Abstract
Background:
Phospholamban (PLN) inhibition enhances calcium cycling and is a potential novel therapy for heart failure (HF). Antisense oligonucleotides (ASOs) are a promising tool for unmet medical needs. Nonviral vector use of locked nucleic acid (LNA)-modified ASOs (LNA-ASOs), which shows strong binding to target RNAs and is resistant to nuclease, is considered to have a potential for use in novel therapeutics in the next decades. Thus, the efficacy of a single-dose injection of LNA-ASO for cardiac disease needs to be elucidated. We assessed the therapeutic efficacy of a single-dose LNA-ASO injection targeting PLN in pressure overload-induced cardiac dysfunction.
Methods and Results:
Mice intravenously injected with Cy3-labeled LNA-ASO displayed Cy3 fluorescence in the liver and heart 24 hours after injection. Subsequently, male C57BL/6 mice were subjected to sham or transverse aortic constriction surgery; after 3 weeks, these were treated with PLN-targeting LNA-ASO (0.3 mg/kg) or scrambled LNA-ASO. Cardiac function was measured by echocardiography before and 1 week after injection. Phospholamban-targeting LNA-ASO treatment significantly improved fractional shortening (FS) by 6.5%, whereas administration of the scrambled LNA-ASO decreased FS by 4.0%.
Conclusion:
Our study revealed that a single-dose injection of PLN-targeting LNA-ASO improved contractility in pressure overload-induced cardiac dysfunction, suggesting that LNA-ASO is a promising tool for hypertensive HF treatment.
Introduction
Heart failure (HF) represents the advanced state of almost all cardiovascular diseases and is the major cause of death worldwide. 1 The etiologies include myocardial infarction, hypertensive and valvular heart diseases, cardiomyopathies, and congenital heart diseases. 2,3 The hallmarks of HF are impaired cardiac contraction and relaxation accompanied by abnormalities in calcium handling and β-adrenergic signaling. 4,5
In cardiomyocytes, cytosolic calcium regulates cardiac contraction and relaxation by an excitation–contraction coupling mechanism. 6,7 The Ca2+ influx via L-type calcium channels elicits Ca2+-induced Ca2+ release from sarco(endo)plasmic reticulum (SR) through ryanodine receptors, and increased cytosolic Ca2+ leads to cardiac contraction. The sequestration of Ca2+ from the cytosol into the SR, which determines active relaxation, is caused by a calcium pump on the SR called SR Ca2+ ATPase (SERCA2a), the activity of which is regulated by a small phosphoprotein, phospholamban (PLN). This Ca2+ cycling is impaired in failing cardiomyocytes and is considered to be an important therapeutic target. 8,9
In physiological conditions, β-adrenergic receptor (βAR) stimulation enhances myocyte contraction by activating cyclic adenosine monophosphate–dependent kinase (protein kinase A [PKA]), which phosphorylates multiple Ca2+ cycling proteins, including PLN. 10 Phospholamban inhibits SERCA2a activity through protein–protein interaction. Phosphorylation of PLN at serine 16 by PKA alters its interaction with SERCA2a to activate Ca2+ reuptake to the SR, resulting in enhanced SR Ca2+ loading and Ca2+ cycling. 11 In the failing myocyte, dysfunctional βAR signaling leads to less PKA activation and activation of alternate pathways, such as calcium/calmodulin-dependent kinase II signaling to cause pathological hypertrophy. 12 -16 Consequently, the usefulness of positive inotropic agents in HF is strongly limited, and direct activation of Ca2+ cycling, which can circumvent dysfunctional βAR activity, is required. Thus, inhibition of PLN is one of the most promising strategies in this context. In the past 2 decades, several reports have demonstrated that PLN inhibition alleviates cardiac failure in various animal models of cardiac pathologies, including myocardial infarction in rats, 17 genetic cardiomyopathy in hamsters, 18 and dilated cardiomyopathy in mice. 19 In addition, modulation of PLN improves contractility in human cardiomyocytes from patients with advanced HF, 20 suggesting that targeting PLN could be a bona fide therapy for failing hearts. However, the limited availability of safe and effective gene transfer tools to the heart has hindered the clinical development of PLN-targeting therapies. Adenoviral/adeno-associated viral (AAV) gene delivery into myocardium is one of the most prevalent tools for gene transfer; however, there are multiple obstacles including safety risks and strict legal regulation. Thus, a novel therapeutic tool for gene modification for the heart needs to be developed.
Recent advances in nucleic acid–based therapeutics elicited high expectations to develop novel treatments for various diseases, including cancer, 21 genetic disorders, 22 liver disease, 23 and HIV infection. 24 However, the clinical use of nucleic acids composed of naturally occurring DNA or RNA has been limited because of their poor binding affinity and specificity, instability caused by degradation by nuclease, and proinflammatory effect via activation of innate immunity. 25,26 Locked nucleic acids (LNAs) are modified RNA molecules in which the ribose sugar moiety is locked by an oxymethylene bridge connecting the C2′ and C4′ atoms. 27 -29 This modification confers high binding affinity to complementary DNA/RNA and reduced off-target effects. 29,30 To date, a plethora of reports showed a broad potential of LNA-modified oligonucleotides in antisense-mediated gene-silencing applications in vitro and in vivo for multiple pathologies including cardiac diseases. 31,32 Thus, this study examines the usefulness of LNA antisense oligonucleotides (LNA-ASOs) in vivo to establish a novel therapy to treat HF by targeting PLN.
Methods
Ethics Statement
The experiments using animals were approved by the Institutional Animal Care and Use Committee of the Graduate School of Pharmaceutical Sciences, Osaka University (approval number: DOUYAKU19-32-6). All animals were treated in compliance with the Osaka University Animal Care guidelines. All experimental procedures conformed to the Guide for the Care and Use of Laboratory Animals, Eighth Edition, updated by the US National Research Council Committee in 2011.
Antibodies and Reagents
Primary antibodies for Western blot included PLN (2D12; Thermo Fisher Scientific, Waltham, MA), SERCA2a (N-19; Santa Cruz Biotechnology, Dallas, TX), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (Millipore, Billerica, MA). TransIT-QR Hydrodynamic Delivery Solution designed specifically for the safe and efficient delivery of nucleic acids was obtained from Mirus Bio Corporation, Madison, WI.
All ASOs in this study were synthesized by the GeneDesign Corporation, Ibaraki, Japan. The sequence of Cy3-labeled LNA-ASO was 5′-Cy3^A(L)^T(L)^ 5mC(L)^a^a^g^a^t^g^a^g^G(L)^ 5mC(L)-3′, where (L) indicates LNA. All internucleotide linkages indicated by “^” are phosphorothioates. Lowercase letters represent natural DNAs. The oligonucleotide sequences of PLN-targeting LNA-ASO (PLN-AS) were as follows: AS-1, A(L)^T(L)^ 5mC(L)^a^a^g^a^t^g^a^g^G(L)^ 5mC(L); AS-2, T(L)^G(L)^ 5mC(L)^a^c^t^t^t^t^t^c^ 5mC(L)^A(L); AS-3, T(L)^G(L)^G(L)^a^g^g^c^t^c^t^c^ 5mC(L)^T(L); and AS-4, T(L)^T(L)^ 5mC(L)^t^g^g^a^g^a^t^t^ 5mC(L)^T(L). The sequence of the scrambled LNA-ASO (scr-LNA) was G(L)^A(L)^T(L)^a^g^c^a^g^a^t^g^A(L)^ 5mC(L).
Immunofluorescence Microscopy
Hearts were harvested, rinsed, and frozen in optimal cutting temperature (O.C.T.) compound (Sakura Corp, Tokyo, Japan). Frozen 5-μm-thick sections were prepared, fixed in 4% paraformaldehyde, and washed with phosphate-buffered saline (PBS) twice. Membrane staining with lectin from Triticum vulgaris conjugated with fluorescein isothiocyanate (FITC; Sigma, St. Louis, MO) was performed for 1 hour at room temperature. The prepared sections were examined using confocal microscopy (Leica, Wetzler, Germany, TCS SP5).
Screening of LNA-ASOs by Cotransfection in HEK293 Cells
HEK293 cells were seeded in 12-well plates and grown to 70% to 80% confluence. The PLN-AS and pcDNA3.1-PLN (mouse) were cotransfected using FuGENE6 (Promega, Madison, WI), according to the manufacturer’s protocols. Cells were incubated for 24 hours at 37°C and harvested to prepare protein samples for Western blotting analysis using anti-PLN antibodies.
Surgical Procedure and Echocardiographic Analysis
C57BL/6 male mice (8-10 weeks of age) were subjected to transverse aortic constriction (TAC), surgery as described previously. 33 Of 38 mice that underwent TAC surgery, 15 died during the procedure, yielding a perioperative survival rate of approximately 60%. In addition, another 8 mice died within 3 weeks after TAC. Two-dimensional and M-mode transthoracic echocardiography was performed for the remaining mice 3 weeks after TAC using an iE33 model equipped with a 15-MHz transducer (Philips Electronics, Andover, MA) under isoflurane anesthesia, as previously described. 34 The mice were allocated to the PLN-AS or the scr-LNA groups, and the corresponding LNA-ASOs (0.3 mg/kg) were administered 3 weeks after TAC using the TransIT-QR Hydrodynamic Delivery Solution (Mirus Bio Corporation), following the manufacturer’s protocol. The follow-up echocardiographic examination was performed 1 week after administration. The investigator who performed the echocardiographic analysis was blinded to the treatments of the mice.
Western Blotting Analysis
Western blotting analyses using protein samples or cardiac homogenates were performed, as previously described. 34 Briefly, cells were washed in cold PBS and scraped into RIPA buffer containing 150 mM NaCl, 0.2% Triton X-100, 0.1% sodium dodecyl sulfate (SDS), 50 mM Tris–HCl (pH 8.0), 0.5 mM Na3VO4, 1 mM dithiothreitol, 0.5 mM NaF, and protease inhibitor cocktail (Roche Diagnostics GmbH, Basel, Switzerland), followed by centrifugation at 1000g for 5 minutes. Supernatants were mixed with an equal amount of sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) sample buffer containing 0.05 M Tris–HCl, 2% SDS, 6% β-mercaptoethanol, 10% glycerol, and bromophenol blue and boiled for 5 minutes. Each sample was separated by SDS-PAGE and transferred to polyvinylidene difluoride membranes (Millipore). Membranes were blocked in Tris-buffered saline containing Tween (0.1%) and 2% nonfat dry milk for 1 hour, followed by incubation with primary antibodies overnight at 4°C. Bound antibodies were visualized using horseradish peroxidase-conjugated secondary antibodies (Santa Cruz Biotechnology), followed by incubation with enhanced chemiluminescence reagent (Promega). Probed membranes were analyzed by ImageQuant LAS 4010 using ImageQuant TL software (GE Healthcare, Chicago, IL).
Serum Biochemical Analysis in Mice
Serum from blood collected from the inferior vena cava after mice were killed was subjected to assays conducted by Oriental Yeast Co, Ltd, to measure serum levels of aspartate aminotransferase (AST), alanine aminotransferase (ALT), blood urea nitrogen, and creatinine, which are biomarkers for hepatic and kidney toxicity.
Real-Time Reverse Transcription-Polymerase Chain Reaction
Real-time reverse transcription-polymerase chain reaction (RT-PCR) was performed as previously described. 34 Total RNA (1 μg) was subjected to first-strand complementary DNA (cDNA) synthesis using the oligodeoxythymidine standard primer. The expression of ANP, BNP, and GAPDH was quantified by RT-PCR using the SYBR Green kit (Applied Biosystems). The following primers were used in this study: ANP, forward 5′-AGTGGGCAGAGACAGCAAAC-3′ and reverse 5′-TGTGTTGGACACCGCACTGT-3′; BNP, forward 5′-AAGCTGCTGGAGCTGATAAG-3′ and reverse 5′-GGTGCTGCCTTGAGACCGAA-3′; and GAPDH, forward 5′-CATCACCATCTTCCAGGAGCG-3′ and reverse 5′-GAGGGGCCATCCACAGTCTTC-3′.
Histological Analysis of Cardiac Hypertrophy and Cardiac Fibrosis
Hearts were harvested 4 weeks after TAC. Frozen sections (5-μm thick), prepared as described above, were stained with FITC-labeled lectin (Sigma) and Masson trichrome. Using FITC-labeled lectin-stained sections, the cross-sectional area of cardiomyocytes was measured as previously described. 33 Photomicrographs of the Masson trichrome-stained sections were taken, and the fibrotic area was measured with the use of ImageJ software [1.47v] (National Institutes of Health, Bethesda, MD).
Statistical Analysis
All data are expressed as mean ± standard error of the mean. Differences between experimental groups were determined using Student t test or 1-way analysis of variance, and P < .05 was considered statistically significant.
Results
Locked Nucleic Acid ASOs Are Distributed to Hepatocytes and Cardiomyocytes in Mice After Intravenous Injection
Several previous reports suggest low efficacy of exogenous gene induction in heart except when viral gene transfer methods are used. 35,36 To evaluate the efficacy of LNA-ASO transfer into the heart in vivo, we injected mice with Cy3-labeled LNA-ASO (Cy3-LNA) intravenously at doses of 0.3 or 3.0 mg/kg and assessed Cy3 fluorescence in the heart using microscopy. First, we assessed the fluorescence in sections prepared from the liver as a control and observed Cy3-LNA accumulation in mice injected with either dose (Figure 1A). Similarly, we observed a distinct Cy3-LNA fluorescence in the heart but less frequently than in the liver (Figure 1B). These results suggested that intravenously injected ASOs could accumulate in the heart and elicit a biological effect. A single injection of 3.0 mg/kg tended to show liver toxicity, as indicated by elevation of AST and ALT (data not shown). Thus, we performed the following experiments using a single dose of 0.3 mg/kg of LNA-ASO.

Distribution of locked nucleic acid (LNA)-modified antisense oligonucleotide (ASO) in mouse hepatocytes and cardiomyocytes after intravenous injection. Cy3-labeled LNA (Cy3-LNA) was administered to mice via tail vein injection; sections were prepared from liver (A) and heart (B) excised 24 hours after injection. Subsequently, the sections were stained with fluorescein isothiocyanate (FITC)-conjugated lectin. A, Biodistribution of Cy3-LNA was examined by immunofluorescence staining with FITC-conjugated lectin (green) and Cy3 (red) in liver tissue from mice injected with the indicated amounts of Cy3-LNA. The control represents sections from mice injected with unlabeled LNA. B, Accumulation of Cy3-LNA was observed in heart sections prepared from mice injected with the indicated amounts of Cy3-LNA. Immunofluorescence staining with FITC-conjugated lectin (green) and Cy3 (red) are shown.
Design and Validation of PLN-Targeting LNA-ASOs Using Cotransfection With PLN Expression Vector in HEK Cells
To choose the appropriate sequences of LNA-ASOs to inhibit PLN expression, we designed multiple LNA-ASOs predicted to minimize off-target effects. We screened for inhibitory effects on protein levels using a coexpression system with PLN-expressing vectors in human embryonic kidney (HEK) cells and found 2 PLN-AS sequences (AS-1 and AS-2) that decreased the expression of PLN (Figure 2A, B). In contrast, a scr-LNA failed to inhibit PLN expression (Figure 2C, D). These results suggest that experiments evaluating the coexpression of targeted genes and LNA-ASOs in HEK cells are a simple and useful screening system to find appropriate sequences for inhibition.

Determination of appropriate phospholamban (PLN)-targeting locked nucleic acid (LNA)-modified antisense oligonucleotide (ASO) in HEK293 cells with induced PLN expression. In vitro silencing properties of PLN-targeting LNA-ASO (PLN-AS). The PLN-AS and pcDNA3.1-PLN were cotransfected into HEK293 cells. After 24 hours of incubation, cells were collected and the expression levels of PLN protein were determined by Western blot analysis. A, The screening of suitable PLN-AS with 4 distinct sequences (AS-1, AS-2, AS-3, and AS-4) was performed by Western blot analysis. The control represents a protein sample from HEK293 cells transfected with pcDNA3.1-PLN without PLN-AS. B, Quantification of the expression levels of PLN in HEK293 cells cotransfected with pcDNA3.1-PLN and the indicated PLN-AS. The PLN-AS-1 displayed the most potent suppression of PLN protein expression. The data are expressed as mean ± standard error of the mean (SEM) of 3 independent experiments. C, Western blot analysis using protein samples from cells transfected without (CNT) or with AS-1/scrambled LNA (scr-LNA). D, Quantification of the expression levels of PLN in HEK293 cells cotransfected with pcDNA3.1-PLN and PLN-AS-1 or scr-LNA. The data are expressed as mean ± SEM of 3 independent experiments.
Intravenous Injection of PLN-Targeting LNA-ASO Reduces PLN Expression in Mouse Hearts
To further elucidate the usefulness of the selected PLN-AS (PLN-AS-1) in vivo, we injected C57BL/6 wild-type mice with PLN-AS-1 or scr-LNA at a dose of 0.3 mg/kg intravenously and collected heart samples at 1, 3, and 7 days after injection. The injection of scr-LNA failed to decrease PLN expression. In contrast, administration of PLN-AS-1 decreased PLN expression in a time-dependent manner until day 7 (Figure 3A, B). These results suggest that this PLN-AS-1 could be a useful tool to inhibit PLN gene expression.
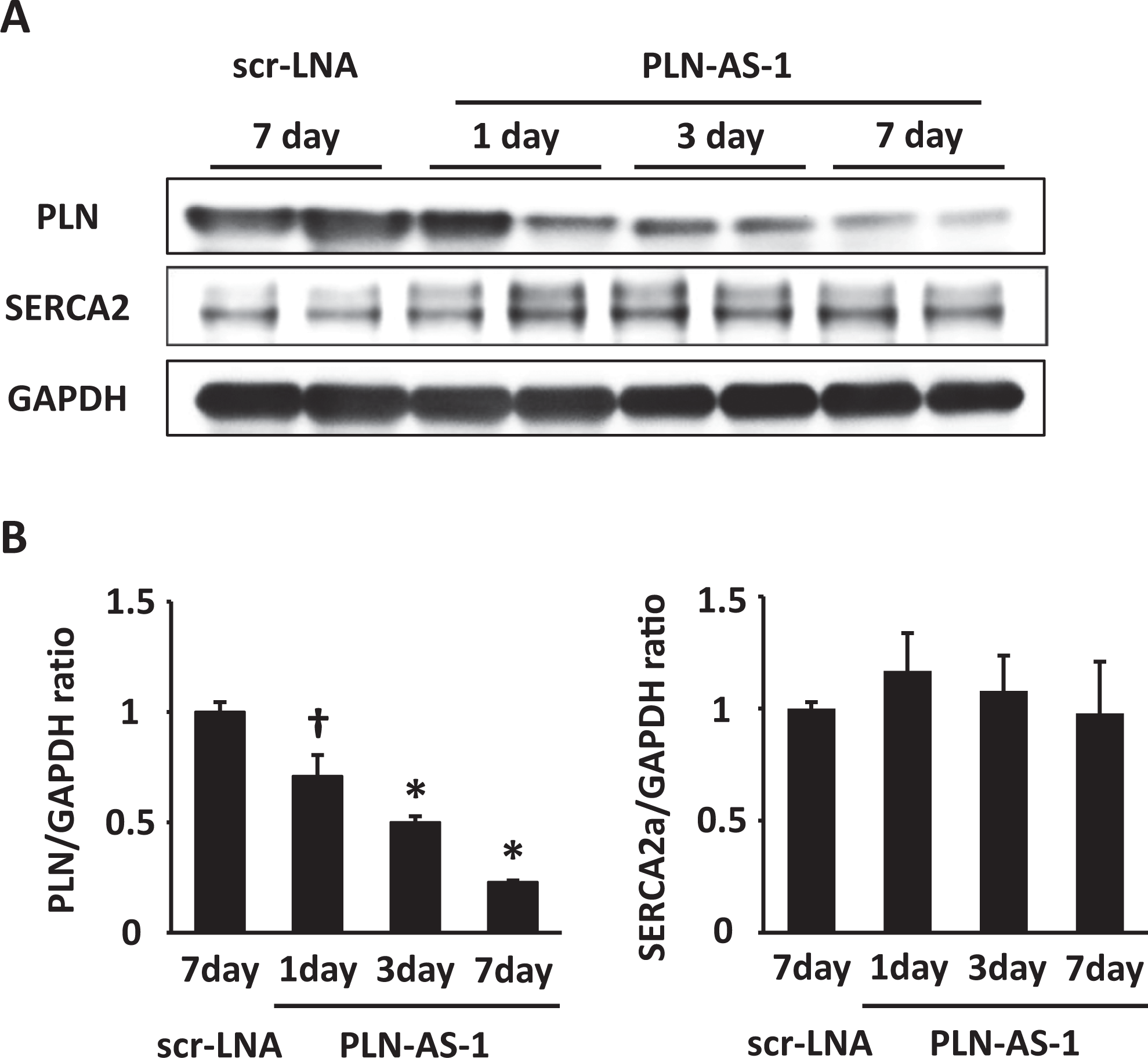
Administration of phospholamban (PLN)-targeting locked nucleic acid (LNA)-antisense oligonucleotide (ASO) decreases PLN protein expression in mouse hearts. Adult male C57BL/6 mice were administered 0.3 mg/kg of scrambled LNA (scr-LNA) or PLN-targeting LNA-ASO-1 (PLN-AS-1) via tail vein injection. Hearts were excised at 1, 3, and 7 days after injection and cardiac homogenates were prepared, followed by Western blotting analysis with anti-PLN, anti-SERCA2, or anti-GAPDH antibodies. A, Representative blots of 2 independent samples from each cohort probed with the indicated primary antibodies. B, Quantification of the PLN/GAPDH or SERCA2a/GAPDH band density ratio. The data are shown as mean ± standard error of the mean of 3 independent experiments. † P < .05 and *P < .01 compared with scr-LNA by one-way analysis of variance.
Intravenous Injection of PLN-targeting LNA-ASO Improves Systolic Function as Assessed by Echocardiography in a Pressure Overload-induced Cardiac Dysfunction Model
To further investigate the efficacy of PLN inhibition by PLN-AS-1 in a cardiac dysfunction model, we performed TAC or sham surgery on C57BL/6 wild-type mice to induce a pressure overload and assessed cardiac function 3 weeks after the procedure. The mice that underwent TAC or sham surgery were allocated into 2 groups based on echocardiographic analyses and were injected with PLN-AS-1 or scr-LNA (Figure 4A).

Administration of phospholamban (PLN)-targeting locked nucleic acid (LNA)-antisense oligonucleotide (ASO) improves systolic function in a pressure overload-induced cardiac dysfunction model. A, Schematic diagram of the experimental protocol. Adult male C57BL/6 mice were subjected to sham or transverse aortic constriction (TAC) surgery, and echocardiographic analyses were performed 3 weeks after TAC. Thereafter, the mice were separated into 2 groups and administered scrambled locked nucleic acid (scr-LNA) or 0.3 mg/kg of PLN-targeting LNA-ASO-1 (PLN-AS-1) via tail vein injection. Echocardiographic and gravimetric analyses were performed 1 week after injection (ie, 4 weeks after TAC). B, Expression levels of PLN and SERCA2a protein in cardiac homogenate obtained from mice 1 week after a single injection of the indicated LNA. C, Quantification of the PLN/GAPDH and SERCA2a/GAPDH ratios. The data are expressed as mean ± standard error of the mean (SEM) of 3 independent experiments. *P < .01 compared with sham-operated mice treated with scr-LNA by one-way analysis of variance. D, Alterations of fractional shortening (FS) assessed by echocardiography between pre- and 1 week after injection. The changes in the TAC groups treated with scr-LNA (n = 10, left panel) or PLN-AS-1 (n = 5, right panel) are displayed. P < 0.05 by paired t test. E, Decrease/increase in FS of scr-LNA TAC or PLN-AS-1 TAC groups. F, Pressure gradient (mm Hg) of each TAC group before administration of LNA. The data are expressed as mean ± SEM (n = 5-10). † P < .05 compared with TAC group injected with scr-LNA by Student t test.
One week after injection of scr-LNA, the protein levels of PLN tended to decrease in the TAC group compared with the sham group (Figure 4B). Of note, the PLN protein levels were significantly lower in the PLN-AS-1-treated TAC groups compared with the scr-LNA sham and TAC groups, despite a similar expression of SERCA2a (Figure 4C), suggesting that SERCA2a activity could be enhanced in PLN-AS-1-treated heart in this experimental model, however, this needs to be experimentally verified. Echocardiographic measurements before administration were not significantly different between the PLN-AS-1 and scr-LNA groups with TAC (Table 1). In the sham groups, no significant differences were observed in fractional shortening (FS) between groups before PLN-AS-1 or scr-LNA administration (Table 1). One week after injection (ie, 4 weeks after TAC), FS in the PLN-AS-1 group was significantly higher than that in the scr-LNA group, suggesting enhancement of contractility by PLN-AS injection (Table 1). In addition, although FS in mice of the TAC group treated with scr-LNA tended to decrease, PLN-AS-1-treated mice displayed significant improvement in FS one week after injection (Figure 4D). The changes in FS values were significantly greater in the PLN-AS-1 TAC group than in the scr-LNA TAC group (6.5 ± 1.6% vs −4.0 ± 2.3%, P < .05, Figure 4E). Notably, no significant differences were observed in pressure gradients across the constriction before administration, suggesting equal pressure overloads were applied in the 2 groups (Figure 4F). Taken together, these results suggest that PLN inhibition by PLN-AS-1 treatment primarily enhances contractile function in a systolic dysfunction model induced by pressure overload.
Echocardiographic Analyses.a

Abbreviations: ANOVA, analysis of variance; ASO, antisense oligonucleotide; FS, fractional shortening; HR, heart rate; LNA, locked nucleic acid; LVIDd, left ventricular internal dimension in diastole; LVIDs, left ventricular internal dimension in systole; PLN, phospholamban; PLN-AS-1, PLN-targeting LNA-ASO-1; scr-LNA, scrambled LNA-ASO; SEM, standard error of the mean; TAC, transverse aortic constriction.
aData are shown as mean ± SEM (n = 5 for scr-LNA-sham; n = 6 for PLN-AS-1-sham; n = 10 for scr-LNA-TAC; n = 5 for PLN-AS-1-TAC).
b P < .05 versus sham treated with corresponding LNA by 2-factorial ANOVA followed by Bonferroni.
c P < .05 versus scr-LNA-TAC (post).
Treatment With PLN-Targeting LNA-ASO Fails to Attenuate Cardiac Hypertrophy
In light of the effect of PLN-AS treatment on pathological remodeling, gravimetric and histological analyses were performed in the mice 1 week after a single injection of the corresponding LNA-ASOs (ie, 4 weeks after TAC). Cardiac hypertrophy assessed by heart weight-to-body weight ratio showed a trend to be lower in the PLN-AS-1 group compared to the scr-LNA group after pressure overload (Figure 5A). In contrast, there were no significant differences in the expression of ANP or BNP between the 2 groups of TAC (Figure 5C, D). Consistently, while assessment of cross-sectional cell surface area of cardiomyocytes showed a trend to be less cellular hypertrophy in PLN-AS-1-treated mice compared to scr-LNA mice 1 week after injection (ie, 4 weeks after TAC, Figure 5E), the difference was not significant. Taken together, these results indicate that treatment with PLN-antisense primarily affects cardiac contractile function and fails to impact cardiac pathology in the model presented here. Regarding other histological changes, there were no differences between the 2 TAC groups regarding fibrosis (Figure 5G) or inflammatory cell infiltration (Supplemental Figure 4).

Treatment with phospholamban (PLN)-targeting locked nucleic acid (LNA)-antisense oligonucleotide (ASO) partially attenuates cardiac hypertrophic responses. The effect of PLN-AS treatment on pathological remodeling, gravimetric analysis, and molecular markers of cardiac hypertrophy was examined in mice 1 week after a single injection of the indicated LNA (ie, 4 weeks after transverse aortic constriction [TAC]). A, Heart weight (HW) normalized to body weight (BW) ratio for the mice treated with scrambled LNA (scr-LNA) or PLN-targeting LNA-ASO-1 (PLN-AS-1) 4 weeks after sham or TAC surgery. The data are expressed as mean ± standard error of the mean (SEM; n = 4-10). B, Lung weight (Lung W) normalized to BW ratio for the mice from the indicated cohort. The data are expressed as mean ± SEM (n = 4-10). C and D, Gene expression levels of ANP (C) or BNP (D) normalized to GAPDH. The data are expressed as mean ± SEM (n = 4-7). E, Representative images of heart sections stained with wheat germ lectin conjugated with fluorescein isothiocyanate are shown. F, Quantification of the longitudinal cross-sectional area assessed from heart with nonoperated mice (non-ope) and TAC mice injected with the indicated LNA. G, Quantification of fibrotic area assessed from heart with TAC mice injected with the indicated LNA. Values are shown as mean ± SEM of 3 to 4 hearts in each cohort. †P < .05 and *P < .01 compared with the corresponding sham group.
Discussion
In the present study, we examined cardiac delivery of LNA-ASOs for PLN using Cy3-labeled LNA-ASO and revealed infiltration of this modified RNA into adult mouse myocardium. We further demonstrated that a single dose of PLN-targeting LNA-ASO leads to enhancement of systolic function as assessed by echocardiography, suggesting that suppression of PLN by LNA-ASO is physiologically effective.
Until recently, the efficacy of the transfer of exogenous oligonucleotides into cardiomyocytes has been considered to be extremely low. 35,37 Although LNA-ASOs are relatively resistant to nuclease and could be much more effective for gene silencing in cardiomyocytes, the redistribution of LNA-ASO in the heart is <5% of the total dose. 35 Therefore, the heart is considered to be a particularly unsuitable organ for gene therapy using oligonucleotide drugs. Consistently, in the present results using Cy3-labeled LNA-ASO, the efficacy of LNA-ASO transfer to cardiomyocytes appeared much lower than that of LNA-ASO transfer to the liver in microscopic analysis. However, several groups previously reported that LNA-ASOs used for silencing microRNAs demonstrated the expected results in mice, 32,38,39 suggesting that the amount of LNA-ASO distributed to the heart is sufficient to suppress target gene expression. Our results indicate that the amount of administered LNA-ASO capable of suppressing gene expression in the heart is much lower than the dose that induces liver toxicity in mice, suggesting that an effective concentration of LNA-modified oligonucleotide for gene silencing in the heart could be useful for mammals, including humans.
The validation of LNA-modified oligonucleotide is an important and sometimes time-consuming process, especially in cardiomyocytes, which are resistant to transfection by nonviral vectors. To select the proper sequence of LNA-ASOs to inhibit protein expression, we used a coexpression system of the PLN gene and LNA-ASO, and the selection of the sequence by this method is suitable for choosing effective LNA-ASOs for in vivo use.
The SERCA2a–PLN interaction is considered an attractive target for the treatment of HF. 7 The efficacy of PLN inhibition in HF has been indicated in a plethora of previous reports. 18,19,40,41 Genetic deletion of PLN alleviates cardiomyopathy in multiple models including muscle LIM protein knockout (MLPKO) 19 and calsequestrin overexpression. 40 Furthermore, inhibition of PLN by viral gene transfer ameliorates cardiac dysfunction in a pathological rodent model. 18,41 Thus, therapeutic approaches that can activate SERCA2a are required. Our results indicate that a single-dose injection to achieve virus-free gene transfer using LNA-ASOs could be as effective as those methods for enhancement of contractility. In addition, as reported in the Calcium Upregulation by Percutaneous Administration of Gene Therapy in Cardiac Disease (CUPID) trial, 42 SERCA2a gene transfer to the myocardium of patients with HF by coronary administration of AAV vectors demonstrated safety and potential efficacy. These results have been extended to phase II multicenter clinical trials as CUPID II to develop a novel treatment. 43 Recently, the results of CUPID II were published, and unfortunately, this largest gene transfer study performed in patients with HF failed to improve outcomes, which might be due to the insufficient dose of AAV1/SERCA2a. 44 Because the inhibitory effect of PLN may have overwhelmed the beneficial effect of upregulated SERCA2a by gene transfer in the CUPID II trial, the present results suggest that PLN-targeting LNA-ASO is a potentially useful tool in combination with or as an alternative to AAV-based SERCA2a gene therapy by modulating the SERCA2a–PLN axis.
In the present study, we assessed a positive inotropic effect of PLN-AS in mice with moderate cardiac dysfunction caused by pressure overload. This experimental model is believed to mimic the condition of patients with HF who require short-term catecholamine treatment. Although enhanced systolic function was observed in mice treated with PLN-AS, improvements in pathological changes such as fibrosis or cardiac hypertrophy were modest. Consistent with the present results, several previous reports indicated that inhibition of PLN failed to attenuate the histological changes in genetically engineered models, despite inducing enhanced myocyte contractility. 45,46 Thus, at least in the model presented here, modulation of PLN mainly elicited alterations of cardiac contractility and demonstrated less effect on the fibrotic change or hypertrophy that developed before the administration of PLN-AS. Our results suggest that direct modification of calcium cycling by PLN inhibition enhances contractility without activation of βAR signaling, which could elicit pathological alterations including cell death.
In the present study, we mainly focused on systolic function modified by PLN-AS; however, improving diastolic function is another issue for PLN-targeting therapy. We attempted to analyze the peak early to late diastolic filling velocity (E/A) ratio of mitral flow obtained by echocardiography in mice and observed a higher E/A ratio in the PLN-AS-treated group after TAC, suggesting that the PLN-targeting approach may be useful to improve diastolic function (Supplemental Figure 2). In the future, further examination using the ex vivo Langendorff system is required to decipher the beneficial effect of PLN-AS against diastolic HF.
As previously described, inhibition of PLN is a promising therapeutic strategy, especially in combination with SERCA2a gene transfer or β-blockade. In the former case, PLN-AS accelerates the improvement of calcium cycling caused by increased SERCA2a expression. In the latter case, PLN antisense therapy attenuates the impairment of contractility, which is the most significant side effect of β-blockade, limiting the usefulness of this therapy. Thus, the development of efficient and safe methods to inhibit PLN expression or function is one of the most promising innovations in the treatment of HF. The PLN-targeting LNA-ASO presented here is considered as a strong candidate for this goal.
Footnotes
Acknowledgments
The authors would like to thank Wakako Okamoto and Chiharu Tottori for their excellent secretarial work.
Author Contributions
H.N. participated in study design. H.M., T.Y., H.O., K.T., D.T. and M.O. conducted the experiments. H.M., H.O., I.K and H.N. performed data analysis. H.M., T.Y., S.O., M.M., Y.F. and H.N. wrote or contributed to the writing of the manuscript. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by MEXT/JSPS KAKENHI Grant Numbers 23390212 and 23659110 to H.N.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
