Abstract
Objective:
To evaluate the therapeutic effects of prostaglandin E1 (PGE1) on residual pulmonary arterial hypertension (PAH) after corrective surgery for congenital heart disease.
Methods:
Thirty-one patients with postoperational PAH were randomly divided into control group (n = 15) and PGE1 group (n = 16, 6 courses of intravenous PGE1 plus conventional therapy). Mean pulmonary arterial pressure (MPAP), right ventricular ejection fraction (RVEF), and left ventricular ejection fraction (LVEF) were measured by echocardiography before and 3, 6, and 12 months after the treatment. Arterial oxygen pressure (Pa
Results:
In both groups, MPAP decreased and RVEF, LVEF, and Pa
Conclusions:
Intravenous PGE1 therapy after corrective surgery for congenital heart disease was associated with a reduction in mean pulmonary arterial pressure and a lower risk of death.
Introduction
Approximately 10% of patients with congenital heart disease develop pulmonary arterial hypertension (PAH), despite advances in diagnosis and therapy. 1,2 After surgical correction of congenital heart disease, PAH is still present in some cases which may affect the long-term outcomes. 3 Management for patients with PAH after corrective surgery is challenging. Conventional pharmacological treatment including vasodilators and anticoagulants do not seem to alter patients’ survival rates. 4,5 Prostacyclin analogues targeting the abnormal contraction of the pulmonary artery have emerged as one of the promising therapies. 6 –9 Continuous infusion of epoprostenol, a prostacyclin analogue, was reported to improve cardiac function, oxygen saturation, and exercise capacity in patients with PAH. 10
Prostaglandin E1 (PGE1), a member of the prostanoid family, has been demonstrated to improve pulmonary hemodynamics and clinical outcomes in patients with PAH. 11 However, long-term effects of PGE1 on the clinical outcomes of patients with residual PAH after corrective cardiac surgery has not been investigated. The aim of this study was to evaluate the effect of continuous intravenous PGE1 on the hemodynamics and long-term outcomes in patients following corrective surgery for congenital heart disease.
Patients and Methods
Patient Selection
This study was approved by the Human Research Ethics Committee of Liaocheng People’s Hospital. Informed written consent was obtained from all participants. Between September 2005 and August 2010, 40 consecutive patients with residual PAH following corrective surgery for congenital heart disease were initially recruited into this study. All patients had clinical symptoms of pulmonary hypertension, which was supported by preoperational Doppler echocardiographic studies. Before surgery, all patients were treated with captopril (0.3-0.5 mg/kg/d) and loop diuretics for 2 weeks. In addition, intravenous infusion of PGE1 (200 μg/d) was administered for 10 to 15 consecutive days in all patients to reduce MPAP to below 70 mm Hg before surgery.
Physical examination, echocardiography, and blood oxygen assessments were performed within 7 days after the surgery. Residual PAH was defined as an increase in pulmonary arterial systolic pressure ≥50 mm Hg at rest, in the absence of other causes of precapillary pulmonary hypertension such as pulmonary hypertension due to lung diseases or chronic thromboembolic pulmonary hypertension. 12,13 Inclusion criteria in this study were mean pulmonary arterial pressure (MPAP) between 25 and 70 mm Hg and MPAP/mean systemic blood pressure ratio ≤0.75. Nine patients who had severe residual pulmonary hypertension (MPAP >70 mm Hg or mean PAP/mean systemic blood pressure ratio >0.75) were also excluded as these patients required intensive antihypertensive management including intravenous infusion of PGE1. In the end, 31 patients entered the trial and were randomized, using random drawing of numbers, into control group (n = 15) and PGE1 group (n = 16). The general characteristics of the patients are shown in Table 1. Most patients were adults who presented to the clinics for corrective congenital heart disease surgery after experiencing significant clinical symptoms.
Baseline Characteristics of the Patients a
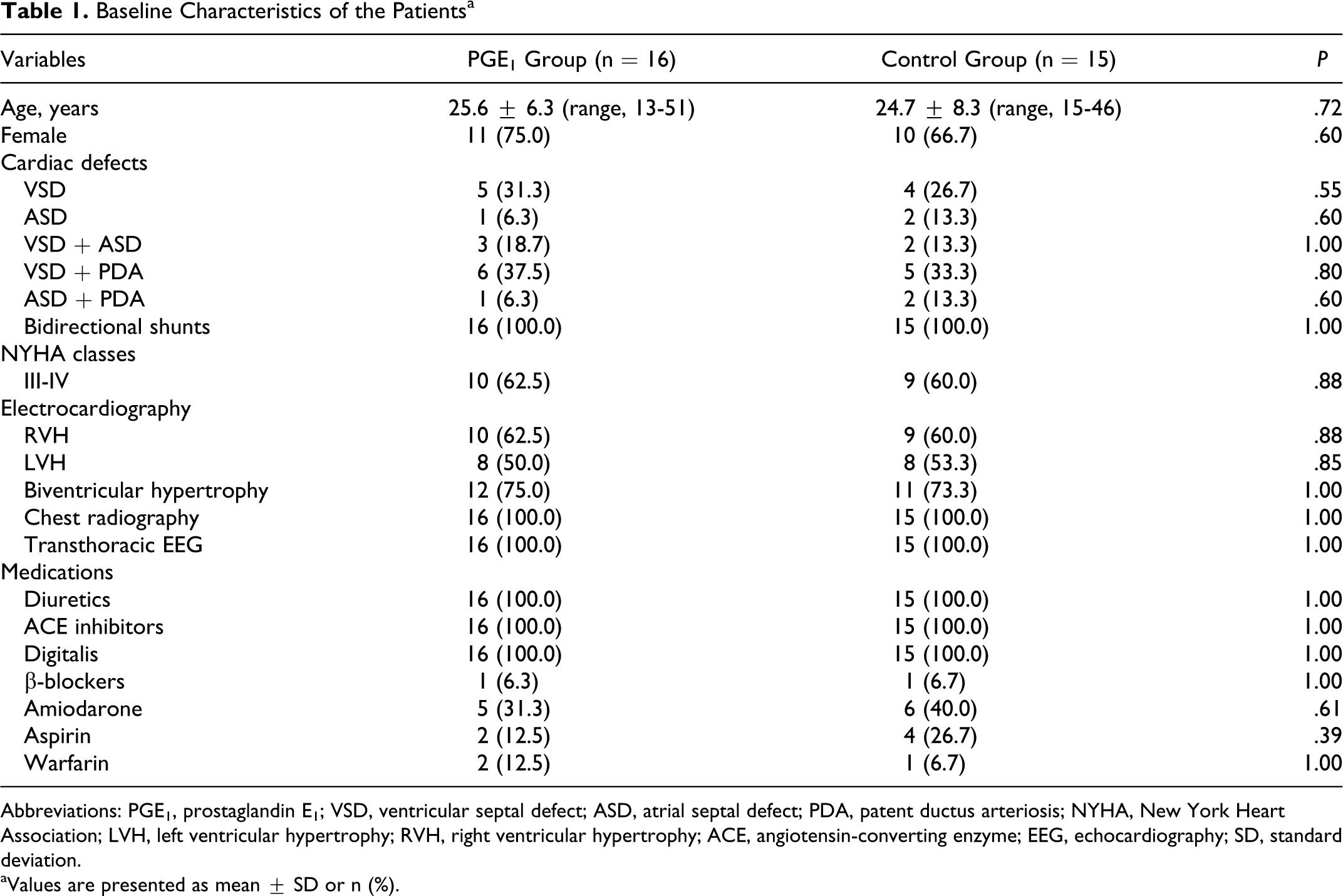
Abbreviations: PGE1, prostaglandin E1; VSD, ventricular septal defect; ASD, atrial septal defect; PDA, patent ductus arteriosis; NYHA, New York Heart Association; LVH, left ventricular hypertrophy; RVH, right ventricular hypertrophy; ACE, angiotensin-converting enzyme; EEG, echocardiography; SD, standard deviation.
aValues are presented as mean ± SD or n (%).
Transthoracic Echocardiography
Both MPAP and ventricular function were evaluated with colored Doppler echocardiography (HP-SONOS 5500). Pulmonary arterial systolic pressure was calculated from the tricuspid regurgitation velocity and pressure gradient, and the estimated right atrial pressure: PA systolic pressure = tricuspid regurgitation pressure gradient + estimated right atrial pressure. 13,14 The MPAP was subsequently calculated as follows: MPAP = 0.61 × pulmonary arterial systolic pressure + 2 mm Hg. 13 Standard parasternal long-axis, short-axis, and apical 4- and 2-chamber views were obtained, and the right ventricular ejection fraction (RVEF) and left ventricular ejection fraction (LVEF) were calculated by a modified Simpson formula. The cardiologists who performed echocardiographic and New York Heart Association (NYHA) function class assessments were blinded to the patient’s groupings.
Pharmacological Management
Captopril was administered to all patients from the study and control group. Captopril (0.3-0.5 mg/kg/d) was the drug of choice in our department because of its low cost and proven effect on pulmonary hypertension. 14 In the PGE1 group, intravenous infusion of PGE1 was immediately commenced in addition to captopril and loop diuretics. In the first 3 months following surgery, PGE1 was intravenously administered daily (200 μg/d, continuous infusion over 10-12 hours) for 10 consecutive days each month. The 10-day treatment was repeated in months 6, 9, and 12 following the surgery. The after-discharge intravenous infusion was conducted at the patient’s home via peripheral veins. The daily dosing regimen and the duration of each treatment course (10 days) of PGE1 were based on the manufacturers’ instructions (Beijing Tide Pharm, Beijing, China). As there was no published data or guidelines on how long PEG1 treatment should be given, 4 courses of intravenous treatment were used in the present study to achieve maximum therapeutic effect.
Clinical Outcomes
All patients were followed up in our clinics for at least 12 months. In both groups, the arterial oxygen saturation rate was measured before and at the end of the 12-month follow-up. The PAP and ventricular function were also assessed with echocardiography at months 3, 6 and 12 following the surgery.
Statistics Analysis
All data are presented as mean ± standard deviation. Continuous variables were compared with Student t-test or analysis of variance. Categorical data were compared with Fisher exact test. Survival rate was calculated using the Kaplan-Meier methods. P value < .05 was considered statistically significant. All data were analyzed by SPSS 13.0 software (SPSS, Inc, Chicago, Illinois).
Results
General Findings
As shown in Table 1, the most common type of congenital heart disease was ventricular septal defect (VSD) combined with patent ductus arteriosis (PDA; n = 11) and VSD alone (n = 9). All patients had bidirectional shunts. Before the surgery, the systolic PAP was more than 90 mm Hg and MPAP of more than 50 mm Hg in all patients. In 27 patients, the mean PAP/mean blood pressure ratio was >0.75. In the remaining patients, the mean PAP/mean blood pressure ratio was between 0.56 and 0.74. All patients had clinical signs and echocardiographic evidence of ventricular dysfunction. New York Heart Association function class III and IV was found in 19 patients (Table 1). After preoperational treatment with intravenous PGE1, captopril and loop diuretics, MPAP was reduced to below 70 mm Hg in all patients before the surgery (Table 1). The congenital defects were successfully repaired in all patients.
At the beginning of the study, there was no difference in age, sex, type of congenital heart disease, or NYHA function classes between the 2 groups (Table 1). The types of cardiovascular medications used (except PGE1 in the study group) were also similar between the 2 groups.
Effect on MPAP and Ventricular Function
As shown in Table 2, in both groups, there were no statistically significant changes in systolic blood pressure (P > .05). The MPAP at 6 and 12 months was lower than the baseline levels (within days following the surgery) in both groups. The 12-month MPAP in the PGE1 group was lower than in the control group (P = .008). The 12-month RVEF and LVEF in the PGE1 group was higher than in the control group (P = .031 and 0.019, respectively). The 12-month Pa
Effects of PGE1 on MPAP, LVEF, RVEF, and Pa
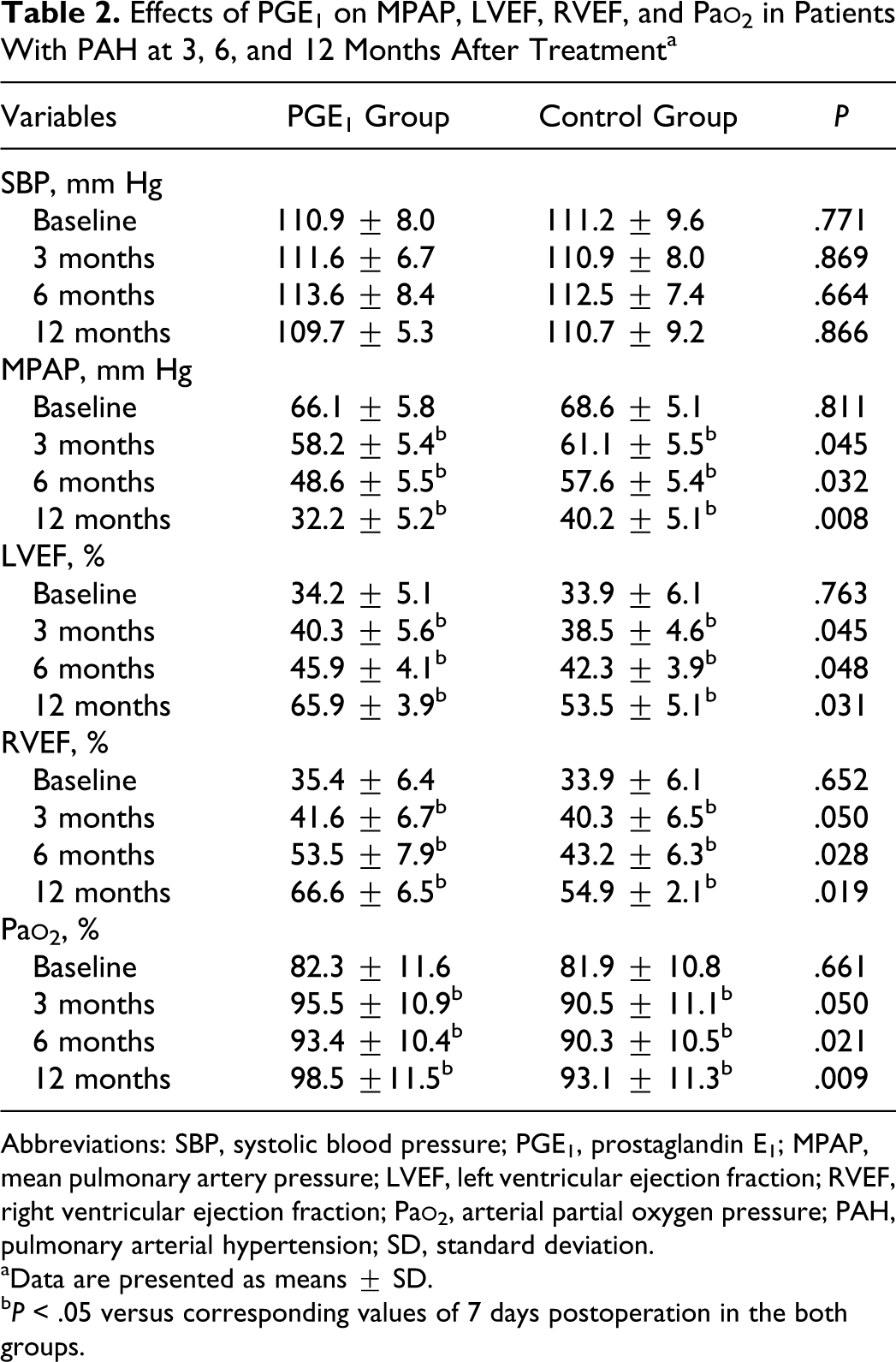
Abbreviations: SBP, systolic blood pressure; PGE1, prostaglandin E1; MPAP, mean pulmonary artery pressure; LVEF, left ventricular ejection fraction; RVEF, right ventricular ejection fraction; Pa
aData are presented as means ± SD.
b P < .05 versus corresponding values of 7 days postoperation in the both groups.
Follow-Up
During the follow-up of 36 months (range 12-60 months), 4 patients in the control group died of PAH crisis at months 1, 3 and years 2 and 3, respectively. All patients in the PGE1 group survived. The survival rate in the PGE1 group was higher than in the control group (100% vs 73.3%, P = .029).Cumulative survival rate in the control group were 86.7%, 80%, 73.3%, and 73.3% at 1, 2, 3, and 5 years, respectively (Figure 1).
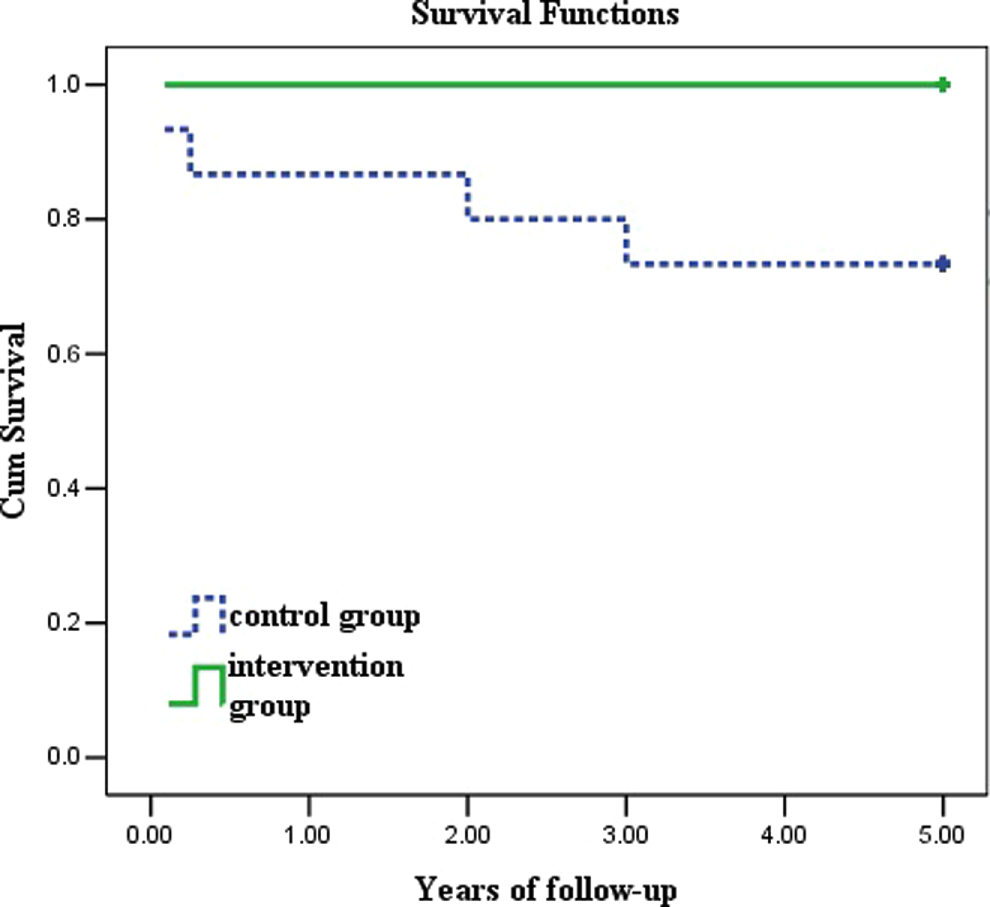
Comparison of survival rates between the 2 groups. Four patients in the control group died at months 1, 3 and years 2 and 3, respectively, in the follow-up. There were significant differences in survival rates between prostaglandin E1 (PGE1) group and control group (P = .029).
In the treatment group, 8 patients (50%) experienced side effects of PGE1, including nausea and vomiting in 2, fever in 3, and pain in the intravenous infusion site in 3.
Discussion
This study demonstrated that in patients with residual PAH after corrective surgery for congenital heart disease, 6 courses of intravenous PGE1 treatment was associated with a reduction in MPAP, an increase in RVEF, LVEF, and an improvement in NYHA functional class. During an average follow-up of 36 months, the survival rate in the PGE1 treatment group was higher than in the control group.
All our patients had significant preoperational PAH and ventricular dysfunction before the surgery due to late presentation, a situation which is common in this inland Chinese community. 14 Even with preoperational pharmacological management of PAH and subsequent surgical correction of congenital cardiac defect, residual PAH persisted in all patients. The residual PAH may be due to a number of reasons, such as vasoconstriction, proliferative and obstructive remodeling of the pulmonary vessel wall, inflammation, or thrombosis. 13 –17 Current therapies of PAH are directed at preventing or reversing vasoconstriction, vasoproliferation, or thrombosis. 7,18 Treatment strategies for patients with PAH associated with congenital heart defect are mainly based on the expert consensus rather than evidence based. 19
Prostanoids have been shown to reduce mortality by 51% in patients with PAH, regardless of the etiology of the PAH. 20,21 Prostaglandin E1, a member of prostanoids family, has vasodilative effects both on pulmonary and systemic arteries. 22 It also has antiplatelet and antiproliferative effects. 23 Prostaglandin E1 has been used to treat pulmonary hypertension after mitral valve replacement 22 or after open-heart surgery in children. 24,25 Previous studies also demonstrated that continuous intravenous infusion of PGE1 improves exercise tolerance and enhances the quality of life of patients with PAH. 11 However, data about medium- to long-term effects of PGE1 on PAH following corrective surgery for congenital heart disease are lacking. In this study, we found that 6 courses of intravenous infusion of PGE1 over 12 months following corrective cardiac surgery reduces mean PAP and improves cardiac function. We also found that the survival rate in the PGE1 group was higher than in the control group of patients. The beneficial effects of PGE1 infusion may be largely related its vasodilative effect on the pulmonary vasculature. 22 Prostaglandin E1 may also suppress the remodeling of the pulmonary arteries, 26 reducing pulmonary hypertension following corrective cardiac surgery.
Repetitive intravenous infusions of PGE1 for PAH over several months appear safe. As PGE1 infusion leads to both pulmonary vasodilatation and systemic vasodilatation, 22 hypotension during or after the treatment may be a concern. However, the reported hypotension rates were low, ranging from 10% to 15%. 26 In our study, at a daily dose of approximately 200 µg, we did not observe hypotension in the 16 patients treated with PGE1. Other reported side effect of PGE1 were fever (15%-55%), apnea (15%-26%), convulsion (10%-15%), diarrhea (<5%), nausea (<5%), vomiting (<5%), and pain in the intravenous infusion site. 11,27,28 Other concerns may be the risk of paradoxical embolism and sepsis if a central line is used. 10,29 In the present study, 50% of the patients experienced one of these adverse effects of PGE1, such as nausea and vomiting, fever, and pain in the intravenous infusion site. These side effects resolved spontaneously after the conclusion of the PGE1 infusion and did not prevent further administration of this drug.
There are several potential limitations of this study. First, there were only a small number of patients enrolled, however, the improvement in hemodynamic variables and long-term results were statistically significant. Second, the patient population was heterogeneous with a spectrum of anatomic defects which could have affected the survival outcomes. A larger and more homogenous patient population may be required to ascertain the survival benefits of PGE1.
Conclusions
Six courses of intravenous PGE1 therapy over 12 months decreased mean PAP and improved cardiac function in patients with residual PAH after corrective surgery for congenital heart defect. Prostaglandin E1 therapy also improved survival rates in these patients. This PGE1 treatment may be used in addition to standard pharmacological therapy to manage residual PAH after corrective cardiac surgery.
Footnotes
Ming-Feng Dong and Zeng-Shan Ma equally contributed to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
