Abstract
Introduction:
The differential response of atrial and ventricular cells to late sodium channel current (late INa) inhibition has not been thoroughly investigated. The aim of the present study was to compare the atrioventricular differences in electrophysiological actions of GS-458967, a potent late INa blocker.
Methods and Materials:
Canine coronary-perfused atrial and ventricular preparations and isolated ventricular myocytes were used. Transmembrane action potentials were recorded using standard microelectrode recording techniques.
Results:
In coronary-perfused preparations paced at a cycle length (CL) of 500 ms, GS-458967 (100-300 nmol/L) significantly abbreviated action potential duration at 50% to 90% (APD50-90) in atria but not in the ventricles. GS-458967 (≥100 nmol/L) prolonged the effective refractory period (ERP) in atria due to the development of postrepolarization refractoriness (PRR) but did not alter ERP in the ventricles. The maximum rate of rise in the action potential upstroke (Vmax) was significantly reduced at concentrations ≥100 nmol/L in atria but not in the ventricles (CL = 300 ms). At slower pacing rates (CL = 2000 ms) and higher concentrations, GS-458967 (100-1000 nmol/L) still failed to abbreviate ventricular APD. However, when APD was prolonged by the rapidly activating delayed rectifier potassium channel blocker E-4031 (1 µmol/L), addition of 1 μmol/L GS-458967 abbreviated APD in the ventricles at slow rates. In contrast, GS-458967 (300 nmol/L) consistently abbreviated APD in untreated isolated ventricular myocytes.
Conclusion:
In canine coronary-perfused preparations, GS-458967 abbreviates APD, induces PRR, and reduces Vmax in atria but has no significant effect on these parameters in the ventricles, indicating an atrial-selective effect of GS-458967 on both peak and late INa-mediated parameters. In multicellular preparations, GS-458967 abbreviated ventricular APD only under long QT conditions, suggesting a pathology-specific action of GS-458967 in canine ventricular myocardium.
Introduction
The electrophysiology and pharmacology of late sodium channel current (INa) have been studied to a much greater extent in the ventricles than in atria. 1 –5 Studies involving ranolazine, a mixed ion channel blocker with preferential inhibition of late INa, have greatly advanced our understanding of the role of this current under physiological and pathophysiological conditions. 2 –6 Recent studies report major atrioventricular differences in the electrophysiological effects of ranolazine, showing atrial-selective depression of peak INa and peak INa-mediated parameters. 7,8 Similar atrial-selectivity has been reported for a number of other sodium channel blockers, including amiodarone and vernakalant. 9 –12 The extent to which the inhibitory effect of these drugs toward late INa is atrial selective is poorly delineated.
GS-458967 is a recently introduced highly selective late INa blocker. 13 The present study was designed to directly compare electrophysiological effects of GS-458967 in coronary-perfused canine atrial and ventricular preparations.
Methods
All experiments were carried out in compliance with the Guide for Care and Use of Laboratory Animals published by the National Institutes of Health (NIH publication No 85-23, Revised 1996) and approved by the Institutional Animal Care and Use Committee. Dogs weighing 20 to 25 kg were anticoagulated with heparin and anesthetized with pentobarbital (30-35 mg/kg, intravenous [iv]). The chest was opened via a left thoracotomy, the heart excised, placed in a cardioplegic solution consisting of cold (4°C) Tyrode solution containing 12 mmol/L [K+]o, and transported to a dissection tray.
Arterially Perfused Canine Right Atrium
The entire right atrium (RA) with a thin rim of right ventricular (RV) tissue was dissected from the isolated dog heart and then the preparation was unfolded. The ostium of the right coronary artery was cannulated with polyethylene tubing (inner diameter, 1.75 mm; outer diameter, 2.1 mm), and the preparation was perfused with cold Tyrode solution (12°C-15°C) containing 8 mmol/L [K+]o. Ventricular right coronary branches and the cut atrial branches were ligated using silk thread. The preparation was placed in a temperature-controlled bath (8 × 6 × 3 cm3) and perfused with Tyrode solution (37.0°C ± 0.5°C).
Arterially Perfused Canine Left Ventricular and RV Wedge
Transmural wedges with dimensions of approximately 3 × 1.5 × 1.5 cm3 were dissected from the anterior-apical aspects of the left ventricle (LV) or anterior-basal part of RV. The tissues were cannulated and perfused with cardioplegic solution. The preparations were then placed in a temperature-controlled tissue bath and perfused with Tyrode solution (37.0°C ± 0.5°C).
For both atrial and ventricular preparations, the composition of the Tyrode solution was the following (in mmol/L): NaCl 129, KCl 4, NaH2PO4 0.9, NaHCO3 20, CaCl2 1.8, MgSO4 0.5, and
Transmembrane action potential (AP) recordings were obtained using the floating microelectrode technique (10-25 MΩ direct current [DC] microelectrode resistance) connected to a high input impedance intracellular amplifier (World Precision Instruments, Sarasota, Florida). The signals were further amplified and digitized (sampling rate = 40 kHz) using Spike2 acquisition system (Cambridge Electronic Design, Cambridge, England).
An electrocardiogram (pseudo-ECG) was recorded using 2 electrodes consisting of AgCl half cells placed in the bath solution 1.0 to 1.5 cm from the opposite ends of the atrial and ventricular preparations.
Effective refractory period (ERP) was measured by delivering premature stimuli after every 10th regular beat at a pacing cycle length (CL) of 500 (with 5-10 ms resolution; stimulation with twice diastolic threshold of excitation (DTE) amplitude).
Postrepolarization refractoriness (PRR) was defined as the difference between ERP and AP duration at 75% (APD75) in atria and APD90 in ventricles (ERP corresponds to APD70-75 in atria and APD90 in ventricles). 7
Maximum Rate of Rise in the AP Upstroke
Stable AP recordings and maximum rate of rise in the AP upstroke (Vmax) measurements are difficult to obtain in vigorously contracting perfused preparations. A large variability in Vmax measurements is normally encountered at any given condition, primarily due to variability in the amplitude of phase 0 of the AP. The effects of GS-458967 on Vmax were determined by comparing the changes in Vmax values upon acceleration from a CL of 500 to 300 ms. Due to a substantial interpreparation variability, Vmax values were normalized for each experiment and then averaged.
Experimental Protocols
Coronary-perfused RA and LV/RV preparations were equilibrated in the tissue bath until electrically stable (30-120 min). The RA beat spontaneously and LV/RV preparations were paced using a pair of thin silver electrodes insulated except at their tips (bipolar rectangular pulses of 2-millisecond duration and twice DTE intensity). Electrophysiological parameters were recorded under baseline conditions and after the addition of progressively higher concentrations of GS-458967 to the coronary perfusate (10, 30, 100, 300, and 1000 nmol/L). A period of at least 20 to 30 minutes was allowed for each concentration of GS-458967 to act before the start of data collection. In a subset of the experiments with LV and RV wedge preparations, E-4031 (1 µmol/L; a specific rapidly activating delayed rectifier potassium channel [IKr] blocker) was added to the coronary perfusate first, followed by addition of progressively higher concentrations of GS-458967 (300 and 1 μmol/L) to the perfusate containing E-4031.
Ventricular-Isolated Myocytes
Single myocytes were obtained by enzymatic dissociation from coronary-perfused ventricular wedge preparations of LV free wall as described previously. 14 Cells from the epicardial, M cell, and endocardial regions of the LV were used in the study. The composition of external solution was (in mml/L) as follows: 140 NaCl, 4 KCl, 2 CaCl2, 1 MgCl2, 10 glucose, and 10 HEPES. Transmembrane AP recordings were obtained using high-resistance microelectrode technique (40-60 MΩ DC microelectrode resistance).
Chemicals
GS-458967, synthesized at Gilead Sciences, Inc (Foster City, California), is a triazolopyridine derivative, 6-(4-(trifluoromethoxy)phenyl)-3-(trifluoromethyl)-[1,2,4]triazolo[4,3-a] pyridine, with a molecular weight of 347.
Drugs
GS-458967 (Gilead Sciences, Inc, dissolved in 100% DMSO) and E-4031 (Enzo Life Sciences, Inc, Farmingdale, New York; dissolved in distilled water) were prepared fresh before each experiment (stock solution = 0.3 or 1 mmol/L for GS-458967 and 5 mmol/L for E-4031).
Statistics
Statistical analysis was performed using a Student t test or 1-way analysis of variance for paired or unpaired data, as appropriate. All data are expressed as mean ± standard deviation.
Results
Electrophysiological Effects of GS-458967 in Atria and Ventricles
In the initial experimental series, we compared the effect of GS-458967 (10-300 nmol/L) in arterially perfused atrial and ventricular preparations paced at a CL of 500 ms (approximating the resting heart rate in canine in vivo). At a pacing CL of 500 ms, GS-458967 (100-300 nmol/L) significantly abbreviated APD50-90 in atria but did not significantly alter APD in the ventricles (Figure 1). Despite its effect to abbreviate APD in atria, GS-458967 (100-300 nmol/L) prolonged atrial ERP secondary to the development of PRR (Figure 2). GS-458967 did not affect ERP or induce PRR in the coronary-perfused ventricular wedge (Figure 2). In the presence of GS-458967 (100 and 300 nmol/L), acceleration of pacing rate from a CL of 500 to 300 ms caused a significant use-dependent reduction in Vmax in the atrium but not in the ventricle (Figure 3). The shortest S1-S1 permitting 1:1 activation was significantly increased in atria but not in the ventricles (Figure 4).

GS-458967 abbreviates action potential duration (APD) in the atria but causes little change in APD in the ventricles at a pacing cycle length (CL) of 500 ms. Upper panels: shown are superimposed action potentials recorded in atrial and ventricular preparations under baseline condition (control) and following the addition of 100 and 300 nmol/L of GS-458967. Bottom panels: summary data of the effect of GS-458967 on APD at 50% repolarization (APD50) and APD at 90% repolarization (APD90) in atria and ventricles. Pacing CL = 500 ms. *P < .05 versus respective control (n = 8-10). CT indicates crista terminalis; PM, pectinate muscle; Epi, epicardium.
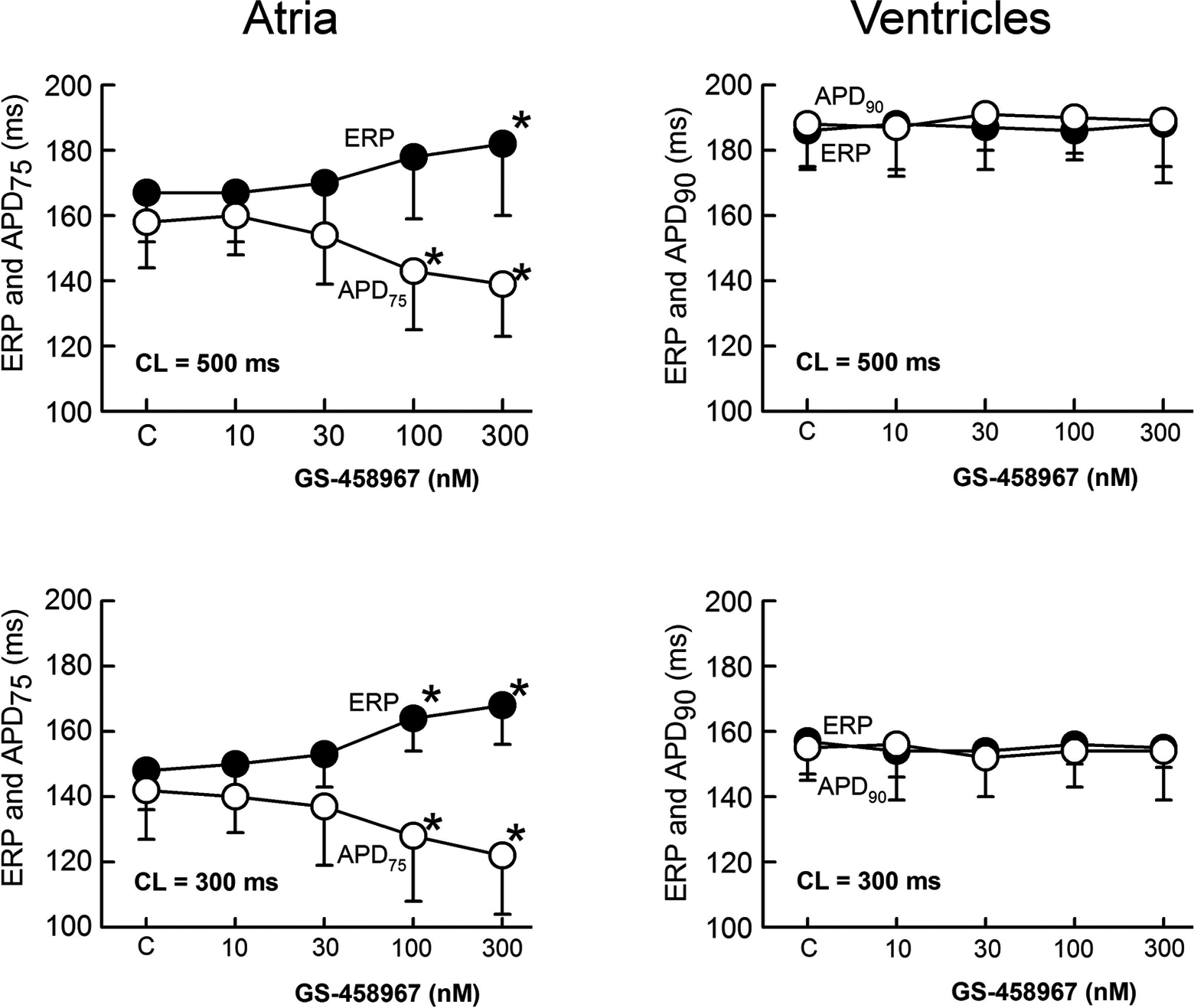
Atrial-selective induction of postrepolarization refractoriness (PRR) by GS-458967. PRR, estimated by the difference between ERP and action potential duration at 75% repolarization (ADP75) in atria and APD at 90% repolarization (ADP90) in ventricles, increases with increasing concentrations of GS-458967 in atria but not in the ventricles. *P < .05 versus respective control (n = 7-8). Atrial data are from the pectinate muscle region of the right atrium. Ventricular ERP was measured in the endocardial surface and APD90 is from the subendocardial region (ie, M cell region).

Atrial-selective use-dependent depression of the maximum rate of rise of the action potential upstroke (Vmax) in the presence of GS-458967. Left panel: representative atrial and ventricular action potentials and accompanying Vmax tracings recorded following acceleration of pacing rate from a CL (cycle length) of 500 to 300 ms. Right panel: summary data expressed as percentage of V value at 500 ms following acceleration of pacing rate from a CL of 500 to 300 ms. *P < .05 versus Vmax change in control (n = 8-10).

GS-458967 significantly increases the shortest S1-S1 permitting 1:1 activation in atria but not in ventricles. The shortest S1-S1 was measured using 2 × diastolic threshold intensity (as determined at a cycle length [ CL] of 500 ms). *P < .05 versus control (n = 4-9).
Because the effects of late INa inhibition on APD are most evident when APD is prolonged, 3 –5 additional experiments were performed in ventricular wedge preparations paced at slow rates. A higher concentration of GS-458967 (1 μmol/L) was tested as well. At a CL of 500 to 2000 ms, GS-458967 (300-1000 nmol/L) produced no significant effect on APD of subendocardial M cells, the cells traditionally displaying the longest APD 15 (Figure 5). When ventricular APD was further prolonged using E-4031 (mimicking long QT type 2 syndrome), addition of GS-458967 (1 μmol/L) significantly abbreviated APD in the M cell region of the ventricular wedge preparation at a CL of 2000 ms (Figure 5). At a CL of 500 milliseocnds, GS-458967 (300-1000 nmol/L) did not significantly abbreviate ventricular APD (207 ± 13 ms in the presence of E-4031 and 204 ± 17 ms following 1 μmol/L GS-458967).

GS-458967 does not significantly affect repolarization in control ventricles but abbreviates it in a long QT2 model (in the presence of E-4031). Upper panel: superimposed action potentials recorded from M cell regions of left ventricle wedge preparations before and after progressively higher concentrations of GS-458967 (300 and 1000 nmol/L) at a pacing cycle length (CL) of 2000 ms. Data from 2 different preparations are shown. Bottom panel: summary effect of GS-458967 on action potential duration at 90% APD90 in subendocardial M cell regions (combined data from left and right ventricular wedge preparations) at a pacing CL of 2000 ms. *P < .05 versus control (n = 4-5).
In contrast to the lack of effect of the drug in untreated ventricular multicellular preparations (Figure 5, upper left panel), GS-458967 (300 nmol/L) consistently and significantly abbreviated APD when studied in myocytes isolated from the epicardial, M cell, and endocardial regions of the LV, with greater APD shortening of the cells isolated from the M region (Figure 6).

GS-458967 consistently shortens action potential duration (APD) in isolated single ventricular myocytes. Upper panel: superimposed action potentials recorded from isolated left ventricular midmyocardium cell (M cell) myocytes before (control) and after addition of 300 nmol/L of GS-458967 at a pacing cycle length (CL) of 2000 ms. Bottom panels: summary data of the effect of GS-458967 on APD at 90% (APD90) in isolated ventricular myocytes at pacing CLs of 500 and 2000 ms. *P < .05 versus control (n = 4-8). Epi indicates epicardium; Endo, endocardium.
Discussion
The principal finding of the present study is that in multicellular preparations, GS-458967 causes APD abbreviation, ERP prolongation, and a reduction in Vmax in atria but not in the ventricles, pointing to atrial-selective effects of GS-458967 on INa-mediated parameters. In the ventricle, GS-458967 leads to abbreviation of APD only under long QT conditions, suggesting a pathology-specific action of GS-458967 in canine ventricular myocardium. Although a greater sensitivity of atria than of ventricles to peak INa block by sodium channel blockers has been previously reported, 7,8,11,12 an atrial-selective response to the inhibition of late INa is a novel finding.
Is GS-458967an Atrial Preferential Peak and Late INa Inhibitor?
GS-458967 is reported to be a highly selective late INa inhibitor when studied in isolated ventricular myocytes. 13,16 The half maximal inhibitory concentration (IC50) for GS-458967 block of endogenous late INa is 0.46 µmol/L in guinea pig ventricular myocytes 16 and of ATX-II-augmented late INa is 0.13 ± 0.01 µmol/L in rabbit ventricular myocytes. 13 Only 7.5% of peak INa and 16% of IKr are inhibited by 10 µmol/L of GS-458967 in rabbit ventricular myocytes. Moreover, GS-458967 does not significantly inhibit ICa-L, ICa-T, IK-ATP, and INa-Ca (at 1-3 µmol/L). 13 Of note, the potency of GS-458967 to block peak INa was measured at a CL of 0.1 Hz and holding potential of −120 mV, which may significantly underestimate its effect under physiological conditions. Under these voltage clamp conditions, 285 to 1329 µmol/L of ranolazine has been reported to cause a 50% reduction (IC50) in peak INa, 6,13 whereas at faster rates and “physiological” holding potentials, IC50 is less than 10% of the lower value. 8 The QRS interval, an peak INa-dependent parameter, was not affected by 3 µmol/L of GS-486967 at pacing rates of 1 to 4 Hz in isolated rabbit heart, indicating lack of effect of this compound in rabbit ventricles at physiologically relevant heart rates. 13
Atrial-selective reduction in Vmax, induction of PRR, and an increase in the shortest S1-S1 interval by GS-458967 in our study are consistent with atrial-selective block of peak INa by this agent. Atrial selectivity of peak INa block has been described for sodium channel blockers that dissociate rapidly from the sodium channel including ranolazine, amiodarone, dronedarone, vernakalant, AZD1305, AVE0118, and Wenxin Keli. 7,9 –12,17 –21 The factors underlying the atrial-selective effects of INa blockers include a more negative steady state inactivation relationship, a less negative resting membrane potential, and a more gradual phase 3 of the AP in atrial versus ventricular cells as well as a relatively rapid dissociation of the drug from the sodium channel. 7,8,22 GS-458967 dissociates from sodium channels (HEK 293 and atrial myocytes) rapidly with a time constant of 300 to 500 ms (holding potential of −120 mV at 23°C), a value approaching that of lidocaine (L. Belardinelli et al, PhD unpublished observation, 2014). Because GS-458967 has rapid unbinding kinetics and considering the fact that all INa blockers inhibit both peak and late INa, commonly with a higher potency for late versus peak INa inhibition, atrial-selective block of late INa by GS-458967 is not unexpected.
GS-458967-induced APD abbreviation in atria but not in ventricles could be due to atrial-selective inhibition of late INa. In support of this hypothesis, ranolazine inhibits sea anemone toxin-enhanced late INa more effectively in atrial versus ventricular myocytes. 23 The atrial-selective abbreviation of APD could also be due to atrioventricular differences in other factors, including AP morphology, late INa density, input resistance, and cellular coupling. Maximum late INa density recorded under voltage clamp conditions has been reported to be greater in atrial versus ventricular cells in one study, 23 but greater in ventricular versus atrial in another (G.X. Yan MD, PhD, and L. Belardinelli PhD, unpublished observation, 2013), and of similar density in a third study. 24 A greater density of late INa in atria is expected based on the finding that peak INa density is much greater in canine atrial versus ventricular cells under identical recording conditions. 7,25 The voltage dependence of late INa peaks at about −20 mV14 approximating the voltage of the AP plateau in atrial but not ventricular cells (Figure 1). Thus, the contribution of late INa during the AP plateau might be greater in atrial versus ventricular cells. A greater input resistance in atria likely contributes to the greater effect of GS-458967 on the APD of atrial cells. 26 –28 Moreover, membrane resistance of single isolated myocytes is much higher than that of cells that are well coupled to each other in a multicellular preparation. These findings can explain the effect of GS-458967 to abbreviate APD in isolated ventricular myocytes but not in multicellular preparations (Figures 1 and 6). Note that isolated single myocytes and tissue slices are generally more responsive than isolated coronary-perfused cardiac preparations to agents that either prolong or abbreviate APD. 29
Atria have an intrinsically greater level of fibrosis than ventricles, 30 contributing to reduced cell to cell coupling. Cellular uncoupling is known to amplify the effect of drugs that prolong or abbreviate APD. 29,31 It is expected that GS-458967 would shorten APD in structurally compromised ventricles.
A greater effect of GS-458967 on M cell versus epicardial and endocardial myocytes (Figure 6) is likely due to a larger late INa density in the M cells. 14 The effect of GS-458967 to abbreviate APD is reported to be prominent in ventricular myocytes isolated from guinea pig 16 but not from rabbit. 13 GS-458967 (1 µmol/L) causes only a small statistically nonsignificant APD50 abbreviation in rabbit isolated ventricular myocytes (−5.9%, at a pacing CL of 1000 ms). 13 The effect of GS-458967 (30-300 nmol/L) to abbreviate APD in Purkinje fibers at all pacing rates 32 may be due to its intrinsically longer APD, relatively high level of late INa and higher input resistance. 33 GS-458967 induced abbreviation of APD in Purkinje fibers but not in ventricular muscle, thus reducing dispersion of repolarization at the Purkinje–muscle junction, which could diminish arrhythmic potential when this parameter is augmented.
The atrioventricular differences in the effect of GS-458967 on APD are not observed with ranolazine. 7 This is likely due to the fact that ranolazine blocks both the depolarizing late INa and repolarizing IKr. 2 Inhibition of IKr causes a greater APD prolongation in atria versus ventricles at normal pacing rates. 9 However, like GS-458967, ranolazine produces atrial-selective depression of Vmax- and INa-dependent parameters. 7,34
It remains to be determined whether other ion channel-specific sodium channel blockers can produce atrial-selective APD abbreviation. The specific INa blocker lidocaine, at concentrations that significantly reduce peak INa in atria and ventricles, abbreviates APD in both ventricular and atrial multicellular preparations. 7,35 Unlike ranolazine, lidocaine is a sodium channel blocker with relatively poor atrial selectivity. 7
Pathology-Specific Effect of GS-458967 in Ventricular Muscle
Our results suggest that the effect of late INa block in the ventricle is enhanced under conditions in which APD is significantly prolonged, suggesting a pathology-specific role for late INa inhibition in cardiac diseases in which APD is prolonged, including heart failure, long QT syndromes, and hypertrophic cardiomyopathy. 5 Heart rate can significantly affect the contribution of late INa in cardiac electrophysiology. The magnitude of late INa is reverse-rate dependent, that is, the faster the rate the smaller is the magnitude of this current. 2,14,36 Late INa blockers commonly produce significant APD abbreviation in ventricular multicellular preparations only when APD is significantly prolonged (eg, at slow rates and/or in the presence of IKr blockers or late INa augmenters), 2,4,13,37 consistent with the effect of GS-458967 in the present study.
Limitations of the Study
Our experiments were performed in isolated coronary-perfused atrial and ventricular preparations perfused with Tyrode solution or in isolated single-ventricular myocytes. These preparations lack autonomic influences and other factors present in vivo, which could influence our results. The atrial selectivity of GS-458967 was determined acutely in “healthy” cardiac preparations. Clinical cardiac arrhythmias are normally associated with pathology-mediated electrical and structural abnormalities, which may significantly modulate pharmacological response and antiarrhythmic efficacy. Thus, the pathology specificity of the drug’s effect remains to be more fully explored. We cannot totally dismiss the possibility that GS-458967 affects other currents in ventricular myocardium that may prevent the expected shortening of APD in response to its inhibition of late INa. Lidocaine and mexiletine, specific INa blockers, are reported to cause small but significant abbreviation of APD in canine coronary-perfused and superfused tissue slice ventricular preparations in control (nonlong QT) setting, 7,35 and this effect is most likely due to inhibition of late INa.
Conclusions
Previous studies have shown that the biophysics and pharmacology of peak INa differ between atrial and ventricular muscle cells. 7,8 The results of the present study demonstrate atrial-selective effects of GS-458967 on both peak and late INa-dependent parameters in canine cardiac preparations. Interestingly, in multicellular ventricular myocardial healthy preparations, the effect of GS-458967 to abbreviate APD is apparent in atria and Purkinje fibers but not in the ventricles. GS-458967 abbreviates APD in canine multicellular ventricular myocardium only under long QT conditions, suggesting a pathology-specific effect of the drug.
Footnotes
Author Contributions
A. Burashnikov contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. J.M. Di Diego contributed to design, acquisition and analysis, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. R.J. Goodrow contributed to acquisition and analysis, critically revised the article, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. L. Belardinelli contributed to conception and design, analysis and interpretation, critically revised the article, and gave final approval. C. Antzelevitch contributed to conception and design, analysis and interpretation, drafted the article, critically revised the article, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Acknowledgment
We gratefully acknowledge the expert technical assistance of Judy Hefferon.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Antzelevitch receives research support and is a consultant to Gilead Sciences, Inc. Dr Belardinelli is an employee of Gilead Sciences, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by grants from Gilead Sciences; HL 47678 from the National Institutes of Health (CA); NYSTEM C026424 (CA); Grant-in-Aid from the American Heart Association 10GRNT4210016 (JMDD); and Masons of New York, Florida, Massachusetts, Connecticut, Rhode Island, Wisconsin, and Maryland.
