Abstract
Obesity has become a growing pandemic of alarming proportions in the developed and developing countries over the last few decades. The most perturbing fact regarding obesity is the increased predisposition for coronary artery disease, congestive heart failure and sudden cardiac death. The modest efficacy of current anti-obesity agents such as orlistat and the increasing withdrawals of several anti-obesity agents such as sibutramine, rimonabant have led to huge gaps in the pharmacotherapy of obesity. Lorcaserin and Phentermine-topiramate combination (phen-top) are two drugs approved by US FDA in 2012. Lorcaserin, a 5HT2C agonist has moderate efficacy with an acceptable safety profile. Clinical trials with Phen-top have shown a reasonable efficacy but at the cost of risks such as teratogenicity and psychiatric disturbances. Cetilistat, a lipase inhibitor is claimed to have superior safety profile to orlistat and is in phase 3 clinical trials. Other promising anti-obesity molecules acting on the gut which are in clinical trials include exenatide and liraglutide. Drugs which act on the monoaminergic and opioid systems include bupropion-naltrexone and bupropion-zonisamide. Other novel first-in-class drugs which have been explored and have limited success in early clinical development include velneperit, tesofensine, and beloranib. Tesofensine is a triple monoamine re-uptake inhibitor, velneperit acts as a neuropeptide Y5 receptor antagonist and beloranib is a methionine amino peptidase 2 inhibitor. Novel targets such as histamine H3 receptor, VEGF, matrix-metalloproteinase, sirtuin receptors are also being investigated. This review is an attempt to describe the new and emerging molecules that are in clinical development for obesity.
Introduction
Obesity is a growing pandemic that has risen to alarming proportions over the last decade in the developed world. The situation is also changing in the developing world with the increasing westernization of food habits and lifestyle. According to the World Health Organization projections, by 2015 approximately 2.3 billion adults will be overweight and more than 700 million will be obese. 1 In addition, the economic cost of obesity-associated diseases is approaching US$100 billion per year in the United States. 2 The most perturbing fact regarding obesity is the increased predisposition for coronary artery disease, congestive heart failure, and sudden cardiac death. Besides cardiovascular (CV) risk, overweight (body mass index [BMI] 25-29) and obesity (BMI >29) are major risk factors for a wide range of diseases such as diabetes, dyslipidemia, obstructive sleep apnea, asthma, musculoskeletal disorders, and cancers. 3 There is compelling evidence to suggest that obesity is associated with more morbidity than smoking, alcoholism, and poverty and is on the verge of overtaking smoking as a leading cause of preventable death in the United States. 4,5
The current methods for reducing body weight include diet, exercise, drug therapy, bariatric surgery, or combinations of those mentioned previously. 6 Unfortunately, failure to maintain changes in diet and exercise patterns is a major reason for weight regain experienced by most people. Bariatric surgery, generally used for patients with morbid obesity (BMI ≥ 40), is effective for sustaining weight loss but is often fraught with catastrophic complications such as increased mortality. 7,8 The drugs that are currently approved for obesity by the US Food and Drug Administration (FDA) include orlistat, phendimetrazine, and diethylpropion, of which only orlistat is approved for long-term use. Although these drugs have existed in the market for several years, it has failed to make any significant impact on the burden of obesity and its consequences, especially on the CV health. Table 1 highlights the limitations of these older antiobesity agents along with their regulatory status. 9 –12 Thus, there is an obvious need for an effective antiobesity agent that causes substantial weight loss thereby reducing the risk of cardiovascular disease (CVD). This article is an attempt to highlight the new molecules that have been recently approved for obesity by the US FDA and the emerging molecules that are in clinical development for obesity.
Abbreviations: FDA, Food and Drug Administration; SR, sustained release.
Antiobesity Molecules Recently Approved by the US FDA
Lorcaserin
Lorcaserin is a new molecular entity with a 2-ringed structure that has high affinity for the 5-HT2C receptor that is present in the hypothalamic proopiomelancortin neurons. Chemically, lorcaserin is described as [1R]-8-chloro-1-methyl-2,3,4,5-tetrahydro-1H-3-benzazepine hydrochloride. 13 On account of its low-molecular weight, nonpolymer structure, and a binding activity that alters biotarget functions, it is considered a “small molecule.” Binding of lorcaserin to the 5-HT2C receptors in proopiomelanocortin neurons promotes satiety and reduces the individual’s food intake. Lorcaserin is selective for the 5-HT2C subtype and does not have significant activity at either 5-HT2B or 5-HT2A receptors; activity at the 5-HT2B receptor has been associated with the development of valvular heart disease with earlier antiobesity agents such as dexfenfluramine. 14,15
Pharmacokinetics of Lorcaserin
The recommended clinical dose of lorcaserin is 10 mg twice daily. Following oral administration, more than 90% of lorcaserin is rapidly absorbed. The time taken to attain maximum plasma concentration is approximately 1.5 to 2 hours, and its half-life (t1/2) is about 11 hours. Approximately 70% of the drugs are bound to plasma proteins. The volume of distribution in the body is ∼252 L in a 92.5-kg patient. Lorcaserin is mainly metabolized in the liver and excreted by the kidney. The major circulating metabolite is the sulfamate of lorcaserin (M1), and the major urinary metabolite is N-carbamoyl glucuronide (M5). 13 –17
Efficacy of Lorcaserin
Lorcaserin has been evaluated in 3 large randomized, placebo-controlled, double-blind studies, the results of which paved way for the drug to receive an FDA approval in June 27, 2012, for the treatment of obesity. The first study investigating lorcaserin, the Behavioral Modification and Lorcaserin for Overweight and Obesity Management (BLOOM) study, enrolled people 18 to 65 years of age with a baseline BMI of 30 to 45 kg/m2 or a BMI of 27 to 45 kg/m2 with at least 1 concomitant weight-related comorbidity, such as hypertension, CVD, dyslipidemia, impaired glucose tolerance, or obstructive sleep apnea. 18 A total of 3182 participants were randomized to receive lorcaserin 10 mg twice a day (n = 1595) or placebo (n = 1587) for 52 weeks. At 52 weeks, those receiving placebo continued for an additional 52 weeks, whereas those receiving lorcaserin twice a day were randomized in a 2:1 ratio to continue receiving lorcaserin or be switched to placebo. The primary outcome, which was the percentage of patients who achieved 5% weight loss of baseline weight after 52 weeks, occurred in 47.5% of those receiving lorcaserin compared with 20.3% receiving placebo (modified intention to treat [mITT] analysis population with last observation carried forward [LOCF], P < .001). The mean decrease in weight (calculated using the least squares mean) was 5.8% (5.8 kg) for lorcaserin compared with 2.2% (2.2 kg) for placebo. After 104 weeks of treatment, more patients who continued taking lorcaserin were able to maintain weight loss of 5% compared with those who transitioned to placebo (67.9% vs 50.3%, P < .001). Secondary outcomes, which included percentage achieving >10% weight loss (P < .001), reduction in waist circumference, blood pressure reduction, and BMI, were improved with lorcaserin after 52 weeks of treatment, although the clinical significance of these differences is not known.
In the BLOSSOM trial, a total of 4008 participants were randomized to receive either lorcaserin 10 mg (n = 801), lorcaserin 10 mg twice a day (n = 1602), or placebo (n = 1601) for 52 weeks in addition to nutritional and exercise counseling provided at each study visit. 19 After 1 year of treatment, 47.2% of those receiving lorcaserin twice a day lost >5% of their baseline weight compared with 40.2% of those receiving lorcaserin every day and 25% of those receiving placebo (P < .001 for both doses of lorcaserin vs placebo). Greater than 10% weight loss was achieved by 22.6% receiving lorcaserin twice a day, 17.4% receiving lorcaserin every day, and 9.7% receiving placebo (P < .001 for both doses vs placebo). Absolute weight loss, calculated by least squares mean, was 5.8, 4.7, and 2.9 kg for lorcaserin twice a day, lorcaserin every day, and placebo, respectively. Although systolic blood pressure, diastolic blood pressure, and heart rate decreased in all the groups, the difference was not statistically significant. Importantly, no differences were noted between lorcaserin and placebo in echocardiographic outcomes.
The BLOOM in Diabetes Mellitus (BLOOM-DM) study enrolled people aged 18 to 65 years with a BMI of 27 to 45 kg/m2 and a diagnosis of type 2 diabetes with a hemoglobin A1c (HbA1c) level of 7% to 10%. 20 Patients were stratified based on baseline type 2 diabetes treatment and randomized to lorcaserin 10 mg twice a day (n = 256), lorcaserin 10 mg every day (n = 95), or placebo (n = 253) for 52 weeks; enrollment into the lorcaserin 10 mg every day group was stopped after 8 months because of slow enrollment. Patients received standardized nutritional and exercise counseling at each visit. At baseline, the mean body weight was 104 kg, and the mean BMI was 36 kg/m2. Approximately 45% of the patients in the lorcaserin once daily group and 38% of the lorcaserin twice daily group achieved atleast 5%weight loss compared with only 16% in the placebo group (P < .001 for both doses compared with placebo). A 10% reduction in weight was achieved by only 4.4% in the placebo group compared with 16.3% of those receiving lorcaserin twice daily and 18.1% of those receiving lorcaserin once daily. (P < .001 for both doses compared with placebo). Absolute weight loss (using least squares means) was 4.7 kg for lorcaserin twice daily, 5 kg for lorcaserin once daily, and 1.6 kg for placebo. Incidence of FDA-defined valvular disease did not differ between the groups.
In February 2007, a draft guidance document was released by the US FDA on the principles of developing drugs for weight loss management. In order for an antiobesity agent to be deemed effective, it needs to fulfill certain benchmarks as laid down by the guidelines. Treatment with the antiobesity molecule for 1 year should result in at least a 5% difference between the active group and the placebo, and this difference must be statistically significant or the proportion of patients who lose ≥5% of baseline body weight in the active product group should be at least 35%, and should approximately double the proportion in the placebo-treated group, and the difference between the groups should be statistically significant. By these standards, the efficacy of lorcaserin that was achieved in the three phase 3 clinical trials was only modest. Additionally, the high percentage of dropouts in all the 3 studies may actually tilt the results in favor of the drug, since the statistical approach used was mITT with LOCF. It is not known whether the results would appear different, if an imputation analysis was performed instead of the former. Nevertheless, considering the fact that the currently available antiobesity agents have only a similar or slightly lower efficacy, the drug regulators in the US FDA have approved the drug for marketing.
Safety of Lorcaserin
Lorcaserin had to face several regulatory hiccups prior to its approval by FDA on account of several safety concerns. Initially, FDA rejected an application for approval of this drug due to concerns of unexplained preclinical carcinogenicity signals in the form of breast tumors in rats treated with lorcaserin. Another disturbing feature with lorcaserin was the rising rates of new valvulopathy. However, subsequent analysis has shown that the absolute numbers of valvulopathy were much less. The most common adverse events with lorcaserin include headache, nausea, dizziness, dry mouth, paresthesia, upper respiratory tract infection, and anorexia. A small number of patients did experience a change in mood. 21 There have not been any carcinogenic signals from the data generated from human studies as of date.
On the basis of the results of these studies, lorcaserin is approved at a dose of 10 mg twice daily in patients with a BMI of 30 or 27 kg/m2 with at least 1 weight-related comorbidity, such as hypertension, type 2 diabetes, or dyslipidemia, in addition to a reduced calorie diet (RCD) and increased physical activity. The labeling indicates response to therapy should be assessed at week 12, and if there is a 5% decrease in weight, use of the drug should be discontinued, because it will be unlikely that the patient will achieve and sustain adequate weight loss with continued treatment. The long-term CV safety of lorcaserin with respect to major adverse cardiac events such as stroke and myocardial infarction (MI) is currently being investigated in postmarketing studies. 22 Although the drug has only a modest efficacy, its favorable safety profile may make it a preferred choice in obesity clinics in the years ahead.
Phentermine–Topiramate
The phentermine–topiramate (Phen-top) combination is a once-daily formulation designed to provide an immediate release of phentermine and a delayed release of topiramate. Phentermine reduces the food intake by enhancement of norepinephrine release and blockade of norepinephrine reuptake. The antiobesity action of topiramate although largely unexplained is believed to be due to increased energy expenditure, decreased energetic efficiency, and decreased caloric intake from preclinical studies. The combination allows lower doses of the individual compounds and is reported to cause fewer side effects than the individual compounds. The fixed dose combination was approved by the FDA in July 17, 2012, for promoting weight loss based on the tolerability and efficacy data from 3 large randomized, double-blind, placebo-controlled trials.
The EQUIP trial (Controlled-Release Phentermine/Topiramate in Severely Obese Adults: A Randomized Controlled Trial) included people aged 18 to 70 years with a BMI of 35 kg/m2 with no upper limit, a fasting blood glucose level 100 mg/dL, and a blood pressure 140/90 mm Hg. 23 Participants were stratified by sex and randomized to controlled-release phen-top (phen-top CR) 15/92 mg (n = 512), phen-top CR 3.75/23 mg (n = 241), or placebo (n = 514) for 52 weeks. The primary outcome was percentage of weight loss at week 52; analysis was by ITT with LOCF. The percentage of weight loss with high-dose phen-top was 10.9% compared with 5.1% in the low-dose phen-top and 1.6% for placebo (P < .001 for both doses compared with placebo). Absolute weight loss based on least squares mean was 12.6 kg for high-dose phen-top, 6 kg for low-dose phen-top CR, and 1.8 kg for placebo.
The CONQUER study (Effects of Low-Dose, Controlled-Release, Phentermine Plus Topiramate Combination on Weight and Associated Comorbidities in Overweight and Obese Adults) included people with a BMI of 27 to 45 kg/m2 or those with type 2 diabetes with any BMI. 24 Participants were stratified based on the gender and presence of type 2 diabetes and were randomized to phen-top CR 7.5/46 mg (n = 498), phen-top CR 15/92 mg (n = 995), or placebo (n = 994) which was continued for 52 weeks. The coprimary outcome of the CONQUER study was mean percentage change in weight and percentage of patients achieving at least 5% weight loss; analysis was by ITT with LOCF. The percentage weight loss was 7.8% (8.1 kg) with low-dose phen-top, 9.8% (10.2 kg) for high-dose phen-top, and 1.2% (1.8 kg) for placebo (P < .001 for both doses vs placebo). Approximately 21%, 62%, and 70% achieved at least 5% weight loss while receiving placebo, low-dose phen-top, and high-dose phen-top CR, respectively (P < .001 for both doses vs placebo). Treatment with phen-top CR improved blood pressure, high-density lipoprotein cholesterol, triglyceride (TG), HbA1c, and fasting glucose compared with placebo (P < .05 for all comparisons), although the absolute differences are of questionable clinical significance.
The SEQUEL study was a 1-year extension of the CONQUER trial; patients were only eligible if they completed the CONQUER study on treatment and complied with protocol requirements. Patients continued their randomized treatment from CONQUER for an additional 52 weeks. In the SEQUEL study, the high-dose group achieved a weight loss percentage of 10.5% (10.9 kg), and this benefit was also observed in the low-dose group [9.3% (9.6 kg)] compared to placebo [1.8% (2.1 kg); P < .001 for both doses vs placebo]. Nearly 80% of participants in the high-dose group attained 5% weight loss compared to 75% in the low-dose group and 30% receiving placebo (P < .001 for both doses compared with placebo). The reduction was sustained even after patients continued the treatment for the second consecutive year. However, the study investigators do acknowledge the inherent bias of the SEQUEL study conclusions since only the adherent patients of the CONQUER study were chosen to participate, thus representing a group that is already well satisfied with therapy. 25
Safety
Although the study investigators were able to document the efficacy of phen-top combination, there were many questions raised by the FDA regarding the safety of the molecule. 20 The most frequently reported adverse events associated with phen-top treatment were paresthesia (17.0%), dry mouth (16.6%), constipation (15.1%), upper respiratory tract infection (13.5%), nasopharyngitis (10.0%), and headache (9.8%). There were concerns of teratogenic risk in women with child-bearing potential receiving topiramate. A retrospective observational study named Fetal Outcome Retrospective TopiRamate ExpoSure Study (FORTRESS) was initiated by the manufacturing agency, using electronic medical health records, to determine the fetal outcome in the offspring of mothers exposed to topiramate. The data showed that exposed women had nearly a 2 times greater risk of having children with oral clefts. 26 There was also a concern about the increase in heart rate with phen-top and whether this increase could lead to increased CV risk, as seen with sibutramine. This concern was laid to rest when data analysis revealed that there were only a small number of major adverse cardiac events among patients exposed to phen-top, and these patients did not have increased heart rates. Taking these factors into consideration and the beneficial effect of phen-top on the BMI, blood pressure, and diabetic status, the CV safety of phen-top appears reasonable. Nevertheless, there is an onus on the regulators to seek data from prospective long-term studies to assess the effects of the drug on the risk of MI, stroke, CV, and all-cause mortality.
On the basis of the results of these studies, phen-top CR was approved by the US FDA for the treatment of obesity in patients with a BMI of 30 or 27 kg/m2 with at least one weight-related comorbidity, such as hypertension, type 2 diabetes mellitus, or dyslipidemia, in addition to an RCD and increased physical activity. The Phen-top CR therapy should be started at 3.75/23 mg, taken once daily in the morning, and after 14 days the dose can be increased to 7.5/46 mg. If after 12 weeks, at least 3% weight loss has not been achieved, use of the drug can be discontinued or the dose can be increased further to 11.25/69 mg for 14 days followed by a final dose increase up to 15/92 mg. Weight loss should be evaluated after an additional 12 weeks, and if 5% weight loss is not achieved, therapy should be discontinued. The Risk Evaluation and Mitigation Strategy has been put in place by the US FDA for phen-top to ensure that the benefits of the drug overcome the risks associated with it such as teratogenicity. Women of child-bearing potential are cautioned regarding the risks such as orofacial clefts to the developing fetus with the use of the drug, counseled on the use of effective contraception during drug use, and to terminate the drug if pregnancy does occur. 27 The drug is yet to receive European Medicines Agency approval. Table 2 summarizes the salient features of lorcaserin and phen-top.
Newly Approved Antiobesity Agents.

Abbreviations: ER, extended release.
Antiobesity Molecules in Clinical Development
Agents Acting Through Central Mechanisms
Bupropion–Naltrexone
Bupropion is an atypical anti-depressant that inhibits the reuptake of dopamine and noradrenaline. Although the drug was detected to have weight loss-inducing potential, the decrease in body weight was a mere 2.8 kg after 6 months of therapy, and hence the drug did not match the FDA standards as monotherapy for obesity. Bupropion additionally stimulates proopiomelanocortin firing in the arcuate nucleus of the hypothalamus which in turn releases α-melanocyte-stimulating hormone that mediates the anorectic effects. However, the stimulation of POMC also stimulates a negative-feedback loop that releases β endorphins, which suppress further POMC firing. One of the reasons attributed to the failure of bupropion as a sole agent for obesity is the negative-feedback loop activation by bupropion. This negative feedback can be suppressed by naltrexone, a µ-opioid receptor antagonist.
A randomized, placebo-controlled trial by Wadden et al in 793 patients demonstrated that weight loss was 5.1% ± 0.6% with placebo + behavioral modification (BM) versus 9.3% ± 0.4% with naltrexone–bupropion combination + BM (P < .001). 28 In a phase 3 clinical trial carried out in 1742 patients by Greenway et al, patients were randomized to receive naltrexone 32 mg + bupropion; naltrexone 16 mg + bupropion; or placebo. The mean change in body weight was −6.1%, −5.0%, and −1.3% in the 3 groups, respectively (P < .001 in both groups compared to placebo). 29 The common adverse reactions that were seen among the trial participants receiving the combination therapy included nausea, headache, constipation, dizziness, vomiting, and dry mouth. Another phase 3 clinical trial performed in 1496 patients by Apovian et al showed that patients on NB-32 combination had a weight loss superior to placebo (−6.5% vs −1.9%; P < .001). More NB-32-treated participants (P < .001) experienced ≥5% weight loss versus placebo at week 28 (55.6% vs 17.5%) and week 56 (50.5% vs 17.1%). There was also an improvement in various cardiometabolic risk markers, participant-reported, weight-related quality of life, and control of eating with NB-32. 30 The drug was in the verge of obtaining FDA approval. However, the regulatory body insisted that the manufacturer carry out long-term CV safety studies before approval. 31
Zonisamide–Bupropion
Zonisamide is an antiepileptic agent that is used in the management of partial seizures. The serendipitous discovery of zonisamide’s weight loss promoting potential in patients receiving the drug led the manufacturers to develop it for the treatment of obesity. Although the precise mechanism is yet to be elucidated, modulation of sodium channel, carbonic anhydrase inhibition, and enhancement of dopamine and serotonin transmission are said to play a role in inducing weight loss. Bupropion causes weight loss by increase in the levels of dopamine that decreases appetite. The detrimental effects of zonisamide such as depression and sedation may be overcome by its combination with bupropion. Conversely, the seizure-inducing property of bupropion could be offset by zonisamide. Combination of zonisamide with bupropion was observed to be superior to bupropion or zonisamide monotherapy in inducing weight loss in a pilot study performed among 18 obese women. 32 A phase 2b study clinical trial in 729 patients with obesity showed that the combination in 2 different doses had a weight loss that was superior to placebo (6.1% and 7.5% vs 1.4%, P < .01). 33,34 Nausea, headache, and insomnia were the main adverse events in this clinical trial with the combination. The manufacturer Orexigen is in the process of initiating phase 3 clinical trials with the fixed dose combination.
Tesofensine
Tesofensine is a monoamine reuptake inhibitor originally developed for Alzheimer disease and Parkinson disease, which was incidentally found to induce weight loss in clinical trials. The drug prevents the reuptake of serotonin, noradrenaline, and dopamine and thereby suppresses appetite and increases thermogenesis. In a phase 2 clinical trial in which patients were randomized to receive tesofensine or placebo for 24 weeks, tesofensine at a dose of 0.25, 0.5, and 1 mg showed a weight loss of 4.7, 9.1, and 10.6 kg, respectively. 35 The drug was fairly tolerated, with dry mouth, dizziness, constipation, abdominal pain, and nausea being the common side effects. A modest increase in heart rate up to 8 bpm has been recorded with the higher dosage of tesofensine (1 mg). 36 This clinical trial that was published in Lancet came under the scrutiny of the Danish drug regulatory agency in 2011, which cited lapses in trial methodologic procedures during a site inspection. 37 The drug is yet to enter confirmatory phase of clinical trial development.
Velneperit
It has been well recognized over the last 2 decades that neuropeptide Y (NPY) stimulates food intake, reduces energy expenditure, and increases body weight by activating NPY receptors, Y1, and Y5 present in the hypothalamus. Velneperit is a Y5 receptor antagonist that prevents the binding of NPY to the Y5 receptors and thus decreases hunger and controls energy balance. The RCD study that was carried out in 656 patients randomized them to receive velneperit 800/1600 mg or placebo along with RCD regimen. Patients receiving 800 mg lost an average of 3.8 kg compared to the placebo group with a mere 0.8 kg (P < .0001). In the 800-mg group, 35% of the patients lost greater than 5% of body weight, while in the placebo group only 12% showed >5% reduction. The low-calorie diet (LCD) study which also evaluated velneperit at a dose of 1600 mg showed a weight loss of 7.1 kg in the study group over placebo (4.3 kg). Among the study patients, 52% in the velneperit group and 35% in the placebo group had a weight loss greater than 5%. 33 Nasopharyngitis, sinusitis, upper respiratory infection, sinusitis, and headache were the common adverse events observed. The lack of significant benefit of velneperit over placebo in the LCD study has raised several questions on the future prospects of the drug and the need for further drug development.
GSK1521498
Opioid neurotransmission plays a stellar role in behaviors reinforced by primary rewards, especially fatty or sugary foods. 38 Opioid antagonists have been shown to reduce short-term food intake and affective or subjective pleasantness of palatable foods in healthy participants. 39 GSK1521498 is a µ-opioid receptor inverse agonist that is being investigated primarily for the treatment of overeating behavior in obesity. A study in healthy volunteers showed that GSK1521498 selectively reduced sensory hedonic ratings of high-sugar and high-fat dairy products and caloric intake of high-fat/high-sucrose snack foods. 40 Another phase 1 study showed the drug to have an acceptable safety profile and pharmacokinetics when the drug was administered for 10 days in multiple doses. 41 It remains to be seen whether the decreased food craving would translate into significant weight reduction when the drug is given for extended periods of time.
Agents Acting Through Peripheral Mechanisms in the Gut
Cetilistat
Cetilistat is a pancreatic lipase inhibitor that is undergoing phase III clinical trials. Pancreatic lipase is a key enzyme that causes breakdown of TGs into free fatty acids that are absorbed in the intestine. Inhibition of pancreatic lipase by cetilistat reduces the conversion of TGs into free fatty acids, and thus the TGs are excreted unchanged in the urine. Cetilistat was administered in 3 different doses of 60, 120, and 240 mg twice daily and compared with placebo. The weight loss obtained was 3.3, 3.5, and 4.1 kg, respectively, at the end of 12 weeks. All 3 groups had a weight loss that was almost similar and was statistically superior to the placebo group. 42 Although cetilistat is similar to orlistat in its mechanism, it is claimed to have a superior safety profile that was demonstrated in a phase 2b clinical trial. 42,43
Liraglutide
In the last decade, glucagon-like peptide 1 (GLP-1) analogs have become a frontline option in the treatment of patients with diabetes. Liraglutide has been the latest of the GLP analogs approved for lowering of blood glucose in diabetes. The GLP-1 enhances insulin secretion, delays gastric emptying, and suppresses appetite. This results in weight loss and decreased energy intake. 44 In a study that evaluated liraglutide in 4 different dosages of 1.2, 1.8, 2.4, or 3 mg, the mean weight loss obtained in each group at the end of the study was 4.8, 5.5, 6.3, and 7.2 kg, respectively. In the same study, patients in the placebo arm showed a weight loss of 2.8 kg, while those on standard therapy orlistat 120 mg twice daily had a weight loss of 4.1 kg. 45 Liraglutide also showed an acceptable safety profile with the most common adverse reactions experienced being nausea and vomiting. Several clinical trials are currently underway (phases 2 and 3) to test the safety and efficacy of liraglutide as an antiobesity molecule. 46 A meta-analysis showed that treatment with GLP-1 R agonists reduces body weight in both diabetic and nondiabetic patients with obesity. 47 The main limitation with both liraglutide and exenatide is the need for parenteral formulation. Since GLP analogs are currently being explored for their CV risk-lowering potential, this may be an added advantage apart from the weight loss sustained due to drug therapy.
PF-04620110
Alteration in fatty acid and TG metabolism is one of the factors implicated in the pathophysiology of obesity. The final step in triacylglycerol formation is catalyzed by the enzyme diacylglycerol acyltransferase 1 (DGAT1), a member of the large family of membrane-bound O-acyltransferases. The DGAT1 causes reesterification of dietary TG in the intestine. PF-04620110 is a selective pharmacological inhibitor of DGAT1. In C57/BL6J mice, acute DGAT1 inhibition alters the temporal and spatial pattern of dietary lipid absorption. The DGAT1 inhibition causes an enrichment of polyunsaturated fatty acids within the TG class of lipids. 48 Phase 1 clinical trials of PF-04620110 have been conducted in healthy volunteers, patients with obesity, and diabetic patients, but the results of these trials could not be retrieved. 49 It is too early to conclude that DGAT inhibition could lead to significant weight reduction; nevertheless, DGAT appears to be evoke interest as an antiobesity and antidiabetic molecule.
Agents Acting Though Metabolic Pathways in Liver and Adipose Tissue
Beloranib
It is a methionine aminopeptidase 2 (MetAP2) inhibitor that has been found to be effective in preclinical studies. The drug inhibits MetAP2 that reduces production of new fatty acid molecules by the liver and helps to convert stored fats into useful energy. 50 A study comparing 3 different intravenous doses of beloranib given twice weekly showed that the median weight reduction was −3.8 kg with beloranib 0.9 mg/meter square versus −0.6 kg with placebo. 51 The drug is currently undergoing phase 2 clinical trials to establish the safety, efficacy, and pharmacokinetics of the drug in patients with obesity. 52 One of the limitations with this molecule is the need for parenteral formulation—subcutaneous formulation and intravenous formulation are presently being studied.
Resveratrol
Sirtuin 1 (SIRT1) is an NAD-dependent deacetylase that is upregulated in the setting of calorie restriction in rodents. The SIRT1 binds to PPAR-δ and suppresses the expression of PPAR-δ genes that are responsible for fat storage. It also activates oxidative metabolism and fatty acid oxidation by virtue of its interaction with peroxisome proliferator-activated receptor α coactivator (PGC-1α). 44 Resveratrol is an allosteric activator of SIRT1 that has been demonstrated to protect against insulin resistance and increase the mitochondrial activity in brown adipose tissue and skeletal muscle from preclinical studies. In a double-blind crossover study done in 11 patients with obesity, 30 days of resveratrol supplementation induces metabolic changes such as activation of adenosine monophosphate-activated protein kinase, increased SIRT1 and PGC-1α protein levels, increased citrate synthase activity without change in mitochondrial content, and improved muscle mitochondrial respiration on a fatty acid-derived substrate. 53 Besides resveratrol, several other sirtuin activators are in early clinical development for obesity.
ALS-L1023
The adipose tissue is a highly vascularized organ that is subject to extensive modification in obesity. The growth of adipose tissue is remarkably affected by adipogenesis, angiogenesis, and extracellular matrix (ECM) remodeling. Modulation of ECM also enhances the maturation of microvessels, and this is regulated by matrix metalloproteinase (MMP). 54 –56 Active tissue remodeling is associated with adipogenesis and increased MMP expression. The serum concentrations of growth factors such as vascular endothelial growth factor (VEGF), fibroblast growth factor, soluble VEGF receptor 2 (VEGFR2), hepatocyte growth factor, angiopoietin-2, and angiogenin are higher in overweight or obese individuals than in normal-weight individuals. 57 ALS-L1023 is a drug obtained from Melissa officinalis that has exhibited enhanced antiangiogenic and MMP inhibitory activities. The messenger RNA-2 expression of angiogenic factors, MMPs, and the resulting adipose tissue growth were modulated by ALS in obese mice. 58 As per the manufacturer Angiolabs, ALS-L-1023 has already reached phase 3 clinical trial development in Korea. 59 The Ob-X is a mixture of 3 herbs M officinalis L (Labiatae), Morus alba L (Moraceae), and Artemisia capillaris Thunb (Compositae). Administration of Ob-X to ob/ob mice for 5 weeks produced a significant reduction in body weight gain by 27% compared with control. The Ob-X also significantly decreased visceral adipose tissue mass by 15%. 60 The Ob-X is currently being promoted in Korea as a dietary supplement. Thus, it appears that inhibition of MMP and angiogenesis could be a potential target for obesity. Table 3 is an overview of the drugs that are in the pipeline for the treatment of obesity.
Antiobesity Molecules in Clinical Development.
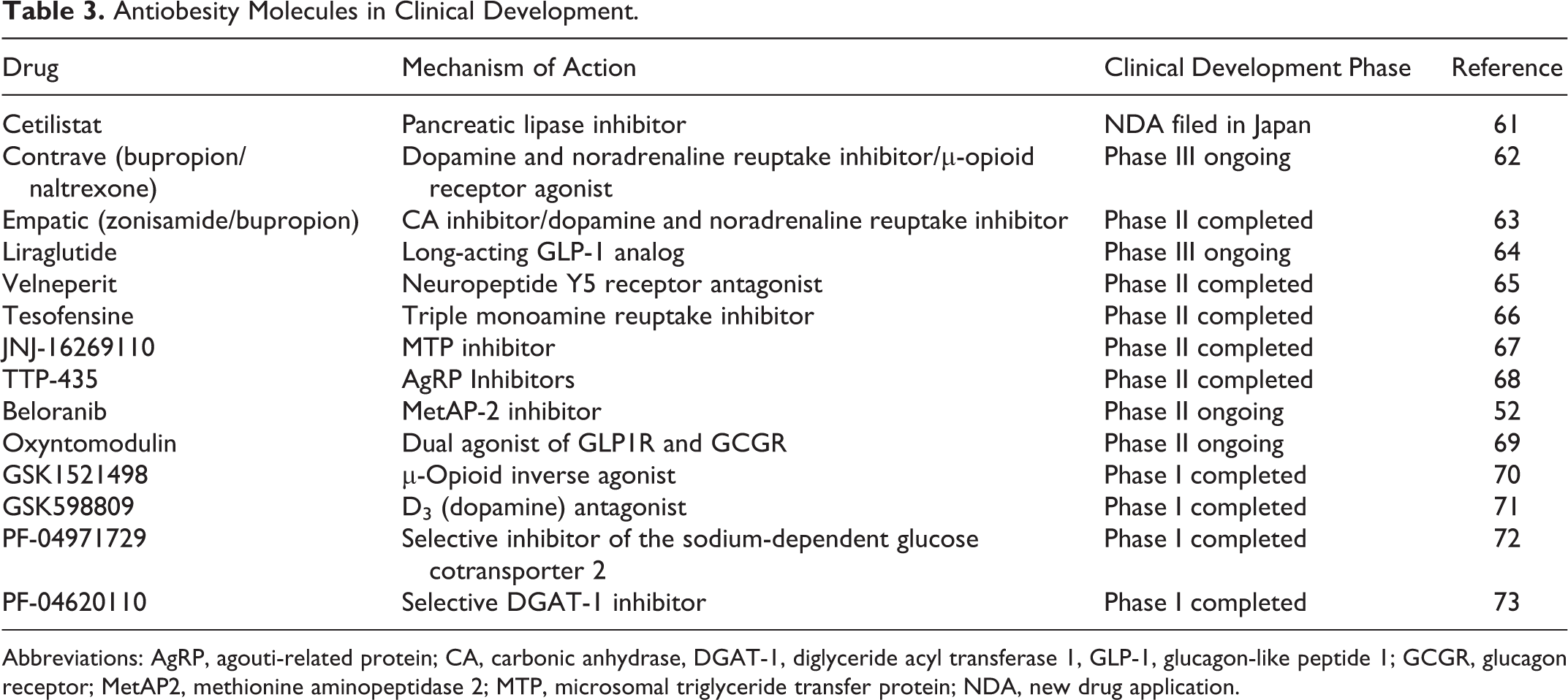
Abbreviations: AgRP, agouti-related protein; CA, carbonic anhydrase, DGAT-1, diglyceride acyl transferase 1, GLP-1, glucagon-like peptide 1; GCGR, glucagon receptor; MetAP2, methionine aminopeptidase 2; MTP, microsomal triglyceride transfer protein; NDA, new drug application.
Obesity Paradox
Although obesity is a strong and well-known risk factor for CVD, there have been some intriguing findings from different investigators that demonstrate the contrary that has been termed obesity paradox. For instance, a study by Uretsky et al in 22 576 treated patients with hypertension showed that all-cause mortality was 30% lower in overweight and patients with obesity, despite the inadequate control of their blood pressure. 83 These findings were replicated in another study performed in 3975 patients, which showed a U-shaped relationship between all-cause, CV and non-CV mortality, and BMI, 84 thereby leading to the hypothesis that obese patients with hypertension have a paradoxically better prognosis. The obesity paradox was also demonstrated in studies that were performed in patients with heart failure. 85 A meta-analyses by Flegal et al also showed that overweight was associated with lower all-cause mortality, while grades 2 and 3 obesity was associated with higher all-cause mortality. 86 This paradoxical relationship is also observed in patients with heart failure having obesity, where higher BMI has been linked with lower mortality. The risk of mortality was lower for every 5-unit increase in BMI (P < .001). 87 It is not known whether weight loss-lowering strategies with the new and emerging agents would cause a difference to this obesity paradox. Nevertheless, there is ample evidence linking weight loss strategies with proven CV benefits. 88 There is a definite need to explore these issues further, and at the moment with our current level of understanding, the most reasonable strategy is to continue achieving weight reduction through pharmacologic and nonpharmacologic therapy.
Challenges in Obesity Drug Development
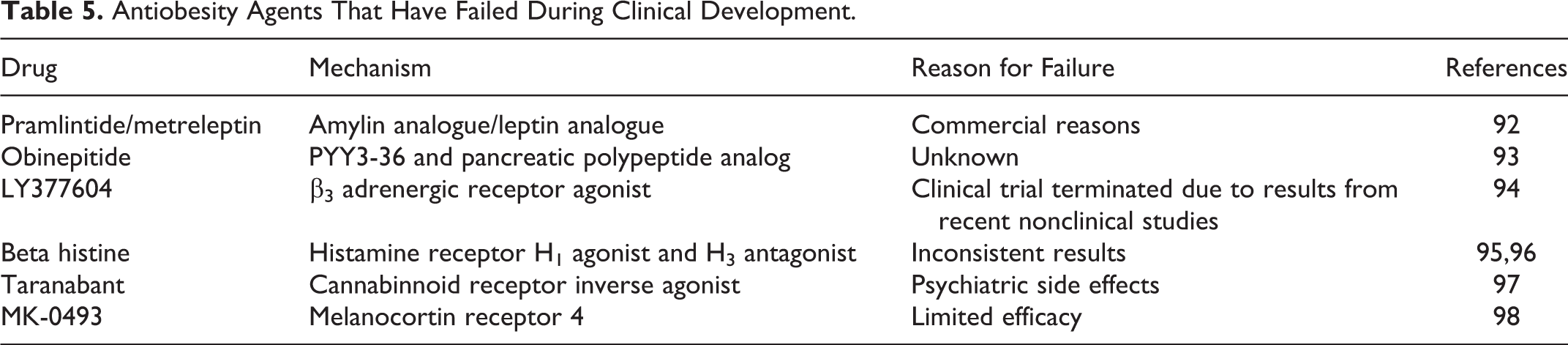
There is almost no other area in drug development, which is replete with as many failures and withdrawals as seen in obesity (Tables 4 and 5). Some of the major challenges in obesity drug development include the regulatory body’s expectations on the risk benefit profile being far superior to other drug classes, considering the high number of drug withdrawals in the past. The lack of widespread reimbursement options for an antiobesity molecule is another serious possible reasons for manufacturers to stay away from antiobesity drug development. 99 The reluctance by many physicians to consider obesity as a disease but more as a risk factor may make them even more hesitant to prescribe antiobesity drug therapy. The regulations also stipulate that antiobesity agents apart from causing significant weight reduction should also cause improvement in blood pressure, lipids, glycemia, or other beneficial outcomes that are commensurate with the degree of weight loss. 100,101
Antiobesity Drugs That Have Been Withdrawn From the Market.

Abbreviation: FDA, Food and Drug Administration.
Antiobesity Agents That Have Failed During Clinical Development.
Conclusion
In spite of the checkered past of obesity drug development, with so many promising molecules ending in failure, the search is still on for the elusive molecule that can target obesity and reduce its consequences substantially. Although the regulatory agencies have rightly expressed concern on certain antiobesity molecules and refused marketing approval in lieu of safety issues, this has not blunted the enthusiasm of pharmaceutical companies to discover new molecules, since the market for an antiobesity drug appears too alluring to resist. Lorcaserin and phen-top are the 2 drugs that have been recently approved for obesity. Although not compared from head-to-head clinical trials, phentermine appears to have greater efficacy than lorcaserin. Lorcaserin may be a safer option, more so in women of child-bearing potential due to the teratogenic risk attributed to topiramate. The need of the hour is to gather data from prospective clinical trials that evaluate the long-term CV outcomes with the use of the newly approved agents. Obesity, being a complex multifactorial disease with several pathways disturbed, would require a molecule or a combination of molecules to act at multiple pathways/targets if it is to produce clinically significant weight reduction. There are several instances of older drugs approved for other indications being explored such as zonisamide, naltrexone, and bupropion. Some of the new targets that have evoked great interest in the last few years include SIRT1, GLP-1, aminopeptidase-2, sodium glucose cotransporter, and NPY, but much needs to be done before effective drugs are discovered that target these receptors. It can be safely surmised that as our knowledge of the pathophysiology of obesity improves, it may be possible to discover the “holy grail” of antiobesity drugs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

