Abstract
Background and Purpose:
Published data on nebivolol reveal selective β1 adrenergic selectively along with novel nitric oxide (NO)-dependent vasodilatory properties. However, the exact molecular mechanism is unknown. Protein S-nitrosylation constitutes a large part of the ubiquitous influence of NO on cellular signal transduction and is involved in a number of human diseases. More recently, protein denitrosylation has been shown to play a major role in controlling cellular
Experimental Approach:
Using cell culture systems, tissue organ bath, and intact animal models, we report that nebivolol treatment leads to a dose-dependent accumulation of nitrosothiols in cells, and this is associated with an enhanced vasodilation by
Key Results:
These data suggest a new mechanism of action of nebivolol that may explain in part the reported NO activity.
Conclusions and Implications:
Because exogenous mediators of protein SNO or denitrosylation can substantially affect the development or progression of disease, this may call for new utility of nebivolol.
Introduction
Nebivolol is a selective β1-adrenergic receptor blocker with approximately 320-fold greater affinity for β1 than β2 receptors that also possesses vasodilator and antioxidant properties. 1 This dual mechanism of action underscores unique hemodynamic qualities of nebivolol, which include reductions in heart rate and blood pressure and improvements in systolic and diastolic function. Although nebivolol reduces blood pressure to a degree similar to that of conventional β-blockers and other types of antihypertensive drugs, it offers selective advantages in populations with resistant hypertension, such as patients with heart failure along with other comorbidities, such as diabetes and obesity, and elderly patients in whom nitric oxide (NO)-mediated endothelial dysfunction may be more pronounced. Nebivolol maintains cardiac output by increasing stroke volume, while decreasing systemic vascular resistance in human volunteers and in animal studies. 2 Nebivolol has also been shown to be effective in pulmonary hypertension (PH) in vitro 3,4 and in animal models of PH. However, the efficacy of nebivolol in patients with PH has not yet been demonstrated. While the vasodilatory properties of carvedilol and labetalol are mediated by α-adrenergic receptor blockade, nebivolol exerts these effects by increasing endothelium-derived NO via stimulatory effect on endothelial NO synthase (eNOS), mediated through β3 agonism. 5 –8 Some work suggests that the vasodilatory effects are mediated through NO 9 but does not involve NO’s primary receptor soluble guanylyl cyclase (sGC). 10 Nebivolol causes an endothelial-dependent relaxation of canine coronary arteries and potentiates adenosine diphosphate–induced, endothelial-dependent relaxation through NO. The vasodilator effect is attributed largely to activation of eNOS and increased NO release, whereas the antioxidant effect is thought to be attributable to inhibition of Nicotinamide adenine dinucleotide phosphate (NADPH) oxidase and reduced superoxide formation. 5,11 Several studies have implicated its mechanism of action to be through increased NO production from eNOS because it can be abrogated by inhibitors of NOS. 5,6 Other investigators have found that nebivolol reduces plasma levels of Assymetric dimethyl L-Arginine (ADMA) in spontaneously hypertensive rats (SHRs). 12 There are clear and demonstrable effects of nebivolol on steady state NO bioavailability, however, its exact mechanism of action has yet to be completed elucidated.
S-Nitrosothiols are thioesters of nitrite with the general structure R-S-N=O (RSNO), naturally occurring examples include S-nitrosocysteine, S-nitrosoglutathione (GSNO), and S-nitrosoalbumin, in which R is an amino acid, polypeptide, and protein, respectively. S-Nitrosation is a ubiquitous redox-related modification of cysteine thiol which transduces NO bioactivity. 13 There is now a large body of literature implicating RSNO as an intermediate in NO-dependent and sGC-independent signaling processes. S-Nitrosothiols are susceptible to decomposition by numerous mechanisms, giving rise to NO and the corresponding disulfide. The rate of decomposition is influenced by a number of factors, including metal ions (particularly Cu+), transnitrosation, that is, the transfer of NO or nitrosonium ion from S-nitrosothiols to other thiols, enzymatic decomposition, photochemical decomposition, thermal decomposition, and reaction with ascorbate. S-Nitrosothiols are actively metabolized by cells 14 and are biologically active as vasodilators and inhibitors of platelet aggregation. 15 Low-molecular-weight nitrosothiols act similar to endothelium-derived relaxing factor, but with a longer circulating half-life than NO and therefore make them an ideal candidate for drug development.
Specificity for
Structurally, nebivolol is similar to known inhibitors of GSNOR (see Figure 1 below) and therefore there is reason to investigate its activity. Broad structural heterocyclic compounds with functional hydroxyl and carboxylic acid groups are potent inhibitors of GSNOR. The emerging picture shows that, in many cases, pathophysiology correlates with hypo- or hyper-S-nitrosylation of specific protein targets rather than a general cellular insult due to loss of or enhanced NOS activity. Exogenous mediators of protein S-nitrosylation or denitrosylation can substantially affect the development or progression of disease and therefore have broad clinical utility. The objective of this study is to investigate nebivolol’s effects on steady state protein S-nitrosylation as a novel inhibitor of GSNOR and its effect in cell culture along with intact animals and vascular reactivity.

Structural comparison between nebivolol and known inhibitors of
Materials and Methods
Tissue Harvest and Vascular Reactivity
Male mice (C57BL/6J) and eNOS knockout mice, aged 10 weeks, were anesthetized with diethyl ether. In a separate set of experiments, male Sprague Dawley rats (6 months) were used for vascular experiments. A thoracotomy was performed to expose thoracic and abdominal aorta. Venous blood was collected through the inferior vena cava. A 25-gauge syringe was then inserted into the apex of left ventricle and perfused free of blood with oxygenated Krebs Henseleit buffer. The right atrium was cut to provide an exit for blood. Heart and liver were excised for NO biochemistry. The aorta was removed and cleaned of fat and adventitia. The aorta was cut into 2-mm long segments and mounted on a 4-channel wire myograph (AD Instruments, Colorado Springs, CO, USA). Vessel rings were maintained in 10 mL organ baths with oxygenated phosphate-buffered saline (PBS; 95% O2 and 5% CO2) at 37°C. Rings were allowed to equilibrate for 80 minutes with the buffer in each organ bath changed every 20 minutes. One and a half grams pretension was placed on each aortic ring (appropriate starting tension for optimal vasomotor function as determined in previous experiments). An 8-channel octal bridge (Powerlab, Colorado Springs, CO, USA) and data acquisition software (Chart version 5.2.2) was used to record all force measurements. After equilibration for 80 minutes, 1 µmol/L phenylephrine (PE) was added to each ring for submaximal contraction as determined by first contraction with KCl. After stabilization, GSNO (1 µmol/L) was added to the rings, and tension was recorded every 4 minutes for 20 minutes. In separate experiments, nebivolol (10 µ) was added to each organ bath prior to the addition of GSNO (1 µmol/L). We selected 10 µmol/L since this concentration alone did not cause vasodilation of the isolated rings. All data reported are average of 4 rings from n = 3 mice from each group.
In a separate set of vascular experiments, C57 and eNOS knockout mice were either pretreated for 7 days with 10 mg/kg nebivolol per day or with 5% gum Arabic as vehicle control. Aorta was then harvested as described above for vascular reactivity experiments and heart and liver harvested for NO biochemistry.
Cell Culture Experiments
Determination of nitroso species accumulation in RAW cells using the triiodide-based chemiluminescence method
RAW 264.7 and HL-1 cells were cultured in DMEM medium supplemented with 10% fetal bovine serum, 200 U/mL penicillin, and 200 µg/mL streptomycin. The cells are incubated at 37°C in an atmosphere containing 5% CO2 and 95% air. For the experiments, 1 to 2 × 106 cells are plated in 6-well plates with or without 33 µmol/L compounds, 16 hours before the experiment (later experiments showed that pretreatment with compounds had no effect on the rate of accumulation of nitroso species inside the cells). On the day of experiment, the medium is replaced with fresh 3 mL of medium, and the cells are treated with compounds for a predetermined length of time. Following the incubation period, the cells are washed 3 times with PBS and scraped off the plate in 250 µL of lysis buffer (50 mmol/L potassium phosphate pH 7.0 containing 50 mmol/L N-ethylmaleimide (NEM) and 1 mmol/L EDTA) and lysed by sonication using a micro tip probe (3 pulses of 30% duty cycle and 2 output control on a Branson Sonicator). The cell debri was pelleted by centrifugation (10 minutes at 16 000
S-nitrothiol determination
Control C57Bl/6 and eNOS knockout mice were purchased from Jackson Laboratories, Inc. (Bar Harbor, ME, USA). Mice were allowed 10 days acclimatization after arrival prior to any experiments. The eNOS knock out mice and control C57 mice were grouped into 2. One group of mice was dosed with 10 mg/kg nebivolol in 5% gum Arabic as a suspension by oral gavage for 7 days, and another group was dosed with vehicle 5% gum Arabic by oral gavage for 7 days. Twenty mg of nebivolol was dissolved in 1 mL 5% gum Arabic. Mice were dosed via oral gavage with 150 to 200 µL depending upon individual weight. Nebivolol was provided by Forest Research, Inc. Nitrosothiols were quantified and detected using ozone-based chemiluminescence (EcoPhysics, CLD88Y). Nitrosothiols were determined by the difference in peak area from sulfanilamide versus mercuric chloride with sulfanilamide treatment as previously described. 23,24
Biotin switch assay
We determined the effects of nebivolol on the modulation of S-nitrosylation through the biotin switch assay. Cayman Chemical Co (item No. 10006518) was purchased and used for the analysis. This method is based on a modification of the Jaffrey et al 25 method.
Statistics
Significant effects of treatment were evaluated by one-way analysis of the variance using Origin 8.5 software. Where significant differences between data sets were observed, post hoc tests (eg, Tukey or Scheffe) were performed to determine differences between individual data sets. In all cases,
Results
To address whether nebivolol had any effect on the metabolism or stability of nitrosothiols, we treated RAW 234.7 murine macrophage cells with 500 µmol/L S-nitrosoglutathione (GSNO) without nebivolol and treated separate cell culture wells with 500 µmo/L GSNO in the presence of increasing concentrations of nebivolol (10-50 µmol/L). After 6 hours, cells were lysed and then analyzed for nitrosothiols. As shown in Figure 2A, treatment with nebivolol lead to a dose-dependent increase in total nitrosothiols in the cell lysate after 6 hours. All concentrations tested beginning at 10 µmol/L nebivolol caused a statistically significant increase in intracellular nitrosothiols after 6 hours (
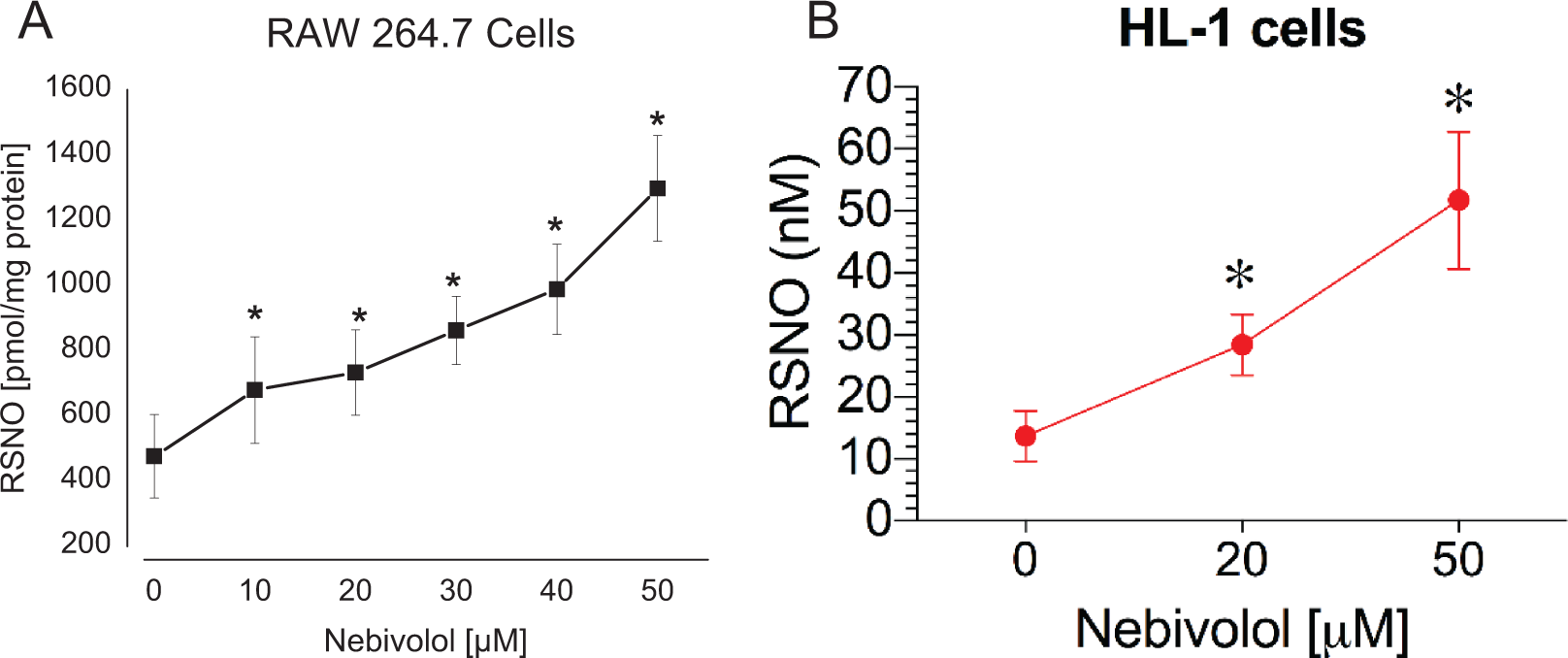
A, Accumulation of nitrosothiols after 6 hours in RAW 264.7 cells after treatment with
To validate the cell culture data in experimental animals, we administered nebivolol (10 mg/kg) or vehicle once daily for 7 days in C57 control and eNOS knockout mice and then euthanized the mice, harvested the heart, and assayed for nitrosothiols. As shown in Figure 3, administration of nebivolol caused a significant increase in total nitosothiols in the heart of C57 mice, whereas nebivolol did not cause any significant changes in total nitrosothiol levels in the heart tissue of eNOS knockout mice.

Accumulation of nitrosothiols in the heart of C57 and endothelial nitric oxide synthase (eNOS) knockout mice after treatment with 10 mg/kg nebivolol daily for 7 days or vehicle. Data are average ± standard error of the mean (SEM) from n = 3 mice from each group.
To corroborate the total tissue nitrosothiol content of the tissues of the mice, we performed the biotin switch assay. This technique allows for detection of specific changes in protein bands of each tissue. As shown in Figure 4, the heart of the C57 mice revealed significant increases in density of bands of the nebivolol-treated mice. There was also a slight increase in density of the bands from the nebivolol treated C57 liver at the 36 kD level. The eNOS knockout mice did not show any changes with nebivolol treatment in heart or liver tissue homogenate.
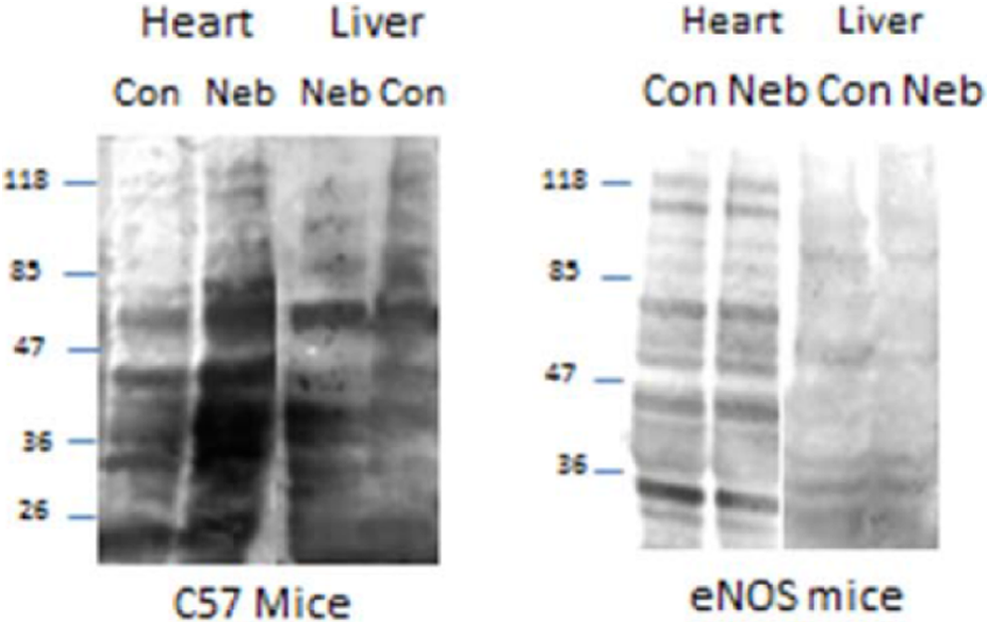
Biotin switch assay showing the increase in protein S-nitrosylation in nebivolol-treated C57 mice compared to control. endothelial nitric oxide synthase (eNOS) knockout mice did not show an increase in S-nitrosylation after treatment with nebivolol. Data are average ± standard error of the mean (SEM) of n = 3 mice in each group.
We next conducted a full dose–response of nebivolol on vascular relaxation in aortic strips from male Sprague Dawley rats. As shown in Figure 5, there is a dose-dependent increase in relaxation beginning at 30 µmol/L concentrations of nebivolol. We also treated aortic rings acutely with an NOS inhibitor NG-nitro-L-arginine methyl ester (L-NAME) to determine whether acute inhibition of NOS would affect relaxation by nebivolol. There was no difference in the relaxation response to nebivolol when treated with L-NAME. This is likely due to the slower turnover of S-nitrosothiols and longer half-life than authentic NO gas. This also suggest that nebivolol is not directly acting on endothelial NO production but rather a thiol-dependent relaxation mechanism that is largely dependent upon NO production but not directly on NO. To investigate this mechanistically, we added 1 µmol/L GSNO to isolated aortic rings and observed that the addition of nebivolol further potentiated the effects of low dose GSNO in a dose-dependent manner (Figure 5). To determine whether nebivolol could enhance the time of relaxation to GSNO, we performed additional vascular relaxation experiments. Preliminary studies revealed that 1 µmol/L GSNO caused about 25% relaxation to precontracted rings and that 10 µmol/L nebivolol alone did not cause any vasodilation to precontracted rings. Based on these data, we treated precontracted aortic rings to 80% maximal constriction with PE and then treated with 1 µmol/L GSNO ± 10 µmol/L nebivolol to investigate whether nebivolol could potentiate the effects of GSNO on the degree of vasorelaxation as well as extend the time of relaxation. As shown in Figure 6, treatment with nebivolol caused a statistically significant increase in vessel relaxation compared to GSNO alone after 15 and 20 minutes.
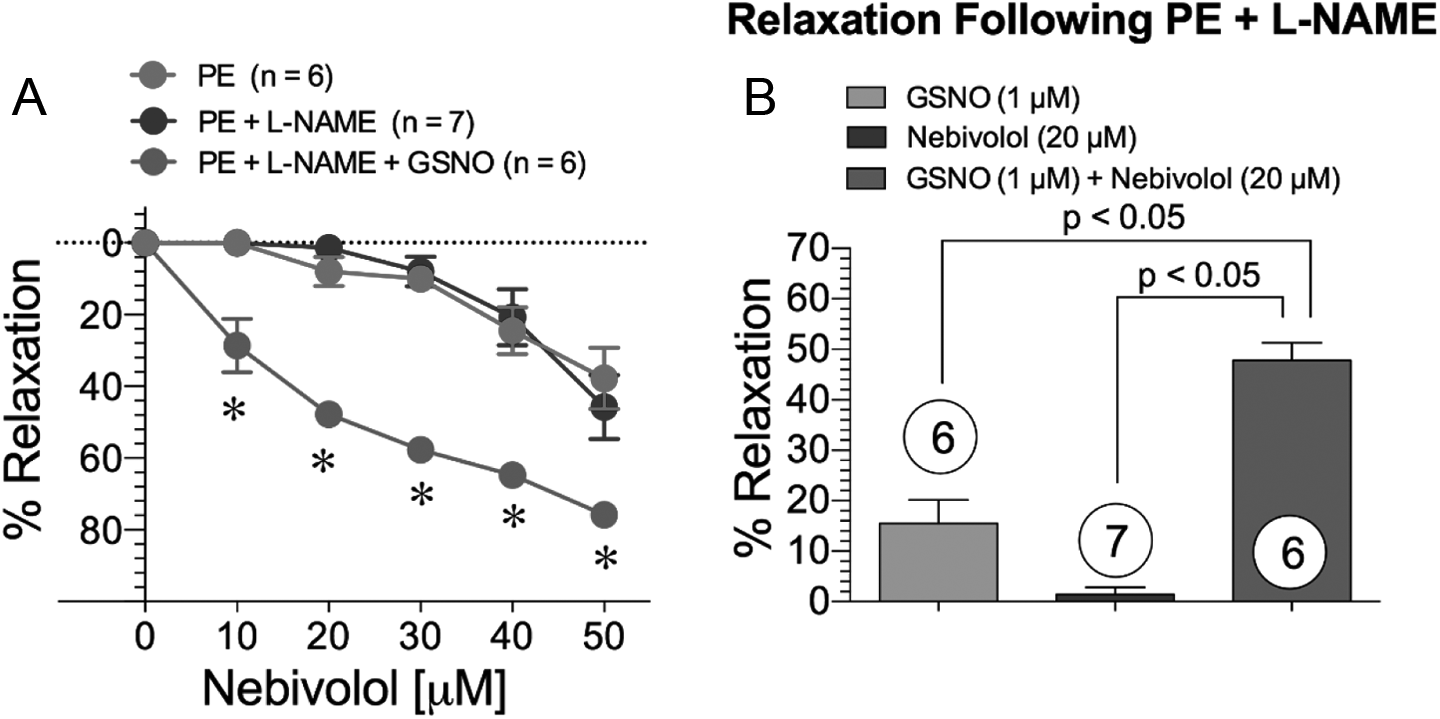
Nebivolol-induced relaxation in isolated phenylephrine (PE)-precontracted rat thoracic aorta. A, Dose-dependent nebivolol relaxation curves in the presence and absence of L-NAME and/or S-nitrosoglutathione. B, Nebivolol and

Effects of nebivolol on S-nitrosoglutathione (GSNO) induced vasorelaxation. Treatment with 10 µmol/L nebivolol to isolated aortic rings caused a significant increase in vasorelaxation due to GSNO after 15 and 20 minutes. Data are average ± standard error of the mean (SEM) of 4 rings isolated from 5 individual mice in each group. (* denotes
In order to determine whether subchronic daily treatment of nebivolol had any effect on vascular reactivity and endothelial function, the abdominal aorta was harvested from C57 mice administered either vehicle control or 10 mg/kg nebivolol once daily for 7 days or eNOS knockout mice administered vehicle or 10 mg/kg nebivolol once daily for 7 days. The C57 control administered nebivolol for 7 days revealed a significant improvement in vascular function based on the percentage relaxation to acetylcholine when compared to vehicle-treated mice at concentrations of 1, 3, and 10 µmol/L. The nebivolol-treated mice had a significantly higher maximal relaxation compared to vehicle-treated mice. There was no difference in the treated versus untreated eNOS knockout mice. These data are illustrated in Figure 7A. Aortic rings were also subjected to a dose–response to GSNO to determine whether the pretreated mice were more sensitive to GSNO administration. As shown in Figure 7B, the nebivolol-treated C57 mice revealed a higher sensitivity to GSNO induced vasorelaxation compared to vehicle control, although there was no difference in maximal relaxation to GSNO. Interestingly, the eNOS knockout mice were much more sensitive to GSNO so lower concentrations were used. The eNOS knockout mice relaxed significantly more than the C57 mice to the same concentration of GSNO. Nebivolol-treated eNOS knockout mice also dilated more to the 0.1 µmol/L dose of GSNO, although the higher concentrations of GSNO caused no significant differences in relaxation. These data are illustrated in Figure 7B.
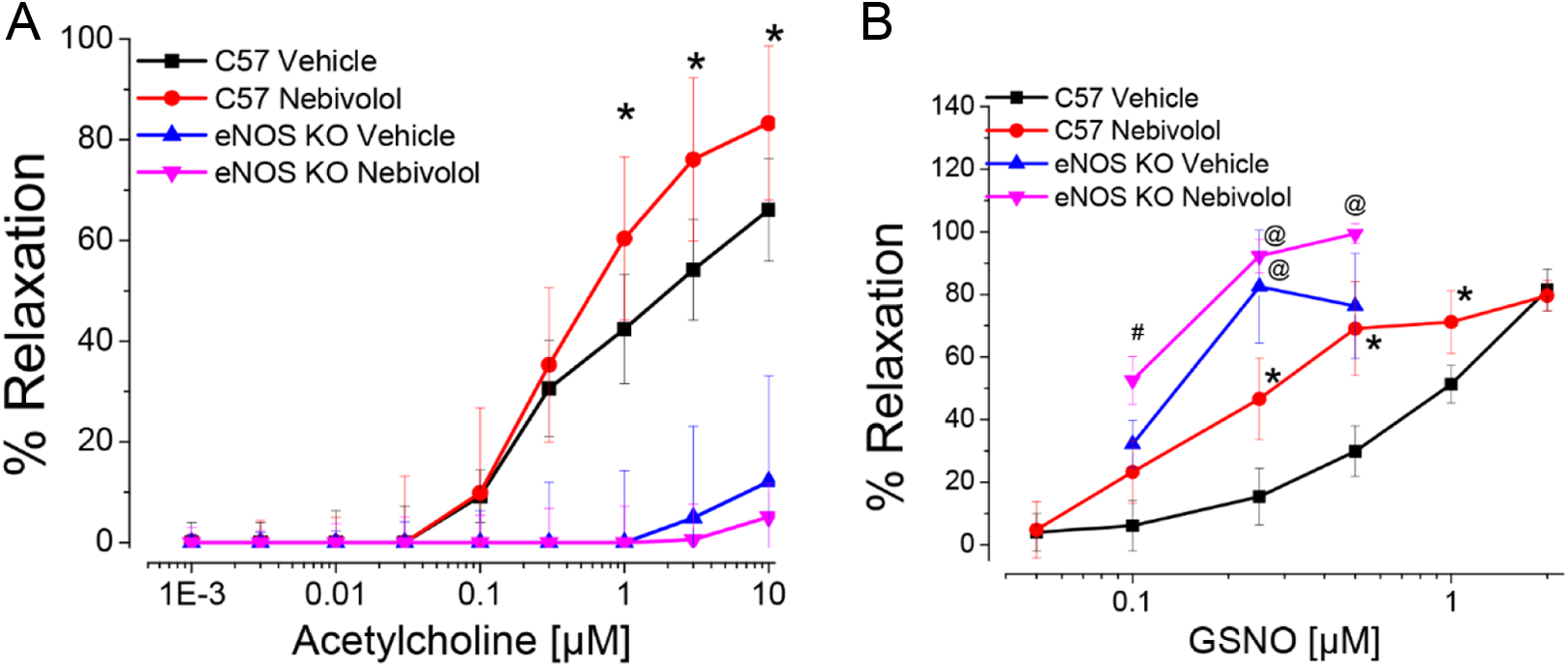
A, Treatment with 10 mg/kg nebivolol leads to significant increase in maximal relaxation to acetylcholine compared to controls. There is no effect of nebivolol in endothelial NO synthase (eNOS knockout mice.) B,. Treatment with 10 mg/kg nebivolol causes a significant increase in relaxation to GSNO. Endothelial NO synthase knockout mice are more sensitive to the relaxing effects of GSNO than C57. Data are average ± SEM of 2 rings each from 3 mice per group (* denotes statistically significant from untreated C57 mice; # denotes statistical significance from treated vs untreated eNOS knockout mice; @ denotes statistical significant difference between eNOS knockout and C57 mice).
Discussion
The vasodilatory β-blocker nebivolol has a pharmacologic profile different from other agents in this class. Unlike 2 other vasodilating β-blockers, which block α1 receptors (carvedilol and labetalol), nebivolol possesses a direct stimulatory effect on eNOS, which results in increased levels of local NO. 26 Although nebivolol has higher β1-selectivity and the highest β1-/β2-selectivity compared with other β-blockers, 2 its vasodilatory properties have been attributed to direct stimulation of endothelial-dependent NO release, 27 but its exact mechanism is still not known. Nebivolol increases NO synthase activity in healthy male volunteers and is blocked by L-NG-monomethyl Arginine. 5 In patients with hypertension, treatment with nebivolol significantly increases forearm blood flow in response to intra-arterial infusion of acetylcholine. 8 The data are quite convincing that NO is involved in the mechanism of action of nebivolol but unclear as to the exact mechanism. NO produced from NOS (either directly through reaction with thyl radicals or from indirectly through oxidation to other nitrosating agents) can modify cysteine thiols on proteins to affect structure and function. 28 The half-life of these posttranslational modifications is highly variable depending on the types of proteins modified and how accessible and susceptible the cysteine thiol may be to metabolism but has been reported to be on the order of 1 hour. 29 Nitrosothiols have been shown to contribute to the systemic vasodilatory effects of NO 30 that last longer than the reported half-life of NO itself (∼1 second). Therefore by affecting the metabolism and turnover of nitrosothiols, the biological activity of NO may be prolonged or conserved once NO is produced and metabolized.
The results from this investigation reveal that nebivolol directly affects the stability and accumulation of nitrosothiols in cell culture. Treatment of mice with nebivolol increases the steady-state concentrations of nitrosothiols in the heart as quantified and detected by chemiluminescence and biotin switch assay. These effects are abrogated in eNOS knockout mice demonstrating the source of nitrosothiol formation is from endogenous NO production. Furthermore, the effects of nebivolol lead to a potentiation of vasorelaxation of GSNO over time. Mice treated for 7 days have significant improvement in endothelium-dependent vasorelaxation and are more sensitive to GSNO induced relaxation. Interestingly, eNOS knockout mice were more sensitive to GSNO than treated or untreated C57 mice. This may be due to receptor sensitization since these mice do not produce NO in their endothelium. Nebivolol-treated eNOS knockout mice also appeared to respond better to GSNO.
These data raise important new considerations about the therapeutic potential of nebivolol. It appears from our mouse studies that nebivolol may work best under conditions where there is sufficient endogenous NO production that can lead to secondary formation of nitrosothiols. It is now known that dietary intake of nitrite and nitrate can lead to appreciable formation of nitrosothiols. 31 –33 An important question arises then as to what effects high nitrite and nitrate diets may have on the efficacy of nebivolol. These data provide new insight into a possible new mechanism of action for nebivolol that may partly explain its biological activity. Due to the structurally similarity with other known GSNO reductase inhibitors, part of the mechanism of action of nebivolol may be through the modulation of intracellular nitrosothiols that are indeed partly dependent upon NOS derived NO. These data may allow for additional indications for nebivolol for human conditions known to have altered nitrosothiol formation, stability, or metabolism.
Footnotes
Author Contributions
HJ, DJP, and ZL conducted animal experiments and analyzed data. ACT and KNI performed cell culture experiments and analyzed data. AP and DJP performed organ bath experiments and analyzed data. NSB and DJL designed experiments, interpreted data, and wrote paper. Jiang, H contributed to acquisition and analysis, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; Polhemus, D contributed to acquisition and analysis, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Islam K contributed to acquisition and interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; Torregrossa, A contributed to acquisition and analysis, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Li, Z contributed to acquisition and analysis, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy; Potts, A contributed to acquisition and analysis, critically revised the manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy. Lefer, D contributed to conception and design, contributed to analysis and interpretation, critically revised the manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy; Bryan, N contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: N.S. Bryan and UTHealth have financial interests in Neogenis, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Forest Research Institute, Inc.
