Abstract
Aim:
Despite clear evidence of immune system involvement in the pathogenesis of myocarditis, the treatment of myocarditis remains nonspecific and supportive. We sought to test the hypothesis that injection of a collagen-based implant into the inflamed myocardium would stabilize the left ventricular (LV) wall and prevent adverse remodeling and dysfunction.
Methods and Results:
Autoimmune myocarditis was induced in 42 male Lewis rats. Development of myocarditis was evaluated and confirmed by serial echocardiography and cardiac magnetic resonance scans, LV wall thickening, global and regional LV wall motion abnormalities, and in some cases pericardial effusion. Sick animals were randomized to either injectable collagen implantation or saline injection into the anterior inflamed myocardium 14 days after immunization. Significantly, injectable collagen implantation improved 31-day survival compared with controls (85.7% vs 50%; P = .03). Furthermore, although injectable collagen significantly attenuated LV systolic and diastolic dilatation and preserved LV geometry and function, control animals developed significant LV dilatation and dysfunction. These favorable effects on LV remodeling were confirmed by postmortem morphometry. Significantly, the injectable collagen implant attenuated cardiomyocyte hypertrophy and infiltration of macrophages and lymphocytes into the myocardium.
Conclusions:
The present study shows, for the first time, that injectable collagen biomaterial improves survival and attenuates cardiac inflammation, cardiomyocyte hypertrophy, LV remodeling, and dysfunction in the early period after myocarditis in rats. Our findings suggest a new biomaterial-based strategy to ameliorate the devastating effects of myocarditis.
Myocarditis is an inflammatory heart disease that progresses to dilated cardiomyopathy in approximately 30% of the cases. 1,2 The overwhelming inflammatory response that takes place in myocarditis leads to early destruction of the extracellular matrix (ECM) architecture, causing substantial alterations in the matrix degradation system. 3 This leads to the development of a dilated and spherical-shaped ventricle, resulting in negative left ventricular (LV) remodeling. These detrimental events advance to progressive structural and functional changes in the ventricles and play a key role in heart failure progression, which is strongly linked to poor outcome after myocarditis. 4 –7
Currently, treatment of myocarditis remains nonspecific and supportive, 2,8,9 highlighting the need to develop effective therapy for myocarditis and its potential complications. The recent introduction of injectable biomaterials and scaffolds for in situ tissue engineering has generated great hope in the field of cardiovascular regenerative medicine. 10 Injectable biomaterials could replace the damaged ECM with the benefit of minimally invasive delivery into the myocardium. 10 We and others have shown that acellular injectable biomaterials can prevent adverse cardiac remodeling after myocardial infarction (MI) in animal models. 11 –15 However, it is still unknown whether this treatment is effective in myocarditis. Therefore, we aimed to test the hypothesis that injection of collagen-based implant into the inflamed myocardium of rats with myocarditis would replace the damaged ECM, stabilize LV walls, and prevent adverse LV remodeling and dysfunction. The rational for this approach was the pivotal role of the damaged ECM in the pathogenesis of LV remodeling after myocarditis 3 and our previous favorable experience with injectable biomaterial in acute MI. 12,13 The ability to scaffold and replace the damaged ECM with injectable biomaterial could provide new options for the treatment of myocarditis and prevent adverse LV remodeling and heart failure.
Method
The experiments were performed in accordance with the guidelines of the Animal Care and Use Committee of the Sheba Medical Center, Tel-Aviv University, and conform to the policies of the American Heart Association and the “Guide for the Care and Use of Laboratory Animals” (Department of Health and Human Services, NIH Publication No. 85-23).
Rat Model of Autoimmune Myocarditis
To induce experimental autoimmune myocarditis (EAM), we anesthetized 42 male Lewis rats (Harlan, Israel), aged 6 to 8 weeks, weighing ∼200 g, with a combination of ketamine–xylazine (40 mg/10 mg/kg) and subjected them to porcine cardiac myosin (PCM) immunization (M0531; Sigma-Aldrich, Israel) as described previously. 16,17 In brief, we emulsified PCM with an equal volume of complete Freund adjuvant (CFA; Difco) mixed with desiccated Mycobacterium tuberculosis H37Ra (Difco, Detroit, Michigan). On days 0 and 7, 0.2 mL of the PCM-CFA emulsion (containing 1 mg PCM) was subcutaneously injected into all animals.
Injectable Collagen Implantation
The collagen implant used in this study is a natural, cross-linked collagen type I dermal filler that has demonstrated unique qualities for in situ tissue engineering 18 and has been used for cosmetic soft tissue augmentation procedures. 19,20 The injectable collagen filler gel (Evolence) is a suspension of cross-linked type I collagen (isolated from porcine tendons) with a natural sugar ribose (Glymatrix Technology, Herzliya, Israel). 18 The resultant material was further suspended in phosphate-buffered saline at a concentration of 35 mg/mL, supplied by Colbar LifeScience Ltd (Herzliya, Israel).
The collagen implant was injected 14 days after the induction of myocarditis. Animals with confirmed myocarditis were anesthetized with a combination of 40 mg/kg ketamine and 10 mg/kg xylazine, intubated, and mechanically ventilated. Then, the chest was opened by left thoracotomy, the heart was exposed, and the pericardium was removed. Animals were randomized for either collagen implantation or saline injection (100 μL, using a 29-guage needle) into the anterior myocardium. The chest was closed and the animals were allowed to recover.
Echocardiography
Echocardiography of both long- and short-axis views was acquired in all animals using a commercially available system (Sonos 5500; Philips, Andover, Massachusetts) equipped with a 12-MHz phased-array transducer. Examinations were performed before induction of myocarditis, before collagen implantation, and 3, 10, and 17 days after collagen implantation. 16,17 All measurements were done blindly by an experienced technician, averaged for 3 consecutive cardiac cycles and blindly reviewed by an echocardiography expert (MSF).
Cardiac Magnetic Resonance Imaging
Cardiac magnetic resonance (CMR) imaging was performed before induction of myocarditis, before collagen implantation, and 10 days after implantation in a subgroup of 13 rats, under isoflurane anesthesia (1.5%; Abbot Laboratories Ltd, Berkshire, United Kingdom) at a constant temperature of 37°C. We used a whole-body human 3-T magnetic resonance imaging (MRI) system (3 T HDxt Ver.15 M4A; GE Medical Systems, Milwaukee, Wisconsin) and a custom-built quadrature cylindrical radiofrequency volume coil, ID 77 mm × 178 mm length (Doty Litzcage Coil-Doty Scientific, Inc, Columbia, South Carolina).
Clinical CMR protocols were adjusted and optimized for the rat myocarditis model as described previously. 16,17 The CMR images were analyzed using a dedicated postprocessing CMR Workstation (Medis Medical Imaging Systems BV, QMass MR 7.4, Leiden, the Netherlands). Cine images acquired in multiple short-axis planes were used for functional analysis. 16,17
Histological and Morphometric Analysis
Histological evaluation was carried out 17 days after collagen implantation. Animals were euthanized with 15% KCl, under deep anesthesia with an overdose of phenobarbital. The hearts were perfused with 4% formaldehyde (15 mmHg) for 20 minutes after which they were sectioned into 3 to 4 transverse slices parallel to the atrioventricular ring. We then fixed each slice with 4% formalin, embedded in paraffin, and sectioned them into 5-µm slices. Serial sections were stained with either hematoxylin–eosin to detect cellular infiltration or Masson trichrome (Sigma, St Louis, Missouri) to assess fibrosis. Then, all slides were digitally photographed and used for comprehensive histological and morphometric analysis (Sigma Scan Pro ver.5; SPSS, Inc, Chicago, Illinois) as described previously. 13 To estimate the severity of cardiomyocyte hypertrophy, sections were stained with wheat germ agglutinin (lectin from Triticum vulgaris, TRIC labeled, L-5266; Sigma, Israel) and 3 to 5 adjacent fields of each section were evaluated for the mean thickness of cardiomyocytes. To assess the extent of macrophages and T lymphocyte accumulation, sections were costained with anti-ED1-Macrosialin (mouse anti-rat CD68, MCA341A647; Abd Serotec, Israel) and anti-CD4 (mouse anti-rat CD4, MCA55R; Abd Serotec) and 3 to 5 adjacent fields of each section were evaluated for the number of cells.
Statistical Analysis
Statistical analysis was done with the GraphPad Prism version 5.00 for Windows (GraphPad Software, San Diego, California). All variables are expressed as mean ± standard deviation. Normality was tested with the Kolmogorov-Smirnov test. The difference between means of groups was compared by a 2-tailed unpaired t test or paired t test, when appropriate. To test the hypothesis that changes in measures of LV function over time varied among the experimental groups, a general linear model 2-way repeated-measures analysis of variance was used. The model included the effects of treatment, time, and treatment-by-time interaction.
Results
Initially 42 rats were subjected to EAM. Of them, 5 rats did not develop myocarditis and were excluded from the study and 11 rats died because of myocarditis prior to injection of collagen or saline. Overall, 26 rats with confirmed myocarditis were included in the functional study. Fourteen days after the induction of myocarditis, the experimental animals were randomized and subjected to either collagen implantation (n = 14) or saline injection (n = 12) into the anterior inflamed myocardium. Of them, 8 rats (2 in the experimental and 6 in the control group) died during the follow-up period.
To evaluate LV remodeling and function, all animals underwent 5 echocardiography studies. In addition, a subgroup of animals (6 from the experimental and 7 from the control group) was evaluated by 3 CMR imaging scans. Final data analysis of echocardiography and CMR studies was carried out on 11 and 4 animals in the intervention group and on 6 and 4 in the control group. The characteristics of EAM in rat have been described previously. 16,17
Collagen Implantation Attenuated LV Dilatation and Dysfunction in the Early Period After Myocarditis
Significantly, injectable collagen implantation improved survival, compared with controls (12 of 14 [85.7%] vs 6 of 12 [50%], P = .03, Figure 1), at 31 days after induction of myocarditis.

Collagen implant improves survival in rats with fulminant myocarditis. Treatment with injectable collagen significantly improved survival in rats, 31 days after the induction of myocarditis.
Serial echocardiography studies have shown that LV systolic and diastolic anterior wall thickness (site of injection) significantly increased by 36% ± 44% and 57% ± 68%, respectively, in collagen-treated animals compared with controls 10 days after collagen implantation (P = .01, Figure 2). However, wall thickness decreased to near baseline levels at 17 days after collagen treatment (Table 1).
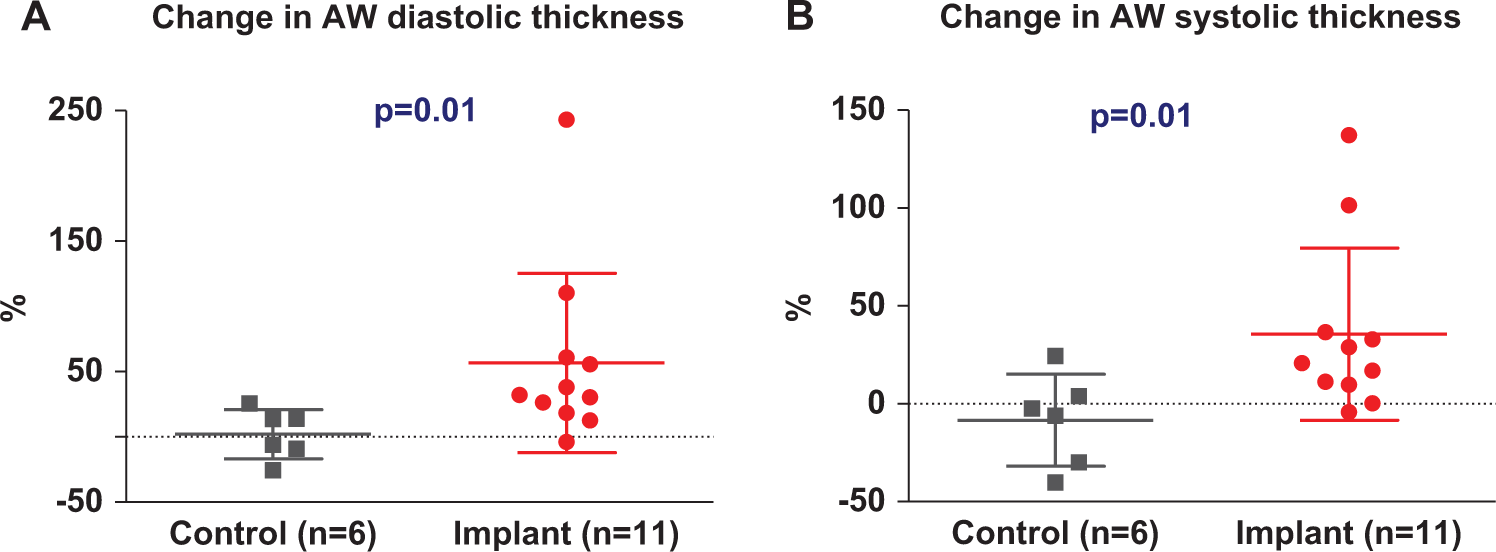
Collagen implant increased LV anterior wall thickness, 10 days after treatment, by 2-dimensional echocardiography. Animals treated with injectable collagen demonstrated significant increases in LV diastolic (A) and systolic anterior wall thickness (B) compared with control hearts, 10 days after collagen treatment. Change is based on the difference between before and 10 days after collagen treatment. The difference between means of groups was compared by a 2-tail unpaired t test (Mann-Whitney nonparametric test). AW indicates anterior wall; LV, left ventricular.
Left Ventricular Variables of Collagen-Treated and Control Rats at Baseline (Day 0), Pretreatment (Day 10), and 10 Days (Day 24) and 17 Days (Day 31) After Injectable Collagen Treatment, by 2D Echocardiography.a

Abbreviations: AW/PW, anterior/posterior wall; d, days; dia, diastolic; FAC, fractional area change; LVDA/LVSA, left ventricular diastolic/systolic area; LVDD/LVSD, left ventricular diastolic/systolic dimension; LVFS, left ventricular fractional shortening; sys, systolic; 2D, two dimensional.
aThe difference between time points within the group was compared by a 2-tail paired t test. The difference between means of groups was compared by a 2-tail unpaired t test.
bThe difference between pretreatment and 10 days after treatment.
cThe difference between pretreatment and 17 days after treatment.
Significantly, treatment with injectable collagen attenuated LV dilatation 10 days after implantation. Although controls demonstrated enlarged hearts and markedly dilated LVs, collagen-treated animals preserved heart size and LV geometry, by echocardiography, 10 days after collagen treatment (Figure 3A and B). Furthermore, the control animals developed progressive increases in LV systolic and diastolic dimensions of 25 ± 8% and 9 ± 5.6%, respectively (Figure 3C and D) and areas (26 ± 13.7% and 7 ± 7.6%), whereas collagen-treated animals developed significant decreases in 7 ± 14% (P = .01) and 10 ± 19% (P = .08) in LV diastolic dimension, respectively, (Figure 3C) and area, and only a minor increase in the systolic dimension (0.5 ± 24%, P = .01, Figure 3D), 10 days after collagen treatment. Notably, this positive effect was diminished 17 days after collagen treatment, with no significant difference between the treatment and the control groups. Of interest, pericardial effusion, typically associated with severe perimyocarditis, was present only in the control animals (in 5 of 6 surviving rats vs 0 of 11, P = .0005).
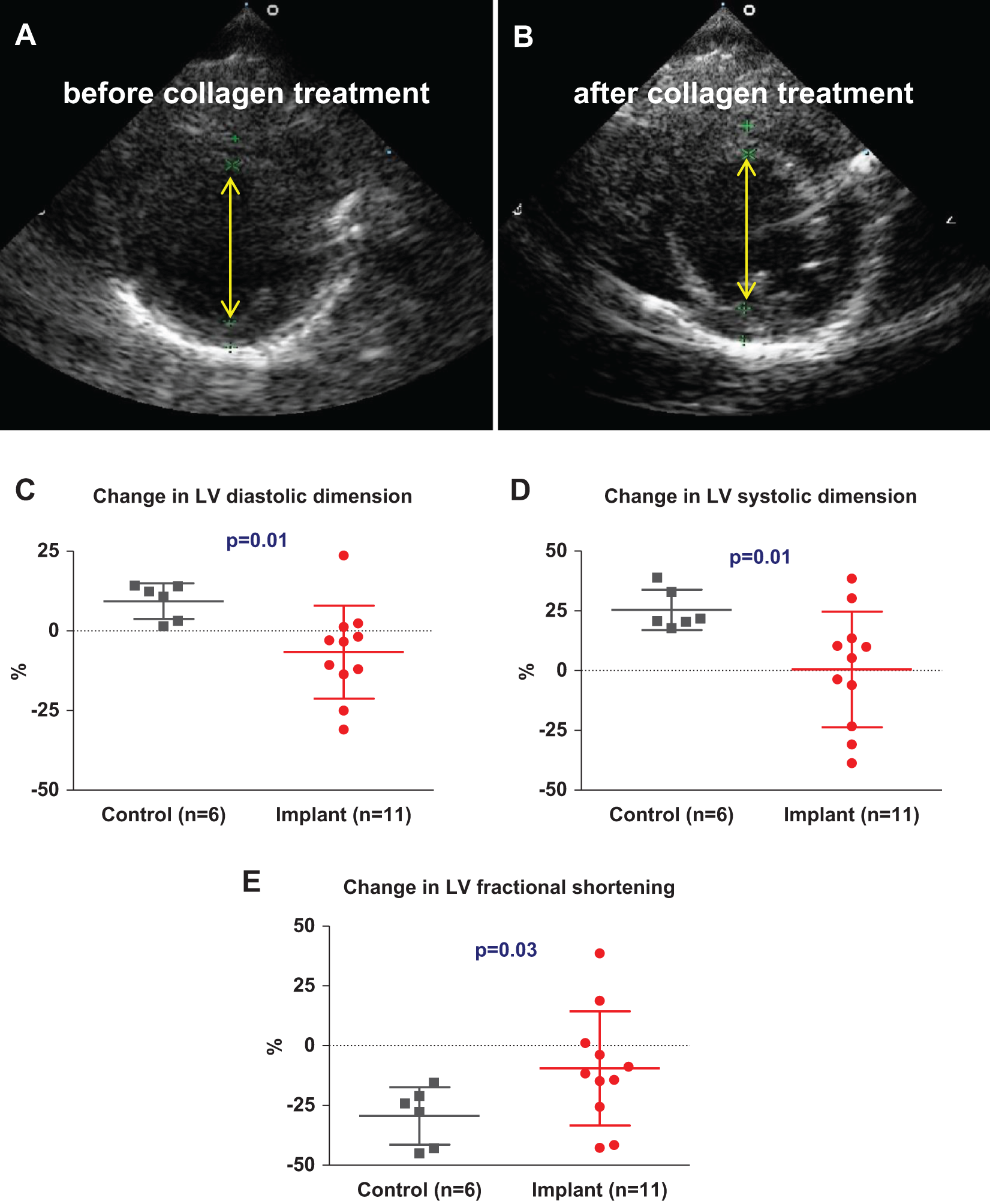
Collagen implant attenuated LV dilatation and dysfunction, 10 days after treatment, by 2-dimensional echocardiography. Representative short-axis end-diastolic images of rat heart with myocarditis (upper panel) before (A) and 10 days after collagen implantation (B). Treatment with injectable collagen preserved near normal heart size and LV geometry 10 days after treatment. In addition, injectable collagen attenuated increases in LV diastolic (C) and systolic dimensions (D) 10 days after collagen treatment. Furthermore, the decrease in LV fractional shortening detected in the controls was significantly attenuated in collagen-treated rats at 10 days after treatment (E). Change is based on the difference between before and 10 days after collagen treatment. The difference between means of groups was compared by a 2-tail unpaired t test (Mann-Whitney nonparametric test). LV indicates left ventricular.
Treatment with injectable collagen improved LV contractility. By echocardiography, a marked deterioration in LV fractional shortening, from 42.5 ± 5.4% to 27 ± 11.7% (P = .03), was observed in control animals 31 days after induction of myocarditis (Table 1). Significantly, this decrease was attenuated 10 days after injectable collagen treatment where the change in the treatment group (−9.5 ± 23%) was significantly smaller than that in the control group (−29.4 ± 12%, P = .03; Figure 3E). Furthermore, a trend for a protective effect on LV function was still noticeable at 31 days after induction of myocarditis (Table 1, P = .4).
The results of echocardiography studies were confirmed and extended by CMR imaging, which demonstrated that injectable collagen reduced adverse LV remodeling and improved LV contractility (Figure 4). The control animals developed significant increases of 108 ± 33% and 34 ± 11% in systolic and diastolic LV volumes, respectively. Significantly, treatment with injectable collagen attenuated the increase in the LV systolic volume 10 days after collagen implantation (P = .007; Figure 4C). In addition, LV ejection fraction (LVEF) and stroke volume (SV) were significantly increased by 18 ± 15.7% and 77% ± 47%, respectively, in the collagen-treated group 10 days following treatment while the control animals demonstrated a marked decrease in both LVEF and SV of 34% ± 7.7% and 11% ± 24%, respectively (P = .001, P = .01; Figure 4D and E) by CMR imaging.
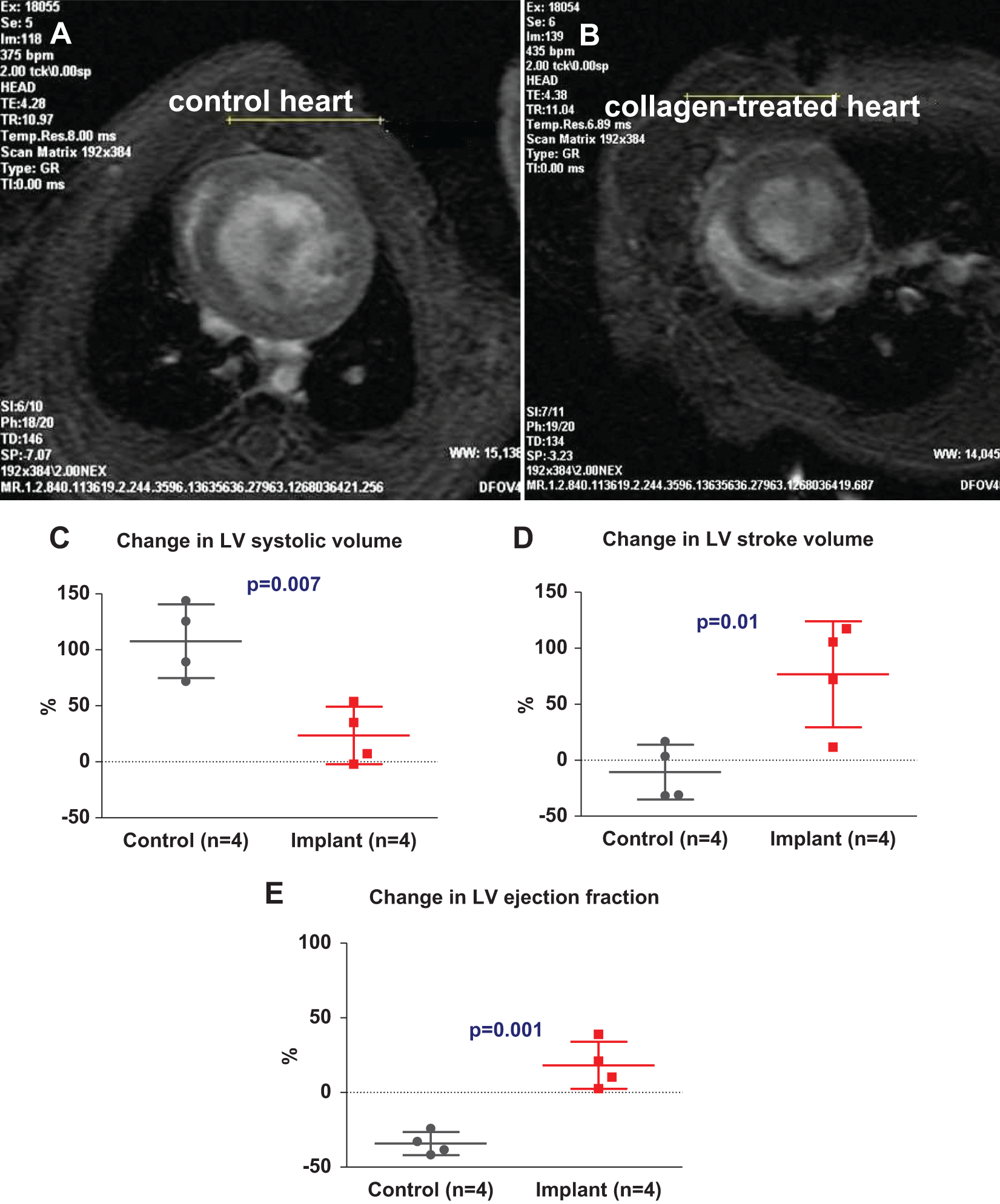
Collagen implant attenuated LV remodeling and dysfunction, 10 days after treatment, by CMR imaging. Representative short-axis end-diastolic cine-CMR images of collagen-treated (B) and control (A) hearts, 10 days after collagen implantation. Heart size, LV size, and LV geometry of the collagen-treated heart is generally normal, while the control heart is enlarged, and the LV is markedly dilated. In addition, treatment with injectable collagen significantly attenuated the increase in LV systolic volume compared with controls, 10 days after treatment (C). Furthermore, injectable collagen significantly improved LV ejection fraction (E) and stroke volume 10 days after collagen treatment (D). Change is based on the difference between before and 10 days after collagen treatment. The difference between means of groups was compared by a 2-tail unpaired t test (Mann-Whitney nonparametric test). CMR indicates cardiac magnetic resonance; LV, left ventricular.
Collagen Implantation Attenuated Inflammation, Hypertrophy, and LV Dilatation
By postmortem morphometry, the control group developed a spherical-shaped chamber, which is a well-recognized manifestation of progressive remodeling that occurs after myocarditis (Figure 5C and D). Furthermore, control hearts were enlarged, pale, and extensively scarred compared to those treated with injectable collagen (Figure 5A and C). In addition, microscopic examinations showed fibrous scars and tissue fibrosis that spread throughout the left and right ventricles (Figure 5B).

Collagen implant attenuated LV dilatation, cardiomyocyte hypertrophy, and inflammation. Hearts were removed and fixed in formalin 17 days after collagen implantation or saline injection. Representative heart sections of controls (upper panel; A and B) and collagen-treated (lower panel; C and D) hearts show that the control heart is enlarged and extensively scarred compared with the collagen-treated heart. Also, the control heart developed into a round-spherical shape, while the collagen-treated heart maintained a near normal elliptical shape. The collagen implant is still present in myocardial tissue at the injected site 17 days after implantation (E) and easily identified as a homogenous pale blue region by Massons trichrome staining. Hematoxylin and eosin staining shows robust cells allocate alongside the border of the collagen implant and infiltrate deep within the collagen matrix, long after the inflammatory phase of myocarditis has rested. Postmortem morphometry showed that the LV maximal diameter (F), whole LV area (G), LV cavity (H), and muscle area (I) were significantly smaller in hearts treated with injectable collagen 17 days after treatment, indicating a substantial attenuation in LV remodeling. Cardiomyocyte hypertrophy was attenuated in those hearts treated with the injectable collagen implant (J–L). In addition, the total number of macrophages and T lymphocytes, distant from the implant, was significantly smaller in the hearts treated with collagen implant, compared to controls (M–R). LV, left ventricular.
By histology, the collagen implant was easily identified on Masson trichrome staining as a homogenous pale blue region with distinct boundaries (Figure 5E). Interestingly, postmortem examinations of the heart sections revealed that a large part of the collagen implant was still present in the myocardial tissue 17 days after implantation (Figure 5E).
Notably, hematoxylin and eosin staining of the collagen-treated hearts showed robust mononuclear cells present alongside the border of the collagen implant, which also infiltrated deep into the matrix of the implant (Figure 5E).
Significantly, the LV maximal diameter, an indicator of ventricular dimension, was smaller in the collagen-treated hearts (4.8 ± 0.6 vs 3.5 ± 0.4 mm, P = .0001; Figure 5F) 17 days after treatment. Furthermore, the whole LV area and LV cavity area were smaller by 27.6% and 43.8% in the treatment group (55.7 ± 9 and 18 ± 4.4 mm2) compared with the control hearts (77 ± 5.4 and 32 ± 7 mm2, P = .0001 and P = .0001; Figure 5G and H). In addition, the muscle area was markedly smaller in the collagen-treated hearts than that in the control hearts (37.7 ± 6 vs 44.6 ± 5.5 mm2, P = .03; Figure 5I). Thus, the results of postmortem morphometry support the favorable effects of injectable collagen on the inflamed heart as indicated by the echocardiography and CMR studies.
Significantly, collagen implant reduced cardiomyocyte hypertrophy and myocardial tissue inflammation. Although sections of the control hearts demonstrated separation and disruption of the muscle fiber arrangement accompanied with hypotrophy of cardiomyocytes, the collagen-implanted hearts showed near-normal arrangement of myocardial striation with no significant separation of the muscle fibers (Figure 5K and L). Moreover, cardiomyocyte thickness was significantly smaller in the collagen-treated hearts compared with controls (7.8 ± 1.7 vs 17.6 ± 2.8 µm, P < .0001; Figure 5J).
Furthermore, collagen implants attenuated inflammation. Although macrophages and T lymphocytes accumulated massively in the control hearts, the number of macrophages and lymphocytes was significantly smaller in the hearts treated with the collagen implant (1059 ± 777 vs 223 ± 112 mm2, P = .002 and 445 ± 213 vs 168 ± 102 mm2, P = .002; Figure 5M–R), as indicated by anti-ED1 and anti-CD4 staining.
Discussion
The major new findings of the present study suggest, for the first time, that injection of collagen-based implant into the myocardium of rat with myocarditis significantly improves survival, attenuates adverse LV remodeling, and improves LV function in the early period after myocarditis. The injectable collagen implant provides temporary structural support and mechanical stabilization to the injured/inflamed myocardial tissue by replacing the physical functions of the damaged ECM. This scaffolding effect can reduce ventricular wall strain, preventing LV thinning, dilatation, negative remodeling, and deterioration in cardiac function and subsequent mortality following myocarditis.
Significantly, the injectable implant reduced both inflammation and cardiomyocyte hypertrophy. The mechanism behind this surprising finding is unclear. Indeed, mechanical strain induced by hypertension and pressure overload can activate immune response and subsequent inflammation. 21,22 It is possible that by reducing wall strain and subsequent remodeling, the injectable implant also reduces cell hypertrophy, cell death, fibrosis, and reactive inflammation, in remote areas.
The importance of our work lies in providing proof of concept for a novel approach that could lead to the development of therapeutic interventions to treat and prevent one of the serious consequences of myocarditis, adverse LV remodeling and dysfunction.
Active myocarditis leads to early degradation of the ECM, disruption of the collagen network, and weakening of the collagen scaffold, resulting in LV dilatation and dysfunction in some patients. 3,6,23 These key events that follow the acute inflammatory course of myocarditis advance to progressive structural and functional changes in the ventricles, leading subsequently to negative LV remodeling. This is further accompanied by the loss of normal bullet-shaped cavities along with the development of spherical-shaped chambers, coupled with increased wall stress in the remaining viable myocardium. Ventricular dilatation, in terms of LV sphericity, is strongly linked to poor outcome. 24 Despite the urgent clinical need, specific treatments of myocarditis are currently unavailable. 3,9 Thus, any intervention that prevents or reverses LV remodeling could reduce mortality.
Recent experimental approaches to diminish negative LV remodeling and prevent heart failure after MI include in situ-gelling biomaterials. 11 –14,25 –27 Different types of injectable biomaterials have been tested in different animal models: natural polymers or hydrogels, which undergo controlled liquid-to-solid transition, such as fibrin (fibrinogen and thrombin), collagen, alginate solution, matrigel, or hyaluronic acid. 25 Regardless of the differences in models and materials, there is general consensus that there is improvement in cardiac function following the thickening of the LV walls, reducing wall strain and hemodynamic stress, and limiting maladaptive cardiac remodeling. 25 –27
Previously, we have shown that alginate-based injectable biomaterial can reduce post-MI remodeling. 12,13 Intracoronary injection of biomaterial solution might be a more clinically relevant approach. However, in the present study we used a rat model, in which selective intracoronary injection is impossible. Moreover, here we used an injectable collagen implant which, due to its potent thrombogenic properties, could not be delivered into the circulation or the coronary artery. Instead, a collagen–gel-based dermal filler, sold to treat facial wrinkles, was injected into the inflamed heart. The biomaterial used is produced by means of a novel cross-linking method. This specific product has demonstrated favorable biocompatibility, weak antigenicity, resistance to degradation, and longer persistence and shape preservation. 18
The efficacy of similar biomaterials to reduce post-MI remodeling has been described previously. For example, Dai and colleagues tested the effect of injecting collagen-based hydrogel into the infarct area of a rat heart. 15 Although mechanical and functional improvements were noted, no significant angiogenesis or cell infiltration was found. In contrast, Huang and colleagues reported that collagen increased capillary density and myofibroblast infiltration into the infarct area of their ischemia-reperfused rat model. 28 Other collagen-based thermal hydrogels known as matrigel also contain basement membrane components that boost bioactivity, and these have been shown to improve cardiac function and increase scar thickness and angiogenesis. 29 Notably, in the present study, the favorable effect of injectable collagen was found to be temporary by echocardiography. It is likely that the high mortality rate in the control group created a “selection bias.” Thus, had the dead animals remained alive at 17 days, their cardiac function would have been low, making the function between the 2 groups different. Nevertheless, postmortem morphometry clearly showed that the collagen implant was effective and reduced LV dilatation for at least 17 days.
Limitations
The first limitation of our study is the relatively short follow-up period. A longer follow-up may be needed to facilitate long-term assessment of treatment outcome, particularly when assuming that degradation of the implant has been initiated. Second, the optimal time for biomaterial injection remains unknown. In this study, the collagen material was injected when robust infiltration of cells into the cardiac tissue was underway and before the peak of inflammation. Accordingly, selecting an earlier or later time for treatment could possibly have induced a different outcome.
Summary and Implications
Our study suggests that injection of a collagen-based implant into the inflamed myocardium of rats with myocarditis improves survival and preserves LV geometry. The collagen implant provides the structural support needed for inducing mechanical stabilization. Our findings open up new options for the treatment of myocarditis and dilated cardiomyopathies. Other biomaterials, with or without conjugated anti-inflammatory or trophic factors, could further promote tissue restoration via generating a favorable local microenvironment for effective tissue repair. 30 Along these lines, hydrogel delivery could be combined with mesenchymal stem cells for immunomodulatory and regenerative therapy or used as a depot for controlled drug delivery. Decreasing wall strain, inflammation, and cellular stress could provide a better environment to foster stem or progenitor cells to promote regeneration, which in turn may result in new therapeutic strategies for myocarditis.
Footnotes
Acknowledgments
We thank Pat Benjamin (echocardiography) and Yair Rubinstein (MRI) for their excellent technical assistance and Mrs Vivienne York for her skillful language editing.
Authors’ Note
This work was performed in partial fulfillment of the requirements for a PhD degree of Shunit Rinkevich-Shop, Sackler Faculty of Medicine, Tel Aviv University, Tel-Aviv, Israel.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jonathan Leor applied for a patent on injectable alginate for myocardial repair via Ben-Gurion University.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the US-Israel Binational Science Foundation (FHE and JL) grant number 2007290 and the Schlezak Foundation (JL), Tel Aviv University, Israel.
