Abstract
Introduction
Prostate cancer is frequently diagnosed at ages when men may also have comorbidity. Access to care may influence both comorbidity management and cancer treatment and recovery. We examined the association between comorbidity and postoperative outcomes among men with prostate cancer in the universal access Military Health System (MHS).
Methods
We identified a cohort of men diagnosed with non-metastatic prostate adenocarcinoma from 2001-2014 who received radical prostatectomy (RP) within 1 year of diagnosis in the MilCanEpi database, which links the Department of War Cancer Registry and MHS Data Repository. We used ICD-9 diagnosis codes to capture 90-day postoperative general and genitourinary (GU) complications and hospital readmissions; and 18-month postoperative GU complications. Poisson regression estimated the adjusted risk ratios (ARRs) and 95% confidence intervals (CIs) for the outcomes associated with comorbidity (0, 1-2, or ≥3) measured using the Elixhauser Index.
Results
The study included 5645 men with non-metastatic prostate cancer (mean age 57.9 ± 7.7 years) who received RP; 39.9% of patients had no comorbidity, 43.9% had 1-2 conditions, and 16.2% had ≥3 conditions. Patients with ≥3 comorbidities had statistically significant higher risks of 90-day general (ARR = 1.88, 95% CI = 1.34, 2.64) and GU (ARR = 1.20, 95% CI = 1.06, 1.36) complications and hospital readmission (ARR = 1.59, 95% CI = 1.12, 2.26) relative to men with no comorbidity. At 18-month post-RP, men with 1-2 comorbidities (ARR = 1.19, 95% CI = 1.05, 1.35) and ≥3 comorbidities (ARR = 1.32, 95% CI = 1.13, 1.55) had statistically significant higher risk of measured GU complications relative to men with no comorbidity.
Conclusions
In the MHS, higher comorbidity was associated with an increased risk of 30-day and 18-month complications and 90-day readmissions following RP for prostate cancer. This study identifies a need for risk management strategies to reduce complication rates among men with higher comorbidity levels diagnosed with prostate cancer and treated by RP.
Keywords
Introduction
Prostate cancers are the most commonly diagnosed solid tumors among men in the U.S. 1 Over 90% of prostate cancers are diagnosed at localized or regional stages where the cancer is confined to the prostate or nearby tissues.1,2 The primary management of these non-metastatic tumors, ie, tumors that have not spread beyond the prostate and surrounding tissue, includes surgery, radiation therapy with or without androgen deprivation therapy, or protocols of active surveillance.3,4 Men often present with prostate cancer during middle or older age, ie, age 50 or older, when they may be receiving care for comorbid conditions such as cardiovascular disease or diabetes.1,5 The presence of comorbidity may influence treatment decisions, response to treatment, or survival for men with prostate cancer.5–10 Thus, understanding the impact of comorbidities on treatment outcomes is an important part of prostate cancer care.
The presence of comorbid medical conditions has been shown to increase the risk of general cardiac, respiratory, and vascular complications and readmissions following cancer surgery.11–14 For men who undergo surgery for prostate cancer, comorbidity may affect experience of both general surgical and genitourinary (GU) complications.12,15–17 Several studies have reported an increased risk of longer hospital stay, infections, incontinence, or sexual dysfunction in the 30-90 day period after prostate cancer surgery associated with higher comorbidity level.16–18 Further, a few studies have reported specific associations with a history of heart disease, diabetes mellitus, or obesity.15,19–21 Models have also been developed to predict GU outcomes following prostate cancer surgery including comorbidity as a contributing factor.22–26 However, these models have been developed and validated in populations with varying levels of underlying comorbidity and access to care. 22
Insurance status and access to care may influence the diagnosis and management of prostate cancer and comorbidities. In the U.S., patients with no or public health insurance are less likely to receive recommended cancer screening, such as PSA screening in men of eligible age, which may lead to later stage at diagnosis relative to those with continuous private coverage.27–32 Regarding treatment, patients with no or public health insurance are less likely to receive treatment or recommended treatment for prostate cancer.31–35 Patients without health insurance may also be less likely to receive encounters for primary or preventive care, thus leaving comorbidities undiagnosed until they are symptomatic or more severe.36,37 In the U.S., health insurance for persons may vary by age, employment status, income, pre-existing health conditions, or other factors. 38 Thus, studying comorbidity and its effects on cancer treatment and outcomes in populations with universal health insurance may help minimize effects of access to care on the results while controlling for other socioeconomic factors.
While there is literature to demonstrate the association between comorbidity and outcomes of prostate surgery, as outlined above, it has several limitations. First, the published studies predominantly include short-term outcomes, eg, 30 or 90-day. Second, some studies include a case-mix of patients undergoing surgery for different conditions (eg, benign prostate hyperplasia) and are not specific to prostate cancers. Third, not all studies include both general and GU complications. Lastly, the published studies may include men with different access to care, which may affect the results. To address the gaps in the literature, we aimed to study the association between comorbidity level and risk of 90-day general and 90-day and 18-month GU complications among men with prostate cancer undergoing surgery in the universal access Military Health System (MHS), which serves Department of War (DoW) beneficiaries, to better understand the impact of comorbidity on postoperative outcomes while minimizing the effects of access to care on results.39,40
Methods
Data Sources
This retrospective cohort study utilized the Military Cancer Epidemiology (MilCanEpi) database, which contains linked data between DoW cancer registry and medical encounter data. 41 The MilCanEpi database was approved for access for research and it was determined that the requirement for informed consent was waived by the Uniformed Services University of the Health Sciences Institutional Review Board, Bethesda, Maryland (FWA 00001628) on 19 July 2019 (Reference #914142) and renewed on 4 March 2025 (Reference #981045). The study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024. The Reporting of studies Conducted using Observational Routinely-collected health Data (RECORD) checklist, an extension of the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) statement, was used in the generation of this report. 42 The study investigators had access to the database population used to create the analytic cohort and perfomed data cleaning, including recoding invalid data entries as ‘missing’ or ‘unknown’.
Study Population
Eligible patients included men aged 18 or older who were diagnosed with a prostate adenocarcinoma [International Classification of Diseases-Oncology-Third edition (ICD-O-3) primary site code C619] between January 1, 2001, and December 31, 2014. Men were included if they were diagnosed with American Joint Committee on Cancer (AJCC) stages I-III tumors that had not metastasized to distant organ sites at time of diagnosis and if they received radical prostatectomy (RP) surgery in the primary management of their cancer (see Appendix Table A1). Patients with metastatic disease (stage IV) or surgery procedures other than RP were excluded. Patients with surgery information only in the cancer registry portion of the data and no medical encounter data in the immediate postoperative period were excluded due to inability to assess the study outcomes. We included patients with RP surgery within 1 year of the cancer diagnosis to limit possible effects of periods of active surveillance on the results (Figure 1). Selection of patients with prostate cancer receiving radical prostatectomy surgery as treatment in the MilCanEpi database, 2001-2014
Study Variables
The primary study variable was total comorbidity burden. The Elixhauser index was chosen to capture total comorbidity because of its comprehensive list of included conditions and performance in predicting adverse outcomes in the general population.43–47 For this study, we excluded solid tumors, hematologic cancers, and weight loss from the original list of conditions since our population included patients with prostate cancer and weight loss may be a symptom of cancer.43,48 We included comorbid conditions when the patient had at least 1 inpatient or 3 outpatient records with the relevant ICD-9-CM diagnosis codes occurring at least 30 days prior to the prostate cancer diagnosis (see Appendix Table A2). For statistical analysis, comorbidity level was classified as 0 conditions (no comorbidity), 1-2 conditions, or 3 or more conditions based on the frequency and distribution of comorbidity in the patient population.
Cancer attributes included date of prostate cancer diagnosis, AJCC tumor stage, AJCC tumor grade, Gleason grade (available for 2004 and later), prostate specific antigen (PSA) level at diagnosis (available for 2004 and later), and lymph node involvement. Cancer treatment information included the date of RP surgery, and the first date of chemotherapy, radiation, or hormone therapy. Additional surgery information included the care setting (military or civilian facility) and admission duration (≥24 hours, ie, inpatient, or <24 hours, ie, outpatient). Adjuvant treatment was considered any radiation, hormone therapy, or chemotherapy that was initiated within 18 months of the index surgery.
Other patient variables included race (Asian or Pacific Islander, American Indian or Alaska Native, Black, White, or multiple races), Hispanic ethnicity (yes, no, or unknown), marital status at cancer diagnosis (married, single, separated, widowed, divorced, or unknown), active duty military status, sponsor military service branch (Air Force, Army, Marine Corps, Navy, or other Department of War), and TRICARE contract region determined from state at diagnosis (U.S. Northeast, South, West or outside the continental United States, OCONUS).
Study Outcomes
The primary study outcomes included 90-day and 18-month postoperative complications and 90-day hospital readmissions. For 90-day outcomes, we queried the data for occurrence of general surgical and genitourinary (GU) complications using ICD-9-CM diagnosis codes for included conditions (See Appendix Table A3) within 90 days of the surgery (index) date. General surgical complications included myocardial infarction (MI), stroke, hemorrhage, blood transfusion, acute renal failure, deep vein thrombosis (DVT), pulmonary embolism (PE), other thrombosis, pneumonia, sepsis or septic shock, and death. Ninety-day GU complications included surgical site infection (SSI), pelvic abscess, hematoma, urinary tract infection (UTI), urinary incontinence (stress, urge, or mixed), bladder neck contracture (BNC), rectal injury, and rectal incontinence. For 18-month outcomes, we queried the data for GU complications occurring at any time between 12 and 18 months after the surgery date using ICD-9-CM diagnosis codes (see Appendix Table A3). Eighteen-month GU outcomes included UTI, urinary incontinence, blood in urine (hematuria), BNC, rectal incontinence or urgency, blood in stool, and erectile dysfunction.
Statistical Analysis
The distribution of patient and cancer variables were compared between men with 0, 1-2, or 3 or more comorbid conditions at time of prostate cancer diagnosis using frequency statistics and Chi-square tests. The frequency of measured outcomes was assessed and modified Poisson regression with log link and robust standard errors was used to estimate the rate ratios (RRs) and their 95% confidence intervals (CIs) in association with comorbidity level and risk of the study outcomes at 90-days and 18-months after surgery. Poisson models included an offset that represented days at risk to account for differential follow-up due to death, loss to follow-up, or end of the data on December 31, 2015. Patients with less than 12 months (365 days) of follow-up after surgery (n = 110) were excluded from the analysis of 18-month outcomes because they did not have data during the period of ascertainment (ie, 12 to 18 months after surgery). Cochran-Armitage trend test was used to examine whether a statistically significant effect (P < 0.05) was observed between comorbidity level and the primary outcomes of general or GU complications and hospital readmissions. Multivariable Poisson models for 90-day outcomes were adjusted for race-ethnicity, marital status, sponsor service branch, geographic region, comorbidities, year of cancer diagnosis, AJCC tumor stage, AJCC tumor grade, PSA at diagnosis, time between cancer diagnosis and surgery, surgery admission type, and surgery care setting. The models for 18-month outcomes were also adjusted for receipt of adjuvant treatment (radiation, chemotherapy, hormone). We then conducted analysis on 18-month outcomes stratified by adjuvant radiation status since radiation itself may be linked to GU complications and is part of standard treatment for prostate cancers.3,49,50 Analyses were conducted in SAS 9.4 (SAS, Inc, Cary, NC).
Results
Characteristics of Patients Undergoing Radical Prostatectomy for Non-metastatic Prostate Cancer in the Military Health System, 2001-2014, by Comorbidity Burden

Cancer Diagnosis and Treatment Characteristics for Patients Undergoing Radical Prostatectomy for Non-metastatic Prostate Cancer in the MHS, 2001-2014, by Comorbidity Burden

aGleason score and PSA was not available in the data prior to 2004. Data shown for patients diagnosed 2004-2014.
In total, 4.9% of study patients experienced a general complication, 27.9% had a GU complication, and 4.3% were readmitted within 90 days of their cancer surgery. The frequency of complications and readmissions differed across comorbidity level (all Chi-square P < 0.001; Figure 2), with increasing rates among each successive comorbidity level (P-trend<0.001). In multivariable regression analyses, there were significant trends for increased risk of general surgical complications (P-trend<0.001), GU complications (P-trend = 0.018), and readmissions (P-trend = 0.013) with increasing comorbidity level (Table 3). Patients with ≥3 comorbidities had statistically significant elevated risks of general surgical complication (ARR = 1.88, 95% CI = 1.34, 2.64), GU complication (ARR = 1.20, 95% CI = 1.06, 1.36), and hospital readmission (ARR = 1.59, 95% CI = 1.12, 2.26) after adjustment for potential confounders (Table 3). When further examining individual GU complications, there were statistically significant trends for increased risk of SSI (P-trend = 0.003) and UTI (P-trend = 0.002) with increasing comorbidity level (Table 3). Patients with ≥3 comorbidities had statistically significant 146% and 65% higher risks for SSI and UTI, respectively, relative to patients with no comorbidity in adjusted models (Table 3). Frequency of 90-day complications and readmissions by comorbidity level at prostate cancer diagnosis for men undergoing radical prostatectomy in the U.S. Military Health System, 2001-2014. Modified Poisson Regression Estimated Rate Ratio (RR) and 95% Confidence Interval (CI) for 90-Day Postoperative Complications Following Radical Prostatectomy Among Patients With Prostate Cancer by Age at Diagnosis Note. Not all genitourinary complications are listed individually due to low frequency (n < 11) overall or among several age groups. Bold text indicates results that are statistically significant at the alpha = 0.05 level. aModels adjusted for age at diagnosis (continuous), race-ethnicity, marital status, sponsor service branch, geographic region, year of cancer diagnosis, AJCC tumor stage, AJCC tumor grade, PSA at diagnosis, time between diagnosis and surgery (days; continuous), surgery admission type, and surgery care setting. bGeneral surgical complications include myocardial infarction (MI), stroke, hemorrhage, blood transfusion, acute renal failure, deep vein thrombosis (DVT), pulmonary embolism (PE), other thrombosis, pneumonia, sepsis or septic shock, and death. cGenitourinary complications include surgical site infection (SSI), pelvic abscess, hematoma, urinary tract infection, urinary incontinence (stress, urge, or mixed), bladder neck contracture (BNC), rectal injury, or rectal incontinence.

At 18 months post-op, overall, 20.2% of patients experienced an 18-month GU complication; 16.6% among men with no comorbidity, 21.3% among men with 1-2 comorbidities, and 25.4% for men with ≥3 or more comorbidities (P-trend<0.001; Figure 3). The most common complications were urinary incontinence and erectile dysfunction, both of which varied across patient groups by comorbidity (Chi-square P < 0.001; Figure 3) and showed significant trends for increased frequency with higher comorbidity count (P-trend<0.001). The frequencies of UTI, blood in urine, BNC, or rectal incontinence or bleeding were low (<2% each) and did not appear to differ across comorbidity levels (Figure 3). In multivariable regression analysis, there was a significant trend for increased risk of complications with comorbidity level (P-trend<0.001) and both men with 1-2 comorbidities (ARR = 1.19, 95% CI = 1.05, 1.35) and ≥3 comorbidities (ARR = 1.32, 95% CI = 1.13, 1.55) had significantly higher risk of any 18-month complication relative to men with no comorbidity (Table 4). In evaluation of individual complications, there were statistically significant trends for higher risk of urinary incontinence (P-trend = 0.008), including both stress incontinence (P-trend = 0.04) and mixed incontinence (P-trend = 0.03), GI complications of rectal incontinence or bleeding (P-trend = 0.045), and erectile dysfunction (P-trend = 0.018) for men with higher comorbidity level. Men with ≥3 or more comorbidities had a statistically significant 48% higher risk of urinary incontinence and 27% higher risk of erectile dysfunction relative to men with no comorbidity in the adjusted models (Table 4). Frequency of 18-month complications by comorbidity level at prostate cancer diagnosis for men undergoing radical prostatectomy in the U.S. Military Health System, 2001-2014. Modified Poisson Regression Estimated Rate Ratio (RR) and 95% Confidence Interval (CI) for 18-Month Complications Following Radical Prostatectomy Among Patients With Prostate Cancer by Age at Diagnosis
a
Note. Not all genitourinary complications are listed individually due to low frequency (n < 11) overall or among several age groups. Bold text indicates results that are statistically significant at the alpha = 0.05 level. aPatients with at least 12 months follow-up after surgery date included for this analysis. bModels adjusted for race-ethnicity, marital status, sponsor service branch, geographic region, comorbidities, year of cancer diagnosis, AJCC tumor stage, AJCC tumor grade, PSA at diagnosis, time between diagnosis and surgery (days; continuous), surgery admission type, surgery care setting, and adjuvant treatment (radiation, chemotherapy, hormone). cComplications include urinary tract infection (UTI), blood in urine, urinary incontinence (stress, urge, or mixed), bladder neck contracture (BNC), rectal incontinence, blood in stool, and erectile dysfunction. dUrinary incontinence includes stress, urge, and mixed incontinence. eGastrointestinal complications include rectal incontinence or blood in stool.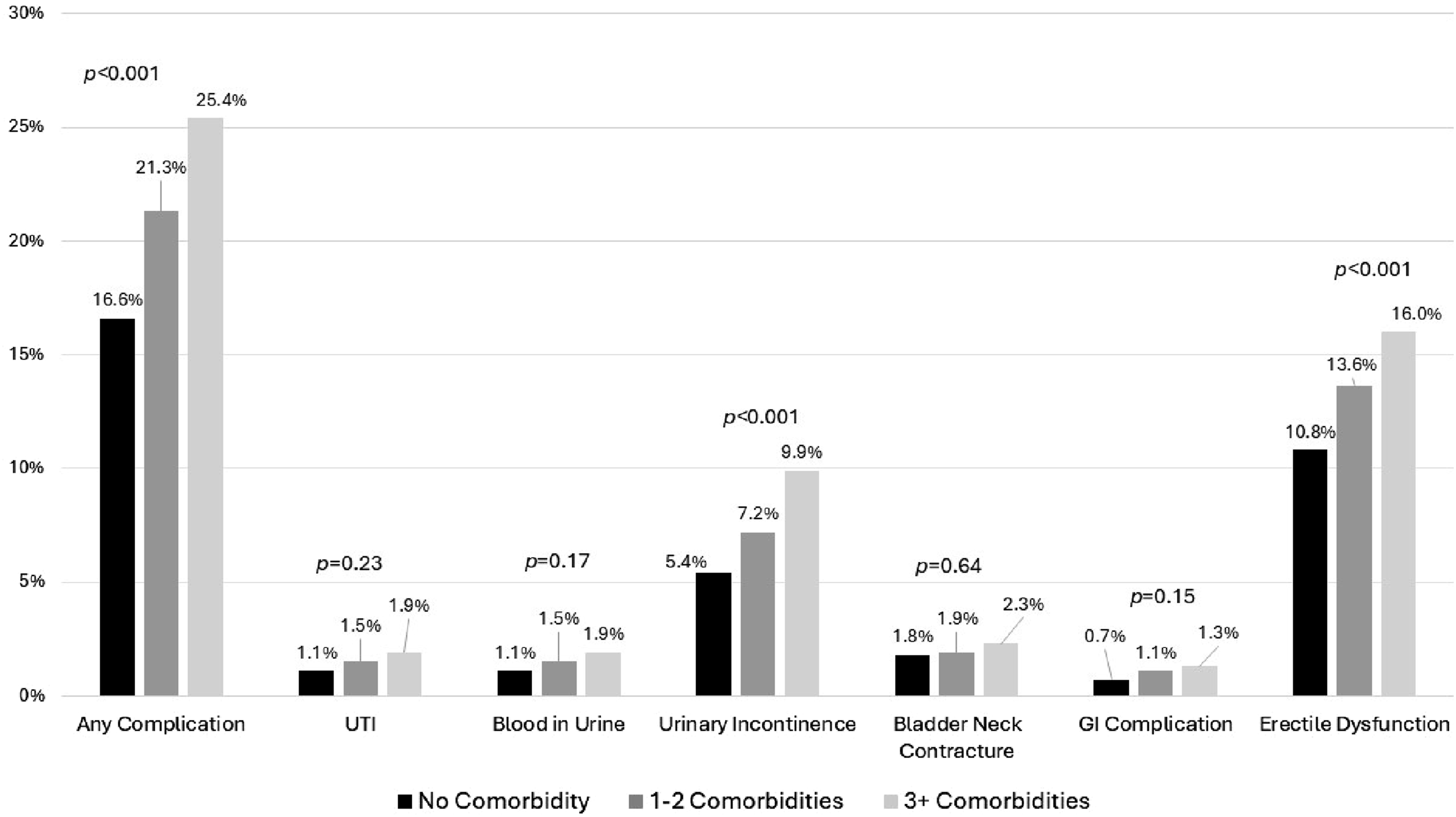

Modified Poisson Regression Estimated Rate Ratio (RR) and 95% Confidence Interval (CI) for 18-Month Complications Following Radical Prostatectomy Among Patients With Prostate Cancer by Comorbidity Level Stratified by Receipt of Adjuvant Radiation
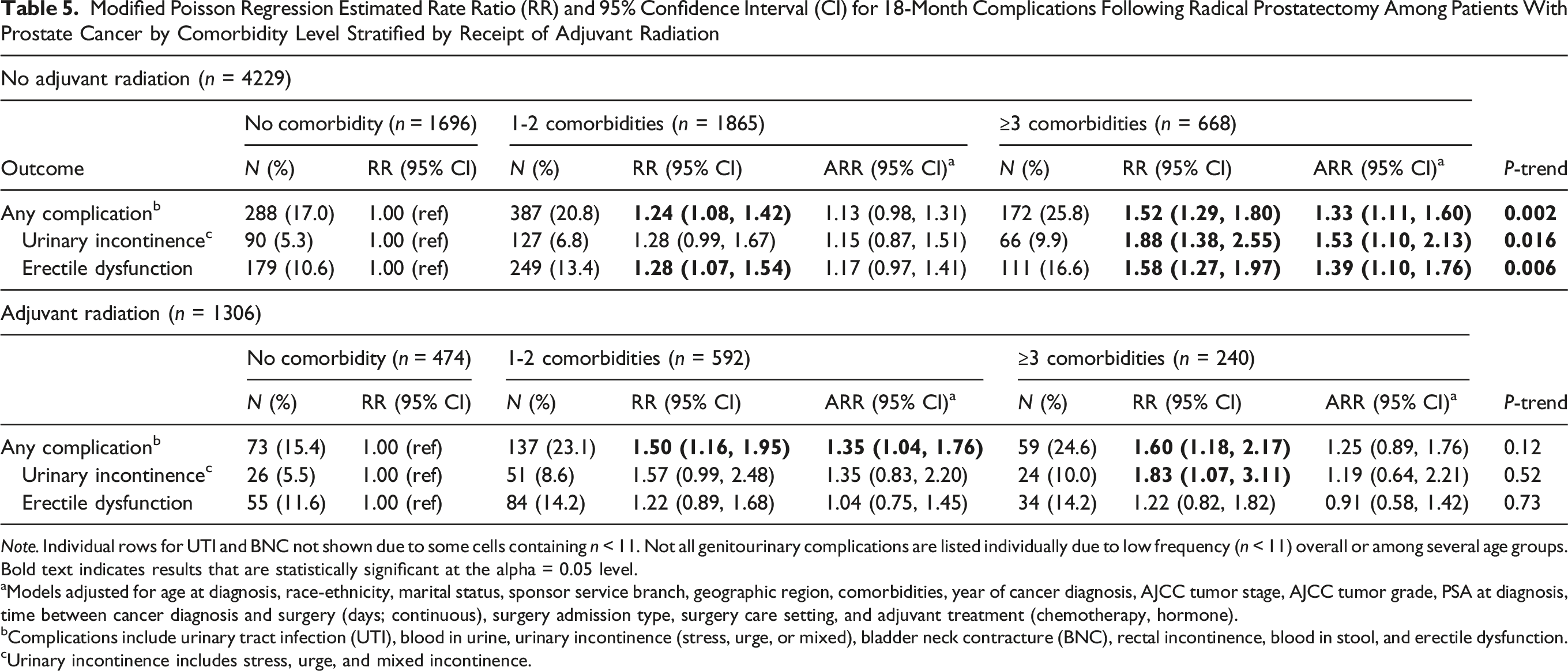
Note. Individual rows for UTI and BNC not shown due to some cells containing n < 11. Not all genitourinary complications are listed individually due to low frequency (n < 11) overall or among several age groups. Bold text indicates results that are statistically significant at the alpha = 0.05 level.
aModels adjusted for age at diagnosis, race-ethnicity, marital status, sponsor service branch, geographic region, comorbidities, year of cancer diagnosis, AJCC tumor stage, AJCC tumor grade, PSA at diagnosis, time between cancer diagnosis and surgery (days; continuous), surgery admission type, surgery care setting, and adjuvant treatment (chemotherapy, hormone).
bComplications include urinary tract infection (UTI), blood in urine, urinary incontinence (stress, urge, or mixed), bladder neck contracture (BNC), rectal incontinence, blood in stool, and erectile dysfunction.
cUrinary incontinence includes stress, urge, and mixed incontinence.
Discussion
In the U.S. Military Health System, we noted differences across comorbidity level in 90-day and 18-month complication rates following radical prostatectomy for men with prostate cancer. Men with ≥3 comorbid conditions at time of prostate cancer diagnosis had higher risks of 90-day general and GU complications and hospital readmissions relative to men with no comorbidity, which remained after adjustment for age and other potential confounders. At 18 months after surgery, men with 1-2 or ≥3 comorbidities had higher risks of GU complications inclusive of UTI, incontinence, BNC, and erectile dysfunction. Findings from this study may inform routine pre- and postoperative management of patients with prostate cancer and identify surgical outcomes most associated with comorbidity level for targeted prevention. 51
Among men receiving RP for prostate cancer in the MHS, patients with ≥3 comorbidities had a statistically significant 88% increased risk of general complications relative to patients with no comorbidity at time of prostate cancer diagnosis. Consistent with our results, a study among men with prostate cancer treated with RP in Ontario, Canada, which also has a universal healthcare system, reported higher risk of 30-day cardiac, respiratory, and medical complications associated with higher comorbidity level. 16 Higher risk of general complications such as myocardial infarction, stroke, thrombosis, or pneumonia among patients with higher comorbidity may be associated with the underlying comorbidity itself. Other studies have demonstrated increased risk for intraoperative and postoperative complications after RP for patients with obesity, diabetes mellitus, or prior heart disease.19–21,26,52,53 A study of the National Inpatient Sample reported higher rates of in-hospital cardiac complications, infections, and postoperative bleeding and longer length of stay (LOS) for patients with prior heart valve replacement receiving RP for prostate cancer. 19 Similarly, a study of the National Surgical Quality Improvement Program reported higher rates of complications, longer LOS, and higher rates of readmissions for patients with congestive heart failure receiving minimally invasive RP. 52 Obesity has also been reported to increase the risk of intra- and immediate postoperative outcomes after RP for men with prostate cancer.20,52 While our study was not designed to test the effects of individual comorbid conditions on the outcomes, the conditions of hypertension, diabetes mellitus, and obesity were among the most prevalent in our study population and may therefore greatly contribute to the observed increased risks of complications in association with comorbidity as previously demonstrated in the literature. Collectively, the current study and published literature support the need for coordinated care and monitoring of patients with higher levels of comorbidity for adverse postoperative events after RP for prostate cancer.
Patients receiving RP for prostate cancer in the MHS with ≥3 comorbidities also had a 59% higher risk of 90-day GU complications relative to men with no comorbidity. The elevated risk was most apparent for surgical site and urinary tract infections. Meanwhile, we did not observe any significant trends or associations between increased comorbidity and occurrence of hematoma, BNC, or urinary incontinence. The presence of comorbidity can impair innate and adaptive immune responses, thus weakening the body’s natural defenses and increasing susceptibility to infection, thus providing biological plausibility to our findings.54,55 Consistent with our results, 1 study found that higher comorbidity was associated with higher risk of complicated UTI in the postoperative period for men receiving transurethral resection of the prostate (TURP). 18 Another study reported higher rates of infection for men undergoing RP with a history of heart disease. 19 Meanwhile, obesity has been shown to increase risk for urinary and surgical site infections in general populations of surgical patients.54,56 If confirmed in other population-based studies, these findings indicate that testing of infection control interventions may be needed to identify effective strategies for reducing complications in the immediate postoperative period for men undergoing RP for prostate cancer with high comorbidity level.
The elevated risks of general and GU complications among men with higher comorbidity may partly contribute to the 59% increased risk of 90-day hospital readmissions among men with ≥3 comorbidities in our study. Although we were limited by a relatively small of readmission events to conduct more thorough analyses on the reasons for admission, other studies have reported higher readmissions related to cardiac and circulatory events which were included among general complications in our data.16,51,52 Also, substantial evidence exists to link pre-existing comorbidities with risk of adverse events following general and oncologic surgery.12–14 While the overall 90-day readmission rate was low (<5%) in our data, it is consistent with other literature on readmission after prostate cancer surgery. 53 Proactive comorbidity management and early identification of complications should continue to be implemented to minimize hospital readmissions following RP.52,53
At up to 18-month post-RP, we observed a higher risk of GU complications, especially urinary incontinence and ED, among men with higher pre-diagnostic comorbidity. Both hypertension and diabetes, which were common in our study population, may negatively affect the vascular and nervous systems and contribute to urologic complications including incontinence and ED.57,58 The results of our study are also consistent with Flores et al who reported lower sexual function and higher rates of ED up to 24-month post RP for men with diabetes mellitus relative to those without. 21 However, in 1 study, baseline erectile function and incontinence were more predictive of outcomes up to 3 years after robotic-assisted RP than comorbidity. 59 Nevertheless, baseline continence and erectile function may be linked to comorbidity such as obesity or diabetes.21,58-61 Yet, Jaber et al reported no association between obesity and potency or urinary incontinence at 12 and 24 months after robotic-assisted RP in a cohort of over 14 000 men. 20 Differences in the association between comorbidity and GU outcomes between studies may be related to inclusion criteria, definitions of comorbidity and conditions included, method of outcomes ascertainment, and length of follow-up. Even so, the evidence suggests that comorbidity at time of prostate cancer diagnosis may impact longer-term continence and potency after prostate surgery. Future research that evaluates the impact of strategies such as pelvic floor therapy, lifestyle changes in diet and physical activity, or pharmacologic interventions on postoperative GU outcomes among men with prostate cancer and higher comorbidity level may be needed.61,62
This study used a comprehensive database to assess the impact of comorbidity at time of prostate cancer diagnosis on 90-day and 18-month postoperative outcomes among men with universal access to care. The extended follow-up beyond 90-days and inclusion of both general and GU complications in a population where the effects of access to care are minimized are strengths of this study and fill gaps in the existing literature. Nevertheless, there are limitations. First, the retrospective database design may introduce selection bias by including only patients treated within the system and who opt for surgical treatment, as opposed to radiation therapy, in the primary management of their prostate cancer. Men who receive surgery vs radiation therapy may differ in overall health status and comorbidity burden as well as other unmeasured confounders which may affect the interpretation of the results. Second, we used ICD-9 diagnosis codes to capture postoperative complications and did not have clinical data to confirm their occurrence. This may underestimate the true rate of complications. However, underreporting of outcomes may not differ between patients by comorbidity level in a universal health system and may not greatly affect the results. Third, we did not have data on patient-reported outcomes such as baseline erectile function or health-related quality of life (HRQoL) and could not assess how comorbidity and experience of complications may affect HRQoL.63–65 This will be an important area for future research in prostate cancer treatment and survivorship. Next, we did not have data to determine physical activity, smoking, or other health characteristics which may be associated with comorbidity, complications, and outcomes in patients with prostate cancer. 59 Thus, we cannot exclude any effects of these unmeasured health aspects on the results. Also, we used the latest data available in the MilCanEpi database (ie, 2001-2014) at the time of analysis. However, this data may not reflect current trends in RP and surgical approaches such as robotic-assisted procedures. Since surgical approach may be influenced by patient comorbidity, we cannot exclude any possible effects on the results. Research in more contemporary datasets is needed to explore the association between comorbidity and postoperative outcomes when considering more modern surgical approaches. Lastly, the DoW beneficiary population may differ in underlying health status and health behaviors than the U.S. general population and thus the results may not be generalizable. Nevertheless, the study provides important details on comorbidity level and risk of postoperative outcomes for men with prostate cancer from a universally insured population.
Conclusions
In the U.S. Military Health System, we noted differences across comorbidity level in complication rates following radical prostatectomy for men with prostate cancer. Men with higher comorbidity burden had higher risks of 90-day surgical complication and readmissions and higher risk of 18-month GU complications. This study identifies a need for well-designed clinical studies that evaluate strategies, such as infection control, pelvic floor therapy, or lifestyle or pharmacologic interventions, for their potential to reduce complication rates among men with higher comorbidity levels diagnosed with prostate cancer and treated by radical prostatectomy.
Supplemental Material
Supplemental Material - Comorbidity Level and Risk of 90-day and 18-month Complications Among Patients Undergoing Radical Prostatectomy for Prostate Cancer in the Military Health System
Supplemental Material for Comorbidity Level and Risk of 90-day and 18-month Complications Among Patients Undergoing Radical Prostatectomy for Prostate Cancer in the Military Health System by Yvonne L. Eaglehouse, PhD, MPH, Christian Dide-Agossou, PhD, Sarah Darmon, PhD, Sean Q. Kern, MD, Molly R. Oroho, BS, Andrea A. Almeida, MA, MPH, Craig D. Shriver, MD, Kangmin Zhu, MD, PhD in Cancer Control.
Footnotes
Acknowledgement
The authors thank the Joint Pathology Center (JPC) for providing the Department of War (DoW) cancer registry data and the Defense Health Agency (DHA) for providing the Military Health System (MHS) data repository (MDR) data. The authors thank ICF International, the Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc (HJF, Inc), and the Uniformed Services University of the Health Sciences (USUHS) for data linkage and hosting.
Ethical Considerations
This retrospective cohort study used data from the Military Cancer Epidemiology (MilCanEpi) database. The MilCanEpi database was approved for access for research by the Uniformed Services University of the Health Sciences Institutional Review Board, Bethesda, Maryland (FWA 00001628) on 19 July 2019 (Reference #914142) and renewed on 4 March 2025 (Reference #981045).
Consent to Participate
The database study was reviewed by the Uniformed Services University of the Health Sciences Institutional Review Board (FWA 00001628; Reference #914142 and #981045) and it was determined that the requirement for informed consent was waived.
Author Contributions
YE, SD, and KZ were employees of the Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc, a 501(c)(3) non-profit organization at the time the work was performed.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Murtha Cancer Center Research Program (MCCRP) of the Department of Surgery, Uniformed Services University of the Health Sciences (USUHS) under the auspices of the Henry M. Jackson Foundation for the Advancement of Military Medicine (HJF, Inc), grant numbers HHU0001-16-2-0014 and HU0001-18-2-0032. The funding agency had no role in the study design; in the collection, analysis, and interpretation of data; or in the writing of the report.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are not publicly available due to the restrictions in the access and use of the MilCanEpi data specified in the data sharing agreements and regulatory approvals. The Department of War Cancer Registry (DoWCR) data and data dictionary may be requested from the Joint Pathology Center and online at https://jpc.capmed.mil/. The Military Health System Data Repository (MDR) data and data dictionary may be requested from the Defense Health Agency and online at ![]() .
.
Disclaimer
The contents of this manuscript are the sole responsibility of the authors and do not necessarily reflect the views, assertions, opinions, or policies of the Uniformed Services University of the Health Sciences, the Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc, the Department of War, or the Departments of the Army, Navy, or Air Force. Mention of trade names, commercial products, or organizations does not imply endorsement by the U.S. government.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
