Abstract
Anacetrapib is a cholesteryl ester transfer protein (CETP) inhibitor that has previously been shown to reduce low-density lipoprotein cholesterol (LDL-C) and raise high-density lipoprotein cholesterol (HDL-C) in patients with or at high risk of coronary heart disease in the 76-week, placebo-controlled, Determining the Efficacy and Tolerability of CETP Inhibition with Anacetrapib (DEFINE) trial. Here, we report the results of the 2-year extension to the DEFINE study where patients (n = 803) continued on the same assigned treatment as in the original 76-week study. Treatment with anacetrapib during the 2-year extension was well tolerated with a safety profile similar to patients on placebo. No clinically important abnormalities in liver enzymes, blood pressure, electrolytes, or adverse experiences were observed during the extension. At the end of the extension study, relative to the original baseline value, anacetrapib reduced Friedewald-calculated LDL-C by 39.9% and increased HDL-C by 153.3%, compared to placebo. The apparent steady state mean plasma trough concentration of anacetrapib was ∼640 nmol/L. Geometric mean plasma concentrations of anacetrapib did not appear to increase beyond week 40 of the 2-year extension of the 76-week DEFINE base study. In conclusion, an additional 2 years of treatment with anacetrapib were well tolerated with durable lipid-modifying effects on LDL-C and HDL-C.
Introduction
Cholesteryl ester transfer protein (CETP) inhibitors are experimental agents that raise high-density lipoprotein cholesterol (HDL-C) levels by inhibiting the 2-way transfer of cholesteryl esters and triglycerides between HDL and apolipoprotein B-containing lipoproteins. 1 Some CETP inhibitors also lower low-density lipoprotein cholesterol (LDL-C) through an unknown mechanism. Anacetrapib is a potent and selective CETP inhibitor in phase III development. The 76-week, placebo-controlled, Determining the Efficacy and Tolerability of CETP Inhibition with Anacetrapib (DEFINE) study evaluated the effects of anacetrapib versus placebo on safety and lipid parameters in patients with coronary heart disease (CHD) or at high risk of CHD, who were treated with statins ± other lipid-modifying agents. 2 The trial revealed no differences in blood pressure, electrolytes, and other safety parameters between the anacetrapib and the placebo treatment groups. Anacetrapib treatment was associated with a placebo-adjusted 39.8% reduction in LDL-C (the primary efficacy end point), as calculated by the Friedewald equation, and a 138.1% increase in HDL-C. A subsequent analysis showed that compared to the reference β-quantification method, the Friedewald equation underestimates LDL-C levels after treatment with anacetrapib, so that the actual degree of LDL-C reduction may have been less than originally reported. 3 A 2-year extension to the DEFINE study was conducted to further evaluate long-term safety, lipid effects, and anacetrapib drug concentrations. The current report describes the results of the DEFINE extension study.
Methods
Study Design
The design and results of the DEFINE base study (clinicaltrials.gov number, NCT00685776), a 76-week, worldwide, multicenter, randomized, double-blind, placebo-controlled trial, have been previously reported. 2,4 During the active treatment phase, patients were randomized to anacetrapib 100 mg/d or placebo, and plasma samples for anacetrapib drug concentrations were collected at the 12, 24, and 76 week visits. Following this base study, there was an off-drug reversibility period of at least 12 and up to 24 weeks duration. 5 A total of 1211 patients completed the base study including the reversibility period (n = 548 on anacetrapib, n = 663 on placebo). 2 The proportion of patients completing the base study was lower in the anacetrapib group than in the placebo group because 18% of patients in the anacetrapib group (n = 142) were discontinued due to protocol-specified criterion when the achieved LDL-C was <25 mg/dL. 2
Patients were eligible to enter the DEFINE 2-year extension study only if they had completed at least 12 and up to 24 weeks of the off-drug reversibility phase at the time the extension study was implemented at their site. Patients who had previously completed the base study, including the off-drug reversibility phase, prior to the implementation of the extension study were not eligible.
In the DEFINE 2-year extension study, patients who had been randomized to treatment with anacetrapib 100 mg/d in the base study resumed treatment with anacetrapib 100 mg/d, while patients previously assigned to placebo resumed placebo. Patients were required to stay on their statin ± lipid-modifying therapy and to maintain their recommended course of diet and exercise throughout the study. There were 8 scheduled clinic visits during the 2-year treatment period to collect blood samples and assess adverse events. Lipid-lowering medications were adjusted if LDL-C levels were above 115 mg/dL as calculated by the Friedewald equation. The 2-year treatment period (extension weeks 0-104) was followed by a poststudy follow-up phone call 12 weeks after early discontinuation or completion of the extension study to query for serious adverse experiences. Patients who discontinued early were also contacted by phone 2 years after entering the extension study (intended extension week 104 visit) to assess for serious cardiovascular adverse events or death.
End Points
Lipid measures included LDL-C (Friedewald-calculation method), HDL-C, non-HDL-C, apolipoprotein B, apolipoprotein A-I, triglycerides, and total cholesterol. Safety variables included adverse experiences, blood pressure, serum electrolytes (potassium, sodium, chloride, and bicarbonate), and hepatic and muscle-related enzyme levels. The prespecified cardiovascular composite end point used to evaluate cardiovascular safety included death from cardiovascular causes, nonfatal myocardial infarction, nonfatal stroke, and hospitalization for unstable angina. An independent Safety Monitoring Committee and an external, independent adjudication committee that had monitored the DEFINE base study also monitored patient safety and clinical events in the extension study, including adverse events.
To determine the time to achieve apparent steady state plasma concentrations of anacetrapib, plasma samples were collected at the beginning of the 2-year extension study (ie, after a 12- to 24-week off-drug period) and then at the weeks 40, 56, 72, 88, and 104 visits during the 2-year extension. Since plasma levels for anacetrapib were available at weeks 12 and 24 during the base study, samples from time points prior to 40 weeks were not collected during the extension study. The interval between dosing and pharmacokinetic sampling was noted for all visits and based on the plasma concentration profile of anacetrapib after multiple-dose studies (unpublished data); samples collected between 19.4 and 40 hours postdose were considered to be trough values.
Statistical Analysis
For all lipid parameters, the durability of the treatment effects of anacetrapib was assessed by examining mean percentage change from baseline estimates and their corresponding confidence intervals, based upon the same repeated measures in the data analysis model utilized in the base study. The All-Patients-as-Treated population was employed for safety analyses. Patient counts were provided for alanine transaminase and/or aspartate transaminase measurements ≥3 × upper limit of normal (ULN) on 2 consecutive occasions, creatine kinase elevations ≥10 × ULN, creatine kinase elevations ≥10 × ULN with muscle symptoms, elevations in sodium, chloride, or bicarbonate levels above the ULN, reductions in potassium levels below the lower limit of normal, myalgia, and rhabdomyolysis, prespecified adjudicated cardiovascular adverse events, death from any cause, and predefined changes in systolic and diastolic blood pressure elevations; these safety evaluations were identified as Tier 1 events in the base study. No formal inferential testing was performed for these safety end points in the extension. The 95% confidence intervals for between-treatment differences in the percentage of patients with Tier 1 events were calculated using the Miettinen and Nurminen method. 6
Individual trough concentration values collected in the 2-year extension study were natural log-transformed and evaluated with a linear mixed-effect model with week as a fixed effect and subject as a random effect. The time to achieve apparent steady state plasma concentrations was evaluated by assessing linear trend (ie, change in natural log-transformed plasma concentration per week) in a stepwise fashion.
Results
Patient Disposition and Initial Baseline Demographics
In all, 803 patients (n = 370 on anacetrapib, n = 433 on placebo) entered the 2-year extension study, and 714 patients (n = 328 on anacetrapib, 386 on placebo) completed the 2 years of the extension period (Figure 1). The baseline patient characteristics (age, gender, and race) and efficacy variables were similar across the treatment arms in the subset of patients who entered the extension phase (Table 1) and were similar to the overall population originally enrolled in the 76-week DEFINE base study (data not shown).

CONSORT Flow Diagram.
Baseline (Week 0 of the DEFINE Base Study) Patient Characteristics in the Extension Study.

Abbreviations: DEFINE, Determining the Efficacy and Tolerability of CETP Inhibition with Anacetrapib; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; CETP, cholesteryl ester transfer protein.
a Plus–minus values are means ± standard deviation.
b Statin information not available.
Safety Analyses
Overall, treatment with anacetrapib over the additional 2 years was well tolerated (Table 2). No notable differences between treatment with anacetrapib and placebo were observed for drug-related or serious adverse events. No patterns of clinically important elevations in electrolytes or in liver or muscle enzymes were observed with anacetrapib versus placebo. There were no significant differences in the continuous change from baseline for diastolic and systolic blood pressure between anacetrapib and placebo groups (Table 2). Although the proportion of patients with categorical blood pressure increases was numerically greater in the anacetrapib versus placebo arms, these absolute differences in proportions were not statistically significant (Table 2). Treatment with anacetrapib decreased fasting plasma glucose relative to patients on placebo (4.7 vs 9.9 mg/dL, placebo-adjusted difference −5.2 (95% confidence interval [CI], −9.9 to −0.6). In patients with diabetes, the change in hemoglobin A1c from initial baseline was 0.03% and 0.2% in the anacetrapib and placebo groups, respectively. The placebo-adjusted difference was −0.2% (95% CI, −0.4-0.0).
Key Safety Parameters During the 2-Year Extension Study.
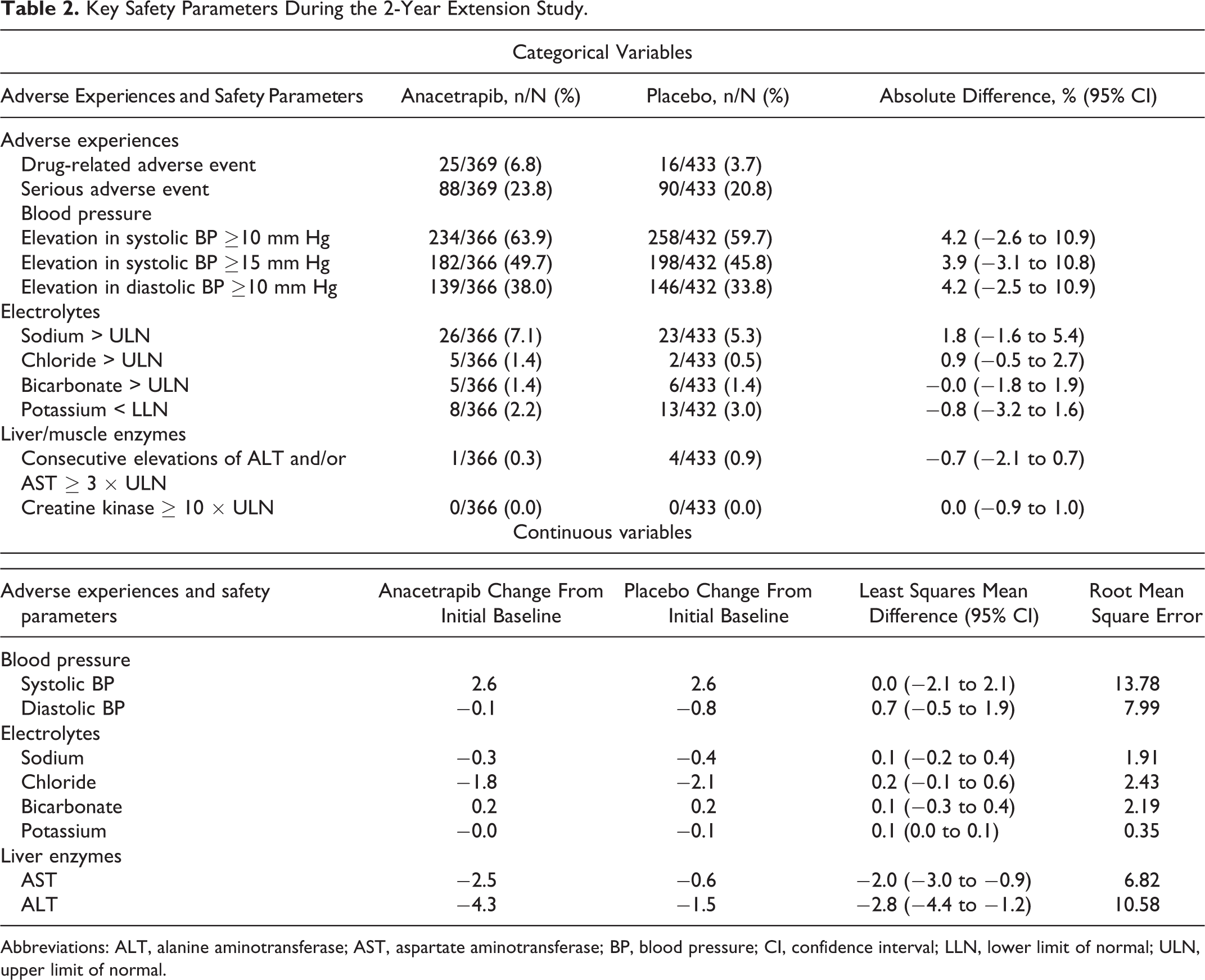

Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; BP, blood pressure; CI, confidence interval; LLN, lower limit of normal; ULN, upper limit of normal.
The prespecified, adjudicated composite cardiovascular safety end point (cardiovascular death, nonfatal stroke, nonfatal myocardial infarction, and unstable angina) and the end points of coronary revascularization, heart failure, and death were similar between the groups (Table 3). During the 2-year extension phase, the median percentage change from initial baseline in C-reactive protein was 0.0% and −14.3% in the anacetrapib and placebo groups, respectively. The placebo-adjusted difference was 14.4% (95% CI, 4.8-25.0; Table 4).
Patients With Adjudicated Cardiovascular Adverse Events and Death During the 2-Year Extension Study.a

Abbreviations: CV, cardiovascular; SAE, serious adverse event; MI, myocardial infarction.
a See reference 4 , appendix B for definitions of end points.
b n = number of patients with events.
Changes in Lipid and C-Reactive Protein Levels During the 2-Year Extension Study.

Abbreviations: CI, confidence interval; cLDA, constrained longitudinal data analysis; FW LDL-C, Friedewald-calculated low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; non-HDL-C, nonhigh-density lipoprotein cholesterol.
a Estimated from the cLDA model including terms for treatment, time, and its interaction with treatments as covariates.
b C-reactive protein was analyzed using nonparametric analysis.
c Hodges-Lehmann estimate of the difference between treatments with a corresponding distribution-free confidence interval based on Wilcoxon rank sum test.
Efficacy Analyses
Evidence of a sustained treatment effect on lipid end points was observed in anacetrapib-treated patients. After 2 years of treatment with anacetrapib, the placebo-adjusted mean percent reduction from the initial baseline was 39.9% for LDL-C, as estimated by the Friedewald equation, and the placebo-adjusted mean percent increase from baseline was 153.5% for HDL-C (Table 4). Mean percentage reductions in non-HDL-C (−30.9%) and apolipoprotein B (−16.3%) and an increase in apolipoprotein A-I (44.5%) were also observed with anacetrapib compared to placebo.
Anacetrapib Concentrations
Concentration time data for patients completing both the DEFINE 76-week base study and the 2-year extension study are shown in Figure 2. This plot represents trough plasma concentrations of anacetrapib at all time points except at the weeks 24 and 76 time points (base study) since time of dosing and plasma sampling were not available for those measures. The trough geometric mean (GM) concentration at week 12 of the base study was 541 nmol/L. The plasma concentration of anacetrapib appeared to increase modestly at weeks 24 (613 nmol/L) and 76 (666 nmol/L), although these were not trough values. Patients were off drug for at least 12 and up to 24 weeks after completion of the 76-week base study before entering the 2-year extension study. The residual mean plasma concentration of anacetrapib at the start of the 2-year extension study (week 0) was 219 nmol/L, about 40% of the on-treatment week 12 trough levels. During the 2-year extension study, anacetrapib levels increased in the plasma and seemed to reach a plateau at the extension week 40 time point (659 nmol/L). The time to achieve apparent steady state plasma concentrations was evaluated by assessing linear trend in log-transformed trough plasma concentrations. Based on the linear trend test, plasma concentrations of anacetrapib reached apparent steady state at around week 40 during the 2-year extension study. The slope of the concentration curve from extension weeks 40 to 104 was −0.0003 log nmol/L/week (90% CI, −0.0015-0.0009; P = .69).

Concentration time data for patients completing the base and extension studies. Anacetrapib C24hours (nmol/L) concentrations for patients participating in the 2-year extension to the DEFINE study (MK859-P11). Geometric means (95% CI) are shown as blue points with error bars. Note: It is not known whether all of the samples collected at weeks 24 and 76 during the Base study were true trough samples, since the time of last dose was not collected at these weeks. DEFINE, Determining the Efficacy and Tolerability of CETP Inhibition with Anacetrapib; CI, confidence interval.
Discussion
During the DEFINE 2-year extension study, no clinically meaningful differences in liver enzymes, electrolytes, plasma glucose, blood pressure, or adverse experiences were detected, and the number of prespecified, adjudicated cardiovascular serious adverse events was similar between treatment groups. The effects of treatment with anacetrapib on LDL-C and HDL-C during this 104-week extension study were similar to those observed during the 76-week DEFINE base study. Compared to placebo, anacetrapib 100 mg/d increased HDL-C levels by 153.5% and decreased LDL-C levels by 39.9% as calculated by the Friedewald formula, both very similar to the 138.1% increase in HDL-C and the 39.8% decrease in LDL-C observed in the base study.
Geometric mean plasma concentrations of anacetrapib did not appear to increase beyond week 40 for patients in the 2-year extension of the DEFINE study, which suggests that apparent steady state was effectively achieved in plasma with an apparent steady state trough level of ∼640 nmol/L. The time scale of the approach to apparent steady state is difficult to predict from the data in the extension study since patients started with significant residual amounts of drug from the 76-week base study. However, plasma concentrations were already ∼85% of this apparent steady-state trough level by week 12 in the 76-week base study.
There are important limitations to this extension study. First, the extension study enrolled a self-selected set of patients who completed the base study. The patient population does not include ineligible patients who finished the base study prior to the implementation of the extension, refused participation in the extension, or were discontinued in the base study due to the prespecified discontinuation criterion of LDL-C <25 mg/dL (0.65 mmol/L). Despite this nonrandom selection of extension study patients, initial baseline LDL-C and HDL-C levels were comparable. Second, patients were off treatment (due to the reversal phase of the base study) for varying lengths of time ranging from 12 to 24 weeks prior to entering the extension study. These limitations may have had effects on the safety, lipid, and drug concentration data. Nevertheless, the data further support the safety and tolerability of treatment with anacetrapib for a period of up to 3.5 years, comprising the 1.5 years of the DEFINE base study and the 2 years of the extension study. The extension results also confirm the favorable effects on HDL-C and LDL-C observed during the base study.
The lack of a significant positive slope in the pharmacokinetic data beyond week 40 of the 2-year extension study demonstrates that anacetrapib does not continue to accumulate in the plasma after 40 weeks of treatment during the 2-year extension study. Further studies will provide additional information on the accumulation and elimination kinetics of anacetrapib.
The Randomized Evaluation of the Effects of Anacetrapib through Lipid-modification (REVEAL) trial is a cardiovascular outcomes trial that has randomized 30 000 patients with occlusive arterial disease to treatment with anacetrapib 100 mg or placebo against a background of atorvastatin therapy (Clinicaltrials.gov number, NCT01252953). Results from this ongoing trial will provide additional, invaluable evidence regarding the clinical efficacy, pharmacokinetics, and safety of CETP inhibition with anacetrapib.
Footnotes
Acknowledgments
We thank Mary Beth Young for coordinating bioanalysis of the MK859 plasma samples.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Antonio Gotto serves on the board of directors of Aegerion Pharmaceuticals, Arisaph Pharmaceuticals, and Esperion Therapeutics; as a consultant for Kowa and Merck; and on an advisory board for Dupont and Vatera Capital. Christopher Cannon receives research grants/support from Accumetrics, AstraZeneca, Boehringer-Ingelheim, CSL-Behring, Essentialis, GlaxoSmithKline, Merck, Regeneron, Sanofi, and Takeda; serves on advisory boards for Alnylam, Bristol-Myers Squibb, Lipimedix, and Pfizer but donates funds to charity; and is a clinical advisor and has equity in Automedics Medical Systems. Eliot Brinton serves on a medical advisory board for Atherotech, Inc; receives compensation for consultancy and/or lectures from Abbvie, Amarin, Arisaph, AstraZeneca, Atherotech, Inc, Daiichi Sankyo Inc, Essentialis, Kowa Pharmaceuticals, Merck & Co, Sanofi-Aventis, and Takeda Pharmaceuticals; and receives research support from Amarin, Health Diagnostic Laboratory, Merck & Co, and Roche. Philip Barter serves on advisory boards for MSD and Kowa; serves as a consultant for CSL-Behring; receives grant support from MSD, Pfizer, and Roche; and receives payments for lectures from MSD, AstraZeneca, Pfizer, and Kowa. Uma Kher, Manash Chatterjee, Yang Liu, Xiujiang Susie Li, Sanskruti Vaidya, Sukrut Shah, Hayes Dansky, and Yale Mitchel are employees of and have stock/stock options in Merck & Co, Inc. Jennifer Moon has no disclosures. All contributors have signed confidentiality agreements with Merck requiring that prior approval is required before publishing the research findings of the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by Merck Research Laboratories, Rahway, New Jersey, USA.
