ready to initiate the shock wave. The local shock wave energy range of the probe was 5.0 mm × 5.0 mm × 30 mm. The rats in the NC+SW group (N = 6) received CSWT (energy: 0.1 mJ/mm2; frequency: 4.0 Hz; shock wave times: 200). The rats in NC group (N = 6) received the same procedure but without shock waves and energy (Figure 1). During the shock wave or sham-CSWT, an electrocardiogram was used to monitor the heart rate and rhythm.

Schematic diagram of rat shock wave therapy. The rats under intraperitoneal anesthesia were fixed on the surgery table, and the probe from the extracorporeal shock wave instrument was placed in the precordium, in close contact with the skin and ready to initiate the shock wave. The local shock wave energy range of the probe was 5.0 mm × 5.0 mm × 30 mm. The electrocardiogram for monitoring is shown to the left of the rat.
Transthoracic Echocardiography
All 12 rats received a complete examination. On day 1, after intraperitoneal injection with compound anesthetic, the rats were fixed in the supine position on the table. A transthoracic echocardiography was performed before either the NC+SW or the NC group received the CSWT treatment. On day 7, after intraperitoneal injection with the anesthetic, the rats were again fixed in a supine position on the table. Transthoracic echocardiography was performed after the NC+SW group rats received CSWT and the NC group received sham-CSWT.
The left ventricular long axis was used as the observation surface for the transthoracic echocardiography, and M-mode ultrasonication (Figure 2) was used to measure the end-diastolic and end-systolic diameters on the papillary muscle level of left ventricular. The built-in ultrasonications of ware were used to measure the left ventricular ejection fraction (LVEF). The end-diastolic diameter, end-systolic diameter, and LVEF in 5 different cardiac cycles were measured, and the values were averaged for statistical analysis.

Rat transthoracic echocardiography showing transthoracic echocardiography M-mode measuring left ventricular ejection fraction and fractional shortening.
Venous Blood Collection and Serum TNI Levels
The blood (0.5 mL) from the caudal vein was drawn before and after the third shock wave on the NC+SW group. The serum TNI levels were assessed in the immunology lab at Beijing Hospital.
Heart Samples
Heart samples were taken from all the rats 8 days after random grouping. The rats were intraperitoneally injected with the anesthetic and, when the reflex response disappeared, were placed on the low-temperature surgery table and intraperitoneally injected with saline containing 15% KCl to stop the heart at the end-diastolic stage. The heart was removed by opening the thoracic cavity through the sternum and clipping and dividing the large cardiac vessels. The chambers of the heart were then washed with saline. The left ventricular was divided into 4 segments along the long axis; a cross-section of the heart was also cut. The cardiac apex segment was discarded, and the second segment was placed and fixed in 10% neutral for malin. After embedding in paraffin and processing, the samples were ready for histopathological observation. The third-segment samples (papillary muscle level) were prepared according to the following TEM steps.
Sample Preparation for TEM
A 2.5% glutaraldehyde solution fixative was prepared, stored, and precooled at 4.0°C. The fixative was dropped on a wax plate, and the third segment of myocardium (papillary muscle level) was cut into blocks with a scalpel (2 antetheca, 1 interventricular septum) and placed in the fixative. Each tissue was then cut to 2.0 mm × 2.0 mm × 1.0 mm in the fixative using the double see-saw method, moved with a tweezer without damaging the samples into vials containing 2.5% glutaraldehyde, shacked thoroughly until they sank to the bottom, labeled, and stored at 4.0°C for >2.0 hours. The samples were prepared in the TEM lab at Peking Union Medical College Hospital, China. There were 18 myocardial tissue samples from each group of rats.
Tissue Pathological Morphology
Myocardial tissue paraffin slices (∼5.0-μm thick) were prepared and stained with hematoxylin and eosin. The slices were dewaxed with xylene, hydrated with gradient ethanol (100%, 95%, 85%, and 75%; and clean water), stained with hematoxylin liquid for 2.0 minutes, differentiated with 1.0% hydrochloric acid alcohol for 3.0 to 5.0 seconds, and stained with eosin liquid for 1.0 minutes. The slices were then dehydrated with gradient ethanol (75%, 85%, 95%, and 100%), cleared with xylene, sealed with neutral gum, and observed under a microscope for histopathological study. The degree of inflammation measured by the distributions of neutrophil granulocytes and lymphocytes in the myocardial tissue was observed.
The myocardial tissue paraffin slices were prepared (∼5.0 μm thick) for Masson staining. The slices were dewaxed with xylene, stained with Weigert hematoxylin iron (hematoxylin iron A and B were mixed 50/50) for 5.0 to 10 minutes, and washed with water. The slices were differentiated with 1.0% hydrochloric acid alcohol and washed with water for a few minutes. The slices were then stained with ponceau acid fuchsin for 5.0 to 10 minutes, washed with water, processed with phosphomolybdic acid solution for ∼5.0 minutes, restained directly with aniline blue for 5.0 minutes, processed with 1.0% glacial acetic acid for 1.0 minutes, dehydrated several times with 95% alcohol, dehydrated with 100% alcohol, cleared with xylene, sealed with neutral gum, and observed under a microscope for any histopathological changes. Myocardial fibrosis was evaluated.
Myocardial Tissue Ultrastructure Analysis
To prepare for TEM, the rat myocardial tissues were cut into blocks of 2.0 mm × 2.0 mm × 1.0 mm, immersed in 2.5% glutaraldehyde (0.1 mol/L phosphate buffer, pH 7.4) at 4.0°C for >2.0 hours, and prefixed. The tissues were then washed with 0.1 mol/L phosphate buffer (pH 7.4) for 3.0 hours, immersed into 1.0% osmic acid for 1.0 to 1.5 hours, and postfixed. The tissues were washed again with 0.1 mol/L phosphate buffer (pH 7.5) for 15 minutes, immersed for 15 minutes each in 50%, 70%, 80%, and 95% alcohol and finally immersed twice in 100% alcohol for 20 minutes to dehydrate. The tissues were then immersed twice in acetone for 20 minutes and in EPON 812 (acetone:resin = 1:2) overnight.
Resin was injected into the embedding plate, into which the processed tissues were placed. The plate was put into an oven under controlled temperatures of 37°C for 12 hours, 45°C for 12 hours, and 60°C for 48 hours. The embedded tissues were then cut into slices 500-µm thick, stained with toluidine blue, and localized under an optical microscope. The excessive resin was cut, and the tissues were clipped into trapezoids for sectioning. Using the ultramicrotome, the localized tissue blocks were sliced into ultrathin sections at a thickness 60 to 70 nm and adhered to a 200-meshgrid. Finally, uranyl acetate–lead citrate was used to perform an electron stain. All samples were observed by TEM and photographed using a digital imaging system.
There were 36 TEM samples from the NC and NC+SW groups, and 10 views (10 000×) were randomly selected from each sample. The myocardial microstructure damage was evaluated by the researcher and an experienced physician from the pathology department, Peking Union Medical College Hospital, China. The myocardial ultrastructure damage was compared between the 2 groups.
Data Analysis
Measurement data were expressed as the mean (SD), and enumeration data were expressed as percentage. SPSS 11.0 (SPSS Inc, Chicago, Illinois) was used to analyze the data. The comparison between the groups and measurement indices were analyzed using Student t test. P < .05 was considered statistically significant.
Results
The NC group received 3 intraperitoneal injections, 3 sham-CSWTs, and 2 transthoracic echocardiography examinations. The NC+SW group received 3 intraperitoneal injections, 3 CSWTs, and 2 transthoracic echocardiography examinations.
Influence of CSWT on Rat Hemodynamics
The electrocardiogram on each rat in the NC+SW group was recorded during CSWT (standard II lead), and no severe arrhythmia was observed. There was no significant change in the electrocardiogram ST–T wave after CSWT (Figure 3).

Electrocardiogram monitoring during shock wave therapy in the NC+SW group. NC+SW indicates normal control with shock wave.
Influence of CSWT on Rat Heart rate and Blood Pressure
After 3 CSWTs, blood pressure and heart rate were measured. The average systolic blood pressures in the NC and NC+SW groups were 129.1 ± 3.14 and 126.3 ± 2.02 (P = .41), respectively. The average diastolic blood pressures were 89.4 ± 1.17 and 88.7 ± 2.01 (P = .35), respectively. The average heart rates were 373.1 ± 6.29 and 380.2 ± 4.27 (P = .53), respectively. The results suggest that CSWT had no significant effect on blood pressure or heart rate; therefore, CSWT had no significant negative effect on the rats’ basic hemodynamic indices.
Influence of CSWT on Rat Serum TNI
The average serum TNI before CSWT was 0.155 ± 0.004 μg/L in the NC+SW group and 0.156 ± 0.0008 μg/L (N = 6, P = .08) after CSWT. This suggests that CSWT did not cause an increase in serum TNI levels.
Influence of CSWT on Rat Left Ventricular Morphology and Function
The ejection fraction (EF) and fractional shortening (FS) in the left ventricle of the rats from the 2 groups are shown in Table 1. There was no significant difference in the front and back left ventricular EF and FS between the 2 groups and before or after CSWT, indicating that the therapy did not influence the rat left ventricular function.
Rat Left Ventricle Function Before and After Shock Wave Therapy.

Abbreviations: NC, normal control; NC+SW, normal control with shock wave group; EF, ejection fraction; FS, fractional shortening.
Myocardial Histopathological Changes After CSWT
The myocardial fiber in the NC+SW group was plump with normal arrangement. The cytoplasm in the myocardial cells and the density of the myocardial cell nucleus were homogeneous. Degeneration and necrosis in the myocardial cells were not observed, and inflammatory cell infiltration was not observed in the myocardial matrix, which was similar to that in the NC group (Figure 4). These results indicate that the morphological characteristics of myocardial tissues did not significantly change after CSWT.

Myocardial hematoxylin and eosin (HE) and Masson staining. The safety of cardiac shock wave therapy (CSWT) has been proved by histopathology, and inflammatory response in the NC and NC+SW groups was not observed by HE staining; Masson staining did not reveal any fibrosis in either the NC or NC+SW group. NC indicates normal control; NC+SW, normal control with shock wave group.
No collagen fibrosis staining was observed in the NC or NC+SW group; therefore, CSWT did not cause an inflammatory response and fibrosis in the myocardial tissue (Figure 4).
More abnormal ultrastructure expressions were observed in the NC+SW group than in the NC group (Figure 5). Abnormal phenomenon, such as many swollen mitochondria and the disappearance of ridge structures, were observed in the NC+SW group. In the same view, the coexistence of degenerated and new mitochondria was commonly observed (Figure 5A and B). Vacuoles were occasionally observed in the intercalated disc structures in the NC+SW group; however, there were still some similarities among 2 groups (Figure 5C). For example, the morphology of most of the mitochondria was normal, although swollen mitochondria were occasionally observed. The myofibril structures in the 2 groups were normal as was the morphological structure of the myocardial cell nucleus. The morphology of the intercalated discs among myocardial cells was normal.

Ultrastructure changes in the NC+SW group. The coexistence of degenerated and new mitochondria was commonly observed (A, B). Vacuoles were occasionally observed in the intercalated disc structures in the NC+SW group (C). NC+SW indicates normal control with shock wave group.
Similar ultrastructure changes were observed in NC and NC+SW groups (Figure 6). Rat myocardial ultrastructures were observed by TEM. Normal morphology of the mitochondria in the NC group (Figure 6 2); occasional swelling (Figure 6 1 and 3); abnormal phenomenon, such as swollen mitochondria and disappearance of ridge structures, in the NC+SW group (Figure 6 4); normal myofibril structure of the 2 groups (Figure 6 1, 2, 4, and5); normal morphological structure of myocardial cell nucleus (Figure 6 2 and 5). The morphologies in most of the NC+SW group and the intercalated discs between myocardial cells in the NC group were normal (Figure 6, arrow).

Ultrastructure changes in NC and NC+SW groups. Rat myocardial ultrastructures were observed by transmission electron microscopy (TEM). (2) Normal morphology of the mitochondria in the NC group;(1, 3) occasional swelling; (4)abnormal phenomenon, such as swollen mitochondria and disappearance of ridge structures, in the NC+SW group; (1, 2, 4, 5) normal myofibril structure of the 2 groups; (2, 5) normal morphological structure of myocardial cell nucleus. The morphologies in most of the NC+SW group and the intercalated discs between myocardial cells in the NC group were normal (arrow). NC indicates normal control; NC+SW, normal control with shock wave group.
Table 2 provides the myocardial ultrastructure damage scoring system proposed by Yildirim 10 for assessing the ultrastructures after CSWT. The average scores of myocardial ultrastructure damage in the NC and NC+SW groups were 1.39 ± 0.982 and 2.42 ± 1.009, respectively (P = .103; Figure 7).
Myocardial Ultrastructure Damage Scoring System.

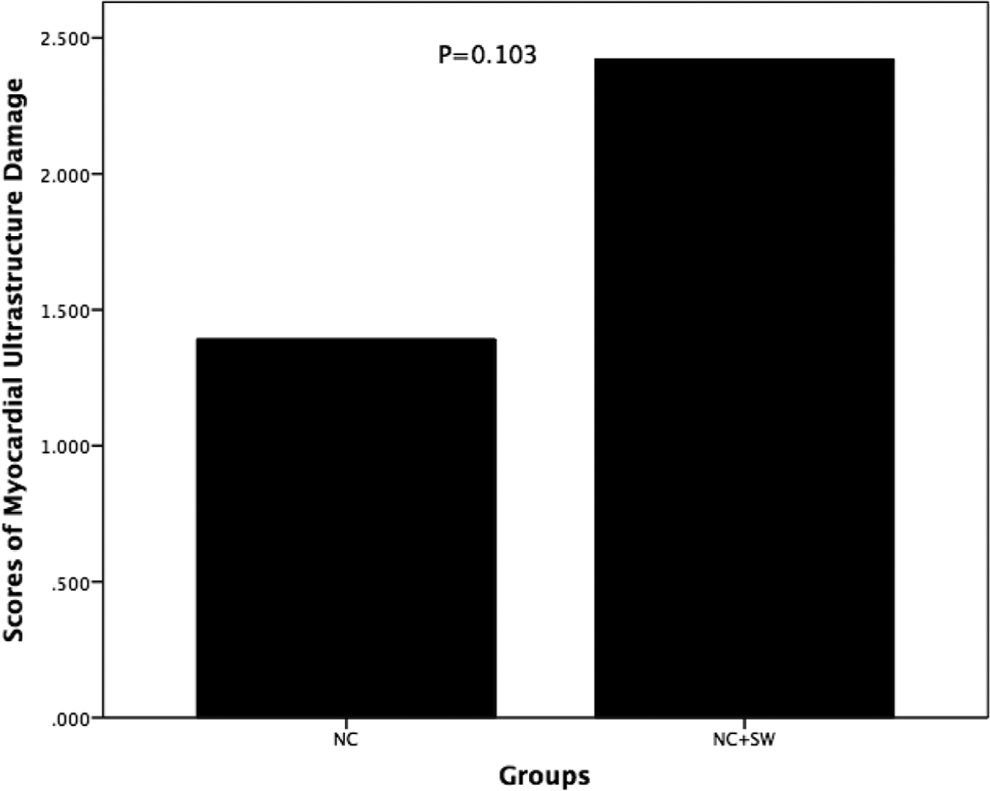
Results of myocardial ultrastructure damage in the NC and NC+SW groups. The average scores of myocardial ultrastructure damage in the NC and NC+SW groups were 1.39 ± 0.982 and 2.42 ± 1.009, respectively. CSWT did not cause significant additional damage to myocardial ultrastructures. NC indicates normal control; NC+SW, normal control with shock wave group; CSWT, cardiac shock wave therapy.
Discussion
Thirty years ago, shock wave was a new therapy to use in clinics, mainly in lithotripsy for kidney stones. Some doctors found that the patients receiving shock wave lithotripsy showed thickening ilium. 11 This stimulated the using the therapy to promote the healing of fractures and soft tissue damage. 12,13 In the 1990s, this technique was widely applied for soft tissue dysfunction, such as rotator cuff calcification fasciitis and external humeral epicondylitis, and extracorporeal shock wave therapy has been used worldwide to treat bone and muscle diseases. In 2000, it was approved by the US Federal Drug Administration to treat chronic plantar fasciitis. 14,15
Shock wave is a monofocal and individual pressure 3-D impulse wave. The period is within milliseconds, and the pressure peak is 35 to 120 MJ. 16 Because shock wave has a nonselectiveness characteristic, while also being therapeutic, the risk of adverse reactions also exists. 16,17
In 2000, Gutersohn et al 18 from the University of Essen found that the shock wave with low energy could upregulate the VEGF expression in epithelial cells for the first time, after which the therapeutic effect of extracorporeal shock wave on promoting angiogenesis and tissue ischemia in the clinic began to attract attention. In 2003, STORZ MEDICAL (Tägerwilen, Switzerland) developed the extracorporeal CSWT system based on extracorporeal shock-wave lithotripsy. It has been reported that the extracorporeally induced shock wave with low energy can make microvesicles vibration the vessel lumen or tissue and form microflow. The asymmetric burst of microvesicles generated jetting action, which generated mechanical shear force and the void effect inside tissues and cells. This further caused a series of biochemical reactions at the subcellular level. 19,20
Shock wave was developed from therapy on damaged tissues (extracorporeal lithotripsy) with high energy sent into the tissue to promote proliferation and low energy to promote repair, and it has been used on patients with refractory coronary heart disease with positive results. In 2010, Jargin 1 expressed doubts on the safety and effectiveness of CSWT from a pathological aspect, and although the support of basic research is limited, the concerns about the possible induction of the injury–repair process in myocardial tissue by CSWT always exist. The outcome of this process is myocardial fibrosis, which further aggravates ventricular remodeling and leads to myocardial function damage; therefore, deeper and more comprehensive exploration on the mechanisms of CSWT is necessary, as well as an evaluation of its safety in clinical settings.
The safety evaluation on rats receiving CSWT indicated that during therapy, arrhythmia did not occur, TNI was not influenced, and the cardiac left ventricular systolic function and structure were not significantly changed, which was in agreement with results from Meglio et al. 2 In other studies on CSWT’s effectiveness, none of the above adverse reactions were reported. 21 –23
Transmission electron microscopy is a mature technique used to evaluate the ultrastructure of various materials. In the middle of last century, TEM was widely used in clinical studies, including on myocardial tissue ultrastructures. 7 The TEM possesses 10 000 times the amplification and nanosized resolution, giving it a significant advantage for evaluating morphology at the cellular level. In the 1950s, TEM was used to evaluate the ultrastructure of normal myocardial tissue in mammals in medical research. 24 –31 Many researchers investigated the ultrastructure of normal rat myocardial tissue; 26,29 –31 however, in these studies, it was difficult to determine which ultrastructure change was caused by anesthesia and which was caused by surgery. Thus, the so-called normal myocardial tissues were taken from the rats that did not receive surgery and anesthesia in the NC group.
In normal rat myocardial ultrastructures, the band phenomenon is very common and believed to be caused by the myocardial fiber at different systolic stages. 32,33 In our study, such phenomenon was commonly observed. The myofiber tunica vaginalis formed by a double membrane structure 26 showed a fan-shaped fold, and regular depressions formed by the Z band crossing the myocardial fiber; the end of it was observed on the surface. 34 –36 In myocardial muscle fiber, there are many mitochondria arranged among the myofibrils. Ridges occupy these mitochondria, and only matrix-like and arranged granular space can be observed. 37 –39 In our study, such expression was very common in the NC group; however, the mitochondria also showed swelling, and the ridge structures had disappeared in some (Figure 6). In the NC+SW group, abnormal phenomena, such as swollen mitochondria and disappearance of ridge structures, were also observed. In the same view, the coexistence of degenerated and new mitochondria was also commonly observed (Figure 5). Sarcoplasmic reticulum was widely distributed in the myofibroblasts, 40 –42 the shape of which was tubular, allowing vacuoles to be observed under TEM. In our study, at high magnification, the swollen sarcoplasmic reticulum was also occasionally observed (Figure 5). The intercalated disk is an end-to-end structure along the myofibroblasts and is longitudinally arranged. It is vertically arranged with along myofiber axis. In the NC group, the normal structure of the intercalated disc was observed often (Figure 6), but vacuoles in the disc were occasionally observed in the NC+SW group (Figure 5).
The TEM is widely used in studies on myocardial cell ultrastructure changes caused by infection, cold stress, toxicant, noise, and pollution 43 –48 ; however, the application for the effect of shock waves on tissue ultrastructures, including on myocardial tissue ultrastructures, is limited. Current studies on evaluating rat myocardial ultrastructure damage focus mainly on the important organelles, such as mitochondria, 9 or on whether the arrangement of the myocardial myofilament is regular. In our study, in the NC and NC+SW groups, abnormal changes, such as sarcomere structure and Z bands, were not observed. Under high magnification (10 000×), observation of mitochondria degeneration in the NC group was limited, and outspread sarcoplasmic reticulum was only occasionally observed. No structural damages in blood capillaries and the myocardial cell nucleus were observed; however, in the NC+SW group, many degenerated mitochondria were seen. In the same view, the coexistence of degenerated and new mitochondria was commonly observed, as well as many swollen sarcoplasmic reticulum. Similarly, no structural damages to blood capillaries and the myocardial cell nucleus were observed. Nevertheless, vacuole-like changes could be seen in the intercalated disc structures (Figure 5); however, the nonquantitative observation could not assess the extra damage by CSWT on the ultrastructure in the myocardial cells because the damage was also observed in the NC group, and damage caused by sampling or stress could not be excluded. 47,48 Thus, a nonquantitative scoring system was needed to further evaluate the differences between the groups.
The TEM was developed and has been used for ∼50 years in medical research. In 1980, Braunwald et al 49 proposed the myocardial cell and microvessel injury classification in a study using a dog myocardial infarction model. The nonquantitative evaluation system was cited by a research on myocardial ultrastructures using a rat septicemia model. 46 In 2004, Yildirim et al 10,50 proposed a scoring system for myocardial ultrastructure damage in a series of studies, which included 3 main organelles, the cell nucleus, the mitochondria, and the sarcoplasmic reticulum. These were scored from 0 to 3 according to the morphological changes observed. Using this system, we scored the ultrastructural changes in the NC and NC+SW groups. The average score for the NC group was 1.39 ± 0.982, and that for the NC+SW group was 2.42 ± 1.009 with no significant differences (P = .103) between them (Figure 7), but the degree of damage in the NS+SW group tended to be aggravated.
Although our study was beneficial to better understand the effects of CSWT, there were some limitations. First, it was an observation of only the morphology of the ultrastructures; therefore, whether the seemingly normal structures perform normal functions is not known and the degree of damage to the ultrastructures could have been under estimated and the assessment of the influence of CSWT could have been misleading. Second, the myocardium was taken on day 8 after randomly dividing the rats into groups, and the observed CSWT was changed compared with the ultrastructures in the acute stage; however, in a clinical setting, the mid- and long-term effects are often observed. Thus, in subsequent studies, TEM is used to observe and evaluate the mid- and long-term ultrastructure.
In our study, we found that CSWT did not have a significant influence on the ultrastructures when observed under a microscope; however, the degree of damage to the myocardial cell ultrastructures based on TEM appeared to increase in the NS+SW group. According to the current semi-quantitative assessment system of the degree of ultrastructure damage, 10 the increase was not remarkably significance. In many current studies, CSWT has not shown negative results. The phenomenon found in our study suggested that CWST could slightly injure myocardial cells, stimulate self-repair, stimulate an increase in new myocardial vessels, 4,51 and alleviate the inflammatory response. This is similar to how physical exercise can slightly injure muscles, but results in an improvement in fitness after the muscles self-repair and increase.
Based on our limited knowledge, TEM was the first time applied to evaluating the ultrastructure of myocardial tissue after CSWT to normal adult rat. We did not succeed in finding reference data about microstructure changing on the animal. In human researches, there was a few of published article concerned with the safety of the CSWT, some of adverse events were observed. 6 –8 The safety of CSWT in healthy rats had been proved in some studies, 2,3 but very few of data was based on ultrastructure or microstructure. Our studies present the safety data by means of the assessment of tissue ultrastructure morphology after CSWT therapy via TEM on rats. Those were puzzles that tissue changes caused by ischemia might be induced by CSWT at same time. Based on our primary date, there are some minor ultrastructure injury observed on myocardium, which may be related to CSWT. Whatever, in normal rat, there is a certain ability to repair itself, it is not clear whether originating from CSWT damage beyond the rat’s ability to repair itself especially in lesions of the myocardium. We will carry on future researches to study.
BL and QH originated the idea and performed preliminary experiments. BL, YHZ, NJ, ML, and LD continued to perform the experiments. DCZ coordinated the laboratory support. BL and NJ was responsible for writing the paper. BL and ML supported the editing of the manuscript and added important comments to the paper. All authors read and approved the final manuscript.
Footnotes
Author Contribution
Bing, L: contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, critically revised manuscript, agrees to be accountable for all aspects of work ensuring integrity and accuracy; Yunhe, Z: contributed to acquisition, agrees to be accountable for all aspects of work ensuring integrity and accuracy; Na, J: contributed to analysis and interpretation, agrees to be accountable for all aspects of work ensuring integrity and accuracy; Ming, L: contributed to interpretation, agrees to be accountable for all aspects of work ensuring integrity and accuracy; Ling, D: contributed to interpretation, agrees to be accountable for all aspects of work ensuring integrity and accuracy; Dachun, Z: contributed to acquisition and analysis, agrees to be accountable for all aspects of work ensuring integrity and accuracy; Qing, H: contributed to conception and design, contributed to acquisition, drafted manuscript, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by grants from projects of the Capital Health Project (Z131100004013032), Beijing Hospital Clinical Research 121 Project (121-2016004), and the National Natural Science Foundation of China (81470427).
