Abstract
Background
Patients with liver cirrhosis are at a higher risk of developing non-hepatic cancers (NHC) than general population. Considering that NHC causes poor outcomes, it is necessary to explore the prevalence of NHC in such patients and identify its associated risk factors.
Methods
Patients with a diagnosis of liver cirrhosis who were consecutively admitted to our hospital between January 1, 2010 and June 30, 2014 were retrospectively screened. They were divided into NHC, primary liver cancer (PLC), and non-cancer groups. After adjusting for age and sex, logistic regression analyses were performed to explore the risk factors of NHC in patients with cirrhosis. Adjusted odds ratios (aORs) with their 95% confidence intervals (CIs) were calculated.
Results
Overall, 2509 patients with cirrhosis were included. Prevalence of NHC and PLC was 6.22% (156/2509) and 22.20% (557/2509), respectively. After adjusting for age and sex, the NHC group had a significantly higher proportion of history of smoking (aOR = 1.994; 95% CI = 1.231-3.231, P = 0.005), but lower proportions of history of encephalopathy (aOR = 0.344; 95% CI = 0.147- 0.804, P = 0.014) and history of heart disease (aOR = 0.300; 95% CI = 0.173-0.519, P < 0.001) as compared to the non-cancer group. Hepatitis B virus infection (aOR = 0.863; 95% CI = 0.581-1.281, P = 0.465), hepatitis C virus infection (aOR = 0.908; 95% CI = 0.503-1.642, P = 0.750), and alcohol abuse alone (aOR = 1.254; 95% CI = 0.779-2.017, P = 0.351) as the etiology of liver cirrhosis were not significantly associated with the risk of NHC.
Conclusions
Smoking may be associated with a higher probability of NHC in patients with cirrhosis, but the probability of NHC may be compromised by encephalopathy or heart disease.
Introduction
Liver cirrhosis can be caused by alcoholic liver disease and chronic hepatitis B virus (HBV) and/or hepatitis C virus (HCV) infections. 1 It can lead to various decompensation events, 2 including esophagogastric variceal bleeding (EGVB), ascites, hepatic encephalopathy, and hepatorenal syndrome, 3 and progress to primary liver cancer (PLC) with an annual incidence of 1.82%. 4 Except for a major risk factor for the development of PLC, 5 liver cirrhosis is also closely associated with the development of NHC. 6 It has been shown that the overall risk for NHC was 2-3 times more common in patients with cirrhosis compared with the general population. 7 HBV infection is associated with a 2.24-fold increased risk of non-Hodgkin’s lymphoma and a 1.49-fold elevated risk of gastric cancer.8,9 Cirrhotic patients with HBV infection have an increased risk of lung, kidney, skin (squamous cell carcinoma), and thyroid cancers, as well as lymphoma and leukaemia10,11; those with HCV infection have an increased risk of gallbladder, stomach, pancreatic, and lung cancers11,12; and those with alcoholic cirrhosis have an increased risk of gastric, colorectal, pancreatic, and esophageal cancers.13,14 Despite a relatively low prevalence of NHC in liver cirrhosis, it deteriorates the outcomes with a high mortality. Additionally, the presence of liver cirrhosis, especially worse liver function reserve, potentially limits the use of anticancer therapy in NHC patients. Notably, it is of importance for clinicians and patients to recognize the risk factors associated with NHC in cirrhosis, because the incidence and mortality of cancer can be effectively reduced by eliminating carcinogenic risk factors.15,16 Age, sex, smoking, and alcohol consumption are well-known risk factors for cancer,17,18 but few studies have explored the risk factors for NHC in patients with liver cirrhosis. Herein, we have conducted a cross-sectional study to estimate the prevalence of NHC and explore its related risk factors in patients with liver cirrhosis.
Methods
Study Design
We retrospectively collected a total of 4221 patients with liver cirrhosis who were consecutively admitted to the Department of Gastroenterology of the General Hospital of Northern Theater Command from January 1, 2010 to June 30, 2014, as previously mentioned. 19 Exclusion criteria were as follows: (1) repeated admissions; (2) unclear origin of cancer; (3) a combination of hepatic and non-hepatic cancers; (4) a suspected diagnosis of cancer; and (5) the age < 18 years old at the first diagnosis of liver cirrhosis. Among the patients with repeated admissions during the study period, only their last admissions were included.
The study protocol was approved by the Medical Ethical Committee of the General Hospital of Northern Theater Command with an approval number [Y (2024) 009] and was carried out following the guidelines of the Declaration of Helsinki. The requirement for informed consent was waived due to the retrospective nature of the study. This study conformed to the STROBE guidelines. 20 All patients’ details were de-identified.
Groups and Definitions
Cirrhotic patients were divided into NHC group, PLC group, and non-cancer group. Liver cirrhosis is diagnosed based on disease history, clinical symptoms and signs, laboratory tests, imaging, liver stiffness measurement, and histopathological examinations, if possible. 21 NHC refers to primary malignancy diagnosed by definitive pathology or radiographic evidence, mainly including cancer of the lung, colon/rectum, non-Hodgkin’s lymphoma, esophagus, pancreas, prostate, and/or breast, etc. 22 PLC is diagnosed based on dynamic CT or MRI scans. If necessary, liver histology may be obtained for further confirmation.23,24
Data Collection
Demographic, clinical, and laboratory data at admissions were collected, including age, sex, smoking, diabetes mellitus (DM), hypertension, history of encephalopathy, history of respiratory, heart, and kidney diseases, etiology of liver cirrhosis (ie, HBV infection alone, HCV infection alone, and alcohol abuse alone), treatment of liver cirrhosis (ie, drug and surgery), red blood cell (RBC), hemoglobin (Hb), white blood cell (WBC), platelet count (PLT), total bilirubin (TBIL), albumin (ALB), serum creatinine (Scr), serum sodium (Na), and international normalized ratio (INR). Child-Pugh and model of end-stage liver disease (MELD) scores were calculated. Viral hepatitis is diagnosed by serological tests for specific viral antigens and antibodies. 25 Smoking focused on burning tobacco, including cigarettes, cigars, and hookah, but not e-cigarettes and smokeless tobacco products.
Statistical Analyses
All statistical analyses were performed by using SPSS version 26.0 statistical software (IBM Corp, Armonk, New York, USA). Continuous variables were expressed as mean ± standard deviation and median (range), and compared by the independent sample t-tests for normal distribution, and the Mann-Whitney U tests for non-normal distribution. The Chi-square test was used for categorical variables to analyze the difference between groups. Univariate logistic regression analysis was used to evaluate the factors associated with NHC and PLC. Odds ratios with their 95% confidence intervals (CIs) were calculated. Multivariate logistic regression analysis was further performed by adjusting for age and sex.26,27 Given that sex is a recognized risk factor for cancer, subgroup analyses were conducted in male and female patients. A two-tailed P value of <0.05 was considered statistically significant. Participants with missing data were removed from the corresponding analyses to ensure the data completeness.
Results
Patients’ Selection
Overall, 1712 patients were excluded due to repeated admissions (n = 1686), unclear origin of cancer (n = 8), NHC complicated with PLC (n = 15), unclear diagnosis of cancer (n = 1), and age <18 years old (n = 2). Finally, 2509 patients were included (Figure 1). The prevalence of NHC and PLC was 6.22% and 22.2%, respectively. There is a consistent upward trend in the prevalence of NHC from 6.95% to 7.65%, while a gradual downward trajectory in the prevalence of PLC from 25.56% to 22.94% during the study period. The median age was 56.60 (18.57-95.13) years old. Flowchart of Patients’ Screening and Grouping. Abbreviations: NHC, Non-hepatic Cancers; PLC, Primary Liver Cancer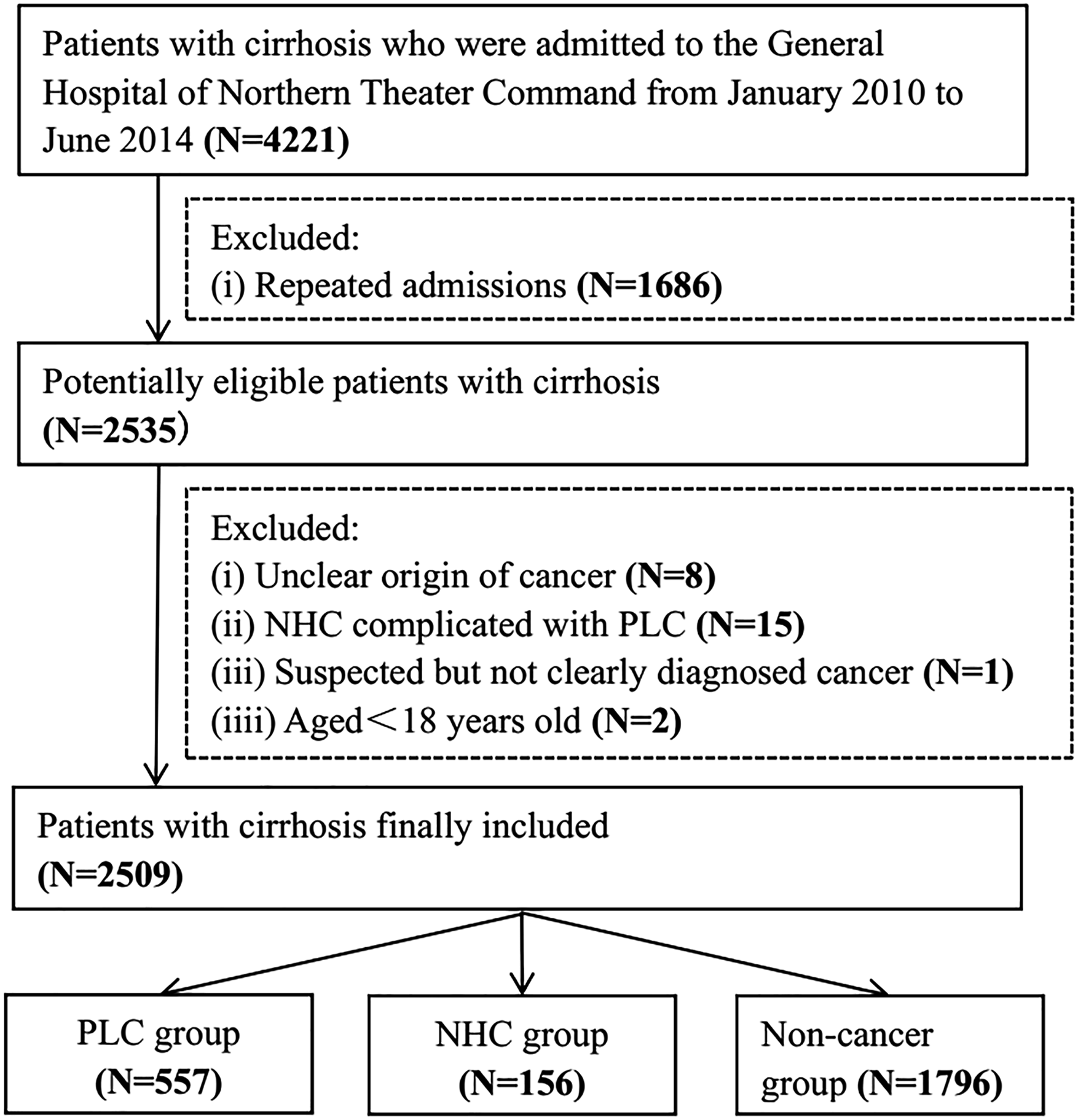
Characteristics of Patients
Characteristics of Patients Among PLC, NHC, and Non-Cancer Groups

Abbreviations: No. pts, numbers of patients; SD, standard deviation; PLC, primary liver cancer; NHC, non-hepatic cancers; MELD, model of end-stage liver disease; HBV, hepatitis B virus; HCV, hepatitis C virus; RBC, red blood cell; Hb, hemoglobin; WBC, white blood cell; PLT, platelet count; TBIL, total bilirubin; ALB, albumin; Scr, serum creatinine; Na, natrium; INR, international normalized ratio. Bold indicates statistically significant of P-value.
aPLC group vs non-cancer group.
bNHC group vs non-cancer group.
In the NHC group, the average age was 63.34 ± 11.38 years, and 62 (39.70%) patients were male. 37 (33.60%) patients had a history of smoking. 28 (18.10%) patients had DM, and 40 (25.80%) had hypertension. HBV infection alone was the most common etiology of liver cirrhosis (n = 37, 23.70%), followed by alcohol abuse alone (n = 28, 17.90%).
In the PLC group, the average age was 57.99 ± 10.93 years, and 445 (79.90%) patients were male. 123 (35.70%) patients had a history of smoking. 93 (16.80%) patients had DM, and 76 (13.70%) had hypertension. HBV infection alone was the most common etiology of liver cirrhosis (n = 272, 48.80%), followed by alcohol abuse alone (n = 59, 10.60%).
In the non-cancer group, the average age was 56.54 ± 12.11 years, and 1194 (66.50%) patients were male. 364 (32.20%) patients had a history of smoking. 140 (20.40%) patients had DM, and 116 (16.90%) had hypertension. HBV infection alone (n = 510, 28.40%) was the most common etiology of liver cirrhosis, followed by alcohol abuse alone (n = 431, 24.00%).
Factors Associated With NHC
Logistic Regression Analyses of Risk Factors of NHCb

Abbreviations: NHC, non-hepatic cancers; OR, odds ratio; CI, confidence interval; HBV, hepatitis B virus; HCV, hepatitis C virus. Bold indicates statistically significant of P-value.
aAdjusted for sex, age.
bNon-cancer group as a reference group.
To further analyze the impact of smoking status on NHC, we further divided patients with a history of smoking into the current and previous smokers. After adjusting for sex and age, multivariate logistic regression analysis showed that smoking status (aOR = 0.466; 95% CI = 0.197-1.101, P = 0.082) was not independently associated with a higher risk of NHC.
Factors Associated With PLC
Logistic Regression Analyses of Risk Factors of PLCb

Abbreviations: PLC, primary liver cancer; OR, odds ratio; CI, confidence interval; HBV, hepatitis B virus; HCV, hepatitis C virus. Bold indicates statistically significant of P-value.
aAdjusted for sex, age.
bNon-cancer group as a reference group.
Subgroup Analyses
Logistic Regression Analyses of Risk Factors of NHC in Male Patientsb
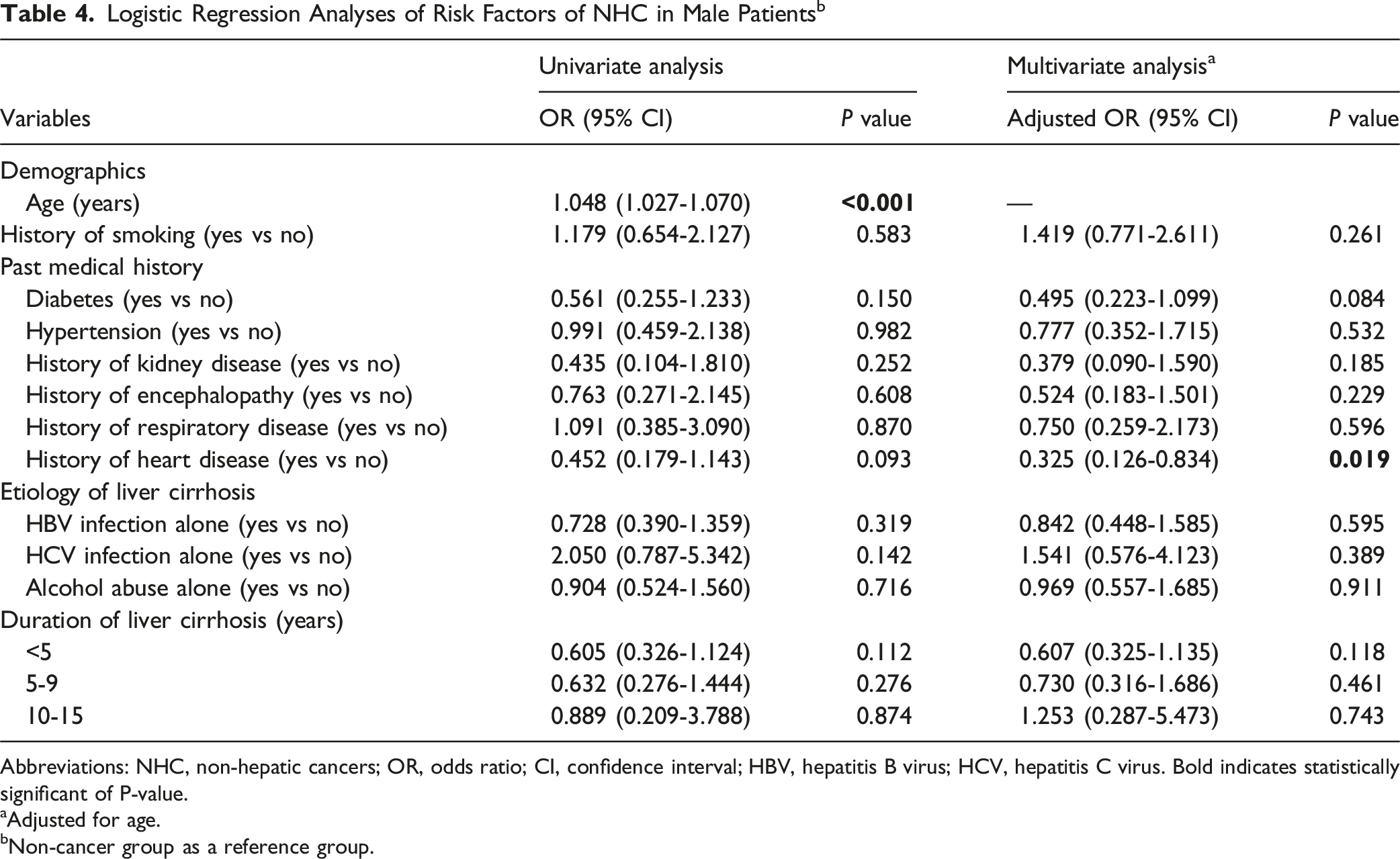
Abbreviations: NHC, non-hepatic cancers; OR, odds ratio; CI, confidence interval; HBV, hepatitis B virus; HCV, hepatitis C virus. Bold indicates statistically significant of P-value.
aAdjusted for age.
bNon-cancer group as a reference group.
Logistic Regression Analyses of Risk Factors of NHC in Female Patientsb

Abbreviations: NHC, non-hepatic cancers; OR, odds ratio; CI, confidence interval; HBV, hepatitis B virus; HCV, hepatitis C virus. Bold indicates statistically significant of P-value.
aAdjusted for age.
bNon-cancer group as a reference group.
Discussion
Our study had several important findings. First, the prevalence of NHC and PLC was 6.22% and 22.20% in cirrhosis, respectively, which should be higher than that of cancer in the general population. 28 Moreover, the overall prevalence of NHC has been steadily increasing from 2011 to 2014, which highlights the necessity of cancer screening as well as surveillance in cirrhotic patients. This was likely driven by gradually increased recognition of various cancers and widespread use of advanced imaging techniques with greater diagnostic performance, and fueled by a higher probability of carrying more risk factors of cancers. Second, cirrhotic patients with a history of smoking had an increased risk of NHC nearly two-fold as compared to those without, and it remains a significant carcinogenic factor regardless of smoking status. Third, cirrhotic patients with encephalopathy or heart disease were less likely to develop NHC.
Previous studies showed that sex and age could influence the occurrence and development of cancer,26,27 which was also confirmed in our univariate logistic regression analysis. Accordingly, we adjusted for sex and age in multivariate logistic regression analysis, showing that the NHC group had a significantly higher proportion of history of smoking than the non-cancer group. There are some possible explanations for this finding, as follows. First, there are many harmful chemicals in cigarettes, including PAH, N-nitrosamines, aromatic amines, 1,3-butadiene, benzene, aldehydes, and ethylene oxide, which are the major causes for various types of cancer. 29 These substances can directly or indirectly damage DNA, leading to genetic mutations in cells and ultimately developing cancer. 30 Second, smoking reduces the formation of cancer-suppressor-related proteins, thereby promoting cell proliferation. 31 Furthermore, chemicals in tobacco can induce inflammatory response, which is closely related to the development of cancers. 32
Interestingly, our study found a reduction in the prevalence of NHC and PLC in cirrhotic patients with heart disease or encephalopathy. This might be because such patients need to take specific medications more frequently, such as aspirin, which can reduce the incidence and mortality of cancer. 33 In addition, cirrhotic patients with heart disease or encephalopathy might undergo screening more comprehensively, thereby initiating the intervention earlier and reducing the incidence of cancer. Another possible explanation is that patients with cardio-cerebral diseases often have lipid disorders, 34 thereby necessitating the use of lipid-regulating drugs, which are also potentially beneficial for anticancer effect by regulating lipids.35-39 Our study also observed a diminished risk of PLC in cirrhotic patients with kidney and/or respiratory disease. This can be explained by the fact that the survival time is substantially shortened in such patients, compromising the development of cancer.
It has been shown that patients with HBV infection, HCV infection, or alcohol related liver cirrhosis have a higher risk of NHC.10,40,41 By contrast, another study by Berman et al 42 demonstrated that patients with cirrhosis secondary to viral hepatitis had a lower risk of NHC, whereas those with alcoholic cirrhosis had a higher risk of NHC. Our study showed no increased risk of NHC in patients with liver cirrhosis secondary to viral hepatitis or alcohol abuse. This discrepancy among studies might be attributed to the heterogeneity in the type of extrahepatic cancer observed and proportion of different cancers. Indeed, there are still some controversies about whether the incidence of certain cancers was increased in cirrhosis. Specifically, Su et al did not show that HBV infection related cirrhosis increased the risk of breast cancer, 43 Duberg et al did not demonstrate that HCV infection related cirrhosis increased the incidence of thyroid cancer, 44 and Sahlman et al 41 did not find that alcohol abuse related cirrhosis increased the risk of lymphomas.
Previous studies suggested that patients with non-alcoholic cirrhosis were more likely to develop PLC.45,46 Similarly, our study found that HBV infection alone was a risk factor for PLC in cirrhotic patients, whereas alcohol abuse alone might be a protective factor. However, in contrast to previous studies, 47 ours did not find that HCV infection alone was a risk factor for PLC in cirrhosis, probably due to a relatively small number of cirrhotic patients with HCV infection alone in our cohort.
We separately conducted an investigation into the risk factors associated with NHC in male and female patients. Female patients who smoked and consumed alcohol had an increased risk of NHC, which is consistent with our current knowledge that alcohol abuse in females can cause more medical problems. 48 Although its specific mechanism is unclear, there is a possible hypothesis that females have a lower proportion of body water than males of similar body weight, thereby causing a higher blood alcohol concentration after consuming an equivalent amount of alcohol. 49 Additionally, females have slower tobacco excretion due to their anatomy and higher level of dependence, thereby contributing to an increased risk of cancer. 50
Our study has comprehensively assessed the risk of NHC in liver cirrhosis, and has for the first time proposed that history of encephalopathy and/or heart disease may reduce the risk of NHC in patients with liver cirrhosis. This conclusion challenges the traditional knowledge that “comorbidities worsen prognosis” and provide new perspectives for subsequent research on the interactions among liver diseases, cancers, and chronic diseases. However, our study had several limitations as follows. First, this was a single-center retrospective study without external validity of its findings, which had selection bias and influenced the reliability of our findings. Second, the risk factors for cancers, such as body mass index, lifestyle habits, and other causes of cirrhosis, were not available and had not been analyzed yet. Third, we could not collect the frequency or amount of smoking and alcohol abuse in cirrhotic patients with a history of smoking and drinking, compromising further quantitative analysis.
Conclusion
The probability of NHC may be increased by smoking in cirrhosis, but compromised by the presence of encephalopathy or heart disease. These findings will be helpful for developing optimal strategies for screening and monitoring of cancer in cirrhosis. Certainly, more multicenter, prospective studies are warranted to further validate these conclusions.
Supplemental Material
Supplemental Material - Prevalence and Risk Factors for Non-Hepatic Cancers in Cirrhosis: A Cross-Sectional Study
Supplemental Material for Prevalence and Risk Factors for Non-Hepatic Cancers in Cirrhosis: A Cross-Sectional Study by Di Sun, Liyan Dong, Yue Sun, Yingchao Li, Junyuan Yin, Ran Wang, Deli Zou and Xingshun Qi in Cancer Control
Footnotes
Acknowledgments
This work has been partially presented as an oral abstract in the section “Public Health” of the Asian Pacific Association for the Study of the Liver (APASL) 2024.
Ethical Considerations
The study protocol was reviewed and approved by the Medical Ethical Committee of the General Hospital of Northern Theater Command [Y (2024) 009] on January 22, 2024. The requirement for informed consent was waived due to the retrospective nature of this study.
Consent to Participate
Because of the retrospective nature of the study, the requirement for written informed consent was waived.
All authors have made an intellectual contribution to the manuscript and approved the submission.
Author Contributions
Conceptualization: Xingshun Qi
Formal analysis: Di Sun, Liyan Dong, Yingchao Li, and Xingshun Qi
Data curation: Di Sun, Liyan Dong, Yue Sun, Yingchao Li, Junyuan Yin, Deli Zou, and Xingshun Qi
Writing–original draft: Di Sun, Liyan Dong, Deli Zou, and Xingshun Qi
Writing–review and editing: Di Sun, Liyan Dong, Yue Sun, Yingchao Li, Junyuan Yin, Deli Zou, and Xingshun Qi
Supervision: Deli Zou, and Xingshun Qi.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by the Independent Research Funding of General Hospital of Northern Theater Command (ZZKY2024018), and Liaoning Provincial Science and Technology Plan Joint Plan (Technology Research and Attack Plan Project) (2024JH2/102600285).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
