Abstract
For older adults with cancer, communication is among the most critical aspects of care. This commentary examines two communication timepoints that may confer disproportionate clinical impact: the initial consultation and major change points, such as progression, hospitalization, or functional decline. We argue that a validating and clear communication style—offering clear recommendations while honoring patient values—anchors trust and improves outcomes. The initial consultation should focus as much on understanding the person as the pathology, incorporating functional status, caregiver capacity, and treatment priorities. Evidence from the Mohile COACH trial demonstrates that geriatric assessment can enrich these conversations and ensure vulnerabilities shape the care plan. While electronic health record (EHR)-supported prompts may strengthen communication, their applicability varies across settings, and such tools require contextual adaptation. At major change points, structured approaches such as best case/worst case framing, agenda setting, and teach-back provide reliability and clarity. Beyond the clinician, learning electronic health records (EHRs) can flag inflection points, nudge best practices, and embed documentation of goals into routine workflows. Framing communication as a measurable intervention—one that is trainable, auditable, and supported by systems—offers a path toward oncology care that is not only evidence-based but also values-based, particularly for older adults, while avoiding mechanistic, tick-box use of communication tools that could undermine authenticity.
Plain Language Summary
Older adults with cancer often face not just medical decisions, but also questions about how treatments will affect their independence, daily lives, and what matters most to them. This article looks at how good communication can guide care during two critical moments: the first visit with an oncologist and major turning points, such as when the cancer progresses, a patient is hospitalized, or there is a noticeable decline in health. At the first visit, the focus should go beyond the tumor. Doctors should ask about memory, walking, social support, and goals for care. This helps build trust and ensures treatment plans fit the patient’s overall health and values. Later, when the disease changes or health worsens, structured communication tools—such as discussing the best and worst possible outcomes—help patients and families prepare and make informed decisions. The authors highlight evidence showing that tools like geriatric assessment, psychosocial support, and palliative care improve both quality of life and medical outcomes. They argue that communication should be seen as a core treatment, not an “extra.” By aligning medical care with the patient’s function, priorities, and values, conversations themselves become a powerful way of delivering better, more person-centered cancer care.
Framing the Challenge
Cancer in older adults is characterized by heterogeneity in function, multimorbidity, and variation in decision-making preferences.1-4 Yet older adults remain underrepresented in clinical trials, creating evidence gaps that heighten uncertainty in treatment decisions.3,5,6 In this environment, clinicians often default to extremes: permissive communication, in which patients are left to shoulder the decisional burden, or authoritarian communication, in which recommendations are imposed paternalistically. Both approaches risk undermining patient autonomy and trust. 7 A more effective path is a clear and validating communication style, in which oncologists offer clear, proportionate recommendations grounded in the patient’s values while affirming the legitimacy of their emotions and perspectives. 8
Effective communication is consistently linked with measurable clinical outcomes, including adherence to treatment, patient satisfaction, reduced decisional regret, and improved quality of life. 9 However, these benefits require avoiding mechanistic, “tick-box” behaviors that may arise when communication is overly quantified.
In oncology, these outcomes are particularly critical given the high stakes of treatment choices, the complexity of therapeutic regimens, and the often-limited time frame in which decisions must be made. Communication failures are also among the most common sources of patient complaints and are tied to decisional regret. 10 Thus, elevating communication from a “soft skill” to a deliberate, evidence-based intervention is not simply aspirational but a clinical necessity.
Beyond outcomes, communication is central to preserving dignity. For many older adults, cancer threatens independence, identity, and roles within family and community. A validating communication style recognizes the patient not only as a host of disease but as a whole person. This reframing shifts oncology from being exclusively tumor-focused to person-centered. 11 In practical terms, it means eliciting what matters, offering a clear recommendation that honors those priorities, and returning to these touchstones at each decision point in a patient’s illness trajectory; from the initial visit, the times of disease progression, until the transition to palliative care.
The Initial Visit: Establishing a Foundation
The initial consultation should focus as much on understanding the biology of the cancer as on understanding the older adult who carries the disease. Beyond staging and pathology, this means appreciating comorbidities, functional status, caregiver capacity, and the patient’s own priorities for treatment and quality of life. A geriatric assessment offers one structured way to bring these domains into the consultation. 12 The geriatric assessment is an evaluation of an older adult’s comorbidities, medications, physical and cognitive function, nutritional status, psychological state, and social support. Its fundamental tenet is person-centeredness, orienting problem evaluation and intervention around an individual’s specific biopsychosocial situation to maximize function and health.
In the Mohile COACH trial, incorporating geriatric assessment findings into oncology visits improved patient–clinician communication, increased discussions of age-related concerns, and led to more individualized care planning. 13 Importantly, GA findings alone do not enhance communication; benefits arise only when clinicians interpret and apply them to refine risk profiles and treatment options. By making vulnerabilities visible—such as falls, cognitive changes, or caregiver strain—the geriatric assessment can redirect conversations from disease alone to the whole person. When used before or in the beginning of the initial visit, it strengthens rapport, surfaces values that should guide treatment, and sets the expectation that both medical and personal contexts will shape care. Notably – it is not only the performance of the GA but the way it is incorporated into the oncology visit that is important. Findings must be interpreted by the oncologist to better classify a patient’s frailty status to help build a risk profile for different treatment strategies and identify areas of vulnerability. Only then can clinicians use the information to predict likely outcomes and tailor treatment options.
At the start of the visit, setting a joint agenda is crucial, including prompting about sensitive issues such as caregiver strain, functional worries, or financial stress that may not be volunteered. As these visits typically include a designated caregiver, first determining the patient’s desired level of involvement in information sharing and decision making helps to create a safe environment and enhances communication efficacy. Clarifying the patient’s preferred decision-making role helps prevent mismatched expectations. Research suggests that when role preferences are not assessed, up to one-third of patients experience discordance between their desired and actual involvement in decisions, which is associated with lower satisfaction and higher anxiety. 14 By then explicitly asking patients what they most wish to address while clarifying the clinician’s priorities, oncologists demonstrate respect for patient agency and reduce the risk of overlooking pressing concerns. This step also enhances efficiency by ensuring mutual clarity. Agenda setting also empowers patients, particularly older adults, to voice concerns that may not otherwise be volunteered, such as worries about caregiver burden, financial toxicity, or functional independence. Agenda setting includes outlining the planned steps of the discussion(learn about the patient, share information about the disease, providing next step options, making a decision with plan for follow up) and clarifying what type of decision would ideally be made by the end of the visit (whether that be chemotherapy choice, pursuing a biopsy for more information, pursuing surgical or radiation intervention).
Understanding and values elicitation follow naturally. Using Ask–Tell–Ask, 15 oncologists can first gauge the patient’s perception of their illness, then provide succinct, jargon-limited information, and finally confirm understanding. Narrative elicitation questions such as “What are you hoping treatment allows you to keep doing?” or “What worries you most?” provide insights into functional goals and existential concerns.
Only after goals are made explicit should treatment options be introduced. While clinicians may best understand options in terms of cure, prolongation or life or symptom relief, translating these options into patient defined goals ensures recommendations are situated within what matters most to the patient. Best case/most likely/worst case frameworks make benefits and risks tangible by linking them to function and place
16
—for example, maintaining independence at home versus requiring hospitalization. Emotional responses should be acknowledged through empathic statements, and some may find the NURSE framework (Name, Understand, Respect, Support, Explore) useful, while recognizing that empathy is not formulaic and must remain authentic.
15
The visit should close with teach-back, asking the patient to summarize the plan in their own words to confirm comprehension and reinforce shared understanding.
17
We summarize the suggested flow of the initial treatment visit in Figure 1. Figure 2 provides examples of concrete situations to qualify treatment success or need for revision. Flow of discussion for initial treatment visit Examples of concrete criteria to define treatment success or failure
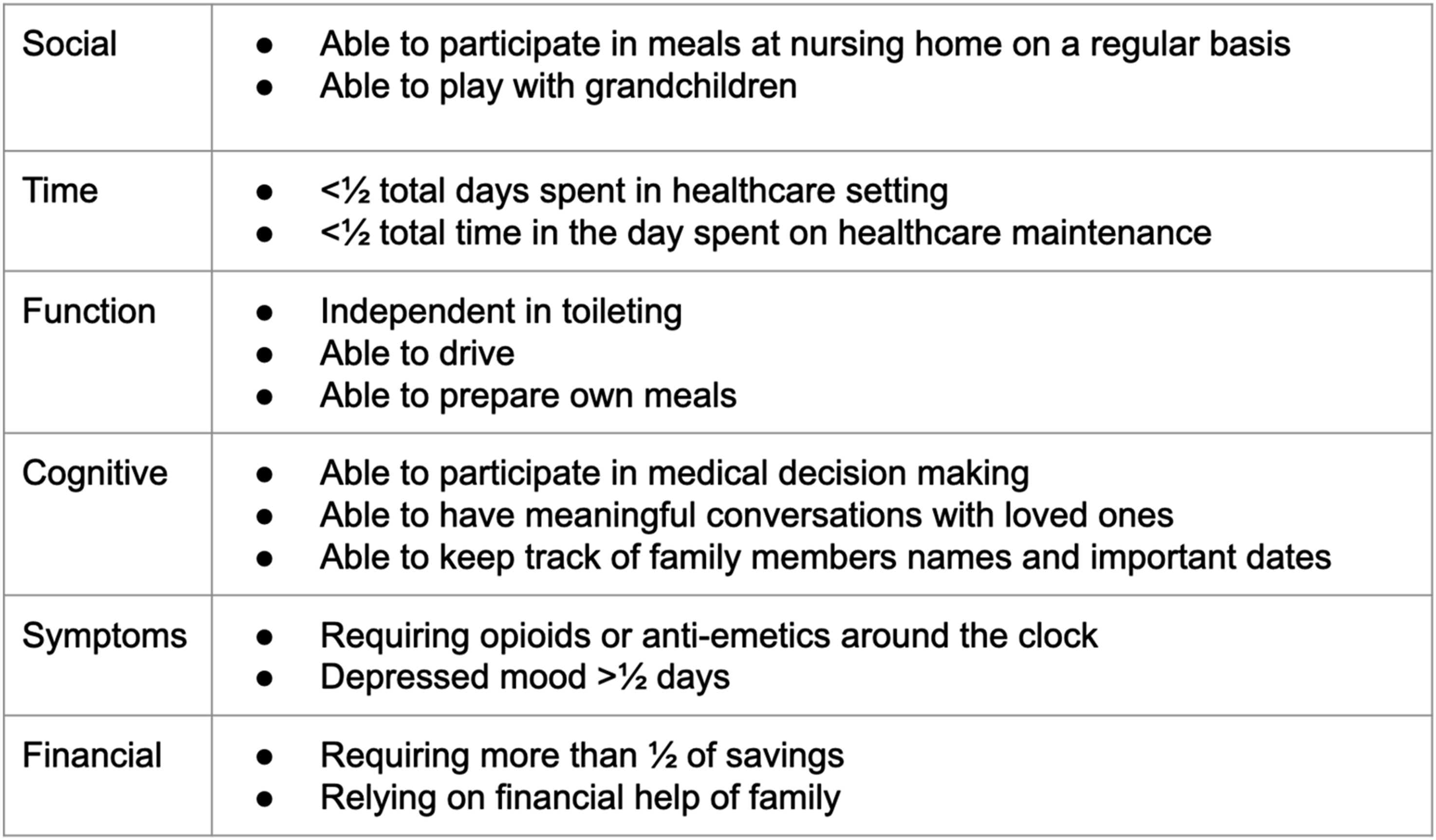
Documenting these conversations in structured EMR fields—including patient goals, preferred role, and key values—ensures continuity and signals to the broader team that communication is a deliberate clinical act. Importantly, as a patient’s care evolves and new allied health care professionals are engaged, such documentation is crucial for effective handoff. This also creates data that can be audited and used for quality improvement efforts. 18
Conversations at Major Change Points
While the initial visit is foundational, major change points in the disease trajectory require recalibration. These include progression of disease, hospitalization, and new functional decline. Each of these moments carries the potential to destabilize the care plan and the patient’s sense of control, making structured communication essential.
Progression of Disease
When progression occurs, oncologists should re-surface patient goals and recalibrate treatment intensity. The SPIKES framework provides structure: setting up the discussion with warning shots, assessing perception, providing knowledge in manageable segments, addressing emotion, and aligning on a strategy. 19 Best case/worst case framing helps contextualize new treatment options or the decision to forgo further therapy. Importantly, geriatric-oncology–specific adaptation of Best Case/Worst Case (BC/WC) highlights several nuances: treat cancer care as a series of decisions rather than a one-off choice; ask permission from the patient before sharing the worst-case scenario; and, when many options exist, present the two most relevant choices to avoid cognitive overload while offering alternatives if a patient expresses concern. 16
An example illustrates the value of this approach. Consider an 82-year-old man with advanced lung cancer who has been on first-line immunotherapy for a year with stable disease. On restaging scans, progression is noted. The permissive approach would be to present all second-line options without guidance, leaving the patient overwhelmed. The authoritarian approach would be to recommend hospice without discussion. The validating approach instead begins with revisiting the patient’s goals—remaining independent at home and avoiding prolonged hospitalization—and then offering a clear recommendation for second-line oral therapy as a time-limited trial, framed in best and worst case terms and paired with concrete criteria for success or stopping. This communication style both guides and honors, and the BC/WC adaptation makes the trade-offs concrete while respecting preferences about hearing worst-case detail.
Post-Hospitalization Debrief
Hospitalizations represent moments of crisis and vulnerability. Post-discharge conversations should integrate the hospital experience into ongoing care. Asking, “What was hardest about your time in the hospital? What worries you most about being home?” invites patients to process the event while giving clinicians insight into new vulnerabilities. Functional recovery (ability to resume baseline activities and independence), caregiver capacity, and fears of recurrence should be reassessed. Options may include de-escalation of therapy, time-limited treatment trials, 20 or a pivot toward comfort-focused care, always tied to what matters most to the patient. Addressing these issues proactively can prevent rehospitalization, reduce caregiver burnout, and improve quality of life. When uncertainty is high, using scenario planning with brief narratives (rather than dense statistics) can foster understanding and anticipatory guidance.
Consider the case of a 75-year-old woman hospitalized for neutropenic fever. After discharge, she expressed fear of returning to the hospital. A validating conversation acknowledges her fear, names the change in her functional status, and frames options in terms of her desire to remain at home. A time-limited treatment trial with built-in review points, coupled with clear home supports, such as home care or home palliative care, may align best with her values.
Functional Decline
Functional decline, such as new dependence or falls, should be explicitly named and validated. This acknowledgment helps address fears of burden or loss of identity. Treatment intensity should be reconsidered, including dose adjustments, regimen changes, or pauses, alongside referral to supportive services such as rehabilitation, nutrition, and psycho-oncology. The aim is to prevent a mismatch between treatment toxicity and patient capacity while affirming dignity. Literature on geriatric assessment underscores that functional status is among the strongest predictors of treatment tolerance and survival.1,3,9,21-23 Integrating these assessments into conversations ensures that decisions reflect both biological and personal realities. As there is a significant risk of evoking anger or confusion if a patient has limited insight into their disease or complicated emotions surrounding independence, an effective discussion of this topic is best performed after appropriate information gathering during the GA or initial session.
Communication as an Intervention at Critical Moments
Understanding that the time required to effectively communicate is not an unlimited resource, careful identification of appropriate times to trigger conversation can help clinicians better utilize their time and the time of supportive staffing. The recent stepped palliative care (PC) trial offers a compelling model for how supportive care can be delivered in a more tailored and scalable fashion. 24 Unlike the traditional “early PC for all” framework, 25 which assumes every patient with advanced cancer requires frequent palliative care visits from the time of diagnosis, the stepped model stratified intensity according to need. Patients received initial PC exposure, but visit frequency and service intensity were escalated only at key inflection points—such as a significant drop in quality of life scores, a hospitalization, or a treatment transition. Results showed noninferior quality of life compared to universal early PC, while conserving scarce workforce resources and directing specialty input where it mattered most. The trial validated a principle long understood in geriatrics and oncology alike: care is most impactful when it is timed to patient need, not simply disease stage.
The stepped PC model underscores that communication is not an accessory to treatment but an intervention in its own right. Well-timed discussions about values, prognosis, and goals alter the trajectory of care just as a drug or procedure might. Evidence demonstrates that serious illness communication reduces unwanted hospitalizations, increases hospice use, and improves patient and caregiver outcomes.26,27 Embedding structured conversations into milestone visits therefore represents not only good practice but a measurable clinical intervention—one with outcomes that can and should be tracked. Figure 3 suggests a possible step up model for monitoring. By incorporating information from patient and caregiver reported outcomes, EMR checks and office visits, the right intervention can be applied earlier. Step up care model for adding interventions based on recurrent needs assessment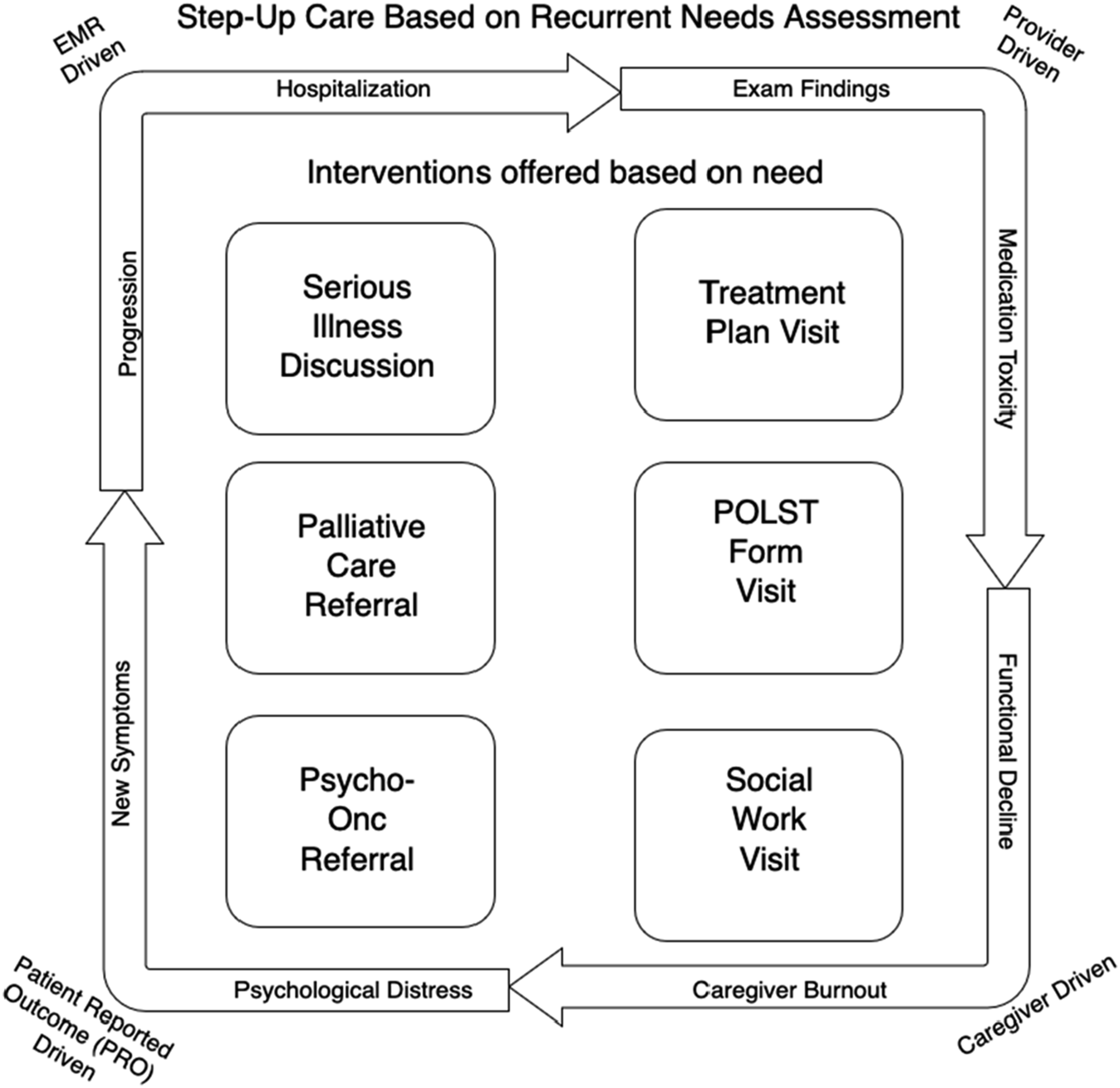
Translating Trial Findings Into EMR Infrastructure
The next challenge is operational: how do we prompt clinicians to address all relevant topics in the initial visits, provide a standardized framing for documenting values and preferences for information sharing, and bring a stepped, need-based approach into real-world oncology practice? The Electronic Medical Record(EMR), and ideally a learning EMR, can serve as the engine.
The Role of a Learning EMR
The demands of oncology practice make it challenging to consistently prioritize communication, even when clinicians recognize its importance. A learning Electronic Medical Record (EMR) can serve as a critical partner by flagging key changes, nudging best practices, and standardizing documentation. • Treatment Plan Changes. Within clinical pathways platforms such as BEACON, used here as an illustrative example rather than a universal system a transition from first-line to second-line therapy—or a pause in treatment—can automatically trigger a communication flag. The system could suggest a “milestone conversation” template, pre-populated with prompts for prognosis, values, and best case/worst case framing - Recognizing that follow-up communication may be shared across allied health professionals and requires reliable interprofessional information flow. • Radiology and Pathology Reports. Natural language processing can scan finalized reports for terms like “progression,” “new lesions,” or “disease advancement.” When detected, the EMR can automatically notify the oncologist and prompt the care team to schedule a progression-focused visit. • Patient-Reported Outcomes (PROs). Stepped models rely heavily on routine symptom and function monitoring. If PROs show a clinically significant decline—such as worsening fatigue, increased falls, or caregiver strain—the EMR can generate an alert, prompting either a phone check-in, an earlier clinic visit, or a specialty referral. Advanced approaches using large language models (LLMs) that have been trained in a patient facing healthcare context could detect concerning sentiment in open-text patient responses, capturing issues that structured symptom checklists might miss. LLMs have essential limitations in sentiment interpretation and require careful governance and contextual adaptation.
Nudging Clinicians at the Right Time
These automated triggers allow the EMR to act as a silent partner in communication. Nudges can be subtle—highlighting a red “communication needed” banner on the patient’s chart—or active, such as inserting a templated checklist for values and goals into the visit note. The goal is not to replace clinician judgment but to ensure that no major inflection point passes, while avoiding checklist-driven communication and supporting skills learned through training.
Examples include: • A prompt to assess caregiver burden after hospital discharge. • A reminder to have a serious illness conversation when documenting progression. • A defaulted order to palliative care referral when a patient meets consensus criteria, such as repeated hospitalizations, refractory symptoms, or progressive functional decline. • A flag suggesting psychosocial oncology consultation, typically provided by psycho-oncology, social work, or behavioral health clinicians, with workload and referral pathways defined, when PROs show persistent distress, loneliness, or caregiver burnout.
Bi-Directional Nudging: Extending to Patients
Communication cannot be a one-way street, and patient-facing nudges must be adapted to local technology, staffing, and patient literacy. A learning EMR can also nudge patients and caregivers through portals, apps, or automated text systems. When a new treatment plan is ordered, patients might receive a short, plain-language summary with a question prompt list (QPL) to bring to their next visit. 28 If a PRO survey shows increasing distress, the system could push tailored resources—such as caregiver support hotlines, psycho-oncology services, or coping strategies—while simultaneously notifying the care team. This dual nudging ensures that patients are both informed and activated, rather than passively waiting for clinicians to interpret data.
Three Pillars of Transforming Practice: An Example
If communication is to be recognized as a clinical intervention, it requires the same infrastructure as chemotherapy or surgery: training, systems integration, and measurement. In this section we offer as example a successful proof of concept with The Serious Illness Care Program (SICP), implemented at Penn Medicine and the Abramson Cancer Center, which illustrates how a structured initiative can transform communication practice at scale. 29 By combining clinician training, leadership support, and electronic health record (EHR) infrastructure, the program quadrupled documentation of serious illness conversations, decreased unwanted end-of-life treatment, and improved hospice utilization. These results highlight that skills alone are insufficient—system design is equally critical. 27
Training and Clinician Support
Training is the first pillar. Communication skills are often assumed rather than taught, leading to variability in practice and clinician discomfort with prognosis, uncertainty, and values-based dialogue. The SICP addressed this gap through small-group experiential workshops, role-play, and observation-based feedback for all oncologists at the beginning of the fiscal year. Clinicians were given RVU credit to participate, and champions across disease teams reinforced cultural buy-in. Importantly, training was not a “one-and-done” activity but supported through coaching, peer feedback, and integration into annual performance review. 29 This model shows how training can normalize communication as part of professional competency, rather than an optional add-on.
EMR Integration and Nudging
The second pillar is embedding communication into workflow. At Penn, a standardized EHR template mirrored the SICP guide, 26 ensuring conversations were not only conducted but also documented in a visible and auditable way. This intersects with the third pillar of measurement, where dashboards tracked performance by disease site and individual clinician, enabling data feedback loops. More recently, Penn deployed machine learning–based mortality prediction tools embedded in the EMR to identify patients at high risk of 180-day mortality. These tools fed into “Conversation Connect,” a program that nudged oncologists via weekly emails, peer comparisons, and same-day text prompts. This intervention increased serious illness conversations nearly fourfold among high-risk patients, with downstream reductions in late-life chemotherapy and health care spending.
Toward Learning Health Systems
Taken together, these initiatives demonstrate that communication quality can be trained, measured, and systematized. A learning EMR becomes the bridge between clinician skill and health system accountability: flagging patients who may benefit from conversations, prompting clinicians to act, and capturing documentation in structured fields that can be analyzed and improved over time. This creates the infrastructure for continuous learning, similar to how tumor boards or clinical trials generate and refine evidence.
Extending to Supportive Services
Finally, integration must extend beyond oncologists; supportive care clinicians must also have access to the same communication tools and EMR prompts. A well-designed EMR can route patients to palliative care, psycho-oncology, or geriatric services when red flags are detected—such as functional decline, distress, or caregiver burden. In this way, communication functions as a triage tool, channeling patients to the right supportive resources at the right time. This step transforms individual conversations into system-level interventions that directly improve outcomes and equity.
Conclusion
Focusing on communication as a clinical intervention has the potential to reframe the standard approaches of geriatric oncology. Optimal care for older adults is not only about drug selection but about aligning treatment intensity with function, independence, and patient priorities. Integrating structured communication approaches with geriatric assessment creates a unified, patient-centered model. Smart EMRs can reinforce this by flagging inflection points such as disease progression or functional decline and nudging clinicians toward timely conversations, referrals to palliative care, or engagement with psycho-oncology, referring specifically to psych-oncology clinical services.
For older adults with cancer, communication is not peripheral to care—it is care. The most consequential conversations occur at the initial visit and during major change points, when treatment goals and values must be recalibrated. By embedding training, structured workflows, and EMR-enabled supports, health systems can elevate communication to the level of other core interventions, ensuring care is both evidence-based and values-based. In doing so, oncology advances not only survival, but also dignity and quality of life.
Footnotes
Author Contributions
Conception and design: CZ, GA, RS
Administrative support: RS
Data analysis and interpretation: All authors
Manuscript writing: All authors
Final approval of manuscript: All authors
Accountable for all aspects of the work: All authors.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
