Abstract
Studies have documented an association between influenza vaccination and risk reduction in myocardial infarction, all-cause mortality, and major adverse cardiac events. This meta-analysis pooled data from 5 trials with a total of 292 383 patients. Influenza vaccination was found to be associated with significant reductions in myocardial infarction, all-cause mortality, and major adverse cardiac events. The risk reduction afforded by vaccination and the lack of major adverse events related to the influenza vaccination makes it particularly important to vaccinate those with a known cardiovascular disease or those at high risk of developing cardiovascular disease.
Introduction
Influenza-related deaths occur more commonly in patients with cardiovascular disease than in patients with other chronic diseases. 1 Studies assessing mortality during influenza season report cardiovascular death is the leading cause of mortality.2–4 Respiratory infections have been shown to be associated with increased risk of cardiovascular events in large cohort studies. 5 ,6 Atherosclerosis is a chronic inflammation of the arteries in which inflammatory cells play an important role in development of atherosclerotic plaques. 7 Various studies have shown the association of infections and atherosclerosis. 8 ,9 However, use of antibiotics has not been demonstrated to be associated with a decrease in cardiovascular events. 10 ,11 Influenza infection can cause progression of atherosclerosis via cellular and humoral autoimmune mechanisms. 12 ,13 Studies have reported an association between community-acquired pneumonia (CAP) and myocardial infarction. 14 ,15 In patients with influenza and pneumonia, symptoms of chest pain, shortness of breath, and fever may be attributed to pneumonia alone and conditions like myocardial infarction may be missed. 16 Few randomized controlled trials have been published which test the efficacy of influenza vaccines in preventing cardiovascular morbidity and mortality. A meta-analysis pools data from these trials.
Methods
Literature Sources, Search Terms, and Study Selection
Medical literature was systematically reviewed by all authors. Studies evaluating the effects of influenza vaccination on cardiovascular events were identified and collected by searching MEDLINE, EMBASE, and the Cochrane Library using Web-based search engines such as OVID. All relevant studies were then assessed for inclusion using a standardized check list. Search terms used included influenza, vaccination, cardiovascular, mortality, morbidity, and various combinations of these terms. The references of identified and collected studies were then used to identify additional studies which were also assessed for inclusion. Figure 1 outlines study selection.
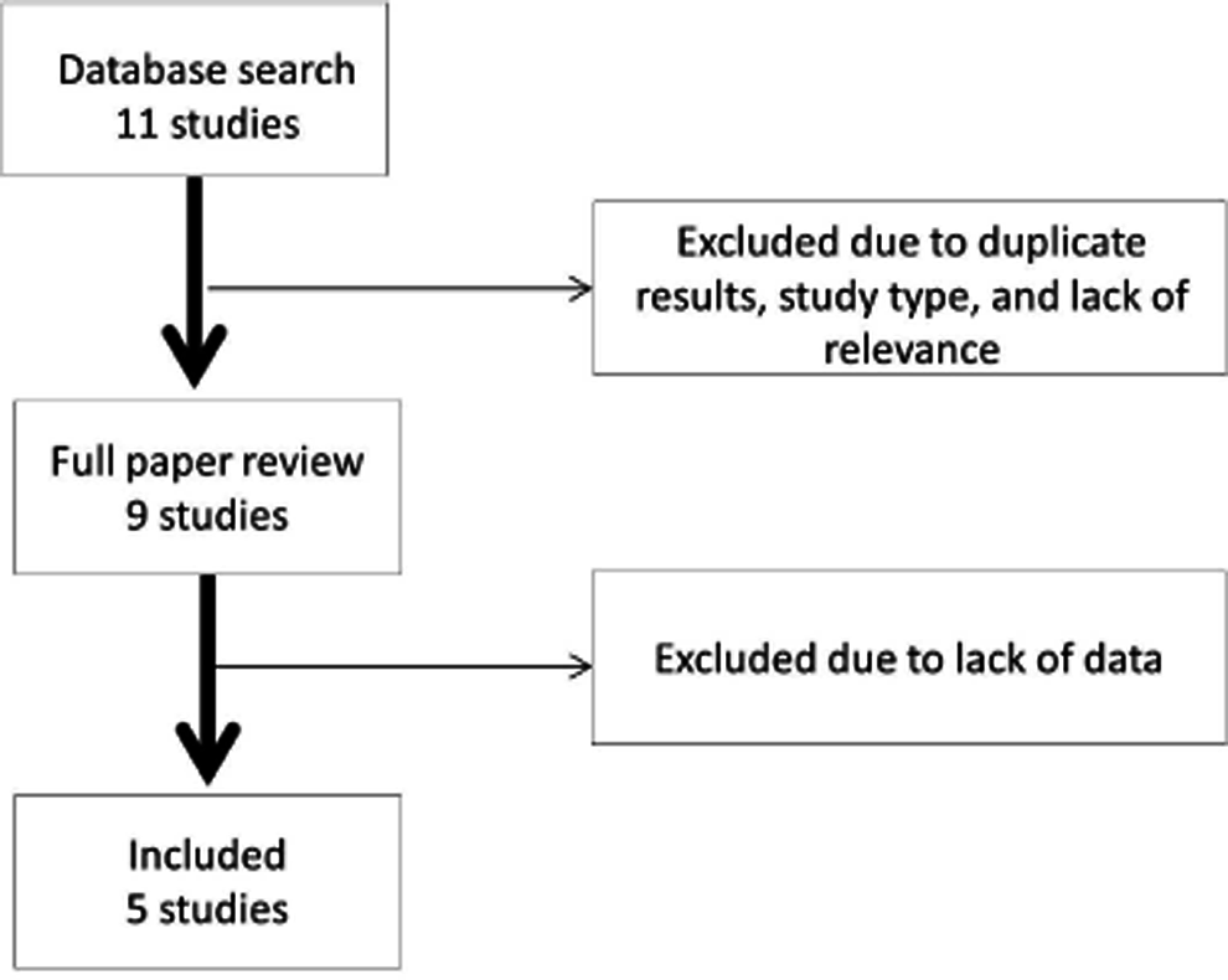
Study selection methodology.
End Points and Definitions
A total of 3 end points were extracted from 5 studies.17–21 End points studied were myocardial infarction, all-cause mortality, and major adverse cardiac events. Only studies with corresponding end point definitions were included in the analysis.
Data Extraction and Quality Assessment
After studies were collected and screened for inclusion, full articles were retrieved for titles fulfilling inclusion criteria. Studies were then scored for quality and data were extracted for studied end points. Included studies had to be randomized trials that were prospective in nature and consisted of only a vaccinated an unvaccinated group. A total of 9 studies were identified for full article review. A lack of usable data or violation of inclusion criteria led to the elimination of 4 studies from the final analysis.22–25
Statistical Analysis
Summary statistics reported in each study were used for the meta-analysis as individual patient data were not accessible. Statistical analysis was performed using the MedCalc software package (Version 11.6.0.0, Mariakerke, Belgium). Cochrane’s Q statistics were calculated and used to determine the heterogeneity of included studies for each end point. The fixed-effects model was used for analysis of end points that were homogenous and the random-effects model was used for analysis of end points that were heterogeneous. Heterogeneity analysis is summarized in Table 1.
Results of Heterogeneity Analysis
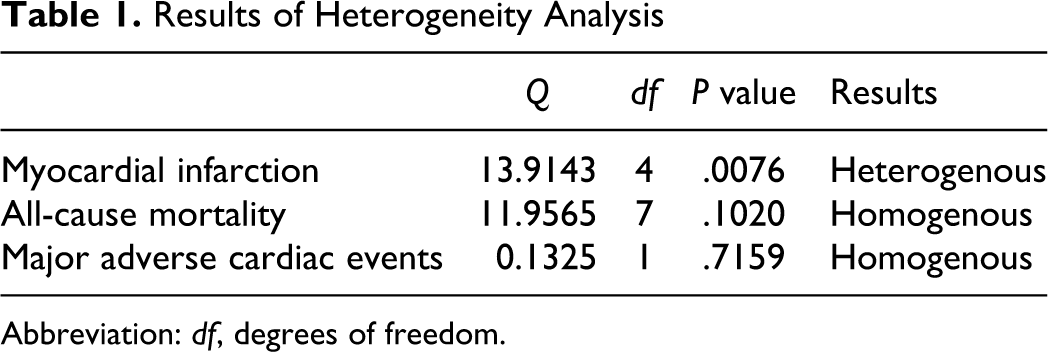
Abbreviation: df, degrees of freedom.
Results
Baseline Characteristics
The included studies did not show significant differences in baseline patient characteristics (Table 2). Results of the analysis are shown in Figures 2 to 4, and there were no significant differences in baseline characteristics within end points once data were pooled.
Baseline Patient Characteristics
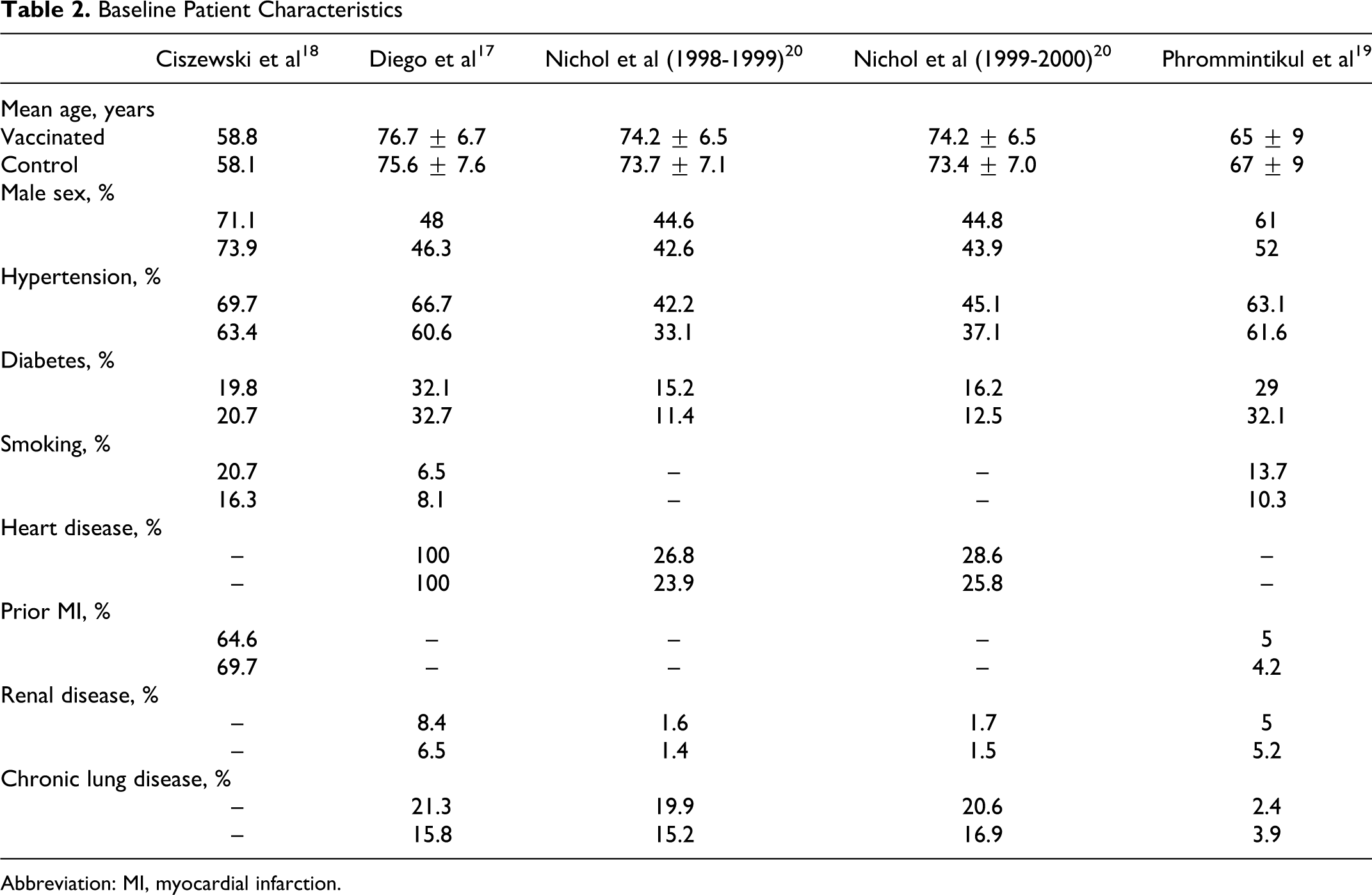
Abbreviation: MI, myocardial infarction.
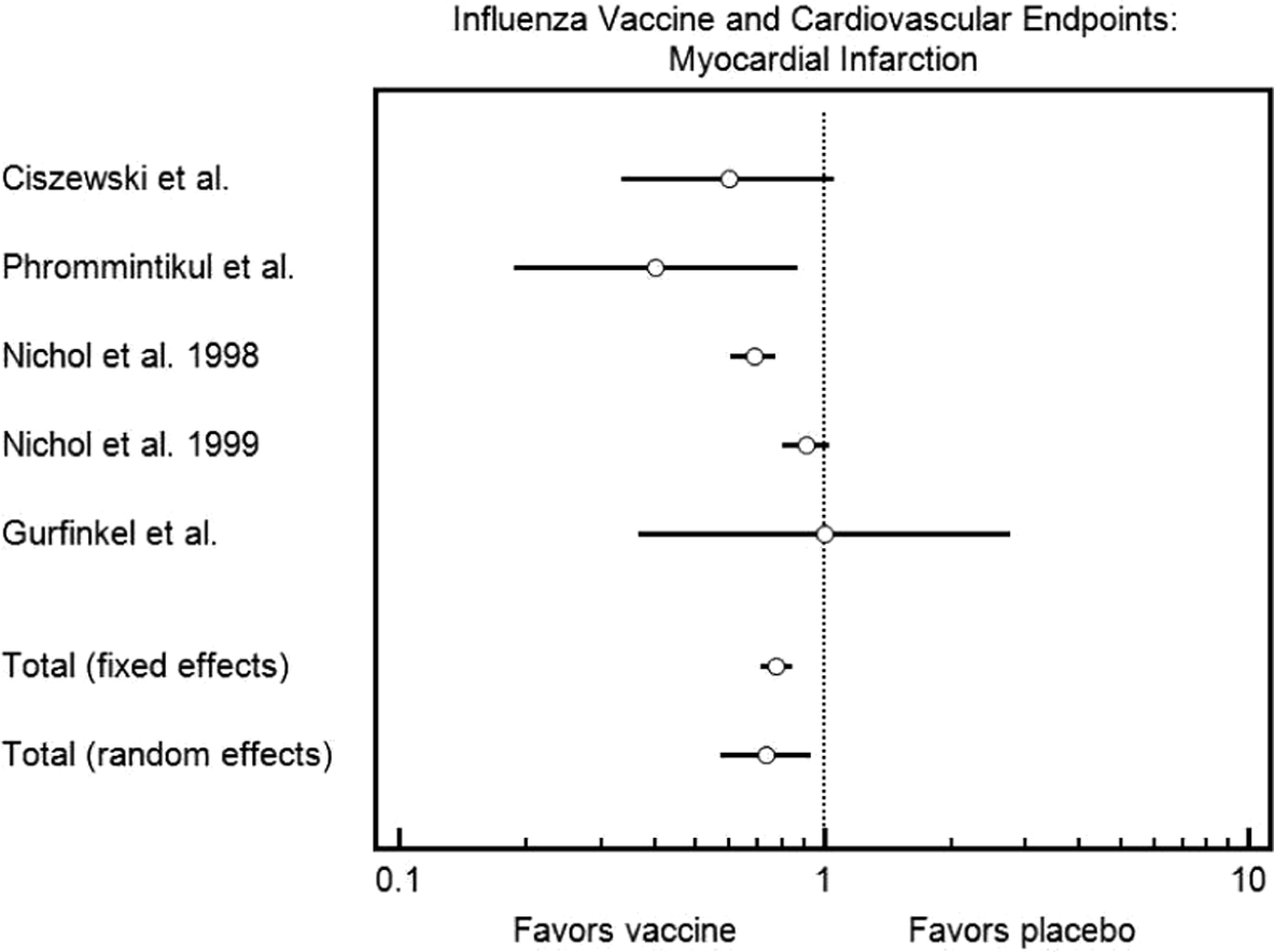
Forest plot demonstrating the effect of influenza vaccination on risk of myocardial infarction.
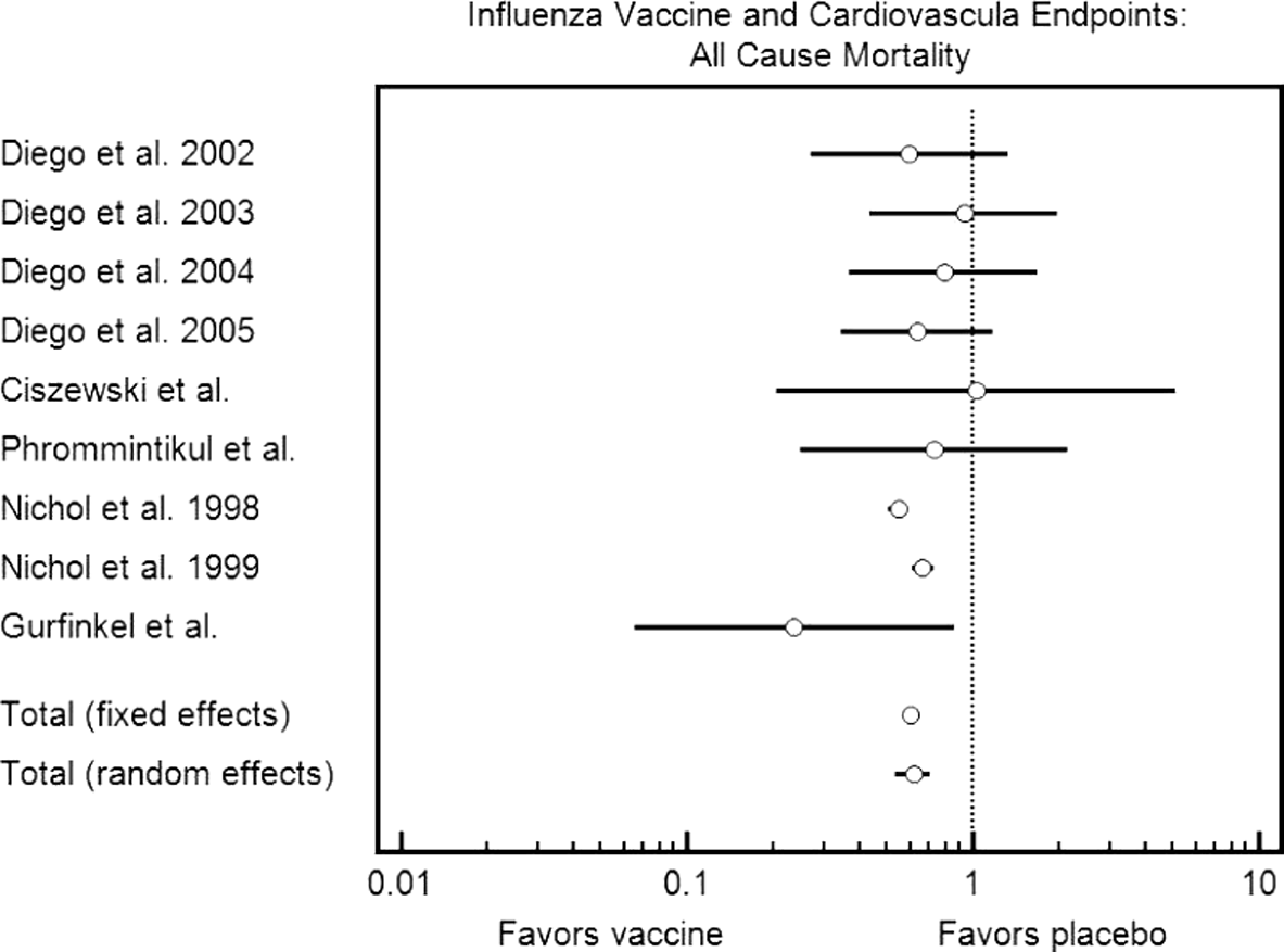
Forest plot demonstrating the effect of influenza vaccination on risk of all-cause mortality.
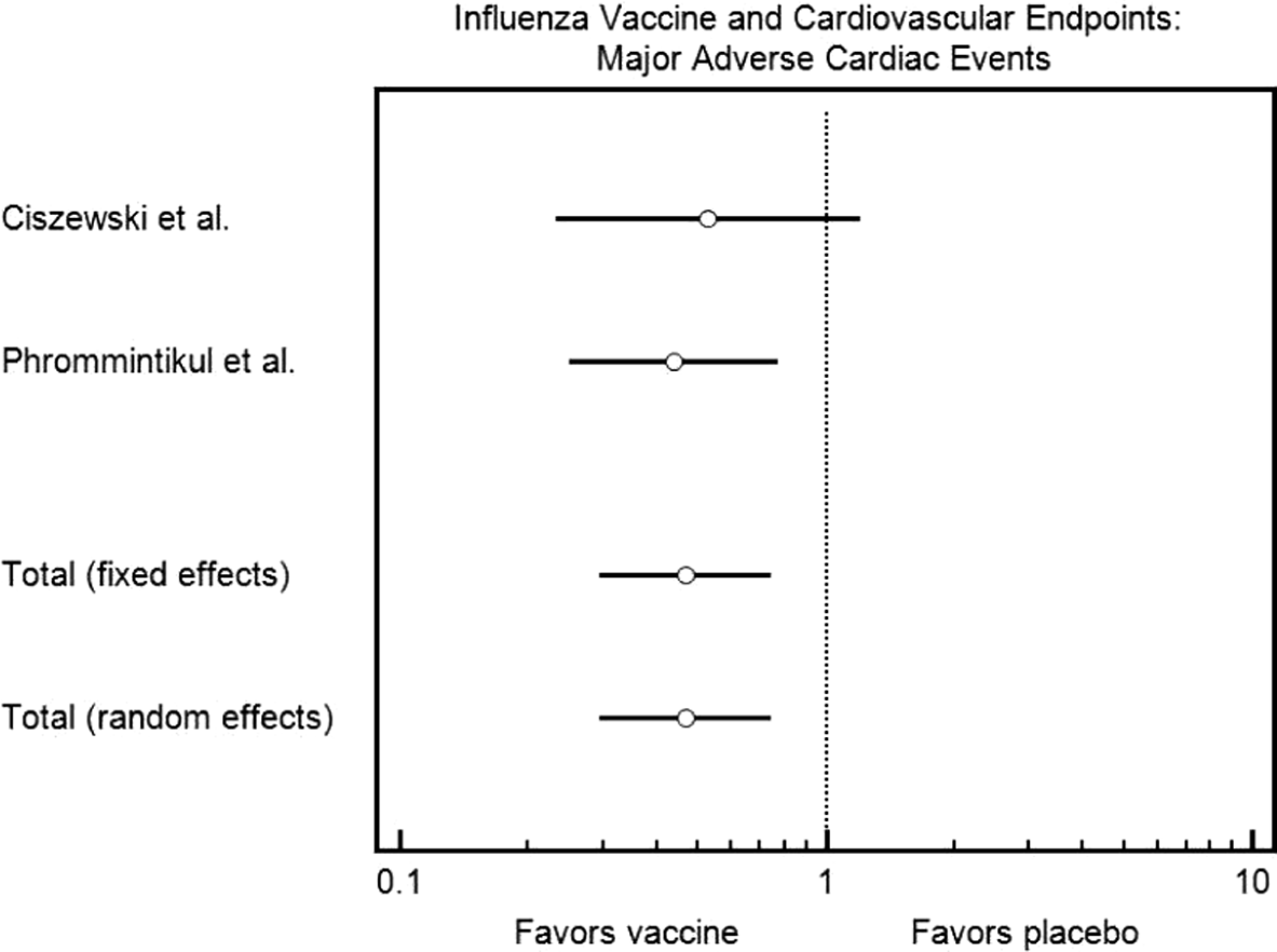
Forest plot demonstrating the effect of influenza vaccination on major adverse cardiac events.
Myocardial Infarction
Data from 4 studies were included for this end point for a total of 165 791 vaccinated patients and 121 990 unvaccinated patients.18–21 Nichol et al reported 2 separate cohorts as to how they were analyzed in the original manuscript. There was a statistically significant reduction in myocardial infarction in those who received the influenza vaccination (odds ratio, 0.731; confidence interval, 0.574-0.931).
All-Cause Mortality
Data from 5 studies were included for this end point for a total of 169 203 vaccinated patients and 123 481 unvaccinated patients.17–21 Diego et al and Nichol et al reported separate cohorts as to how they were analyzed in the original manuscript. There was a statistically significant reduction in all-cause mortality in those who received the influenza vaccination (odds ratio, 0.606, confidence interval, 0.571 to 0.643).
Major Adverse Cardiovascular Events
Data from 2 studies was included for this end point for a total of 546 vaccinated patients and 551 unvaccinated patients. 18 ,19 There was a statistically significant reduction in major adverse cardiac events in those who received the influenza vaccination (odds ratio, 0.467; confidence interval, 0.294-0.742).
Discussion
This meta-analysis pooled data from 5 studies for a total of 292 383 patients. When compared with those who did not receive the influenza vaccination, those who did receive the vaccination had a significantly decreased risk of myocardial infarction, all-cause mortality, and major adverse cardiac event. Individual studies did not reach statistical significance for some end points. By pooling all data, we were able to reach statistical significance for all 3 end points. Not all the outcomes were reported in each individual study included in this analysis.
American Heart Association/American College of Cardiology guidelines recommend influenza vaccination for the secondary prevention of coronary and other atherosclerotic vascular diseases (class 1, level of evidence B). 23 ,26 The influenza vaccine is available in 2 forms: an inactivated form and a live attenuated form. The inactivated vaccine is currently recommended for secondary prevention. 27 Although the optimal time for influenza vaccination has been proposed to be September to November, it should continued till January or even later if vaccination does not occur earlier in the year. 27
The mechanism by which influenza causes cardiovascular-related morbidity is unclear. It might be due to the production of autoantibodies against modified low-density lipoprotein or it might be due to direct vessel wall colonization causing local autoimmune reactions. 28 Influenza infection has also been reported to increase macrophage infiltration and impair anti-inflammatory properties of high-density lipoprotein. 29 ,30 Fever, tachycardia, and dehydration associated with acute influenza may also contribute to the increased risk of cardiovascular events. 5 ,6 Additionally, influenza virus may cause smooth muscle proliferation, platelet aggregation, and thrombus formation and cause angina destabilization. 31 ,32
Major cardiac complications have been reported in a significant proportion of patients with CAP, especially in hospitalized patients. In 1 study, the pooled incidence rates of overall cardiac complications, incident heart failure, acute coronary syndrome, and incident arrhythmias in hospitalized patients with CAP were found to be 17.7%, 14.1%, 5.3%, and 4.7%, respectively. 33 In a systematic review, influenza infection was reported to trigger acute myocardial infarction and cardiovascular death. Also, the review postulated that influenza vaccines are effective at reducing the risk of cardiac events in those patients with established cardiovascular disease. 34
In animal studies, infection with the herpes virus caused development of atherosclerotic lesions in chickens. 35 Immunization against the herpes vaccine prevented the development of atherosclerosis in these chickens. 36 In a human experimental study, it was shown that smooth muscle cells infected with the herpes virus had increased cholesterol deposition, supporting the hypothesis that viral infection may lead to onset, or progression, of atherosclerosis. 37
Various observational studies in humans have come to different conclusions with regard to the association of influenza vaccination and cardiac outcomes. In a study of patients with cardiac arrest, Siscovick et al reported that influenza vaccination in the previous year was associated with reduced risk of cardiac arrest. 38 Naghavi et al studied patients with recurrent myocardial infarction and found that vaccination was associated with a reduction in the risk of myocardial infarction. 24 Lavallee et al found that the patients admitted with stroke were less likely to report influenza vaccination than similar controls. 39 Grau et al also assessed vaccination status in patients admitted with stroke and concluded that vaccination was associated with decreased risk of stroke. 40 In a large cohort study, Nichol et al concluded that influenza vaccination was associated with decreased need for hospitalization for heart disease, cerebrovascular disease, pneumonia, and influenza as well as risk of all-cause mortality during influenza seasons. 20 Armstrong et al assessed that all-cause mortality, respiratory mortality, and even cardiovascular mortality was strongly associated with index of influenza circulating in the population in unvaccinated individuals, while no association was found in vaccinated individuals. 41 In another large study, Wang et al found that influenza vaccination was associated with decreased risk of all-cause mortality, renal disease, stroke, and heart diseases. 42
In contrast, Jackson et al found no significant association between influenza vaccination and risk of recurrent coronary events in patients with an initial nonfatal myocardial infarction, while Meyers et al studied patients discharged after myocardial infarction and found that influenza vaccination in the previous year was not associated with reduced risk of myocardial infarction. 19 ,20,43,44 In a study done in individuals with incident myocardial infarction, Heffelfinger found no association between influenza vaccination and the risk of myocardial infarction at any time of the year. 45
Although increasing, the influenza vaccine coverage rate in the United States is still low. Greater use of pharmacies, schools, and workplaces as venues and better use of evidence-based practices at health facilities are required to improve the coverage. 46
The adverse effects of the influenza vaccination are relatively minor and self-resolving. Contraindications to influenza vaccine are egg allergy and those with a severe allergic reaction to prior influenza vaccination. Those with a severe, acute illness should be vaccinated after resolution of symptoms while precaution should be taken in vaccinating those with a history of Guillain-Barré syndrome within 6 weeks of resolution of their symptoms. 27 No complications associated with the vaccine were reported in any of the included studies.
Limitations of this meta-analysis include publication bias and heterogeneity in specific end points. Publication bias is noted with respect to the absence of smaller studies, demonstrating no significant difference in end points between those who did and did not receive vaccination across all 3 end points. Heterogeneity was present only in the myocardial infarction end point with no impact on the statistical significance of the result. The use of individual cohorts from certain studies does not impact the statistical results of the study.
Conclusion
This analysis demonstrates that influenza vaccination led to significant reductions in the risk of myocardial infarction, all-cause mortality, and major adverse cardiac events. While vaccination should ideally be given to all except for those with contraindications, particular emphasis should be placed on vaccinating patients with known cardiovascular disease or those at high risk of cardiac events.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
