Abstract
Objective:
The purpose of this study is to investigate the potential in vivo antiplatelet and thromboprotective properties of the antihypertensive drug losartan in mice.
Methods:
Aggregometry studies were performed on platelets obtained from mice administered losartan for 5 days, via tail vein to examine the ex vivo effects (dose dependence) of this agent and to select an appropriate dose for the in vivo studies. Next, the tail bleeding time test and the time for occlusion in a carotid artery injury thrombosis model (ferric chloride) were also performed to assess the in vivo effects of losartan treatment.
Results:
These data indicate that the antihypertensive agent losartan exerts dose-dependent inhibition of the thromboxane receptor-mediated (U46619/agonist)-induced platelet aggregation (ex vivo), whereas it produced no detectable effects on aggregation triggered by adenosine diphosphate or the thrombin receptor activating peptide 4. Findings from the in vivo analysis revealed that tail bleeding time of losartan-treated mice was not different from vehicle-treated mice. On the other hand, in the carotid artery injury thrombosis model, it was found that the losartan-treated mice had significantly longer time for occlusion in comparison with those treated with vehicle control.
Conclusions:
These findings provide evidence that administration of the antihypertensive drug losartan into live mice produces thromboxane A2 receptor–specific antiplatelet effects. Furthermore, interestingly, this antiplatelet activity appears to translate into thromboprotective properties, without resulting in a bleeding phenotype. Consequently, aside from its potential use as an antithrombotic agent, losartan’s chemistry may provide a “blueprint” for designing or repurposing novel derivatives which may have the potential to serve as an antiplatelet and thromboprotective agents but are deprived of the usually concomitant bleeding adverse effects.
Keywords
Introduction
Platelets are anuclear cells derived from mature megakaryocytes and are responsible for maintaining hemostasis. Hemostasis is described as the ability for an injured blood vessel to seal the damage by utilizing aggregation of platelets and mobilization of a fibrinogen network to seal an injury site and prevent unnecessary hemorrhage. 1 Although this system is elegant, and an essential component of protecting blood vessels, it can also contribute to harmful disorders. For example, improper hemostatic function may cause excessive thrombus formation and occlusion of blood vessels; which may lead to a host of medical issues including myocardial infarction, 2 stroke, and peripheral vascular occlusions. 3 Thus, proper understanding of the pathways that participate in platelet activation and aggregation is essential to the discovery of new and novel antiplatelet drug therapies.
One pathway that has attracted attention in the last few decades involves the 7-transmembrane protein, thromboxane A2 receptor (TPR), 4 which has been shown to be a vital mediator of platelet aggregation. 5 In this connection, one of the most prominent and classically used drugs, targeting this pathway, is aspirin, which was shown to exhibit antiplatelet activity 6 in the 1960s. The mechanism of action of aspirin derives from inhibiting the synthesis of TPR’s endogenous agonist (ie, thromboxane A2; TXA2) by inhibiting platelet cyclooxygenase 1. Thus, aspirin inhibits TPR-mediated aggregation and is often endorsed by the medical community as a thromboprotective, prophylactic agent. Despite significant gains in knowledge of TPR’s structural biology and function, up to this date, a clinically relevant antagonist is yet to become available for use in antiplatelet therapy. To this end, while few drugs have shown TPR antagonistic activity, 7 such as Z-335, which is currently under investigation, they are yet to find their way onto the market. One of the most commonly prescribed medications for antiplatelet therapy is clopidogrel (Plavix), which acts by antagonizing the P2Y12 adenosine diphosphate (ADP) receptors. While it is still under debate, this activation pathway is thought to be less potent than that of TPR, with regard to platelet aggregation. Furthermore, clopidogrel use poses a host of harmful issues including dangerous gastrointestinal (GI) bleeding, inefficient and unpredictable pharmacological activity in certain patients (ie, biological variability), and concern over negative drug–drug interaction between the commonly co-prescribed GI protective proton pump inhibitors (PPIs). 8
To this end, utilizing a target-based drug rediscovery approach and bypassing the traditional high-throughput screening methods, we proposed drug repurposing as an alternative approach to explore new indications for the current Food and Drug Administration (FDA)–approved drugs. Thus, we have recently shown that an antidiabetic drug, namely glybenclamide, exhibits TPR-specific antiplatelet activity, 9 which translated into antithrombotic effects. 10 Similarly, the antihypertensive agent losartan has been previously shown to exhibit in vitro antiplatelet properties, which were TPR specific (ie, blockade of the potent TXA2 mimetic, U46619-induced aggregation). 11 Interestingly, similar inhibition of human platelet aggregation was only found at higher concentrations of the active metabolite 12 of losartan, EXP3174. The importance of clearly identifying the in vivo significance of losartan and inhibition of platelet aggregation is directly related to a potential for being thromboprotective.
We first sought to characterize the (ex vivo) dose-dependent inhibition of TPR-mediated aggregation by losartan. Next, we examined its in vivo effects by measuring tail bleeding times and the time for occlusion in a well-established carotid injury thrombosis model in mice. The mouse tail bleeding time assay has been an important tool even to this day in determining negative excessive bleeding phenotypes, 13 whereas the (ferric chloride [FeCl3]) carotid artery injury model and the time for occlusion will signify any thromboprotective effects losartan may exhibit.
Materials and Methods
Reagents and Materials
U46619 (15(S)-hydroxy-11,9-epoxymethanoprosta-5Z,13E-dienoic acid) and arachidonic acid were from Cayman Chemical (Ann Arbor, Michigan). Model 700 aggregometer, ADP, stir bars, and other disposables were from Chrono-Log (Havertown, Pennsylvania). Protease-activated receptor 4 (PAR4) activating peptide (TRAP4; AYPGKF-NH2) was purchased from Anaspec Corporation (Sane Jose, California). Isoflurane and indomethacin were from Sigma Aldrich (St Louis, Missouri). Losartan, sodium citrate (3.8% w/v) solution, 50 mL, and 15 mL conical tubes were obtained from Fisher Scientific (Hanover Park, Illinois). The C57BL/6 mice were obtained from Jackson laboratory (Bar Harbor, Maine).
Mouse Platelet Ex Vivo Functional Studies
The ex vivo functional studies were primarily performed to establish the dose–response curve for losartan’s antiplatelet effects and for proper dosage selection for the in vivo studies. Dosages for losartan used are comparable to the previously published studies in rodent models, which focused (mainly) on investigating its antihypertensive effects 14 –16 ; thereby avoiding the use of suprapharmacological or excessive concentrations. For the preparation of mouse platelets, 4 to 5 (6-8-weeks old) mice of either sex were injected, utilizing the intravenous (IV) tail vein route, with 1, 10, or 25 mg/kg losartan (dissolved in phosphate-buffered saline [PBS]) or with equal volume of vehicle (PBS), once daily for 5 days. Mice were then anesthetized with 5% isoflurane, sacrificed, and their blood collected into 50-mL conical tubes containing 3.8% sodium citrate as an anticoagulant (9 parts blood to 1 part citrate), before being pooled. Platelet-rich plasma (PRP) was then isolated by differential centrifugation of pooled blood at 170g for 10 to 15 minutes. Platelet counts were performed, and the counts adjusted to 2 to 3 × 108/mL (with Hepes/Tyrode buffer). Platelets were then stimulated with 2 μmol/L U46619, 20 μmol/L ADP, or 40 μmol/L TRAP4, after establishing baseline light transmission for at least 1 minute. All aggregation experiments were performed after incubation with 10 μmol/L indomethacin for 2 minutes to prevent TXA2 generation. The aggregation traces were captured using Aggrolink8 software (Chronolog Corporation; Havertown, Pennsylvania). All experiments were performed in compliance with the institutional guidelines and relevant laws which were previously approved by the Western University of Health Sciences Institutional Animal Care and Use Committee (IACUC).
Mouse Tail Bleeding Time Assays
These studies were performed as described previously. 17,18 Briefly, a group of 4 to 8 (6-8 weeks old) mice were injected (IV) with 10 mg/kg losartan or vehicle control (PBS), once daily for 5 days. Mice were anesthetized with 5% isoflurane and anesthesia maintained with 2% isoflurane, before a 5 mm segment was amputated off the distal portion of the tail with a scalpel. The exposed vein was immediately submerged into a beaker containing 0.9% physiological saline which is maintained at 37°C. Bleeding from tail was observed and timed until bleeding cessation, which is defined as no bleeding or rebleeding for at least 60 seconds. It is expected that <300 µL of blood will be lost each time even if bleeding continues for 10 minutes. Normal mice have bleeding times of 2 to 4 minutes. For mice where bleeding did not stop within 10 minutes, the experiment was stopped to avoid unnecessary blood loss and harm to the animal. In that case, pressure was applied to the tail using unrolled cotton and styptic powder was used to help to seal the wound, and the data point was recorded as 10 minutes.
Mouse Carotid Artery Injury Thrombosis Model
The thrombosis model studies were performed as described before. 17,18 C57BL/6 mice were injected with losartan (10 mg/kg) or vehicle (PBS), as described earlier by IV route. Mice were anesthetized with 5% isoflurane and anesthesia was maintained with 2% isoflurane. The left common carotid artery was surgically exposed and a miniature Doppler flowprobe (Model MA-0.5PSB nanoprobe; Transonic Systems, Ithaca, New York) was placed on the surface of the artery. After adding 0.9% NaCl solution in the surgical wound to allow Doppler monitoring, baseline blood flow was recorded using the Transonic perivascular flowmeter. Thereafter, NaCl solution was removed and a filter paper (0.5 × 1.0 mm; saturated with 7.5% FeCl3) was applied to the surface of the carotid artery immediately proximal to the flow probe. After 3 minutes, the filter paper was removed, saline solution was placed in the wound, and carotid blood flow was monitored (using LabChart software from ADInstruments, Colorado Spring, Colorado). As the FeCl3 starts to produce the thrombosis, there will be a subsequent blood flow reduction. The time to occlusion is calculated as a difference in time between the removal of the filter paper and stable occlusion (no blood flow for 2 minutes). Measurement and outcome analysis is based upon the recording of vessel patency following injury with FeCl3.
Analysis of Data
All ex vivo experiments were performed at least 3 times, with blood pooled from at least 5 mice each time. Data were analyzed using GraphPad PRISM statistical software (San Diego, California). Results were compared using unpaired 2-tailed Student t test, with P < .05 considered to be statistically significant. Statistical analysis for the bleeding time and time for occlusion studies was performed using the Mann-Whitney test.
Results
Losartan Inhibits U46619-Induced (TPR-Mediated) Platelet Aggregation, Ex Vivo, in a Dose-Dependent Fashion
To establish the proper dosage of losartan for use in the in vivo studies, an ex vivo platelet aggregation analysis was performed. At a low dose of losartan, that is, 1 mg/kg, we observed approximately 29% inhibition of aggregation induced by the TPR agonist U46619 (2 µmol/L), in comparison to vehicle (PBS)-treated mice (Figure 1). This inhibitory response was found to be dose dependent, as 10 mg/kg of losartan produced around 83% inhibition, whereas 25 mg/kg almost completely inhibited aggregation induced by 2 µmol/L U46619 (Figure 1). This effect is not surprising given that it has been previously demonstrated under in vitro experimental settings.
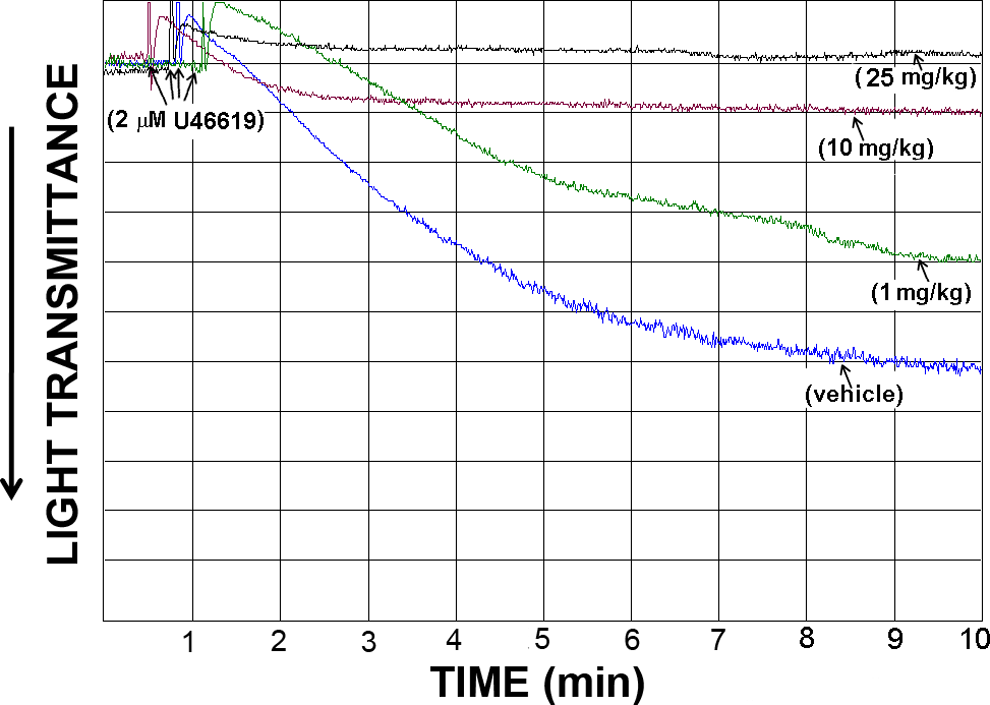
Effect of tail vein intravenous injections of losartan on mouse platelet aggregation stimulated by the TPR agonist U46619, ex vivo. Indomethacin-treated platelet-rich plasma from mice repetitively injected with vehicle, 1, 10, or 25 mg/kg of losartan was stimulated with 2 μmol/L U46619. Each aggregation curve is representative of multiple traces obtained from 3 separate platelet preparations (n = 3), using blood obtained from 3 groups of (4-5) mice (P < .05).
Losartan Fails to Inhibit Platelet Aggregation, Ex Vivo, in Response to Non-TPR-Specific Agonists (ADP and TRAP4)
We next tested whether losartan’s inhibitory effects are specific to the TPR pathway. Thus, the same analysis was performed using non-TPR agonists, namely ADP and TRAP4. Adenosine diphosphate is a physiological agonist for the P2Y1 and P2Y12 receptors and TRAP4 is an activating peptide for PAR4, on platelets. Our results revealed that losartan lacked any detectable effects on aggregation triggered by 20 µmol/L ADP, even at a concentration (ie, 25 mg/kg) that almost completely inhibited aggregation mediated by TPRs (Figure 2). Interestingly, when the same experiment was repeated in the absence of indomethacin, losartan was found to inhibit aggregation stimulated by ADP, albeit to a much smaller degree compared with U46619 (Figure 2). We next examined the effect of losartan on a separate signaling pathway, namely that induced by TRAP4. Similar to our finding with ADP (in the presence of indomethacin), losartan did not exert any apparent effects on aggregation induced by 40 µmol/L TRAP4 (Figure 3), even at 25 mg/kg, which almost completely inhibited aggregation triggered by U46619.
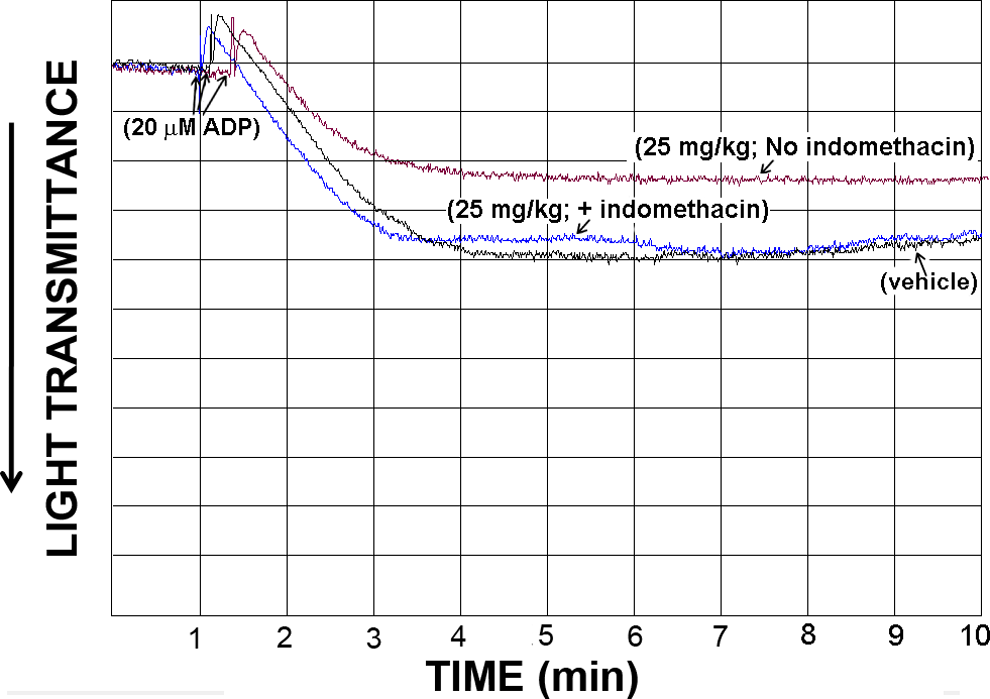
Effect of tail vein IV injections of losartan on mouse platelet aggregation stimulated by ADP, ex vivo, in the presence or absence of indomethacin. Platelet-rich plasma from mice repetitively injected with vehicle or 25 mg/kg of losartan was stimulated with 20 μmol/L ADP, in the presence or absence of indomethacin. Each aggregation curve is representative of multiple traces obtained from 3 separate platelet preparations (n = 3), using blood obtained from 3 groups of (4-5) mice (P > .05).
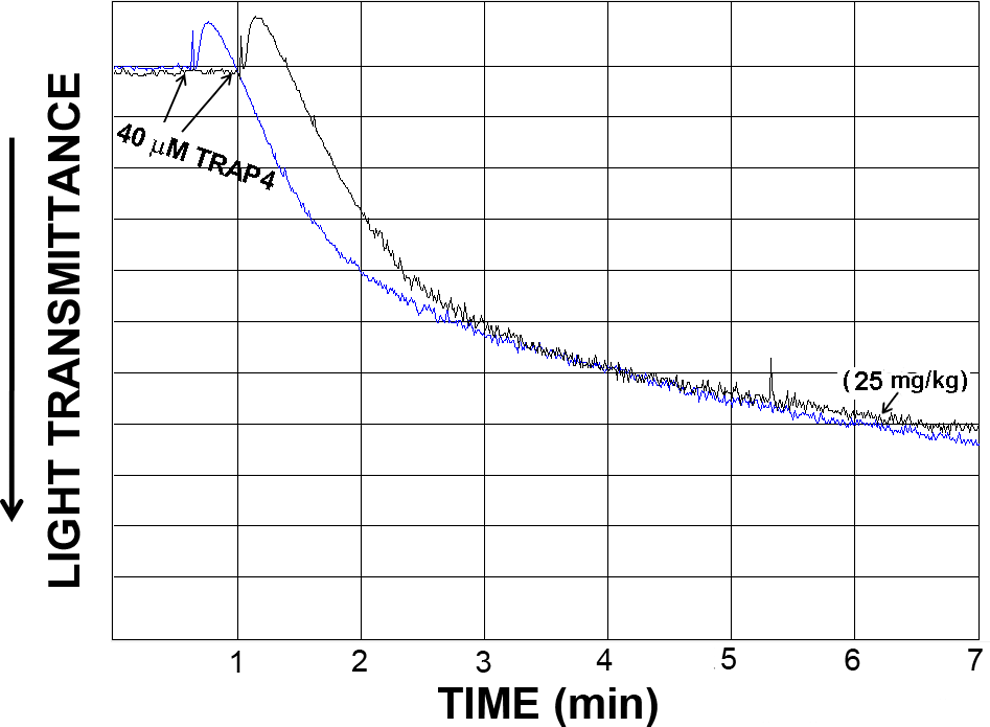
Effect of tail vein IV injections of losartan on mouse platelet aggregation stimulated by TRAP4, ex vivo. Platelet-rich plasma from mice repetitively injected with vehicle or 25 mg/kg of losartan was stimulated with 40 μmol/L TRAP4. Each aggregation curve is representative of multiple traces obtained from 3 separate platelet preparations (n = 3), using blood obtained from 3 groups of (4-5) mice (P > .05).
Losartan Inhibits Thrombosis Development
We next wanted to investigate whether the ex vivo antiplatelet/anti-TPR effects of losartan would translate into in vivo effects. Thus, to determine whether a potential clinical phenotype can be observed, a mouse carotid artery injury model was utilized, and the time for occlusion was measured after FeCl3-induced injury. Losartan was injected at 10 mg/kg (selected based on the ex vivo data) for a 5-day period. It was found that the losartan-injected animals exhibited an increase in the time for occlusion of the injured vessel (P < .001), in comparison to those injected with the PBS vehicle control (Figure 4). This result provides evidence that losartan does indeed possess the capacity to delay the development of thrombus occlusions and hence may provide thromboprotective properties.
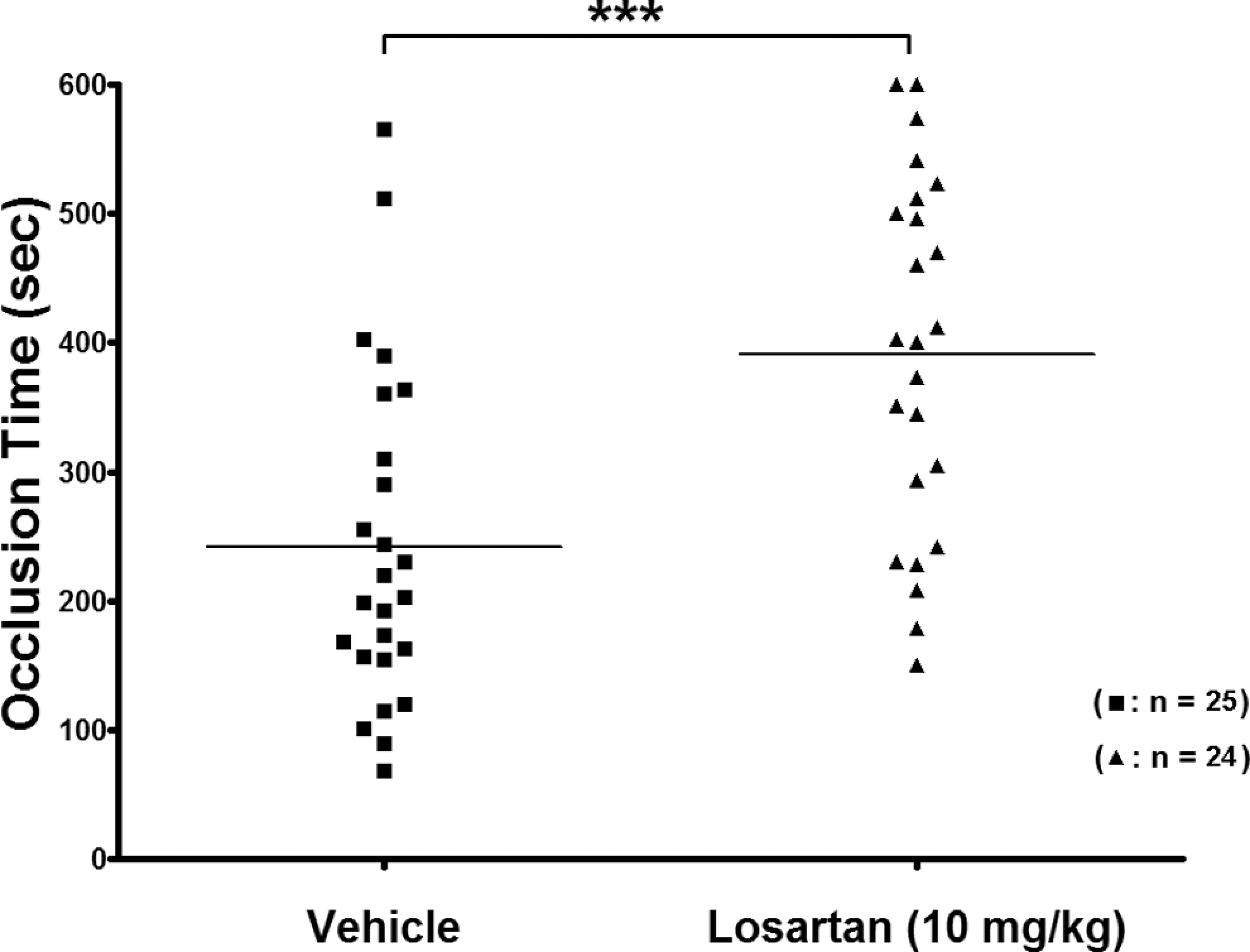
Effect of losartan tail vein IV injections on the time for occlusion in a carotid artery injury thrombosis model. Mice were repetitively injected (5 days) with vehicle control or 10 mg/kg losartan. Losartan treatment resulted in an increased time for occlusion, when compared with vehicle-treated mice (P < .001). Each point represents a single animal.
Losartan Does Not Impair Hemostasis
To determine whether losartan’s antithrombotic activity is associated with bleeding, we measured the tail bleeding time in mice. Again, mice were administered a daily IV injection of 10 mg/kg of losartan, for 5 days. Interestingly, losartan-treated animals did not have a significantly longer bleeding time (P > .05) when compared with the PBS (vehicle)-injected mice, over the same period of time (Figure 5). Thus, it appears that there is no impairment of hemostasis at the dose of 10 mg/kg losartan, even though ex vivo results showed antiplatelet activity. This result combined with ex vivo and the thrombosis model data points to a potential antiplatelet (and antithrombotic) activity, which is not commensurate with a detrimental bleeding phenotype often associated with traditional therapies such as aspirin or clopidogrel.
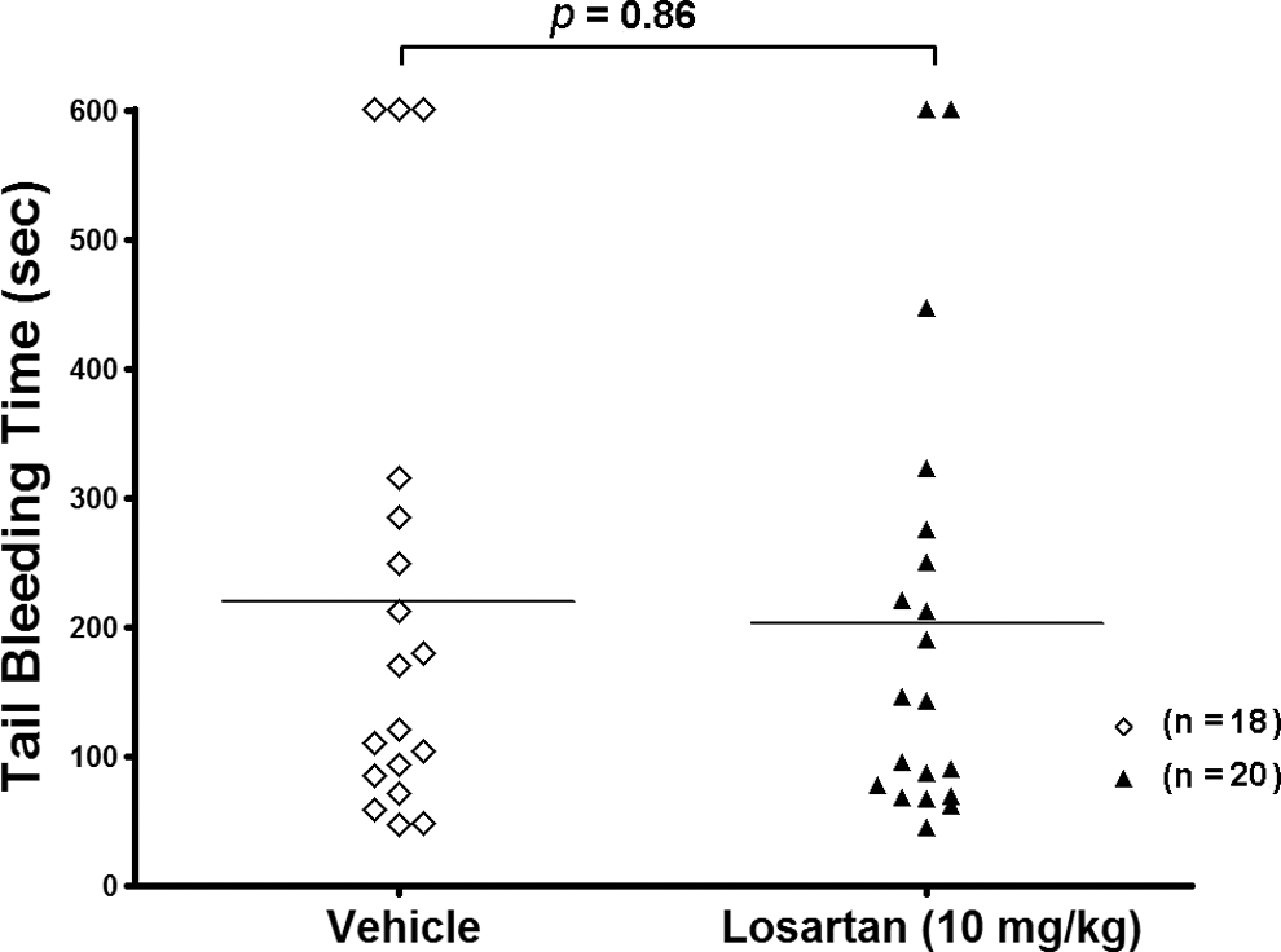
Effect of losartan tail vein IV injections on bleeding time/hemostasis. Mice were repetitively injected (5 days) with vehicle control or 10 mg/kg losartan before their tail bleeding times were measured. Losartan treatment resulted in no significant (P > .05) increase in the bleeding time of mice and thus no significant effect on hemostatsis. Each point represents a single animal.
Discussion
The understanding of the biology of platelet activation pathways and the role of various surface receptors is essential for designing novel antiplatelet drugs. In this connection, the cost of the research and development involved in traditional drug discovery is extremely high and fraught with pitfalls and bureaucratic obstacles. The alternative approach of drug repurposing provides an avenue for research and development in which a drug that is used as an antidiabetic, for example, can be a potential candidate for antiplatelet effects. 10 However, for a drug to be repurposed, certain requirements should be met, such as sufficient similarity in the structure/chemistry with other known ligands. From this method, homologous derivatives of current drugs may be modeled as well.
We speculated that the antihypertensive angiotensin (AT1) receptor antagonist losartan may prove to have significant ex vivo and in vivo antiplatelet and thromboprotective properties, given the previously published in vitro work. Specifically, losartan has already been shown to prevent TXA2-induced contraction of the carotid artery in canine models, as well as inhibit human platelet aggregation (in vitro). 19,20 Hence, the (in vivo) significance of losartan/TPR interaction in mouse models has not been done and would be very beneficial considering the high degree of homology between the mouse and human TPR. 21
To optimize the in vivo experimental design, an ex vivo analysis of the potential dose-dependent inhibition of platelet aggregation by losartan was initially conducted. Chronic administration of losartan into live animals revealed an inhibitory profile of platelets that was specific to the TPR pathway. For example, while the highest dose is used, that is, 25 mg/kg showed nearly complete inhibition of TPR-dependent platelet aggregation, it failed to inhibit aggregation triggered by ADP or TRAP4. Interestingly, in the absence of indomethacin, losartan did inhibit ADP-induced aggregation. This finding is consistent with the notion that ADP-mediated aggregation involves a TXA2/TPR component. Taken together, these results are in agreement with the previously published work where it was shown that losartan and its active metabolite EXP3174 competed for binding to TPRs. 19 However, the same probably cannot be said for similar class drugs such as valsartan and candesartan, at least in vitro. 22 Interestingly, valsartan has been shown to exert inhibitory effects against ADP in human platelets 23 ; which does not appear to be the case with losartan.
Since 10 mg/kg was established to produce “sufficient” inhibition of platelet aggregation (eg, >80%) in the ex vivo mouse trials, we employed this dose in our in vivo studies to determine whether these antiplatelet effects would translate into a clinically relevant antithrombotic phenotype. The results obtained do indeed point toward losartan having thromboprotective properties as the time to occlusion was significantly prolonged in the losartan-treated animals compared with the vehicle control. This finding is similar to that found with the previously mentioned experimental TPR antagonist 24 Z-335, which had also shown some promise in clinical trials in healthy volunteers. 7
Given that the potential application of any drug as antiplatelet therapeutic is often hampered by excessive bleeding, an adverse effect usually associated with common antiplatelet drugs, 25 we next examined losartan’s effect in this regard. Data from the tail bleeding time assay demonstrated no significant difference in bleeding time between the losartan-treated and the control mice. This is an important finding when considering the potential cost versus benefit of any treatment option for thrombotic disorders. A lower incidence of bleeding reduces the potential for hemorrhage or complicated ulcers in the GI tract, which is a definite positive under such circumstances. Proponents of clopidogrel, one of the most common antiplatelet drugs, which has high ulcer tendencies and excessive bleeding in the GI, would suggest that concomitant use of an acid reducing PPI, such as omeprazole, can safely combat the dangers of clopidogrel. 26 However, the medical community and the FDA have established black label warnings regarding the concomitant use of PPIs and clopidogrel. It was feared that the PPI, while trying to solve the GI issue, 27,28 may interfere with clopidogrel’s metabolism by cytochrome P450 2C19 to the active form, thus reducing the protective benefits. Based on these considerations, we believe that antiplatelet drug discovery is far from being complete, considering the dangers that are present in the current therapies.
It is also important to comment on the role of the active metabolite EXP 3174. Losartan is not the active form which exhibits antihypertensive properties, rather it is known that EXP 3174 shows a much more potent antihypertensive effect. 29 Conversely, the antiplatelet activity of EXP 3174 was found to be of much lower effect than that of losartan, in vitro . 12 Considering losartan is clinically administered as an oral tablet, 30 one would expect “lower” losartan levels would become available in the blood to interact with platelets and provide a thromboprotective effects. This issue perhaps should be addressed if it is to be repurposed for human antiplatelet use.
It is noteworthy that the PERFORM trial found no advantage in efficacy or safety for the TPR antagonist terutroban over aspirin for the secondary prevention of ischemic stroke or transient ischemic attack. 31 Nevertheless, the authors acknowledge that whether there are other clinical settings in which selective TPR blockade may provide an advantage over aspirin remains unknown. 31 This trial was discontinued prematurely, and subsequently, the development of the drug and all other trials of it in progress were halted.
In conclusion, the present findings demonstrate that losartan, an antihypertensive agent, has the capacity (1) to block TPRs in platelets, at least in part, by directly interacting with the receptor protein (Figure 6) and (2) to protect against the development of occlusive thrombi. Thus, losartan would provide dual benefits particularly in hypertensive patients, who are under high risk of developing thrombosis. Consequently, aside from its potential use as an antithrombotic agent, losartan’s chemistry may provide a “blueprint” for designing/repurposing novel derivatives which may have the potential to serve as antiplatelet and thromboprotective agents but are deprived of the usually concomitant bleeding adverse effects. Such avenue for drug repurposing or rediscovery should utilize the structural similarity, particularly, the “pharmacophores” losartan shares with TPR ligands currently limited for research use. Nonetheless, clearly, more work must be done to decide on the viability for including losartan in the discussion of potential/future antiplatelet therapies, and perhaps the fact that it appears not to pose a bleeding phenotype, would expedite this process and make it worthwhile.
A schematic model describing the proposed mechanism by which losartan blocks platelet thromboxane A2 receptors (TPRs).
Footnotes
Fadi T. Khasawneh: experimental design, data interpretation/analysis, and manuscript preparation; John P. Murad: performed experiments and manuscript preparation; Enma Veronica Paez Espinosa: performed experiments and manuscript preparation; Harold J. Ting: performed experiments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Intramural Funding from the College of Pharmacy at Western University of Health Sciences (to FTK).
