Abstract
Introduction
Gestational trophoblastic neoplasia (GTN) is the most curable gynecologic malignancy. However, resistance to first-line chemotherapy remains common, and mortality is high in developing countries.
Objective
To evaluate remission, relapse, survival, and associated factors among GTN patients in northwest Ethiopia.
Methods
A hospital-based, multi-center retrospective cohort study was conducted on 205 patients treated between August 1, 2019, and July 31, 2024. Data were collected from medical charts and phone interviews. Logistic regression identified factors affecting remission. Kaplan–Meier analysis was used to estimate the overall survival (OS) probabilities. Prognostic factors were determined using Cox regression. Results were interpreted as adjusted odds (AOR) and adjusted hazard ratios (AHR) with 95% confidence intervals. Statistical significance was declared at P ≤ .05.
Results
The overall remission rate was 62.4%, with 41.0% of low-risk GTN patients developing resistance to monotherapy. Chemo-resistance was independently associated with FIGO/WHO scores of 5-6 (AOR = 2.4, 95% CI: 1.2-5.6), disease duration ≥4 months (AOR = 1.7, 95% CI: 1.0-3.3), metastases (AOR = 2.2, 95% CI: 1.1-4.6), and choriocarcinoma histology (AOR = 3.0, 95% CI: 1.3-6.7). The 5-year OS was 77%, with metastases independently predicting poor survival (AHR = 4.1, 95% CI: 1.0-15.9). Choriocarcinoma and high-risk GTN were associated with increased relapse.
Conclusion
Chemoresistance was notably high among low-risk GTN patients. Particularly, patients with metastasis, choriocarcinoma histology, a WHO-FIGO risk score of 5-6, and longer disease duration may benefit from early combination therapy. In addition, GTN patients have shown relatively low survival rates. Metastases significantly affect survival. These findings highlight the importance of early detection, risk-adapted therapy, and therapeutic advancements to improve outcomes in resource-limited settings.
Plain Language Summary
Gestational trophoblastic neoplasia (GTN) is a rare cancer that usually develops after a pregnancy. Although it is highly curable with proper treatment, many women in low-resource settings face delays in diagnosis and challenges in getting the right medicines. In this study, we followed 205 women with GTN who were treated at three major hospitals in Northwest Ethiopia. We found that women with low-risk GTN had lower chances of complete recovery compared to what is reported in other countries. The chance of treatment not working was higher if the cancer had lasted four months or longer, had spread to other parts of the body, or was a specific type called choriocarcinoma. Patients with very high scores were also more likely to have the cancer come back after treatment. Our findings suggest that some patients might benefit from starting stronger treatment earlier and being monitored more closely. Improving follow-up care and tailoring treatment to each patient could help more women fully recover and live healthy lives.
Keywords
Introduction
Gestational trophoblastic neoplasia (GTN) is a rare malignancy that arises from placental trophoblastic tissue, accounting for less than 1% of gynecologic cancers worldwide. 1 The incidence of gestational trophoblastic diseases (GTD) exhibits marked geographic variation, with disproportionately higher rates reported in low- and middle-income countries (LMIC). In Western nations, GTD occurs in approximately 2 to 7 cases per 100 000 pregnancies. 2 In contrast, the pooled prevalence in East Africa has been documented as high as 22% among pregnant women, 3 with Ethiopia reporting incidence between 1.8 and 11.4 per 1000 deliveries. 4 Indeed, recent findings indicate that 5-10% of GTD cases progress to GTN, whereas rates may surpass 20% in high-risk populations. 5
Risk stratification of GTN traditionally relies on the International Federation of Gynecology and Obstetrics (FIGO) staging and World Health Organization (WHO) prognostic scoring systems, which categorize patients into low-risk (score <7) or high-risk (score ≥7).6-8 Though this binary model may not fully capture GTN’s clinical heterogeneity. A refined four tier system has been proposed to improve prediction of treatment response and relapse. 9 Additionally, molecular profiling, such as androgenetic diploidy in complete mole, diandric triploidy in partial mole, and p53/microRNA alterations in choriocarcinoma, which correlate with tumor behavior and inform individualized therapy.10,11
Despite its malignant nature, GTN is among the most curable human cancers, owing to its high chemosensitivity and favorable therapeutic response. Reported survival rates approach 90%, with cure rates nearing 100% in low-risk cases and reaching 80%-90% in high-risk patients treated at specialized oncology centers.12,13 However, treatment failure remains a significant clinical challenge. For instance, chemoresistance to the first-line single-agent regimen has been inconsistently observed in 25% to 52% of patients with low-risk GTN.13-18 In contrast, resistance to multi-agent regimens among high-risk patients ranges from 10-40%.14,19,20 Moreover, disease recurrence following initial chemotherapy (CT), often associated with prior drug resistance, 21 has been documented in 2.7%-11.3% of cases.22-26 Thus, these treatment failures may necessitate salvage interventions, which are accompanied by prolonged hospitalization, increased healthcare costs, and potentially compromised survival rates.27,28 Nonetheless, most patients can attain remission after receiving second-line or intensified multi-agent CT. 29
However, there is a paucity of data on GTN treatment outcomes across SSA, including Ethiopia, where constraints in diagnostic and treatment availability may perhaps negatively affect clinical outcomes. 30 Furthermore, factors such as delayed presentation, limited access to oncology care, and disparities in health-seeking behavior could further affect the patient prognosis. 30 Therefore, this multi-center retrospective cohort study was designed as a descriptive assessment of treatment outcomes and determinants of remission, relapse, and mortality among women with GTN in northwest Ethiopia, with the additional aim of generating evidence that may inform future treatment policy and support clinical decision-making in resource-limited settings.
Methods and Materials
Study Design and Reporting Guidelines
This study was conducted and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology guidelines (STROBE) for cohort studies. 31 It employed a 5-year, hospital-based, multi-center retrospective cohort design.
Study Setting and Period
The study was conducted at three oncology centers in the Amhara region of northwest Ethiopia: the University of Gondar Comprehensive Specialized Hospital (UoGCSH), Tibebe Ghion Comprehensive Specialized Hospital (TGCSH), and Felege Hiwot Comprehensive Specialized Hospital (FHCSH). This study included all GTN cases managed at these participating centers from August 1, 2019, to July 31, 2024. Data were retrospectively extracted from patient medical charts and telephone interviews conducted between August 7 and September 25, 2024.
Patient Selection and Eligibility Criteria
All women with GTN treated at participating centers during the study period who met the inclusion criteria were enrolled. Inclusion criteria were: (1) histologically, imagining, and clinically confirmed GTN, (2) age ≥18 years, and (3) receipt of CT with a minimum follow-up period of 12 months after treatment initiation.
Exclusion criteria: (1) women who became pregnant during the follow-up period and (2) patients with incomplete charts (>10% missing critical data) to avoid biased estimates. 32
According to hospital records, 264 women with GTN were treated and followed during the study period—62 at TGCSH, 93 at FHCSH, and 109 at UoGCSH. After applying the eligibility criteria, 205 cases were included in the final analysis.
Study Variables and Outcome Definitions
In this study, the treatment outcomes were complete remission and death. Complete remission was defined as three consecutive weekly serum beta-human chorionic gonadotropin (β-hCG) levels within the normal range (<5 IU/L). Death was confirmed through medical charts or telephone interviews.
Independent variables included sociodemographic factors (age, education status, and health insurance), baseline clinical characteristics (FIGO/WHO prognostic score, presence of metastasis, histopathology, disease duration, prior failed CT, and prior pregnancy type), and treatment-related factors (CT regimen and number of cycles). Pre-treatment β-hCG level were categorized based on clinical cut-offs established in previous studies. 33
Chemoresistance was defined as a <10% decline over two cycles or a >10% rise after one cycle. Relapse was defined as two consecutive rising β-hCG levels after remission (in the absence of pregnancy). 34 Survival status was categorized as alive, lost to follow-up (>3 months without visit), or dead. Overall survival (OS) was defined as the proportion of patients alive at the end of follow-up.
Treatment Protocols and Follow-Up
Patients were treated according to each hospital’s standard protocol. Low-risk patients received methotrexate (MTX) (50 mg IM on days 1, 3, 5, and 7) with folinic acid (FA) (15 mg PO/IV on days 2, 4, 6, and 8) every 2 weeks. Actinomycin-D (0.5 mg IV push daily for 5 days) was used when MTX was contraindicated, unavailable, or ineffective. High-risk patients received the EMA/CO regimen (etoposide, methotrexate, actinomycin-D, cyclophosphamide, and vincristine) every 2 weeks. When the normalization of β-hCG levels was achieved, additional 2-3 cycles of consolidation therapy were given to prevent recurrence.
After achieving remission, patients were monitored monthly with β-hCG levels for six months, followed by every 2 months for another six months. All patients had at least six months of post-treatment follow-up. They were counselled to avoid conception for 1 year post-treatment. Adverse drug reactions were graded using the Common Terminology Criteria for Adverse Events (CTCAE - Version 5.0). 35
Data Collection Procedure and Quality Assurance
A structured data abstraction tool was developed by the principal investigator, based on a comprehensive review of the existing literature,14,15,19,25,36 to ensure that all relevant variables were captured. The tool, which was prepared in English, systematically collected data across three domains: socio-demographic characteristics, baseline clinical and pathological features, and treatment-related information.
Primary data were extracted directly from patient medical records using a pre-tested abstraction sheet. For patients lost to follow-up, telephone interviews were conducted first with the patient, and if the patient did not respond, with their primary caregivers (parents, spouses, or adult children ≥18 years) to ascertain the patients’ survival status. The interview questionnaire was developed in English, translated into Amharic by a language expert, and then back-translated into English to ensure conceptual equivalence. All interviews were conducted using a standardized and pretested interview guide to ensure consistency across participants, with minor wording adjustments for relatives. The interviewers were trained to address sensitive issues, such as patient death, in an empathetic manner, allowing respondents to pause, skip, or discontinue the question if needed. All participants provided informed verbal consent before the interview, as approved by the ethics committee. After data collection, all charts were returned to their respective files to maintain data integrity and confidentiality.
Data Management and Analysis
Data were coded, cleaned, and analyzed using STATA version 17. Descriptive statistics were summarized as frequencies, percentages, and measures of the central tendency and dispersion. Chi-square or Fisher’s exact test compared patient characteristics by relapse status, and logistic regression were applied to identify factors associated with remission. Model adequacy was assessed using the Hosmer–Lemeshow goodness-of-fit test (P > .05), and Multicollinearity was evaluated using the variance inflation factor (VIF <10). Survival analysis was performed using the Kaplan–Meier method, and the log-rank test was used to compare survival distributions between the groups. Cox regression was used to identify the predictors of mortality. The proportional hazards assumption was checked using Schoenfeld residuals (Global test = 0.08), and the model fit was further assessed using Cox–Snell residuals. Variables with P < .20 in the bivariable analyses were included in the final multivariate model. A P-value ≤0.05 with a 95% confidence interval was considered statistically significant.
Missing Data and Sensitivity Analyses
Charts with >10% missing critical data were excluded during case selection. For sporadic missing data, we assessed its pattern and mechanism. If deemed missing at random and affecting key covariates (>5%), we conducted multiple imputations and compared the results with the complete-case analyses. Minimal missing data (<5%) was handled using the available data, with sensitivity checks. Additional sensitivity analyses excluded influential cases and tested alternative variable coding to evaluate robustness.
Ethical Considerations
Ethical approval was obtained from the Ethical Review Committee of the University of Gondar (Ref. No.: SOPS/281/2016), which also approved the verbal informed consent procedure. Administrative permissions were granted by the oncology departments of FHCSH, TGCSH, and UoGCSH for chart review and data access. Additionally, informed verbal consent was obtained from the patients or their caregivers through phone before interviews. Confidentiality and privacy were strictly maintained by anonymizing all data and restricting access to authorized personnel. The study adhered to the ethical principles of the Declaration of Helsinki. Patients who were alive but lost to follow-up were referred back to their treating clinicians for continued follow-up.
Results
Patient Follow-Up Characteristics
Among the 205 female GTN patients included in the study, 112 (54.6%) were under follow-up. In comparison, 6 (3.0%) patients were found dead in their death reports. A total of 87 patients (42.4%) had lost their follow-up; therefore, telephone interviews were conducted with the patients or their relatives. Of these, 15 participants (7.3%) were lost to follow-up and could not be reached by phone. Accordingly, the outcomes of 190 (92.7%) patients were confirmed: 174 (84.9%) were alive and 16 (7.8%) had died (Supplemental Figure S1).
Participant Treatment Pattern Flow Diagram
Of the study participants, 142 (69.3%) were classified as having low-risk GTN. The preponderance of patients, 124 (60.5%), received MTX/FA as first line therapy, while all high-risk patients, 63 (30.7%), were treated with EMACO. Among the low-risk patients, 84 patients (40.9%) achieved remission following first-line therapy, whereas 58 patients (28.3%) developed chemoresistance. Notably, all patients eventually attained complete remission following second and third-line therapies (Figure 1). Treatment Pathways and Outcomes of 205 GTN Patients
General GTN Patient Characteristics
Not all sociodemographic variables were available from the medical records. Data on marital status documented for 200 patients; of whom 164 (82%) were married. Occupational information was available for 198 patients: 60 (30.3%) were employed and 87 (43.9%) were housewives. Regarding education, data from 195 patients showed that 57 (29.2%) were unable to write and read, and 55 (28.3%) had completed primary school. Of the 193 patients with residency data, 101 (52.4%) lived in urban areas (Supplemental Table S1).
Sociodemographic, Baseline Clinical Characteristics, and Chemotherapy Cycle of Women With GTN (n = 205)
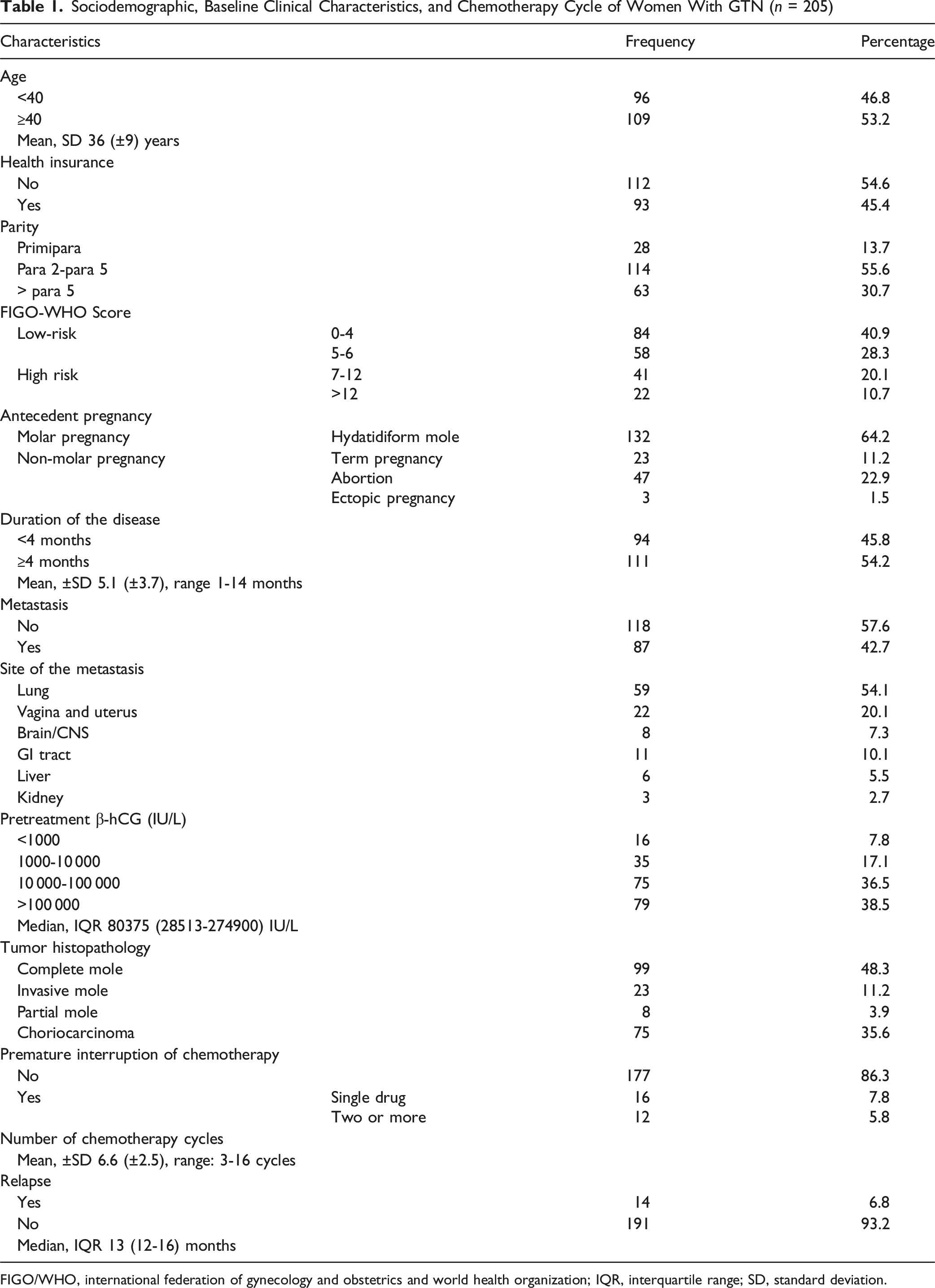
FIGO/WHO, international federation of gynecology and obstetrics and world health organization; IQR, interquartile range; SD, standard deviation.
Treatment Outcome of the GTN Patients
Remission Status and Survival Outcomes Among Women With GTN During Follow-Up

Comparison of Patient General Characteristics with Relapse Status
Comparison of Characteristics and Clinical Features With Patient Relapse Status

aMann–Whitney test.
Bold indicates variables significant in sub group analysis. Asterisk (*) denotes p < 0.05.
Reported Side Effects of the First-Line Therapy
Concerning side effects, more than one-quarter, 58 (28.3%) of the patients experienced chemotherapeutic-related side effects. The most common side effects with MTX were mucositis (Grade I/II) 16 (16.3%) and alopecia (Grade I/II) 8 (8.3%), whereas for EMACO, the frequently reported side effects were anemia 21 (Grade I/II/III) (21.4%) and neutropenia (Grade III/IV) 18 (18.3%) (Supplemental Table S2).
Overall Survival Rate of GTN Patients
The 5-year OS rate was 77% (95% CI: 62%-86.6%). In the first, second, third, and fourth years, the estimated cumulative survival rates were 98.2%, 93.2%, 87.8%, and 81%, respectively. The probability of survival was highest in the first year of diagnosis, whereas it declined as the follow-up time increased (Figure 2). Kaplan–Meier for the Overall Survival Function in Months of GTN Patients
Log-Rank Test Among Various Subgroups of GTN Patients
A log-rank test identified significant differences in median survival based on FIGO/WHO scoring, pretreatment β-hCG levels, and metastasis (Supplemental Table S3).
Kaplan–Meier Curve for Variable Groups of GTN Patients
According to this study, patients with GTN who presented with metastasis had a higher probability of dying than those without metastasis (Figure 3). Patients with FIGO-WHO scores of 0-4 showed a better survival rate than the other groups (Figure 4). Patients with a pre-treatment β-hCG level of >100 000 IU/L had a considerably greater mortality probability than the other categories (Figure 5). Kaplan–Meier Overall 5-Year Survival Curve Depending on the Presence of Metastasis in Patient With GTN Kaplan–Meier Overall 5-Year Survival Curve Regarding the FIGO-WHO Risk Score of GTN Patients Kaplan–Meier Overall 5-Year Survival Curve Depending on Pretreatment β-hCG Level of GTN Patients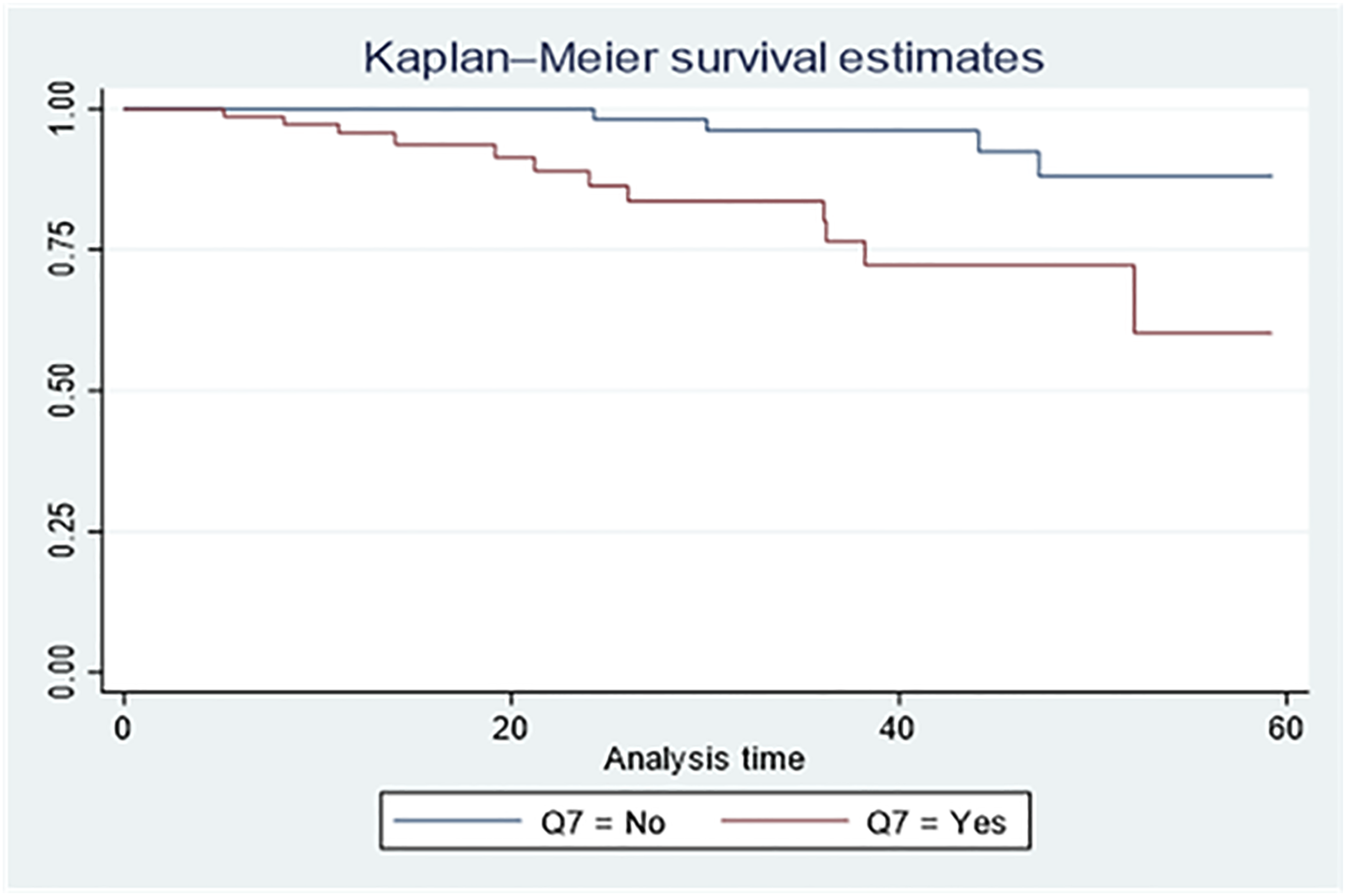


Factors Associated with Chemotherapy Remission in GTN Patients
Multivariate Analysis of Factors Associated With Chemotherapy Remission Among Women With GTN

The final model fit the data, ꭓ2 (8) = 8.6, P = .37. Bold
Prognostic Factors for the Overall Survival of GTN Patients
Prognostic Factors of the Overall Survival of GTN Patients

Bold indicates variables significant in multivariable analysis. Asterisk (*) denotes p < 0.05.
Comparative Remission and OS Rates for GTN Across LMIC and HIC Cohorts
Comparison of Remission and OS Rates Between the Current Cohort and Selected LMIC and HIC GTN Cohorts

Act-D, actinomycin-D; MTX, methotrexate; FA, folinic acid (Leucovorin); EMA/CO, etoposide, methotrexate, actinomycin-D/cyclophosphamide, vincristine; MAC, methotrexate, actinomycin-D, cyclophosphamide; EMA/EP, etoposide, methotrexate, actinomycin-D/etoposide, cisplatin; VMP, vincristine, methotrexate, prednisolone; HIC, high-income countries; LMIC, low and middle-income countries.
Discussion
Despite the high incidence of GTN, this multicenter study provides the first comprehensive evaluation of GTN remission, survival, and prognostic factors in Northwest Ethiopia. By incorporating data from multiple oncology centers, it overcomes the limitations of earlier single-center studies and presents a regionally representative profile of GTN burden and treatment response. Although GTN is highly chemosensitive, our findings showed that the overall remission rate was 62.4%, with 41.0% of low-risk patients showing resistance to single agent regimen. Chemoresistance was significantly associated with intermediate FIGO/WHO scores, prolonged disease duration, the presence of metastasis, and choriocarcinoma histology. The 5-year OS rate was 77%, with metastasis as the sole independent predictor of poor survival. In addition, choriocarcinoma and high-risk GTN were linked to increased relapse rates.
The mean age of this study was 36 years, consistent with other reports,45,46 but slightly older than those reported in Uganda. 47 More than half (55.6%) of our patients had between two and five children, consistent with parity being a known GTN risk factor.47,48 The majority of cases followed molar pregnancies (64.2%), aligning with previous studies,49,50 and 35.6% were choriocarcinoma, slightly higher than other studies. 51 Metastases were observed in 42.7% of cases, predominantly pulmonary, aligning with global patterns. 20
In the current study, up to 41.0% of low-risk patients developed chemoresistance to first-line CT, most of whom initially received MTX/FA. This rate is consistent with findings from other resource-limited settings, where chemoresistance is reported to exceed 40%.16-18 Likewise, a study from Vietnam reported a chemoresistance rate of 33.7%, 37 and Alazzam et al. documented a 45% of resistance rate in this population. 14 These rates contrast with those in high-income countries, where CR approaches 80-90%.39-41,52 These cross-setting differences are further illustrated in Table 6, which compares remission outcomes across LMIC and HIC cohorts. These observed discrepancies in resource-limited settings may be attributed to systemic factors, including delayed diagnosis, late referrals, limited availability of first-line regimens, inadequate follow-up, and treatment interruptions due to financial and logistical constraints. 8 The relatively small sample size and patient selection of our cohort may also have contributed to this variability. Furthermore, resistance was more common in patients with WHO/FIGO scores of 5-6, echoing prior evidence that these intermediate-risk groups respond less favorably to the respective monotherapy. 53 Contrariwise, findings from Taiwan demonstrated CR rate of 83% when using a vincristine, methotrexate, and prednisolone (VMP) regimen as first-line CT. 38 This indicated that the choice of initial regimen plays a greater role in achieving remission.
High-risk patients in our cohort were treated with EMA/CO; however, 30.2% developed chemoresistance, a finding in congruence with studies from China and Egypt.18,19 Alazzam et al. also indicated 30-40% of chemoresistance for multi-agent regimen. 14 In contrast, this finding is higher than those of studies from the United States 54 and India. 16 This high rate in our study may be explained by the high proportion of high-risk patients who had brain and liver metastases, which confer a poor prognosis and are known to be biologically more aggressive and harder to treat. 55 A study from Iran reported CR rates up to 88% when EMA/EP was used as the first-line multi-agent CT. 42 Conversely, a study from Egypt reported a higher chemoresistance rate; only 40% of them achieved CR, 20 due to the use of the MAC regimen (Methotrexate, Actinomycin-D, and Cyclophosphamide) as first-line CT.
In this study, patients classified as low-risk GTN with FIGO-WHO scores of 5-6 showed a significantly higher likelihood of chemoresistance compared to those with scores of 0-4, aligned with previous studies.14,36,53,56 These sub-groups are frequently related to larger tumor sizes, persistently elevated serum β-hCG levels, and the presence of metastatic lesions, all of which may contribute to reduced chemosensitivity. Although the WHO-FIGO system designated these patients as low risk, emerging evidence suggested that a FIGO score of 5-6 may represent an intermediate risk subset with distinct therapeutic implications. 57 Consequently, initial treatment with multi-agent CT may be more appropriate for these patients than conventional monotherapy, with potential benefits in reducing resistance and improving treatment outcomes.
Disease duration also emerged as an important predictor of chemoresistance. Patients presenting four months or more after the antecedent pregnancy exhibited higher chemoresistance rates, consistent with prior studies.15,40 Delayed presentation likely allows further tumor progression to more advanced stages, thereby decreasing responsiveness to CT. Notably, more than half of our cohort presented late, reflecting persistent health system barriers, including limited public awareness, delayed referral pathways, and insufficient diagnostic capacity, particularly in resource-constrained settings.
Similarly, metastatic involvement of lung, liver, or brain was significantly associated with increased chemoresistance, corroborating previous evdences.28,58,59 This association may reflect both aggressive biology of metastatic GTN and micro-environmental factors that reduce drug penetration and thereby diminish drug efficacy. Nonetheless, the resistance rates observed in our cohort were lower than those reported in the Iranian study, 15 possibly due to variations in sample size, baseline risk profile, and treatment protocol.
Finally, patient with choriocarcinoma histology exhibited significantly higher odds of developing chemoresistance compared with those diagnosed with complete mole pathology. This finding is in agreement with previous studies,14,25,36,58 and is likely attributable to the tumors’ inherently aggressive proliferation and distinct molecular features, including vascular endothelial growth factor (VEGF) overexpression and disruption in DNA repair pathways. 60 These results highlight the clinical importance of integrating histopathological and molecular diagnostics into risk stratification frameworks to support individualized therapeutic strategies.
The relapse rate in this study was 6.8%, which agreed with other studies,24-26 but was slightly higher than that reported in the United Kingdom (3.2%) and China (2.7%).22,23 This disparity might be due to differences in patient adherence, healthcare infrastructure, and follow-up protocols. The median time to relapse was 13 months (range: 12-16), comparable to the 7-month median (range: 4-32 months) reported by Maria et al. 25 in Egyptian women with GTN. While previous studies have linked relapses primarily before drug resistance, 21 our finding did not demonstrate a direct association. Furthermore, consistent with earlier research,21,28,61 our study found that elevated β-hCG levels, choriocarcinoma histology, and higher FIGO scores were significantly associated with relapse, highlighting the need for closer follow-up and early intervention for patients with these risk profiles.
In this study, we did not observe a statistically significant relationship between chemoresistance and medication side effects. However, severe adverse reactions often led to treatment interruptions, which may indirectly facilitate the likelihood of chemoresistance. About 43.1% of chemoresistant cases experienced Grade III/IV hematologic toxicities, including neutropenia and anemia, consistent with previous studies.42,62 These findings underline the importance of routine monitoring of complete blood counts and proactive management of myelosuppression, including blood transfusions and granulocyte colony-stimulating factor support when indicated, to optimize treatment adherence and minimize the risk of resistance. 63
The 5-year OS rate observed in this study was 77%, which was comparable to previously published data.18,25,43 This rate was notably higher than those reported in studies from France and South America,64,65 where the majority of participants had FIGO/WHO scores >12 and frequent brain or liver metastases, factors known to be associated with poorer survival rates.66,67 Conversely, our OS rate was lower than that demonstrated in cohorts from the United States 40 and the United Kingdom, 44 where all patients were classified as low-risk (FIGO scores 0-4), a group with cure rates approaching 100% when treated with single agent CT and lower rates chemoresistance.7,68 These cross-setting differences are further illustrated in Table 6, which summarizes remission and survival outcomes across selected LMIC and HIC cohorts. These comparisons highlight the significant impact of baseline risk profiles, tumor burden, and treatment responsiveness on survival outcomes.
We also observed that the presence of metastasis to the lungs, brain, and other organs at diagnosis was associated with an approximately four-fold higher hazard of death, consistent with previous evidences.12,67,69 Reports from France and China similarly demonstrate poor outcomes among patients with multi-organ or renal metastases,43,67 reflecting the biologic aggressiveness and therapeutic resistance characteristics of advanced disease. 70 These findings are mainly relevant for resource-limited settings, where diagnostic delays, constrained treatment options, and limited follow-up capacity remain persistent challenges.
This study has the following strengths: providing one of the first region-specific evaluations of GTN treatment outcomes in northwest Ethiopia, based on multicenter data from three oncology centers. The inclusion of multiple institutions enhances the representativeness of the findings and offers valuable insights into GTN management in a resource-limited setting. Nonetheless, several limitations have been acknowledged. The retrospective design, comparatively small sample size, and exclusion of incomplete records may have introduced selection bias. Information bias related to chart abstraction and secondary data extraction may also have affected data quality. The 7.3% loss to follow-up introduces uncertainty in survival estimates, potentially leading to either overestimation or underestimation of true outcomes. However, the findings offer a meaningful baseline for enhancing GTN care and informing future research in Ethiopia.
These findings also have important implications for health policy. Strengthening diagnostic capacity, optimizing referral pathways, and ensuring equitable and sustained access to evidence-based treatment protocols for GTN are crucial for improving outcomes in low-resource settings. Furthermore, establishing a prospective national GTN registry would enhance surveillance by enabling systematic case identification, longitudinal monitoring of treatment response and survival, and the generation of high-quality data to inform policy development and guide resource allocation. Such a registry would also facilitate earlier detection of high-risk cases and promote adherence to standardized management guidelines, thereby contributing to improved quality of care and better clinical outcomes.
Conclusion
In this multicenter observational study, the remission rate of low-risk GTN patients treated with single-agent CT was lower than global averages, whereas remission among high-risk patients was comparable to international reports. Chemoresistance was independently associated with a FIGO-WHO score of 5-6, the presence of metastases, prolonged disease duration (≥4 months), and choriocarcinoma histology, with metastasis also predicting poorer survival. These findings highlight that patients with intermediate risk scores may benefit from earlier initiation of combination CT, while ultra-high-risk patients (score ≥13) require intensive follow-up and tailored therapeutic strategies. Strengthening early referral pathways, timely diagnosis, and adherence to treatment protocols could improve remission and survival outcomes. Overall, the study provides evidence to inform risk-adapted treatment policies and supports the implementation of personalized GTN management in resource-limited settings.
Supplemental Material
Suppplemental Material - Gestational Trophoblastic Neoplasia in Northwest Ethiopia: A Multi-Center Retrospective Cohort Study
Suppplemental Material for Gestational Trophoblastic Neoplasia in Northwest Ethiopia: A Multi-Center Retrospective Cohort Study by Abel Temeche Kassaw, Mekuanent Kassa Birara, Ephrem Tafesse Teferi3, Eliyas Adissu Taye, Desalegn Addis Mussie, Fissha Nigussie Dagnew, Getachew Yitayew Tarekegn, Samuel Berihun Dagnew, Tilaye Arega Moges, Belayneh Yitayew Wallie, Woretaw Sisay Zewdu, Tigabu Eskeziya Zerihun in Cancer Control
Supplemental Material
Suppplemental Material - Gestational Trophoblastic Neoplasia in Northwest Ethiopia: A Multi-Center Retrospective Cohort Study
Suppplemental Material for Gestational Trophoblastic Neoplasia in Northwest Ethiopia: A Multi-Center Retrospective Cohort Study by Abel Temeche Kassaw, Mekuanent Kassa Birara, Ephrem Tafesse Teferi3, Eliyas Adissu Taye, Desalegn Addis Mussie, Fissha Nigussie Dagnew, Getachew Yitayew Tarekegn, Samuel Berihun Dagnew, Tilaye Arega Moges, Belayneh Yitayew Wallie, Woretaw Sisay Zewdu, Tigabu Eskeziya Zerihun in Cancer Control
Footnotes
Acknowledgments
I want to express my sincere gratitude to the School of Pharmacy, College of Medicine and Health Sciences, University of Gondar, for their unwavering support. Also, I would extend my heartfelt thanks to the oncology staff at the University of Gondar, Tibebe Ghion, and Felege-Hiwot hospitals for their cooperation and assistance. Last but not least, our special thanks go to all patients and relatives involved in the phone interviews.
Ethical Considerations
Ethical approval was obtained from the Ethical Review Committee of the University of Gondar (Ref. No.: SOPS/281/2016), which also approved the verbal informed consent process. Administrative permissions were secured from the oncology departments of FHCSH, TGCSH, and UoGCSH for chart review and data access. Informed verbal consent was obtained from patients or their caregivers before telephone interviews. Confidentiality and privacy were strictly maintained by anonymizing all data and restricting access to authorized personnel only. The study adhered to the ethical principles of the Declaration of Helsinki. Patients who were alive but lost to follow-up were referred back to their treating clinicians for continued follow-up.
Author Contributions
AT: Conceptualization, Formal Analysis, Investigation, Methodology, Project administration, Data interpretation, Writing–original draft, Writing–review and editing, MK, ET, EA, DA, FN, and GY: Data curation, Formal Analysis, Visualization, Writing–review and editing, SB, TA, BY, WS and TE: Data interpretation, Methodology, Supervision, Writing–review and editing. All authors read and approved the revised manuscript for publication.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data for this study were obtained from the corresponding author on a reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
