Abstract
Introduction:
Angiotensin-converting enzyme inhibitor (ACE-I) and angiotensin receptor blocker (ARB) discontinuation during acute heart failure (AHF) is associated with increased mortality following hospitalization. Although the etiology of acute kidney injury (AKI) in type 1 cardiorenal syndrome (CRS) has been linked to renal venous congestion, ACE-I/ARB withdrawal (AW) theoretically promotes renal function recovery. ACE-I/ARBs are dose-reduced or withheld in approximately half of patients with CRS, but the subsequent impact on renal function remains largely uninvestigated. This study compared AW to ACE-I/ARB continuation (AC) during CRS.
Methods:
This was a retrospective, single-center chart review. Patients aged 18-89 years admitted from April 2018 to August 2019 with AHF and AKI were identified using discharge ICD-10 codes. All patients were treated with an ACE-I/ARB before admission. Key exclusion criteria included shock, pregnancy, and end-stage renal disease. The primary endpoint was change in serum creatinine (SCr) from admission through 72 hours. Data were analyzed utilizing chi-square and Mann-Whitney U tests with SPSS software.
Results:
A total of 111 admissions were included. AW occurred in 68 patients upon admission. AW patients presented with a higher blood urea nitrogen (P = 0.034), higher SCr (P = 0.021), and lower ejection fraction (P = 0.04). Median SCr change from admission to 72 hours did not differ between groups (AW −0.1 mg/dL vs AC 0.0 mg/dL, P = 0.05). There was no difference in SCr reduction ≥0.3 mg/dL at 72 hours, 30-day readmissions, or ACE-I/ARB prescription at discharge.
Conclusions:
In patients with type 1 CRS, AW was not associated with improved renal function at 72 hours. A larger sample size is necessary to confirm these results.
Keywords
Introduction
Type 1 cardiorenal syndrome (CRS) represents acute kidney injury (AKI) secondary to acute heart failure (AHF). 1,2 Approximately one-quarter to one-third of patients admitted to the hospital with HF present with concurrent CRS. 3,4 This presentation complicates AHF management, as clinicians attempt to optimize cardiovascular hemodynamics without worsening renal function.
Angiotensin-converting enzyme inhibitors (ACE-Is) and angiotensin receptor blockers (ARBs) decrease symptoms, reduce hospitalizations, and improve mortality in patients with HF with a reduced ejection fraction (HFrEF). 5,6 ACE-Is/ARBs cause efferent renal arteriole vasodilation, thereby increasing tubular blood flow and slowing the progression of chronic kidney disease; however, these hemodynamic changes may also precipitate pre-renal AKI, although this phenomenon is predictable and reversible. 7
The mechanism of AKI in CRS is distinct from that of ACE-I/ARBs. Current evidence suggests that CRS results from a combination of decreased renal perfusion secondary to increased preload and increased renal venous congestion secondary to hypervolemia. 8 Despite these differences, as well as limited evidence to support the practice, ACE-I/ARBs are often withheld in patients hospitalized with type 1 CRS. 9 -11
The purpose of this study was to test the clinical hypothesis that ACE-I/ARB withdrawal improves renal recovery in patients with type 1 CRS.
Methods
This was a single-center, retrospective review of electronic medical records at an urban, academic medical center. Patients 18 to 89 years old hospitalized with type 1 CRS from April 10, 2018 to August 1, 2019 were identified through ICD-10 discharge diagnostic codes for HF and AKI (see Supplementary Appendix). Determination of CRS was adjudicated by the lead investigators. Patients were required to be prescribed either an ACE-I or an ARB (including sacubitril/valsartan) on admission. Exclusion criteria included shock, active malignancy, pregnancy, peripartum cardiomyopathy, and end-stage renal disease (see Figure 1). Patients who had their ACE-I/ARB continued (AC) were compared with patients who had their ACE-I/ARB withdrawn (AW). AC was defined as administration of an ACE-I/ARB within 24 hours of admission. The protocol was granted exempt status by the Institutional Review Board of both participating institutions.

Flow diagram.
The primary endpoint was the change in serum creatinine (SCr) from admission to 72 hours. Secondary endpoints included a reduction in SCr ≥0.3 mg/dL from admission at 72 hours, return of SCr to within 0.3 mg/dL above median baseline at 72 hours, change in serum potassium from admission to 72 hours, and change in systolic blood pressure (SBP), diastolic BP (DBP), and mean arterial pressure (MAP) from admission to 72 hours. Dichotomous secondary endpoints included initiation of IV vasodilators, inotropes, mechanical ventilation, or renal-replacement therapy (RRT), 30-day hospital readmission, and ACE-I/ARB prescription on discharge.
Baseline SCr was defined as the median SCr documented for a patient during the 6 months prior to admission. IV vasodilators included nitroglycerin, nitroprusside, and nesiritide. Inotropes included dobutamine, dopamine, and milrinone. Administration of potentially nephrotoxic medications was also collected.
Statistical Analysis
Mean ± SD, median [IQR], or number (percentage) were used for baseline characteristics as appropriate. Categorical variables were analyzed with the chi-square or Fisher’s exact test when appropriate. Continuous variables were tested for normality with the Kolmogorov-Smirnov test and analyzed with the Mann Whitney U test or Student’s t test for nonparametric and parametric data, respectively. A pre-defined sample size of 128 patients (64 patients in each group) was needed to detect a 50% difference in the primary endpoint with 80% power and a 2-sided significance level of 0.05, assuming a median admission SCr of 1.2 mg/dL. A P value <0.05 was considered statistically significant. All analyses were conducted using SPSS Statistics for Windows, version 25 (IBM Corp., Armonk, NY, USA).
A post-hoc analysis was performed to reduce confounding secondary to patient group crossover. Patients in the AC group who only received a single ACE-I/ARB dose and patients from the AW group who were restarted on an ACE-I/ARB after 24 hours were excluded from the post-hoc analysis. Additionally, 2 subgroup analyses compared outcomes among patients with an ejection fraction (EF) ≤ 40% and in patients who did not receive contrast during the admission.
Results
During the study period, 524 admissions were identified with ICD-10 codes for AHF and AKI. Of these, 111 admissions from 105 unique patients met inclusion criteria and were included in the analysis. Lack of treatment with an ACE-I/ARB prior to admission was the primary reason for exclusion (Figure 1).
Baseline characteristics are shown in Table 1. Demographic variables and comorbid conditions were similar between the groups. Overall, 50.5% of the patients had coronary artery disease, 48.6% had atrial fibrillation, and 36.9% had chronic kidney disease. ACE-inhibitors were the more commonly (58.6%) prescribed prior to admission than ARBs (32.4%). Most patients were prescribed concomitant beta-blockers (89.2%) and loop diuretics (69.4%).
Baseline Characteristics.*

* Results presented as no. (%), mean ± SD, or median [IQR].
† Out of 106 total (40 in RAAS given, 66 in RAAS not given).
‡ Out of 58 total (29 in each group).
Patients in the AW group were more likely to have lower systolic blood pressures (133.2 vs 146.8 mm Hg, P = 0.047), higher BUN (1308 vs 793 mg/dL, P = 0.034), and higher SCr (1.7 vs 1.4 mg/dL, P = 0.021) on admission. A baseline SCr could only be assessed for 58 (52.3%) patients in the sample. Of the 58 patients with a median baseline SCr, AW patients had a larger increase in SCr from baseline (0.6 vs 0.2 mg/dL, P = 0.030). AW patients also had a lower ejection fraction (35% vs 43%, P = 0.040).
All patients in the AC group received ACE-I/ARBs within 72 hours of admission compared to 18/68 (26.4%) of AW patients. Intravenous loop diuretics were administered to 91.8% of patients within 72 hours of admission, while 82.9% received beta-blockers. Additional pharmacologic agents administered upon admission, including intravenous vasodilators, inotropes, fluids, and nephrotoxic medications, were similar between groups (Table 2). No patients required vasopressors during the hospitalization.
Medications Administered During Admission.*
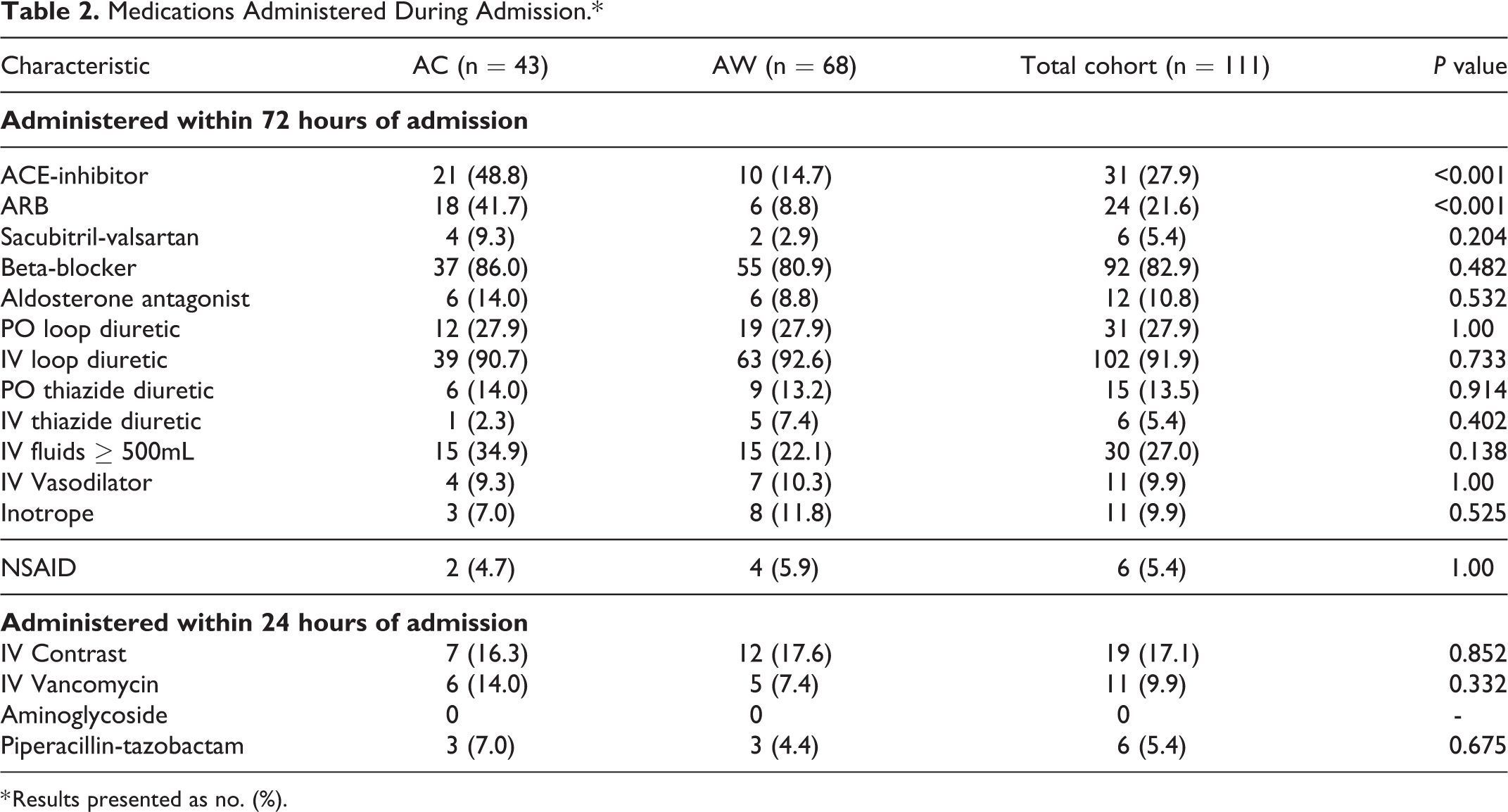
* Results presented as no. (%).
There were no differences between comparison groups in any renal endpoints, including the primary outcome (Table 3). The median change in SCr from admission to 72 hours was 0.0 mg/dL in the AC group compared to −0.1 mg/dL in the AW group (P = 0.050, Figure 2). Numerically more patients in the AW group experienced a SCr decrease of at least 0.3 mg/dL from admission to 72 hours, although this difference was not statistically significant (37% vs 21%, P = 0.078). Only one patient in the AW group required renal replacement therapy within 72 hours.
Primary and Secondary Renal Endpoints.*

Abbreviation: RRT, renal replacement therapy.
* Results presented as no. (%) or median [IQR].
† Out of 58 total (29 in each group).

Change in serum creatinine from admission to 72 hours by comparison group.
After excluding patients for the post-hoc analysis (Table 4), change in SCr at 72 hours (AC 0.1 vs AW −0.1, P = 0.018) was significantly different in favor of the AW group. Additionally, more patients in the AW group achieved a reduction in SCr of at least 0.3 mg/dL from admission at 72 hours (AC 7.7% vs AW 48%, P = 0.003). The subgroup analysis of renal outcomes in patients with HFrEF was consistent with the analyses in the overall cohort (Tables 5 and 6). After excluding patients who received contrast, median change in SCr remained the same as the overall cohort (AC 0.0 mg/dL vs AW −0.1 mg/dL, P = 0.047) except the result was statistically significant.
Primary and Secondary Renal Endpoints in Post-Hoc Analysis.*

* Results presented as no. (%) or median [IQR].
† Out of 38 total (18 AC, 20 AW in each group).
Renal Endpoints Among Patients With HFrEF (EF ≤ 40%).*

* Results presented as no. (%), median [IQR], or mean ± SD.
† Out of 33 total (14 AC, 19 AW).
Renal Endpoints Among Post-Hoc Analysis Patients With HFrEF (EF ≤ 40%).*
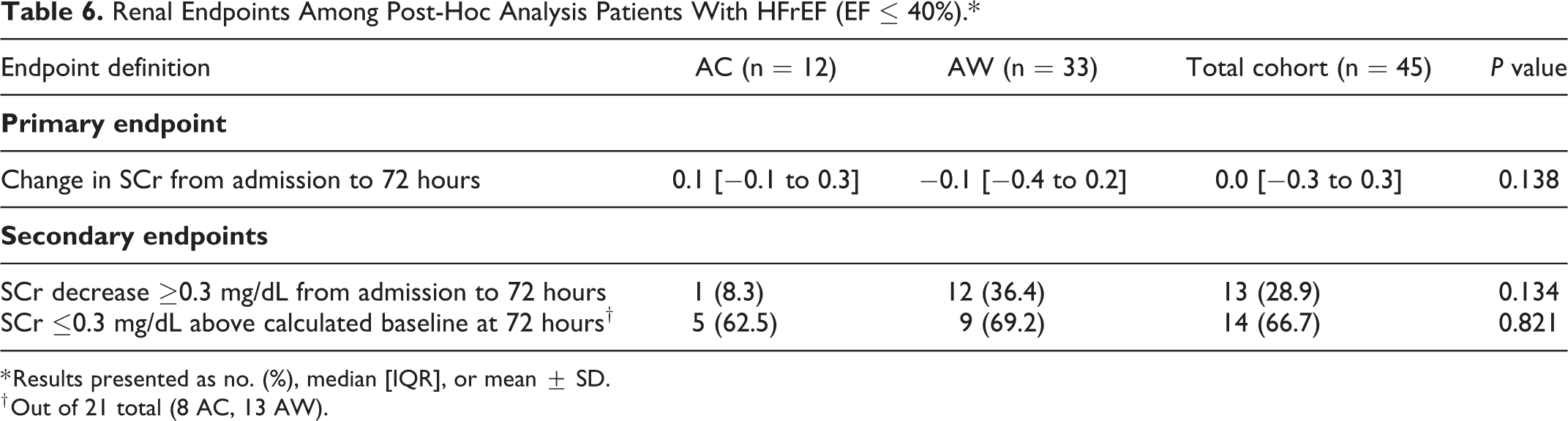
* Results presented as no. (%), median [IQR], or mean ± SD.
† Out of 21 total (8 AC, 13 AW).
Blood pressures were lower in both groups at 72 hours relative to baseline with a mean change in MAP of −14.5 mm Hg (Figure 3). Patients in the AC group experienced a more profound reduction in blood pressure over the 72-hour period compared to patients in the AW group, although this difference was not statistically significant (mean change SBP AC −20.9 vs AW −10.7, P = 0.101).
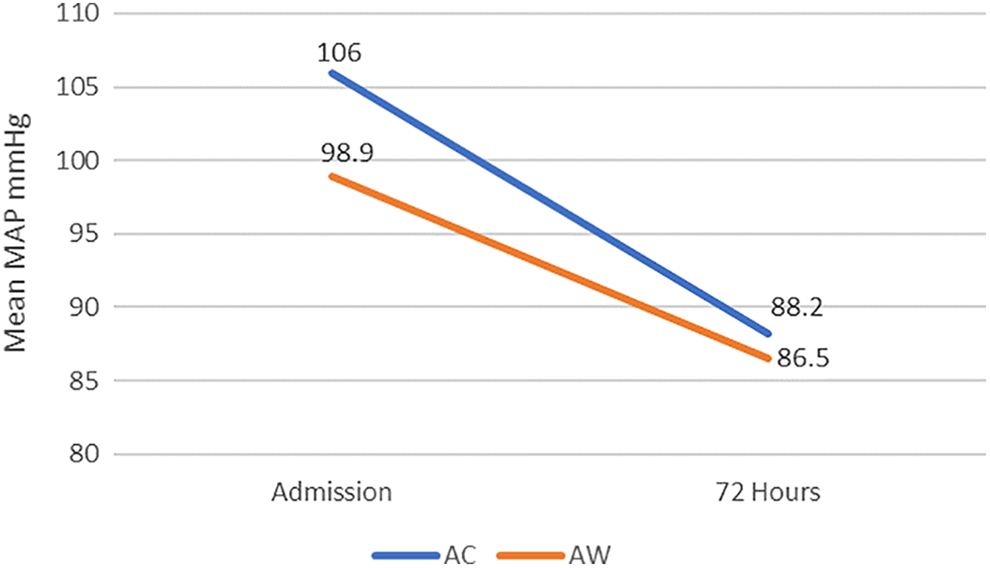
Mean change in MAP from admission to 72 hours by comparison group.
There were no differences in any clinical outcomes between treatment groups, including in-hospital mortality, 30-day readmission, total hospital length of stay, and ICU days (Table 7). There was also no difference in the number of patients receiving escalated medical care, such as intravenous vasodilators and inotropes. Numerically more patients in the AC group had an ACE-I/ARB ordered on discharge compared to patients in the AW group, although this difference was not statistically significant (69.8% vs 61.8%, P = 0.509).
Clinical Outcomes.*

Abbreviations: LOS, length of stay; ICU, intensive care unit.
* Results shown as no. (%) or median [IQR].
Discussion
In this single-center, retrospective study, we found no difference between ACE-I/ARB continuation or withdrawal on renal outcomes in the overall cohort of patients with type 1 CRS, although there was a trend toward better renal function recovery in the AW group (P = 0.05). A significantly greater reduction in SCr was observed among the AW group when patients with ACE-I/ARB administration after 24 hours were excluded from the analysis. Additionally, among patients with a baseline SCr over the previous 6 months, there remained no difference between groups in the proportion of patients within 0.3 mg/dL of their baseline SCr. The data presented here should be considered hypothesis generating and not used to change clinical practice. Further investigations are necessary to determine the optimal management of ACE-I/ARB during type 1 CRS.
Guideline-directed medical therapy (GDMT) for HFrEF includes ACE-I/ARBs to decrease mortality and hospitalizations. 12 Afterload reduction afforded by arterial vasodilators, such as ACE-inhibitors and ARBs, during AHF increases stroke volume and cardiac output. 13 Efferent renal arteriole vasodilation by ACE-I/ARBs improves distal renal perfusion but reduces filtration pressure, thereby causing elevations to SCr. 7 In contrast, high preload (hypervolemia) leading to left ventricular dysfunction and reduced renal perfusion pressure drives AKI in type 1 CRS. 8 Increased renal venous pressure proportionally raises renal tubular hydrostatic pressure resulting in renal arteriole vasoconstriction, decreased renal blood flow, and decreased glomerular filtration. 14 Accordingly, withdrawal of ACE-I/ARBs during type 1 CRS may increase afterload, thereby further depressing left ventricular stroke volume and worsening renal perfusion.
The association between ACE-I/ARBs and hemodynamically mediated AKI leads to a perception that ACE-I/ARBs will worsen renal function in type 1 CRS, resulting in withdrawal of GDMT. Acute discontinuation of GDMT has been associated with reduced survival in patients hospitalized with decompensated HFrEF, 15 possibly because patients are discharged prior to therapy optimization. 16 Non-persistence with HF therapy is also associated with an increased risk of hospitalization. 17 Previous research investigating ACE-I/ARB management in CRS is limited. In a recent prospective study analyzing the impact of incident hyperkalemia in patients hospitalized with HF, researchers found that ACE-I/ARB down-titration in response to incident hyperkalemia was associated with a worse 180-day prognosis compared to patients with stable ACE-I/ARB doses. 18 Similar to our study, Boulos and colleagues 10 noted that patients with hypotension and a greater increase in SCr were more likely to have the ACE-I/ARB dose-reduced or discontinued entirely. Consequently, patients with ACE-I/ARB, beta-blocker, or diuretic down-titration or intravenous fluid administration suffered higher 6-month mortality in the unadjusted analysis compared to patients that did not experience these changes to medical therapy (HR 1.71, 95% CI 1.12-2.61; P = 0.01). 10 Therefore, studies assessing the safety of ACE-I/ARB administration during type 1 CRS are necessary to determine whether the theoretical risk of worsening renal function secondary to ACE-I/ARB administration is valid and warrants acute withdrawal of this vital pharmacologic therapy.
This is the first study to our knowledge to determine the sole impact of ACE-I/ARB withdrawal on renal function in patients with type 1 CRS. Our results did not definitively confirm that withholding ACE-I/ARBs promotes renal function recovery at 72 hours in this patient population. Additionally, we noted that a third of all patients in this cohort had the ACE-inhibitor or ARB discontinued at discharge, and numerically more patients in the AW group experienced this change to medical therapy following hospitalization. This finding may highlight clinician hesitancy to continue ACE-I/ARBs in patients with recent AKI and may also suggest a dependence on clinic follow-up following hospital discharge. A similar urban, academic medical center has reported clinic no-show rates as high as 76% for patients recently hospitalized with HF, 19 and thus, maintaining GDMT on hospital discharge is paramount. Furthermore, acute withdrawal of ACE-I/ARBs may exacerbate the deleterious effects of clinical inertia, the phenomenon in which medications discontinued in the hospital are not restarted in the clinic. 20
This study has several limitations. First, because of the retrospective design, there was inherent selection bias that favored ACE-I/ARB withdrawal in patients with lower blood pressures and higher SCr and BUN levels on admission; however, this finding highlights factors that influence clinical decision-making regarding ACE-I/ARB management in patients presenting with type 1 CRS. Identification of patients via ICD-10 codes introduces the potential that other types of CRS were present in the sample, since there is no specific code for CRS, but each admission was adjudicated to minimize this risk. Data suggesting restoration of euvolemia (i.e., intake and output, daily weights) was inaccurate and not included in the analysis due to a concern for confounding study results. Medication doses were not collected, and this may have influenced renal endpoints; however, there were no significant differences in concomitant nephrotoxin use between groups. There was approximately 25% cross-over from AW to AC within 72 hours which may confound the interpretation of the primary endpoint. While the exclusion of these patients in the post-hoc analysis did yield statistically significant results favoring AW, the clinical importance of a difference of 0.2 mg/dL may be negligible and needs to be assessed with a larger sample of patients. Lastly, our assessment of 30-day hospital readmissions was limited to regional hospitals that share electronic health records with our institution.
In this study, AW was not associated with improved renal function at 72 hours in patients with type 1 CRS. Adequately powered studies of this population in observational databases are necessary to confirm study results, and prospective investigations are required to provide more conclusive evidence to guide practice in CRS.
Supplemental Material
Supplemental Material, sj-pdf-1-cpt-10.1177_10742484211022625 - Impact of Angiotensin-Converting Enzyme Inhibitors and Angiotensin Receptor Blockers on Renal Function in Type 1 Cardiorenal Syndrome
Supplemental Material, sj-pdf-1-cpt-10.1177_10742484211022625 for Impact of Angiotensin-Converting Enzyme Inhibitors and Angiotensin Receptor Blockers on Renal Function in Type 1 Cardiorenal Syndrome by Daniel T. Ilges, Morgan L. Dermody, Caitlyn Blankenship, Valerie Mansfield and Joseph S. Van Tuyl in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
All work was conducted at SSM Health Saint Louis University Hospital, St. Louis, Missouri.
Acknowledgments
The authors would like to recognize Emily Rousseau for her assistance with data collection.
Author Contributions
DTI and JSVT were responsible for study conception, design, data analysis, initial manuscript draft, critical review, and revisions. VM contributed to study design, manuscript review, and revisions. DTI, MLB, and CB were responsible for data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
