Abstract
Objectives:
We aimed to comprehensively evaluate poly-lactide polymer degradation and sirolimus release kinetics from a drug-eluting stent matrix in the in vivo setting using a nuclear magnetic resonance (NMR) method.
Methods:
In 22 domestic swine, 18 biodegradable polymer-only coated stents (BPSs) and 36 biodegradable polymer-coated sirolimus-eluting stents (BP-SES) were implanted in coronary arteries with 115% overstretch. The animals were sacrificed at 1, 3, 7, 14, 28, and 56 days following baseline procedures. Vessel segments with BPS were harvested to evaluate polymer degradation with a NMR method, whereas BP-SES to analyze sirolimus tissue uptake and retention. Additionally, 8 BP-SES were implanted for histological analysis for 90 days of follow-up.
Results:
The NMR showed a gradual absorption of the polymer over the 6 consecutive time points, from 5.48 µg of the polymer on the stent at 1-day follow-up, through 4.33 µg at 3 days, 3.16 µg at 7 days, 2.42 µg at 14 days, 1.92 µg at 28 days to 1.24 µg in the last day of the study. The curve of polymer degradation corresponds well with the pharmacokinetic profile of sirolimus eluted from its surface and measured at identical time points. In histopathology, at 90 days, complete healing and biocompatibility were reported.
Conclusions:
The utilization of NMR method for BP absorption kinetics evaluation is a useful tool, which may be widely adopted to test other biodegradable implants. Further, it may substantially improve their safety and efficacy by facilitating programmed polymer and drugs elution.
Introduction
Efficacy of drug-eluting stents (DESs) in reducing restenosis and decreasing the risk of revascularization has been shown in numerous clinical trials. 1 –3 Unfortunately, in many cases it has been achieved at the cost of delayed endothelialization, impaired vessel healing, and prolonged inflammatory reactions. 4 –8 Theoretically, these negative consequences of DES might be averted by the use of stents coated with biodegradable polymers (BPs), which can be completely resorbed from the stent surface leaving only bare metal stent behind. Although many experimental and clinical studies of BP-based technologies were performed, they are mostly focused on clinical safety and efficacy. There is still lack of quantitative data showing in vivo degradation kinetics of a BP. Therefore, the aim of this study was a complex evaluation of in vivo BP (poly-lactic acid) absorption and sirolimus release kinetics from the surface of a DES using a nuclear magnetic resonance (NMR) method.
Methods
Device Description
The biodegradable polymer-coated sirolimus-eluting stent (BP-SES; Alex, Balton, Warsaw, Poland) is a balloon expandable stent dedicated for coronary atherosclerotic lesions. The stent platform is made of a laser-cut cobalt–chromium alloy with a wall thickness of 70 μm. It has a closed cell design for uniform drug distribution. The profile of the whole implantation system, including stent, is 0.034 in. The stent is covered with a multilayer structure containing a poly-
Study Design
The experiment was performed in the Center for Cardiovascular Research and Development of American Heart of Poland. The study protocol was approved by the Local Ethics Committee for Animal Research in Krakow (Poland). Study flow chart is presented in Figure 1. Twenty-two male and female, nonatheroslerotic, healthy domestic swine, weighing 30 to 48 kg, were included in the study. In 18 animals, 2 BP-SES and 1 biodegradable polymer-only coated stent (BPS) were implanted into 3 different coronary arteries. Therefore, 36 BP-SES designated for pharmacokinetics analysis and 18 BPS for polymer biodegradation analysis were implanted. The animals were sacrificed at 1, 3, 7, 14, 28, and 56 days of follow-up. Subsequently, arterial segments containing stents were harvested (a total of 6 BP-SES Standard and 3 BPS per time point), stored in liquid nitrogen, and sent for biodegradation and pharmacokinetic analysis. Additionally, 5 unimplanted BPSs were sent for NMR analysis as a reference.

Study design.
It was followed by the complementary histological phase in which 8 BP-SESs were implanted in 4 animals in order to assess tissue effects. Coronary angiography was performed 90 days after baseline procedure. Then animals were sacrificed, all arterial segments were dissected and harvested for pathological analysis. In all animals, identical procedures were used as described previously. 9 Briefly, all animals received dual antiplatelet therapy consisting of oral acetylsalicylic acid (325 mg of initial dose and 150 mg subsequently) and clopidogrel (300 mg of initial dose and 75 mg subsequently), starting 3 days prior to intervention and continuing throughout the study. Percutaneous femoral artery access utilizing the Seldinger technique was used for stents introduction and implantation into the following coronary arteries: (1) right coronary artery and (2) left anterior descending and left circumflex. All stents were implanted under live quantitative coronary angiography guidance at inflation pressures sufficient to ensure a range of balloon to artery ratio of 1.15:1.0 to induce mechanical injury of the vessels. All pigs were anesthetized and prepared in the same fashion as described previously at terminal follow-up to perform control coronary angiography and subsequently humanely sacrificed with pentobarbital overdose. Following vessel harvesting, stented segments were immersed in normal-buffered formalin 10%.
Biodegradation Analysis
Quantitative assessment of degradable polymer coating was performed using a novel, high-resolution, proton nuclear magnetic resonance (1H-NMR) spectroscopy with an internal standard. Before analysis, the stent was placed in 5-mm NMR sample tubes with 500 μL deuterated chloroform (CDCl3) and agitated for 24 hours at room temperature for complete dissolution of polymer coating. As the internal standard, 5 μL of known concentration of lactide was applied. The 1H-NMR spectroscopy was recorded at 600 MHz with AVANCE II Ultra Shield Plus Spectrometer (Bruker BioSpin GmbH, Rheinstetten, Germany) using cryoprobe and CDCl3 as a solvent. The analysis was performed with the following acquisition parameters: 2048 scans, 2.66 seconds acquisition time, and 5.5 μs prescan delay time. Spectroscopy of the following 5 kinds of samples were analyzed: (1) CDCl3, (2) baseline polymer coating of studied stents, (3) baseline polymer coating of studied stents with internal standard, (4) vessel tissue of studied coronaries with internal standard, and (5) polymer coating of stents after 1, 3, 7, 14, 28, and 56 days with internal standard.
Lactidyl unit is a monomeric sequence of polymer obtained in a ring opening polymerization of lactide. The molar mass of monomer and polymeric units is the same, but chemical shift of resonance lines in NMR spectra of lactide and lactidyl unit is different (Figure 2). Therefore, comparison of integration of resonance signals arising from protons of methine groups of lactide at 5.05 ppm and lactidyl units at 5.13 to 5.23 ppm enables for quantitative assessment of polymer coating, which is composed of lactidyl units.

Resonance lines in nuclear magnetic resonance (NMR) spectra.
Calculation of Polymer Mass on a Stent
In the case of lactide/x copolymer with molar ratio 1:1 of comonomers, total mass in μg on stent (mTotal) may be calculated from the following equation:
where mST—mass of internal standard substance, ILL—resonance line intensity in H-1 NMR spectrum of lactidyl units in copolymer (about 5.2 ppm), ILact—resonance line intensity in H-1 NMR spectrum of
Pharmacokinetic Analysis
Sirolimus pharmacokinetic analysis was performed at the Chemistry Department of Mikolai Kopernik University in Torun with the use of standard high-performance liquid chromatography method. The samples were collected at different time intervals according to the study design. Sirolimus extraction from the tissue and stent surface was performed using ethanol solution. After ethanol evaporation, samples containing extracted drug were reconstituted with previously prepared mobile phase. 1100 Liquid Chromatography (Agilent Technologies, Santa Clara, United States) was used for analysis and detection. Separation was achieved using phenomenax C-18 column (150 × 4.60 mm2, particle size: 3.5 µm). The mobile phase containing acetonitrile, methanol, and water (24/50/26%, v/v/v, respectively) was prepared and degassed. Chromatographic separations were performed at 50°C. The flow rate was set to 0.5 mL/min. Ultraviolet detection of rapamycin (sirolimus) was at 278 nm.
Histological Analysis
All segments designated for histological analysis were embedded in glycol methacrylate and cut to obtain approximately 6 μm sections. Subsequently, these sections were stained with hematoxylin and eosin. All sections were analyzed by pathologist. The following standard histomorphometric parameters were calculated: external elastic lamina (EEL) area, internal elastic lamina (IEL) area, lumen area, stent area neointimal area, and area stenosis (AS).
Previously described, semiquantitative scoring system was used to evaluate endothelialization, injury, extent of inflammatory reaction, and fibrin deposits. 10 –12 To evaluate the amount of injury, the following criteria defined by Schwartz et al 10 were utilized: 0 = IEL intact, 1 = IEL lacerated, 2 = media lacerated, and 3 = EEL lacerated. To evaluate the extent of peristrut inflammatory reaction the following grade by Kornowski et al 11 was used: 0 = minimal inflammatory response around strut, 1 = few inflammatory cells around strut, 2 = mild to moderate inflammation, can extend into but do not efface surrounding tissue, and 3 = dense and thick peristrut aggregate of inflammatory cells, effacing surrounding tissue. The following endothelialization score was described as percentage endothelial coverage of the arterial circumference: 0 = <25%, 1 = 25% to 75%, 2 = 76% to 95%, and 3 = complete. The fibrin deposition was assessed as 0—none to focal, 1—mild involving <10% of artery circumference, 2—moderate involving of 10% to 25% artery circumference, and 3—heavy, involving >25% of artery circumference. Each strut in the section was scored, and the mean inflammation and injury score for each section was calculated and reported.
Statistical Analysis
Statistical analyses were performed using GraphPad Prism 5 (GraphPad Software, Inc, USA) software. For continuous data, variance and normality tests (Shapiro-Wilk test) were initially performed. Normally distributed parametric data are expressed as average and standard deviation and as median and interquartile range in cases of skewed distribution. Significance of the differences between 2 groups was tested using the Student t test for normally distributed variables and the Mann-Whitney U test for nonnormally distributed variables. A P value of ≤.05 was considered statistically significant.
Results
Procedural and Postoperative Data
The angiographic data are summarized in Table 1. The overstretch in different time points was comparable. All stents were easily introduced and implanted into selected coronary segments. By visual assessment, the stents kept their dedicated size and shape and conformed well to the anatomy of the target vessels. In 1 coronary segment, margin dissection occurred, which required additional stent implantation—this segment was excluded from further analysis. There were no cases of thrombus formation, slow-flow, or access site complications. During the entire follow-up period, no serious adverse events were noted, and all animals remained in good general condition with steady body weight increase.
QCA Analysis.
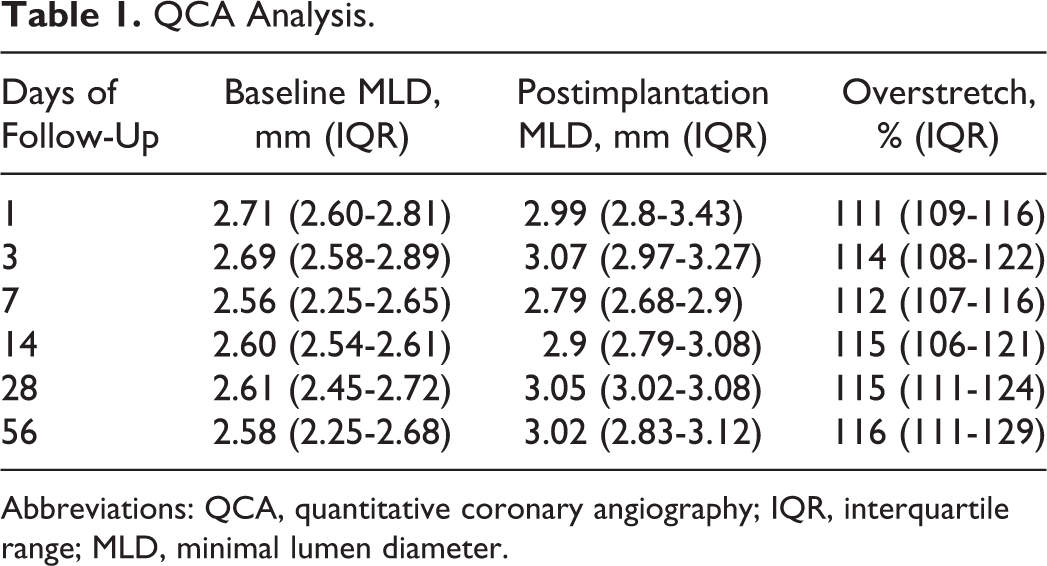
Abbreviations: QCA, quantitative coronary angiography; IQR, interquartile range; MLD, minimal lumen diameter.
Polymer Degradation and Pharmacokinetics
The NMR method showed a gradual absorption of the polymer over the 6 consecutive time points, from 5.48 µg of the polymer on the stent at 1-day follow-up through 4.33 µg at 3 days, 3.16 µg at 7 days, 2.42 µg at 14 days, 1.92 µg at 28 days to 1.24 µg in the last day of the study (Figure 3). Sirolimus was released from the stent over a period of 28 days. At 1 day, mean sirolimus amount on stent surface was 193.73 ± 86.9 ng. This amount decreased gradually to 148.64 ± 105.5 ng at 3 days, 118.77 ± 79.8 ng at 7 days, 35.65 ± 44.9 ng at 14 days, and 9.54 ± 8.0 ng at 28 days. At 56-day follow-up, sirolimus was undetectable on stents surface. There was a good penetration and retention of the drug in the arterial wall. At 1- and 3-day follow-up, sirolimus amount in the tissue was on a similar level of 4.36 (4.1-4.56) and 3.95 (3.72-4.12) ng/mg, respectively. This amount decreased to 2.12 (1.98-2.21) ng/mg at 7 days, 1.02 (0.95-1.1) ng/mg at 14 days, 0.3 (0.25-0.33) ng/mg at 28 days, and 0.28 (0.2-0.32) ng/mg at 56 days (Figure 4). Polymer degradation and pharmacokinetics data are summarized in Table 2.

Polymer degradation and sirolimus release from stent surface over time in porcine coronary arteries.

Sirolimus concentration in coronary artery tissue over time (ng/mg).
Polymer and Sirolimus Mass on a Stent and Sirolimus Concentration in Arterial Tissue in Different Time Points.

Abbreviation: IQR, interquartile range.
Histological Analysis
The histopathological results are summarized in Table 3. Vessel lumen was widely patent without evidence of thrombus and no binary in-stent restenosis. The mean AS at 90 days of follow-up was 42% ± 9% and neointimal area was 3.53 ± 1.08 mm2. Despite high injury score, signs of peristrut inflammation were not detected. The healing process expressed by fibrin deposition and completeness of endothelialization was advanced after 90 days. The endothelialization was finished or almost finished (endothelial coverage more than 75% of the arterial circumference) in 100% of examined cross-sections. In only 2 cross-sections, moderate fibrin depositions were noted. There was no evidence of necrosis (Figure 5).
Histopathological Results of Vessel Morphometry, Biocompatibility, and Healing at 90-Day Follow-Up.
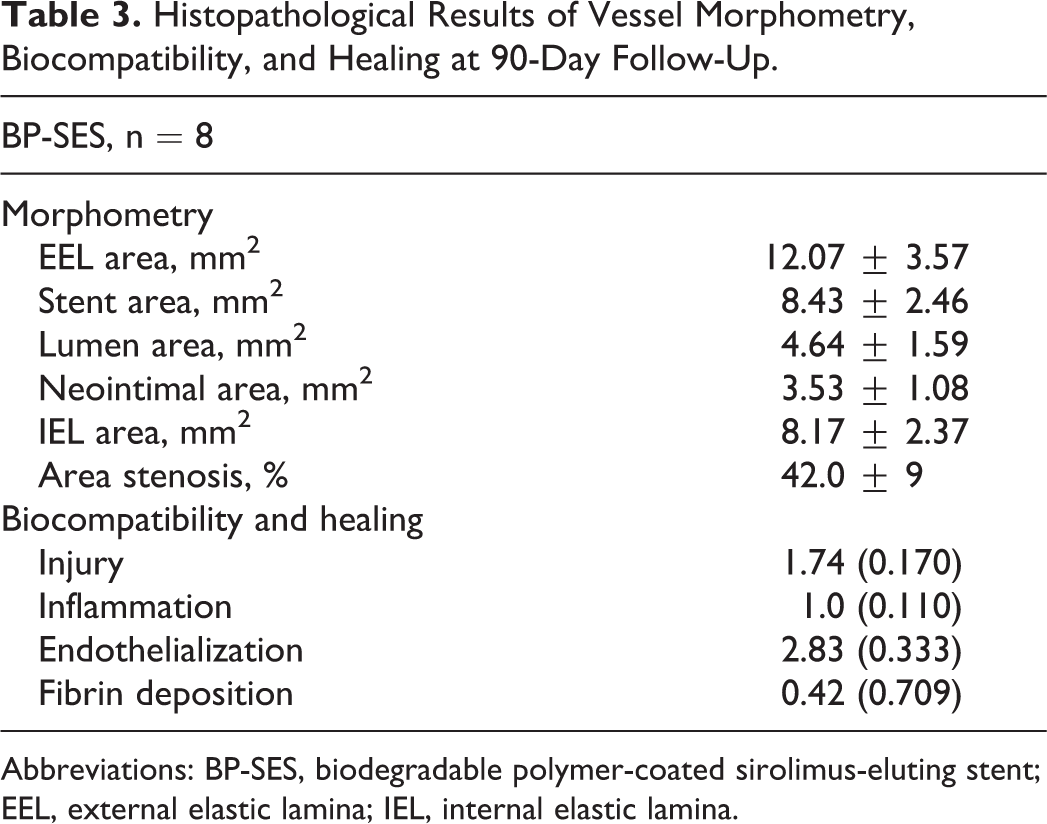
Abbreviations: BP-SES, biodegradable polymer-coated sirolimus-eluting stent; EEL, external elastic lamina; IEL, internal elastic lamina.
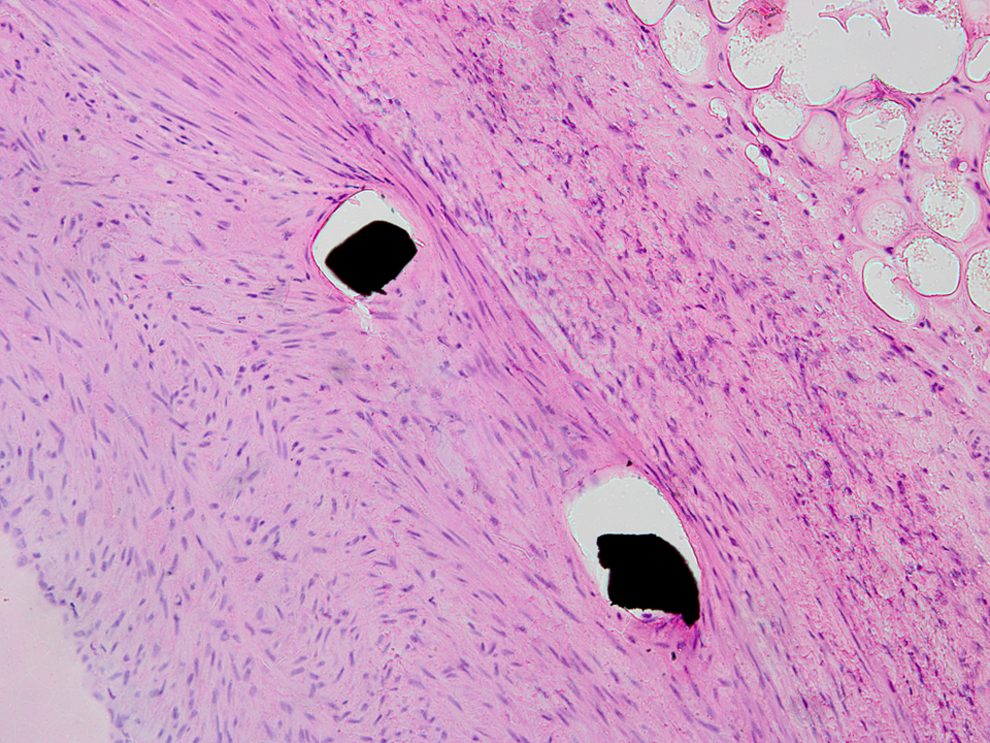
Light microscopy, peristrut 20× magnification picture with no inflammatory reaction. Hematoxylin and eosin staining.
Discussion
Although many BP-based technologies, including vascular scaffolds, 12 –17 are widely used and evaluated in the clinical setting, still there is lack of experimental in vivo data showing degradation kinetics of polymers and their relationship to a drug release pattern. The assessment of polymer degradation in isotonic saline solution (0.9% NaCl) is a common method in ex vivo settings, 18,19 however, it has several drawbacks such as the absence of pulsatile blood flow, artery movement, local pH, and temperature changes as well as lack of tissue enzymes. Moreover, the limited data from in vivo studies were assessed observationally using microscopy and semiquantitative scoring system, evaluating only the presence of polymer on stent surface. 20
To the best of our knowledge, our study represents for the first time the preclinical evaluation of in vivo polymer degradation using a novel quantitative NMR method. This method tracks polymer degradation process in arterial tissue, giving an excellent opportunity to test many polymer-based bioresorbable technologies used in medicine. The major advantage of quantitative NMR is its primary analytical nature. The resonance signals from polymer coating, internal standard, and vessel tissue are well separated without any interference (Figure 2). Therefore, the surrounding tissues do not need to be completely removed, diminishing the risk of polymer loss during preparation procedure.
The tested BP used as a platform for DESs has demonstrated favorable angiographic and clinical results. 15,16 Moreover, we showed that BP-coated paclitaxel-eluting stent (PES) is comparable to the durable polymer-coated PES, with regard to the thrombotic events despite lower compliance to double antiplatelet therapy (DAPT). 17 However, the in vivo evaluation of BP degradation time, which may be important in the decision of shortening DAPT, has been not evaluated.
In the present study, the NMR spectroscopy showed relatively fast and predictable biodegradation kinetics with 9% of baseline polymer mass after 14 days. In following weeks, there was a gradual decrease in polymer mass on a stent. Similarly, the sirolimus release profile demonstrated approximately 95% elution within first 2 weeks and the drug mass on a stent declined below the lower limit of detection by 56 days after stent deployment. Despite the stent delivered its load of sirolimus relatively early, the drug retained in arterial tissue for 56 days. This elution profile of sirolimus is faster than previously reported for the Cypher stents, where approximately 70% of drug was released within the first 2 weeks, while remaining amount was released by 90 days. 21
There are conflicting results with regard to differences in in vivo and in vitro degradation. Lu et al demonstrated a faster in vivo than in vitro degradation rate, 22 while others did not observe any differences. 23 However, contrary to our study, in these studies, the BP was implanted in the mesentery or subcutaneous tissue.
The favorable healing profile of tested stents was confirmed. There were no signs of peristrut inflammation and foreign body giant cells, thus proving absence of polymer on struts at 90 days and presence of fully biocompatible stainless steel. Slightly delayed endothelialization and signs of fibrin deposition are typical footprints of sirolimus. 24
Although in the present study we demonstrated degradation of BP from the surface of metal stent, NMR spectroscopy can be widely adopted to test other biodegradable applications. Absorption kinetics of completely bioresorbable, lactide–polymer-based scaffolds can also be evaluated by NMR method. Besides stent technologies, poly-lactide polymer is approved for use in numerous clinical situations ranging from absorbable sutures, orthopedic plates, and screws. Preclinical studies utilizing simultaneous NMR and histology analysis may substantially improve safety and efficacy of bioresorbable devices by facilitating programmed polymer degradation. Furthermore, this method may facilitate development of novel intravascular technologies evaluating poly-
Study Limitations
There are several limitations of this study. First, the animals with healthy coronary arteries were used. The lack of the biological components seen in atherosclerotic plaque neglects their potential impact on polymer degradation and drug release. Therefore, the results found in our studies may not correlate with the diseased vessels in humans. Furthermore, the significant variability observed in pharmacokinetic studies using animal models suggests the possibility of similar effect according to polymer degradation. A study with higher number of animals is required to investigate foregoing issue.
Conclusions
The novel NMR method for BP absorption kinetics evaluation is a useful and feasible tool, which may be widely adopted to test other intravascular BP applications consisting of poly-
Footnotes
Author Contribution
B. Orlik contributed to acquisition and interpretation, drafted the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. P. P. Buszman contributed to interpretation, drafted the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. J. Pająk contributed to analysis and interpretation, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. A. Krauze contributed to acquisition, drafted the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. P. Gąsior, P. Desperak, and G. Handzlik-Orlik contributed to analysis, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. A. Janas and M. Jelonek contributed to acquisition, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. J. Kasperczyk contributed to conception and design, contributed to acquisition, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. P. P. Buszman and K. Milewski contributed to conception and design, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported with an unrestricted grant by the Balton Company, Warsaw, Poland.

