Abstract
A large body of evidence implicates the immune system in the pathogenesis of atherosclerosis. Both active and passive immunizations have been tested as immunomodulation strategies to confer protective effect against atherogenesis. This review focuses on the current knowledge of the complex role and the potential for immune modulation therapy via active immunization for atherosclerosis.
Introduction
Atherosclerosis is currently regarded as a chronic immune-mediated inflammatory disease of the arterial wall as evidenced by the presence of many components of innate and adaptive immune system such as dendritic cells, B- and T-lymphocytes, macrophages, toll-like receptors, immunoglobulins, inflammatory cytokines, and complement in atherosclerotic plaques. Given the role of immune dysfunction in the pathophysiology of atherogenesis, the idea of an immunomodulatory approach such as active immunization has been developed to modulate atherosclerosis. This concept of “active immunization against atherosclerosis” was first suggested in the 1950s. 1 However, only in the past decade have we observed a vigorous effort in this field, resulting in better understanding of the underlying pathophysiological role of immune responses in atherosclerosis. Many investigators have reported promising results using active immunization to reduce atherosclerosis in preclinical studies. These proof of concept of preclinical studies have raised the possibility that such an approach might be feasible in humans.
Search for Antigens in Atherosclerosis
The most important element of immunization approach is to know which antigens are responsible for the disease. The search for antigens is relatively straightforward in the field of infectious disease, because (1) most disease-causing pathogens are well defined and (2) there has been a long history and experience of successful vaccine development in the field. The antigens used in vaccine formulation also evolved with the advancement of biomedical technology, ranging from live-attenuated virus, nonliving inactivated organism, subunits or toxins of organism, and genetically engineered products (such as protein and DNA plasmid) to synthetic peptides. 2,3
The search for antigens that can be used in vaccine formulation for atherosclerosis is not easy, because (1) although lipids are generally considered to play an important role in atherogenesis, the exact causes of atherosclerosis are not completely known and are likely to be multifactorial; (2) although given the multifactorial causes for atherosclerosis, a comprehensive knowledge of the immune responses against various antigens in atherosclerosis is lacking; (3) unlike infectious diseases, atherosclerotic disease has a long latency before clinical manifestation, making the ideal timing for intervention difficult to determine. Despite these obstacles, many antigens have been implicated in atherogenesis. If used as antigens via active immunization, some promote atherosclerosis, whereas others are atheroprotective (Table 1).
Antigens That Have Been Tested in Preclinical Active Immunization Studies.

Abbreviations: ApoB, apolipoprotein B; LDL, low-density lipoprotein; MDA, malondialdehyde.
Active Immunization With Low-Density Lipoprotein or Apolipoprotein B-100-Related Peptides to Reduce Atherosclerosis
The use of lipoprotein as an antigen via active immunization to reduce atherosclerosis was suggested in the 1950s 1 but major efforts in this direction were not started until mid-1990s. Many laboratories have reported that active immunization with low-density lipoprotein (LDL; in native or modified form) or cholesterol-related antigens reduced atherosclerosis in hypercholesterolemic animals. 4 –9 These promising results provided proof-of-concept evidence that active immunization with lipoprotein particles can reduce atherosclerosis. The LDL contains heterogeneous molecules such as apolipoprotein (apo), cholesteryl ester, triglyceride, and phospholipids. At the time of these publications, the exact nature of immunogenic epitopes within LDL was not known.
The use of whole LDL particle as an antigen is acceptable in preclinical experiments to establish proof-of-concept evidence, but it may not be acceptable if ever this approach has an opportunity to move into clinical practice, due to many limitations such as (1) safety risks to isolate a large amount of LDL for formula preparation, (2) stability of LDL as an antigen, and (3) unknown immunogenic epitopes in LDL precluding advancement and refinement of vaccine preparation. Given that LDL has been used as an antigen in the majority of the investigations, enormous efforts have been dedicated to identify potential antigenic epitopes within apoB-100, the major protein component in LDL, which may be used as antigens to achieve atheroprotective effects via active immunization.
First, a library of 302 peptides (20 amino acids long with a 5-amino acid overlap with the preceding sequence) using sequences from the human apoB-100 molecule was established for screening peptide antibodies that can be detected in pooled human plasma. 10 The logic behind this approach was that positive antibody response against peptides indicates that such peptides were immunogenic in humans and could be responsible for modulating atherogenesis. The screening process identified 102 peptides that can be recognized by antibodies. These peptides were then subjected to further screening via active immunization in a series of animal experiments to determine their atheromodulating potential. Active immunization with vaccine formulations using several such peptides, such as p2, p143 and p210, as antigens resulted in a 40% to 70% decrease in atherosclerosis and reduction in plaque inflammation in hypercholesterolemic mice without a significant change in circulating cholesterol levels. 11,12
Mechanisms of Action Underlying Active Immunization Using ApoB-100 Peptides
Most of the work related to this aspect was performed using p210 as an antigen in the vaccine formulation. Humoral immunity was initially hypothesized to be responsible for the protective effect of active immunization; however, data from the published literature did not support this hypothesis. Fredrikson et al reported that the atheroprotection of apoB peptide immunization can occur without an increase in peptide-specific immunoglobulin (Ig) G. 13 Intranasal immunization with p210 elicited p210 IgG antibody, but the titers were not correlated with the lesion size. 14 We also reported that mice immunized with p210 vaccine developed smaller aortic atherosclerotic lesions with lower circulating p210 IgG antibody titer when compared to mice receiving adjuvant. Additionally, B cells from p210 immunized mice adoptively transferred to nonimmunized donor did not convey the atheroprotection effect. 15 Taking these together, it appears that humoral immunity is not likely to be an important mediator of the atheroprotective effect of active immunization with p210.
Cellular immunity was also investigated as an underlying mechanism. Immunization with p210 in apoE(−/−) mice-activated CD8+ T cells, reduced dendritic cells at the site of immunization and within the plaques, decreased macrophage immunoreactivity in plaques, and significantly reduced aortic atherosclerosis compared to phosphate-buffered saline (PBS) or adjuvant controls. Adoptive transfer of CD8+ T cells from p210 immunized mice into naive, nonimmunized mice recapitulated the atheroprotective effect of active immunization. The CD8+ T cells from p210-immunized mice developed a preferentially higher cytolytic response against p210-loaded dendritic cells in vitro. Adoptive transfer of CD4+CD25+ T cells from p210 immunized donors did not provide atheroprotection in the naive recipient mice. 15
In addition to the above–mentioned report that CD8+ T cells mediated the atheroprotective effect of p210 immunization, other investigators also reported change in CD4+ T cell functions with p210 immunization. In male, apoE(−/−) mice immunized with an immunization schedule and p210 preparation similar to ours, p210 immunization reduced atherosclerosis associated with a functional CD4+CD25+ T cell response. The increase in CD4+CD25+ T cells and the associated atheroprotection by p210 immunization were ablated by administration of CD25 blocking antibody. 16 Several limitations, as acknowledged by the authors, existed in their study including lack of the control group of mice receiving adjuvant and the possibility of CD25 blocking antibody ablating both CD25+ effector and regulatory T cells together. 16 Klingenberg et al introduced p210 immunization differently by generating a recombinant protein by fusing p210 with cholera toxin B (p210-CTB) and delivered it to female apoE(−/−) mice, intranasally twice weekly for 12 weeks. The control group received ovalbumin peptide fused with CTB (OVA-CTB) or PBS. This intranasal approach reduced atherosclerosis in aortic sinuses in mice receiving p210-CTB when compared to the other 2 groups. There was no difference in FoxP3, interleukin (IL)-10, or TGF-β mRNA expression in aorta between p210-CTB and OVA-CTB groups and no difference in the numbers of FoxP3+ cells in aortic lesions or CD4+FoxP3+ T cells in lung mucosa. Splenic CD4+ T cells from p210-CTB immunized mice contained higher percentage of IL-10+ subset and were functional to suppress the effector CD4+ T cells. Hence, the authors suggested that the type 1 regulatory T cell response (CD4+IL-10+ T cells) could be responsible for the beneficial effect of intranasal immunization with p210-CTB. 14 However, their study did not provide direct evidence to support such notion nor did they characterize CD8+ T cell response. Another group of investigators delivered a mixture of apoB-100 peptides (p210, malondialdehyde (MDA)-p210, and p240, 3.3 μg each, total 10 μg) for 2 weeks subcutaneously via an implanted miniosmotic pump in young mice and reported such treatment reduced atherosclerotic lesions in aortic sinus 10 weeks later when compared to those receiving control peptide infusion. Such protection could be recapitulated by delivering p210 peptide alone suggesting that p210 was the major peptide responsible for the atheroprotection among the 3 tested peptides. This approach also retarded the progression of established atherosclerotic lesions in old female mice. 17 This effect was associated with less activation of CD4+ T cells as evidenced by reduced interferon γ, IL-10, and IL-4 cytokine production upon stimulation ex vivo and increased CD4+CD25+FoxP3+ subset of T cells in lymph nodes. Similar to the study by Wigren et al, this group of investigators also reported that ablation of CD25+ T cells by CD25-depleting antibody abrogated the atheroprotection by apoB-100 peptides infusion. Figure 1 summarizes the aforementioned mechanisms responsible for the atheroprotective effect of p210 immunization.
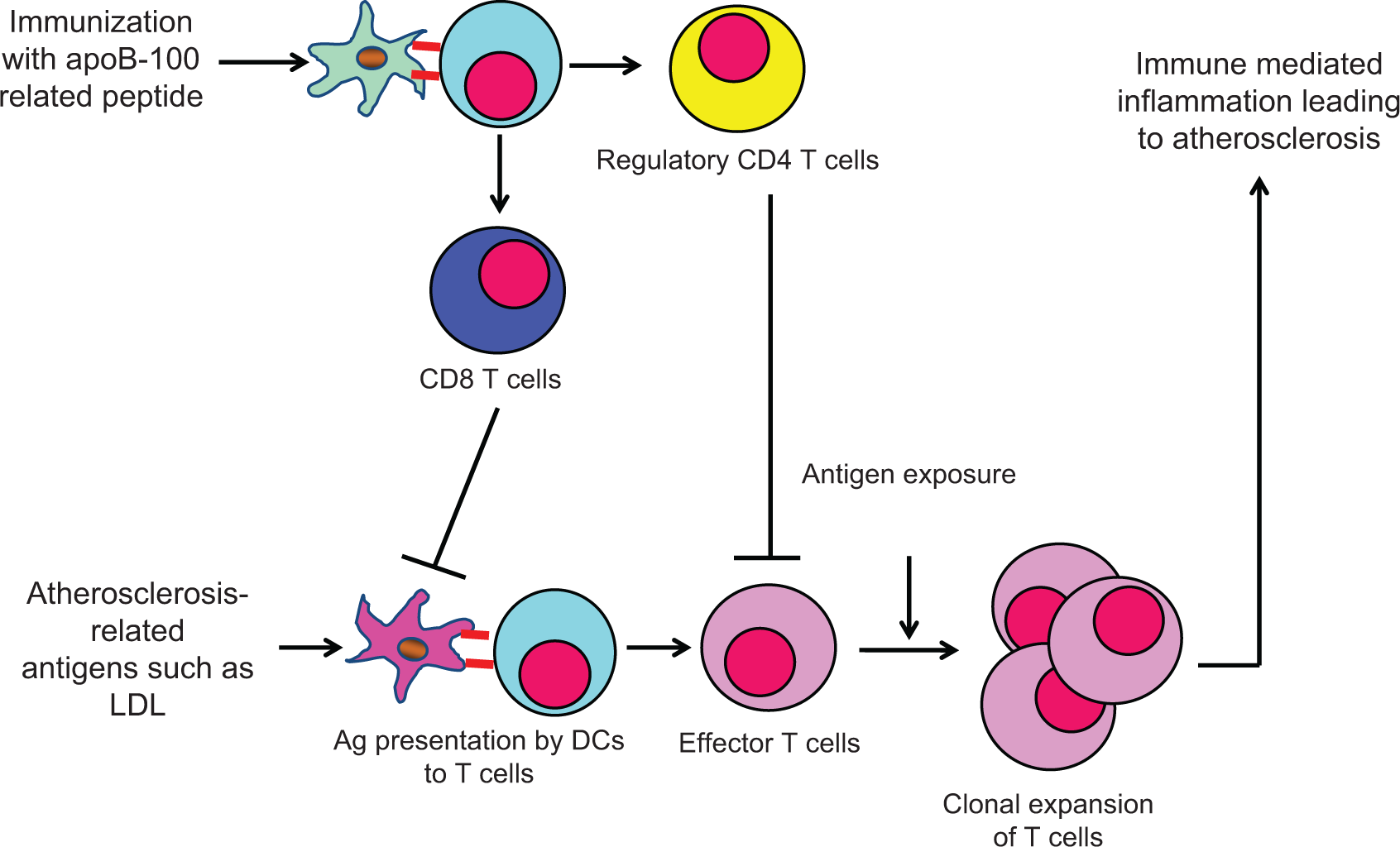
Upon encountering atherogenic antigens, the dendritic cells present such antigens to naive T cells with subsequent generation of effector T cells that eventually lead to inflammation in the arterial walls. Immunization with apoB-100 related peptide, especially p210, a subset of CD8+ T cells, are elicited and confer negative effect on dendritic cells to dampen atherogenic immune response. Regulatory CD4+ T cells can also be elicited and suppress effector T cells.
Taking these together, these data suggested that p210 is a strong candidate antigen for the development of active immunization strategy into future human application, given that immunization with p210 consistently reduces atherosclerosis even when it is administered via different routes and in different forms. It is difficult to reconcile these different cellular immune responses elicited by p210, but they may be due to the difference in (1) the form of p210 delivered—carrier conjugated, recombinant with CTB, or free form; (2) route of delivery—subcutaneous versus mucosal; (3) doses of p210; and (4) length of p210 delivery—long-term depot effect of carrier-conjugated p210 in subcutaneous tissue versus infusion for 2 weeks. It is also possible that p210 immunization elicits both CD4+ and CD8+ T cell responses, and both atheroprotective and atheropromoting T cells exist within each T cell type. The overall beneficial effect of p210 immunization depends on the balance of quality, quantity, and timing of appearance of all the elicited immune responses. Additional studies are needed to test these hypotheses.
What makes the mechanistic study more challenging is a recent observation reporting that p2, p45, or p210 immunization elicited quite different immune responses. Immunization with p2 induced the strongest CD4+CD25+FoxP3+ T cell response, whereas p210 induced the least. Both p2 and p45 elicited CD8+CD25+FoxP3+ T cell response, but p210 decreased it when compared to the carrier control group. 18 Hence, what has been observed in p210-related studies cannot be universally applied to the studies using other peptides.
Active Immunization with Antigens Other Than LDL or ApoB-100-Related Peptides to Reduce Atherosclerosis
Witztum and his colleagues described a natural IgM antibody recognizing the epitopes in oxidized LDL
19,20
and phosphorylcholine (PC) headgroups on the surface of apoptotic cells and inhibiting uptake of oxidized LDL and apoptotic cells in macrophages.
19,21,22
Given that anti-phosphorylcholine antibodies protect against infection from
Hansson and his colleagues obtained T cell clones after immunizing human apoB-100 transgenic mice with human oxidized LDL. These T cells recognized only native LDL or purified apoB-100 and showed uniform expression of a single T cell receptor (TCR) variable β chain (TRVB31). Immunization with a peptide from TRVB31 generated anti-TRVB31 antibodies that blocked T cell recognition of apoB-100 and reduced atherosclerosis. 26
An orally administered DNA vaccine against CD99 or VEGFR2, as reported by Kuiper and his colleagues, was able to generate antigen-specific cytotoxic CD8+ T cells with associated decrease in murine atherosclerosis. 27,28
Active immunization against cholesteryl ester transfer protein (CETP), a key enzyme involved in the HDL metabolism, induced neutralizing antibodies against CETP and resulted in marked elevation of HDL with concomitant reduction in atherosclerosis in hyperlipidemic rabbits, 29,30,31 but human studies were disappointing. 32
Heat shock proteins (HSPs), classified based on their molecular weight, have been implicated in atherogenesis 33 –36 but immunizing with different HSPs resulted in different effects on atherosclerosis. Immunization with HSP 65 was reported to induce atherosclerotic lesions in several reports, 37 –39 whereas others reported reduced atherosclerotic lesions. 40,41 Atheroprotective effect by immunization with apoB-100 and HSP60 peptides together was also reported. 42,43
What is Missing in Developing Vaccines Against Atherosclerosis?
Although preclinical experiments indicate that the strategy of active immunization against atherosclerosis is feasible, the development of a working vaccine for human application is still in its infancy. Many components of a working vaccine formulation are still unknown requiring more in-depth exploration. These include the delivery system for vaccine, choice of carrier to conjugate peptide antigen, adjuvant, route, dose, or schedule. Even more difficult questions to answer are (1) what is the proper population to vaccinate, (2) what are the efficacy end points and how to evaluate them, and (3) given atherosclerosis is a chronic disease, what is the proper timing to evaluate end points.
Our laboratory has conducted experiments to answer some of these questions. Using a single native apoB-100 peptide, p2, we observed that active immunization with p2 was not only effective in reducing atherosclerosis when given early in life before atherosclerosis formed but was also able to retard the progression of atherosclerosis when given after atherosclerosis had been established. 44
Can we Apply Preclinical Data to Human Application?
Although promising results from preclinical studies indicated that active immunization strategy reduces atherosclerosis, whether this approach in humans has similar beneficial effect still awaits clinical trials. We believe active immunization with apoB-100-derived peptides may play a role in reducing atherosclerosis in humans. Murine atherosclerotic plaques bear similar pathological features to those from humans. 45,46 Immune responses to LDL or apoB-100 exist in both murine models of atherosclerosis and humans. 47 –50 Given these similarities between murine and human atherosclerosis, we are cautiously optimistic that active immunization with apoB-100 peptides, which is beneficial in reducing murine atherosclerosis, would provide similar beneficial effect in human atherosclerosis.
Conclusions
The concept of an atherosclerosis vaccine for the prevention and treatment of atherosclerotic cardiovascular diseases is extremely appealing. Scientists in this field have accumulated encouraging preclinical data and showed that many different antigens, especially LDL-related antigens, have the potential to be incorporated into vaccine formulation to reduce atherosclerosis. As summarized earlier, this is only a beginning of the endeavor to advance such knowledge from preclinical area to clinical application. The search for an appropriate vaccine formulation for atherosclerosis will be a long-term journey but highly beneficial and rewarding from global health care point of view if it succeeds.
Footnotes
Authors’ Note
KYC and PKS are co-inventors on patents for immune modulation therapies for atherosclerosis along with collaborator, Dr Jan Nilsson of Sweden.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Cedars-Sinai Medical Center has received research grant support from Cardiovax in support of projects related to vaccine research.
