Abstract
Introduction:
Limited data exist comparing the efficacy and safety of bumetanide- or metolazone-based diuretic regimens to furosemide in acute heart failure (HF). Our purpose was to evaluate the comparative effect on urine output (UO) and renal function between these regimens.
Methods:
A retrospective study of hospitalized HF patients treated with continuous infusion furosemide (CIF), combination furosemide plus metolazone (F + M), or continuous infusion bumetanide (CIB). Primary end points were between regimen comparisons for change in mean hourly UO versus baseline and incidence of worsening renal function.
Results:
Data on 242 patients with acute HF (age 58 ± 12 years, 63% male, left ventricular ejection fraction 38% ± 17%) were analyzed (160 CIF, 42 F + M, 40 CIB). The mean duration of diuretic regimens was 41 ± 32 hours. Compared to baseline, all regimens increased mean hourly UO (P < .0001 for all), with greater increases with F + M (109 ± 171 mL) and CIB (90 ± 90 mL) compared to CIF (48 ± 103 mL; P = .009). Incidence of worsening renal function was not different between regimens; however, blood urea nitrogen (BUN) tended to increase more with F + M (4.4 ± 9.8 mg/dL) and CIB (4.3 ± 9.7 mg/dL) than CIF (1.8 ± 10.8 mg/dL), P = .09. The incidence of hyponatremia was higher with F + M and CIB. Differences in UO, BUN, and hyponatremia were retained in the subgroup analysis limited to patients with baseline serum creatinine <1.5 mg/dL, where renal function between the groups was not different.
Conclusion:
Compared to CIF, F + M or CIB was associated with greater increases in UO. No difference in the incidence of worsening renal function was found; however, electrolyte abnormalities may be more prevalent when furosemide is combined with metolazone or when bumetanide is used. These therapeutic differences warrant prospective study.
Introduction
Patients with heart failure (HF) are frequently hospitalized for acute decompensation, most often associated with volume overload and elevated filling pressures. 1 In these patients, diuretic therapy is considered first line, according to American College of Cardiology/American Heart Association guidelines for the management of HF. 2 However, in patients who present with more advanced stages of HF or those who have been on chronic diuretic therapy, administration of intermittent loop diuretics may fail to control intravascular volume status despite the use of escalating doses, a phenomenon known as diuretic resistance. 3 Several different approaches to the use of diuretics have been suggested in guidelines and are commonly used in an effort to enhance diuresis in these instances. This includes increasing the frequency of administration or initiating a continuous infusion of a loop diuretic, further escalating dose by changing to a more potent loop diuretic (ie, bumetanide), or combining a loop diuretic with a thiazide diuretic. 4
Increasing the frequency of administration or utilizing a continuous infusion reduces intervals of subtherapeutic diuretic concentrations, which prevents periods of relative inadequate blockade of sodium reabsorption following intermittent dosing. Continuous infusions also eliminate periods of compensatory sodium retention. 3 Escalating the dose of loop diuretic by switching to bumetanide, a more potent (on a mg per mg basis) loop diuretic, may also enhance natriuresis and diuresis; however, this assumption has not been formally studied. Combination diuretic therapy is postulated to prevent or attenuate postdiuretic sodium retention due to the longer half-lives of thiazides in relation to loops. 4 In addition, thiazides may potentiate loop diuretics by inhibiting sodium reabsorption at the distal convoluted tubule, blocking the effects of compensatory hypertrophy and hyperplasia of distal tubular cells, which occur with chronic loop diuretic exposure. 3
The recent Diuretic Optimization Strategies Evaluation (DOSE) trial provided insight into 2 of these methods, comparing intermittent bolus versus continuous infusion administration of furosemide and dose escalation (high doses vs low doses) of furosemide. The study found no major differences in improvement of symptoms, changes in renal function, and urine output (UO) between intermittent and continuous infusion administration. 5 However, higher doses of furosemide were associated with greater diuresis at the expense of more transient worsening of renal function. The DOSE trial is widely considered a landmark study for guiding acute diuretic use in HF, however gaps remain. It did not evaluate escalating diuretic dose by switching to a more potent diuretic, the efficacy of adding metolazone, or the comparative efficacy of each treatment approach. Few studies have evaluated the individual efficacy and safety of these diuretic-enhancing regimens in patients with acute HF, and none has attempted to compare them to each other.5–16 As a reflection, there are no recommendations for the selection of more aggressive diuretic regimens in the setting of acute HF. The purpose of this exploratory study was to evaluate for potential signals of clinical differences between these common therapeutic regimens used for enhancing diuresis.
Methods
Study Design
A retrospective cohort study was conducted using data from patients hospitalized for acute HF who initially received intravenous intermittent bolus furosemide therapy upon admission to the hospital and were subsequently escalated to one of the following “more aggressive” refractory diuretic regimens: (1) continuous infusion furosemide (CIF), (2) combination of furosemide plus metolazone (F + M), or (3) switched to continuous infusion bumetanide (CIB). The choice of diuretic medications and administration regimen were solely at the discretion of the primary treating physician. Data from patients admitted to the intensive cardiac care unit (ICCU) of Los Angeles County and University of Southern California (LAC + USC) Medical Center with acute congestive HF between July 2007 and May 2010 were eligible for inclusion in the study. The study was approved by the LAC + USC Institutional Review Board.
Patient Population
Patients aged 18 years and older who received at least 2 separate bolus doses of furosemide without additional intervention related to diuresis and then were prescribed a “more aggressive” diuretic regimen usually reserved for patients considered refractory to intermittent bolus furosemide (as described above) were included in the study. The diuretic-enhancing regimen must have been continued for at least 6 hours. At our institution, the usual dose of furosemide when administered by intermittent bolus ranges from 20 to 80 mg given every 12 hours. Those patients who were transferred out of the ICCU received more than one of these diuretic-enhancing regimens during a hospitalization, had a baseline serum creatinine (SCr) ≥5.0 mg/dL, or were placed on mechanical renal replacement therapy were excluded from the study.
Data Collection
Data were extracted using the ICCU Eclipsys computer system and verified with the paper charts when necessary. The ICCU utilizes a computerized system for daily records of each patient, which includes all information related to medication orders and administration, laboratory tests, hourly clinical (vital statistics, hemodynamics, fluid balance, etc), and nursing notes. Data collected that were related to the study objectives included baseline demographics (age, admission weight, height, race, gender, past medical, and social history), HF characteristics (New York Heart Association functional classification prior to admission, left ventricular ejection fraction [LVEF], etiology, and years since diagnosis), daily laboratory values (serum chemistries, liver function tests, and complete blood counts), hourly and daily UO and daily weight changes, in-hospital diuretic and medication regimen, and patient outcomes (in-hospital mortality). Mean hourly UO was calculated by dividing a given patient’s total UO by the number of hours for the entire ICCU length of stay.
Statistical Analysis
The primary efficacy end point was the difference in mean hourly UO (calculated by taking the difference in UO before and after initiation of the diuretic-enhancing regimen) compared across the 3 treatment regimens. Secondary efficacy end points included comparison of differences in mean hourly net fluid balance and total UO at 24 and 48 hours between the regimens.
Safety end points included incidence of worsening renal function (0.3 mg/dL increase in SCr or 25% over baseline), absolute change in SCr, and blood urea nitrogen (BUN), incidence of hyponatremia, hypokalemia, and hypomagnesemia, incidence of hypotension (incidence of systolic blood pressure [SBP] <90 mm Hg and <80 mm Hg or mean arterial pressure [MAP] <50 mm Hg at any time), and in-hospital mortality.
Due to a lack of previously published data to guide the expected magnitude of difference, we targeted a mean difference of 100% (double the effect; standard deviation equal to the mean difference) with 80% power, α = .05, in the primary end point of change in mean hourly UO after initiation of the diuretic-enhancing regimen. Therefore, data on a minimum of 40 patients were needed in each treatment arm. The comparisons between the treatment arms and possible covariance were also incorporated into the analyses later.
Descriptive statistics were computed for each diuretic-enhancing regimen. For all comparative analyses across the 3 diuretic regimens, 1-way analysis of variance (ANOVA), and chi-square test (or Fisher exact test) were performed for continuous variables or categorical variables, respectively. If either the normality or equal-variance assumptions underlying the traditional ANOVA tests were violated, the Kruskal-Wallis test that does not depend on the normality or equal-variance assumptions was utilized. To identify the predictors of the response (UO), a series of univariate linear regressions was conducted to examine each independent variable separately. Multivariable linear analyses were then conducted to identify the independent risk factors. A stepwise model selection method was employed. All statistical significance was assessed using α = .05 level. Statistical analysis was performed using SAS 9.2 (Cary, North Carolina).
Results
Study Population
A total of 242 patients had data meeting inclusion criteria, with 160 receiving CIF (median dose 7 mg/h, range 2.5-15 mg/h), 42 receiving intravenous F (median daily dose 80 mg/d, range 0-160 mg/d) + M (metolazone median dose 5 mg, range 2.5-10 mg), and 40 receiving CIB (median dose 1 mg/h, range 0.5-3 mg/h). Overall, the mean age was 57 ± 12 years, and 63% were male. Baseline characteristics were similar between the groups except for history of dyslipidemia, which was higher in patients who received F + M, and ischemic etiology, which was lower in those receiving F+M (Table 1). There were several differences in concomitant in-hospital medication use, notably, patients who received CIB were more likely to also receive intravenous nitroglycerin (but use of any intravenous vasodilator was not different), any inotrope, any vasopressor, and amiodarone (Table 2). They were also less likely to receive a β-blocker and angiotensin-converting enzyme inhibitor or angiotensin receptor blocker. Digoxin use was higher in patients who received CIF.
Baseline Characteristics by Diuretic-Enhancing Regimen.

Abbreviations: HTN, hypertension; IHD, ischemic heart disease; LVEF, left ventricular ejection fraction; NYHA FC, New York Heart Association Functional Classification prior to hospitalization.
aSignificant at .05 level, chi-square test.
In-Hospital Medication Use.

Abbreviation: ACE, angiotensin-converting enzyme.
aMetolazone added to furosemide intermittent boluses or continuous infusion.
bSignificant at .05 level, chi-square or Fisher exact test.
Efficacy
The difference in mean hourly UO for each individual regimen represented a significant increase over its respective baseline (Table 3), P < .0001 for all. For the primary end point, the increase in mean hourly UO was significantly greater for F + M and CIB when compared to CIF.
Comparative Effect on Urine Output and Net Fluid Balance While on Diuretic-Enhancing Regimen.

Abbreviation: UO, urine output mean ± standard deviation.
aSignificant at .05 level, 1-way analysis of variance (ANOVA) test or Kruskal-Wallis nonparametric test.
Mean hourly net fluid balance was significantly different at baseline, with patients who received CIF having a greater negative fluid balance compared to the other 2 groups (Table 3). When comparing between the regimens, the difference in mean hourly net fluid balance was significantly greater (indicating a greater loss of fluid) with F + M and CIB compared to CIF. A total of 159 (66%) and 63 (26%) patients received a diuretic-enhancing regimen for at least 24 and 48 hours, respectively. In those patients, there were no between-regimen differences in total UO at 24 or 48 hours.
Results of the multivariable linear regression and logistic regression in 167 patients with complete data are illustrated in Table 4. Only factors that showed statistical significance in the univariate model were included in the multivariable analysis. Our results indicate that treatment with F + M or CIB was predictive of a greater change in hourly UO, independent of other variables. On average, F + M was associated with a 68-mL greater difference in UO compared to CIF. The CIB was associated with a 53-mL greater difference in UO compared to CIF. The only other variable significantly associated with UO was LVEF, with a 1% increase in LVEF being associated with a 1.2-mL increase in UO difference.
Multivariate Analyses for Predictors of Urine Output.
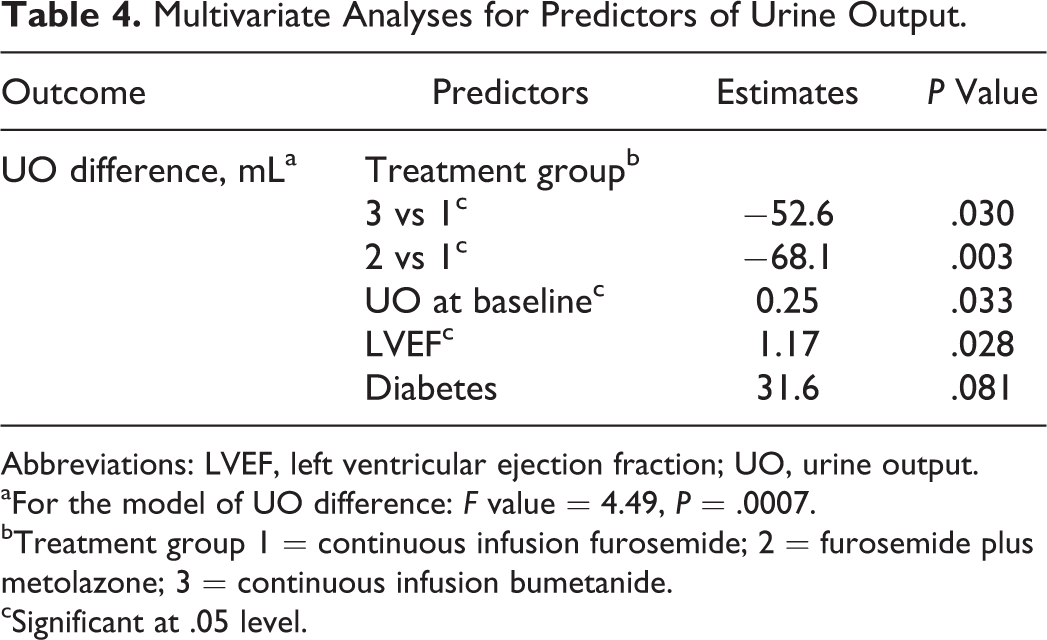
Abbreviations: LVEF, left ventricular ejection fraction; UO, urine output.
aFor the model of UO difference: F value = 4.49, P = .0007.
bTreatment group 1 = continuous infusion furosemide; 2 = furosemide plus metolazone; 3 = continuous infusion bumetanide.
cSignificant at .05 level.
Safety
The incidence of worsening renal function occurred in 21% of the study population; however, there were no differences between the regimens. There was a suggestion of differences in baseline SCr between regimens, although the differences did not reach statistical significance (Table 5). Mean SCr at 24 hours after initiation of the diuretic-enhancing regimen and mean change in SCr from baseline were not different between the regimens. Mean BUN at baseline and 24 hours after initiation of the regimen was significantly lower in patients who received CIF compared to the other 2 regimens. The mean change in BUN from baseline was not different between the 3 regimens; however, it tended to increase to a greater degree with F + M and CIB (Table 5).
Comparative Effects on Renal Function, Electrolytes, and Blood Pressure.a
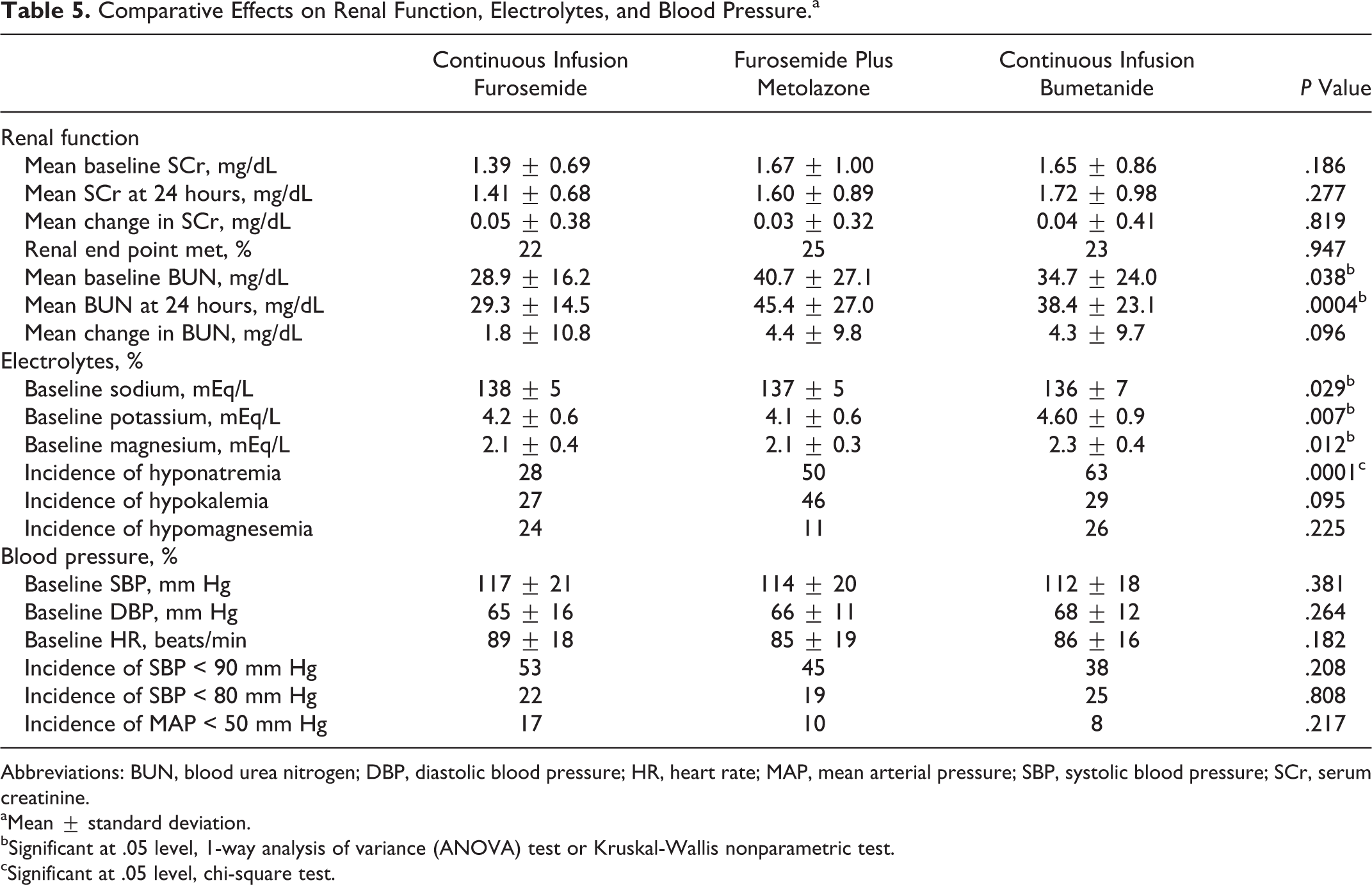
Abbreviations: BUN, blood urea nitrogen; DBP, diastolic blood pressure; HR, heart rate; MAP, mean arterial pressure; SBP, systolic blood pressure; SCr, serum creatinine.
aMean ± standard deviation.
bSignificant at .05 level, 1-way analysis of variance (ANOVA) test or Kruskal-Wallis nonparametric test.
cSignificant at .05 level, chi-square test.
There were baseline differences between the regimens in terms of electrolytes (Table 5). Serum potassium and magnesium were significantly higher in patients who received CIB, whereas serum sodium was slightly higher in the CIF group. During treatment with each diuretic-enhancing regimen, the incidence of hyponatremia (defined as a serum sodium < 135 mmol/L) was higher with F + M and CIB compared to CIF.
Systolic and diastolic blood pressures were not different between regimens at baseline (Table 5), and there were no differences in the incidence of hypotension (defined as SBP < 90 mm Hg, SBP < 80 mm Hg, or MAP < 50 mm Hg) during treatment witheach regimen.
Subgroup With Baseline SCr < 1.5 mg/dL
Due to the baseline differences in SCr between regimens, we conducted an exploratory analysis of the 149 patients who had a baseline SCr < 1.5 mg/dL (106 having received CIF, 22 F + M, and 21 CIB). There were no differences in baseline characteristics between regimens (data not shown). The use of nitroglycerin, any inotropes, any vasopressor, and amiodarone was higher in patients who received CIB, whereas digoxin was used more frequently in patients who received CIF. Unlike the overall study population, baseline SCr, BUN, and serum sodium concentration were not different between regimens (Table 6).
Comparative Effect in Subgroup of Patients With Serum Creatinine < 1.5 mg/dL.

Abbreviations: BUN, blood urea nitrogen; DBP, diastolic blood pressure; HR, heart rate; MAP, mean arterial pressure; SBP, systolic blood pressure; SCr, serum creatinine; UO, urine output.
aSignificant at .05 level, 1-way analysis of variance (ANOVA) test or Kruskal-Wallis nonparametric test.
bSignificant at .05 level, chi-square test.
The results related to efficacy and safety in this subgroup mirrored those of the entire study population. Compared to baseline, each regimen was associated with an increase in UO (P < .0001 for all). The increase in UO was significantly higher with F + M and CIB compared to CIF, as was the mean hourly net negative fluid balance. Incidence of worsening renal function and mean change in SCr were not different, whereas mean change in BUN was higher with F + M and CIB compared to CIF. The incidence of hyponatremia remained highest in patients who received CIB. No differences in blood pressures were noted in baseline or during treatment with each regimen.
Discussion
The HF guidelines recommend 3 regimens to enhance diuresis in patients with acute HF above the use of intermittent bolus furosemide. However, no studies had evaluated the comparative efficacy and safety of bumetanide- and metolazone-based diuretic-enhancing regimens to furosemide in a single patient population, and hence it is unknown whether these regimens are truly more effective, and if so, whether there may be clinical differences between them.
Diuretic-Enhancing Regimen Efficacy
In this retrospective cohort study, we found all 3 diuretic-enhancing regimens (CIF, combination of F + M, and intravenous bumetanide) were associated with greater mean hourly UO and net fluid loss compared to the initial period of time each patient received intermittent bolus furosemide. Our analysis indicated that the increase in diuresis was greater with the combination of intravenous F + M and intravenous bumetanide when compared to CIF. This was evident in the entire study population as well as in the large subset of patients with preserved renal function (baseline SCr of < 1.5 mg/dL). In the subset of patients (N = 167) who remained on each regimen a full 24 hours, a statistically significant difference between the regimens in total UO was not detected. This could be a reflection of the inherent selection bias with the observational study design in which specific diuretic-enhancing regimens may have been chosen for patients, depending on how poorly they were responding to initial therapy. This may explain the higher baseline UO in the CIF group, resulting in less of a difference in absolute and between regimens UO despite the greater relative changes with F + M and bumetanide. In addition, although not statistically significant, the absolute mean difference in total UO at 24 hours was numerically higher (415 and 668 mL) with these 2 regimens. The wide confidence intervals may have precluded the effect from reaching statistical significance.
The results suggest that escalating loop diuretic dose by switching from furosemide to bumetanide may provide a diuresis advantage. 17 The limited data comparing the more potent loop diuretics to furosemide in patients with HF have been reviewed previously. 18 However, the data are limited to outpatient oral dosing, and comparative effect of intravenous bumetanide is lacking. These results are however consistent with the DOSE trial that demonstrated 37% greater diuresis in patients who received high-dose intravenous furosemide (2.5 times their previous oral dose of furosemide) compared to low-dose intravenous furosemide (equivalent to their previous oral dose of furosemide). In this study, median dose of bumetanide (1 mg/h) represented a dose approximately 5.7 times the median dose of CIF (7 mg/h), resulting in an 88% greater hourly diuresis.
Our analysis indicates that the addition of a thiazide diuretic, specifically metolazone, may also augment diuresis. In the current study, the median dose of intravenous furosemide (80 mg/d) was approximately half of the median dose of furosemide administered via continuous infusion (7 mg/h or 168 mg/d). Despite the lower daily dose of furosemide, the combination with metolazone was associated with a 127% greater hourly diuresis. This is an important addition to the limited data evaluating the value of combining loop and thiazide diuretics in HF. Despite there being numerous published reports, the majority of the data reflects small sample sizes. 19 Thiazides mentioned in these reports include hydrochlorothiazide, chlorothiazide, bendroflumethiazide, and quinethazone, in addition to metolazone, which was the most common. Our findings are consistent with the existing literature suggesting that the combination of thiazide and loop diuretic can be used to augment diuresis in patients resistant to intermittent administration of a single agent. The largest randomized trial evaluated 33 patients with severe HF unresponsive to furosemide 80 mg twice daily. 11 The addition of metolazone or bendrofluazide 10 mg once daily conferred a diuretic response in 93% of the patients.
Diuretic-Enhancing Regimen Safety
The current study showed that the comparative renal safety of these diuretic-enhancing regimens appears similar. In our study population, there was a 21% incidence of worsening renal function, with no difference between the regimens. Mean SCr 24 hours after initiation and mean change in SCr from baseline were not different between the 3 regimens. However, mean increase in BUN 24 hours after initiation was significantly lower in patients who received CIF compared to of combination of F + M or CIB. The apparent neutral effect on SCr and development of worsening renal function is consistent with previous prospective continuous infusion studies5,15,20 but discordant from the DOSE study demonstrating a higher incidence of increases in SCr of >0.3 mg/dL during the first 72 hours. 5 Data on combination diuretics and renal function have been mixed; however, minimal effect on SCr was seen in the largest study. 11 The BUN changes were not routinely reported in previous studies; however, in one prospective CIF study, no change in BUN was observed, 20 while 2 small observational studies of combination diuretics suggest a risk of increased BUN and azotemia.21,22 The greater increase in BUN with a combination of F + M, and bumetanide, may reflect the greater mean hourly UO and contraction of the intravascular volume.
There were some differences in terms of propensity for development of electrolyte abnormalities between the 3 regimens as well. Hyponatremia, defined as a serum sodium less than 135 mEq/L, was approximately twice as common in the F + M and the bumetanide groups compared to CIF. There was a signal for hypokalemia being more common with F + M compared to the other 2 regimens; however, it did not reach statistical significance. Our results are consistent with the limited data that exists for patients with HF. The CIF has not been previously shown to be associated with an increased risk of electrolyte abnormalities 13 ; however, studies of combination diuretic regimens do suggest an increased risk. 19 The most recent studies of CIF in HF found no significant difference in hypokalemia and hyponatremia risk but less effect on serum sodium compared to intermittent bolus administration.5,15 Importantly, no difference in UO was observed in either study, and thus the difference in electrolyte abnormalities was not attributable to the extent of diuresis. Previous literature with bumetanide is lacking. Overall, our results add to the existing data that support a higher risk of electrolyte disturbances with combination diuretics, compared to CIF regimens. However, our results must be interpreted under the context of greater UO in the combination diuretic and bumetanide cohorts and the existence of baseline differences in electrolyte concentrations.
Despite the observed differences in UO and net fluid balance, no differences were detected in the incidence of hypotension defined using several blood pressure thresholds. No differences were detected for in-hospital mortality, although this study was not powered to properly assess clinical outcomes and cannot exclude the potential for worse outcomes that were observed in the bumetanide-treated patients.
Limitations
There are limitations in this analysis, and the study results should be considered hypothesis generating and thought provoking. The retrospective nature of the study does not allow us to establish a clear cause and effect, and factors other than the diuretic regimen used may explain the differences in the efficacy and safety end points. However, our multivariate analysis suggests a significant association between the regimen used and the achievement of enhanced diuresis. The lack of randomization also led to several important differences in baseline characteristics. An important difference was in baseline renal function that could directly influence the effectiveness of the refractory regimens. The evaluation of the contribution of baseline renal function to the observed differences was considered in 2 analyses, the subgroup analysis of patients with an SCr < 1.5 mg/dL and the multivariate analysis. The latter effectively eliminated the baseline differences, while demonstrating similar findings to the analysis of the total study population. The multivariate analysis did not find parameters of baseline renal function to be a significant predictor of UO. Therefore, it is unlikely that baseline renal function was the primary variable driving the observed differences. Finally, data for other aggressive diuretic regimens, such as combination of bumetanide and metolazone and vasopressin antagonists, were not included in this study due to the limited use at our institution. Comparison to these regimens, and mechanical modalities such as ultrafiltration, was not possible but remain warranted.
In conclusion, this analysis suggests that there may be some important clinical differences between the 3 commonly employed diuretic-enhancing regimens used in patients with acute HF. The results suggest the combination of F + M or CIB may represent superior regimens in affecting diuresis compared to low-dose CIF. The enhanced diuresis was achieved without an increased incidence of renal insufficiency or hypotension; however, electrolyte abnormalities may be more prevalent when metolazone is added to furosemide or bumetanide is used. These therapeutic differences require confirmation in a prospective study, and our results support specific prospective investigations into the utility of bumetanide- and metolazone-based regimens.
Footnotes
Acknowledgments
The authors would like to thank Tesfa Ghebreyesus for his help in extracting study data from the CCU computer system.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
