Abstract
Introduction
This meta-analysis aims to evaluate the impact of increasing the use of metformin in neoadjuvant treatment for breast cancer (BC) on the rate of pathological complete response (pCR) in patients.
Methods
A systematic search was conducted in four electronic databases: PubMed, Web of Science, Embase, and Cochrane Library. The search scope covered all the literature from the establishment of the databases to April 2025. The risk ratio (RR) and 95% confidence interval (CI) were calculated. The outcome indicator was the pCR rate.
Result
This meta-analysis included a total of 8 randomized controlled trials (RCTs), involving 474 patients. The results showed that there was no statistically significant difference in the pCR rate between the experimental group containing metformin and the control group (RR = 1.21, 95% CI: [0.85, 1.71], P = 0.28). Subgroup analysis revealed that there were no significant differences in the pCR rate between the two groups in patients with metabolic syndrome (RR = 2.09, 95% CI [0.55, 7.85], P = 0.28), patients without metabolic syndrome (RR = 1.12, 95% CI [0.81, 1.55], P = 0.49), patients from Eastern countries (RR = 1.15, 95% CI [0.63, 2.11], P = 0.65), and patients from Western countries (RR = 1.32, 95% CI [0.75, 2.32], P = 0.34).
Conclusion
This study did not observe any effect of increasing the use of metformin on the pCR rate of patients in neoadjuvant treatment for BC.
Introduction
According to the 2022 GLOBOCAN data, breast cancer (BC) ranks second among all malignant tumors in terms of global incidence, only after lung cancer; among women, it has the highest incidence rate. In the same year, there were approximately 2.3 million new cases and 670 000 deaths from BC worldwide. 1 Currently, the treatment options for BC are diverse, mainly including surgery, chemotherapy, radiotherapy, endocrine therapy, targeted therapy, and immunotherapy. Neoadjuvant therapy plays a crucial role in the comprehensive management of BC: it can effectively reduce the tumor stage, narrow the scope of breast and axillary surgery, and provide real-time assessment basis for treatment response. 2 The rate of pathological complete response (pCR) is regarded as a good indicator for evaluating the efficacy of neoadjuvant therapy, and patients who achieve pCR usually have a better long-term prognosis. However, there is some controversy regarding its application in Luminal B-type BC.
At present, neoadjuvant therapy for BC is based on chemotherapy and adopts differentiated strategies according to different molecular subtypes: for triple-negative breast cancer (TNBC), common regimens include AC-T (doxorubicin and cyclophosphamide first, followed by paclitaxel), platinum-based regimens or immunotherapy combined with chemotherapy; for human epidermal growth factor receptor 2 (HER2)+ BC, dual-targeted therapy (trastuzumab combined with pertuzumab) along with chemotherapy is recommended; while for hormone receptor-positive (HR+)/HER2- BC, chemotherapy is the main treatment approach. 2 The axillary pCR rates vary significantly among patients with different molecular subtypes, in descending order: HR-/HER2+ subtype (60%), TNBC subtype (48%), and HR+/HER2- subtype (18%). 3
Metformin is a classic biguanide antidiabetic drug, which has attracted attention due to its wide clinical application, good safety profile, and potential anti-tumor activity demonstrated through multiple mechanisms. 4 The cumulative evidence from both in vivo and in vitro studies5,6 indicates that the potential anti-tumor mechanism of metformin can mainly be classified into the following two non-exclusive categories: (1) Indirect effects, which involve inhibiting the phosphoinositide 3-kinase (PI3K)/mitogen-activated protein kinase (MAPK) signaling pathway associated with cell proliferation by lowering insulin levels and blood glucose; (2) Direct effects, which directly impact cancer cells through pathways such as activating the adenosine monophosphate-activated protein kinase (AMPK) pathway. 7 These two mechanisms typically act in concert and influence each other, providing new insights and potential strategies for BC treatment. However, there is still uncertainty regarding the efficacy of metformin in neoadjuvant therapy for BC, and no systematic meta-analysis specifically addressing this field has been published yet. Therefore, this study aims to deeply explore the impact of metformin on the pCR rate of patients in neoadjuvant treatment for BC by reviewing relevant randomized controlled trials (RCTs).
Methods
This study was conducted in strict accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 8 This meta-analysis has been registered on PROSPERO (Registration Number: CRD420251035097).
Search Strategy
This study searched four electronic databases: PubMed, Web of Science, Embase, and the Cochrane Library, from inception to April 2025. The purpose of this study is to retrieve information regarding the efficacy of adding metformin in neoadjuvant therapy for BC patients. The search strategy employs a combination of Medical Subject Headings (MeSH) terms and free words, with the specific search formula as follows: (((((((((((((((((((((((((((((((((((((Breast Neoplasm) OR (Neoplasm, Breast)) OR (Neoplasms, Breast)) OR (Breast Tumors)) OR (Breast Tumor)) OR (Tumor, Breast)) OR (Tumors, Breast)) OR (Breast Cancer)) OR (Cancer, Breast)) OR (Cancer of Breast)) OR (Cancer of the Breast)) OR (Malignant Neoplasm of Breast)) OR (Breast Malignant Neoplasm)) OR (Breast Malignant Neoplasms)) OR (Malignant Tumor of Breast)) OR (Breast Malignant Tumor)) OR (Breast Malignant Tumors)) OR (Mammary Cancer)) OR (Cancer, Mammary)) OR (Cancers, Mammary)) OR (Mammary Cancers)) OR (Mammary Neoplasms, Human)) OR (Human Mammary Neoplasm)) OR (Human Mammary Neoplasms)) OR (Neoplasm, Human Mammary)) OR (Neoplasms, Human Mammary)) OR (Mammary Neoplasm, Human)) OR (Breast Carcinoma)) OR (Breast Carcinomas)) OR (Carcinoma, Breast)) OR (Carcinomas, Breast)) OR (Mammary Carcinoma, Human)) OR (Carcinoma, Human Mammary)) OR (Carcinomas, Human Mammary)) OR (Human Mammary Carcinomas)) OR (Mammary Carcinomas, Human)) OR (Human Mammary Carcinoma)) AND ((((((((Metformin) OR (Dimethylbiguanidine)) OR (Dimethylguanylguanidine)) OR (Glucophage)) OR (Metformin Hydrochloride)) OR (Hydrochloride, Metformin)) OR (Metformin HCl)) OR (HCl, Metformin)). The search results were imported into EndNote software for deduplication processing. In this study, two researchers independently conducted the literature search. Any discrepancies in the search results were resolved by a third researcher to ensure consensus. Additionally, the reference lists of the included articles were indexed to identify any potentially missed potential studies.
Inclusion and Exclusion Criteria
Inclusion Criteria: the development of the inclusion criteria strictly adhered to the PICOS principle. Only articles that fully meet all the following criteria will be considered for inclusion in this study. 1. The study subjects were patients with resectable BC (with or without diabetes, and regardless of their HR/HER2 status). 2. The experimental group received neoadjuvant therapy that included metformin before surgery (regardless of the dosage and frequency of administration of metformin). 3. The control group received neoadjuvant therapy without metformin before surgery. 4. The study reported data related to pCR rate. 5. The study design belongs to RCT.
Exclusion Criteria: any articles that meet any of the following criteria will be excluded. 1. Studies involving metastatic BC. 2. Articles for which the full text cannot be obtained. 3. Articles published in languages other than English. 4. For the same clinical trial with multiple updated publications, the most comprehensive or recent literature will be selected.
Definition
The definition of pCR rate is the absence of invasive cancer at the primary site and in the axilla following the completion of neoadjuvant systemic chemotherapy, and it also includes the presence of residual ductal carcinoma in situ.
Data Extraction
Data extraction was independently conducted by two researchers based on a pre-designed form and included the following information: (1) basic information, including authors, year of publication, country, BC stage (TNM), national clinical trial number, and name of the clinical trial; (2) study design and implementation, including treatment regimen, recruitment period, and number of participants recruited; (3) study design and criteria, including inclusion and exclusion criteria for patients as well as the primary outcome.
Risk of Bias Assessment
This study utilized the Cochrane Risk of Bias Assessment Tool (Risk of Bias 1.0) to assess the risk of bias across six domains. Each indicator was assessed as “low risk of bias,ˮ “unclear,ˮ or “high risk of bias.ˮ The six domains included: (1) selection bias (random sequence generation and allocation concealment); (2) performance bias (blinding of researchers and participants); (3) detection bias (blinding of outcome assessment); (4) attrition bias (completeness of outcome data); (5) reporting bias (selective reporting of results); (6) other potential sources of bias.
Statistical Analysis
RevMan 5.4 software was used for the meta-analysis. For dichotomous outcomes, the risk ratio (RR) and its 95% confidence interval (CI) were used for evaluation. When RR >1, the statistical results support the experimental group; when RR <1, the statistical results support the control group. Given the potential heterogeneity among the included studies (eg, different countries, treatment regimen), a random-effects model was employed for statistical analysis to enhance the feasibility of the results. In this study, the chi-square test and the I2 statistic were used to assess heterogeneity among studies. When I2 ≤ 50% and P ≥ 0.05, it is considered that the heterogeneity is low; when I2 > 50% and P < 0.05, it is considered that there is significant heterogeneity. Subgroup analyses were performed to explore the potential sources of heterogeneity, and funnel plots were used to assess publication bias. Additionally, sensitivity analyses were conducted by excluding each study one by one to evaluate the stability of the results. All statistical tests were two-sided, with statistical significance set at P < 0.05.
Results
Literature Screening Results
Based on the pre-defined search terms, 8247 entries were retrieved from four electronic databases, and no additional entries were found through other search methods. After duplicate removal using Endnote software, 6128 entries remained. Subsequently, 6085 entries were excluded by reading the titles and abstracts, leaving 43 articles for full-text review. After full-text review, 35 studies were further excluded, and 8 studies were finally included for analysis. The reasons for excluding the 35 studies were as follows: 15 were non-RCTs, 14 had unavailable data, 2 could not be obtained in full text, and 4 did not report the outcome of interest. The detailed screening process is shown in Figure 1. Flow Diagram Illustrating the Study Selection Process
Characteristics of the Articles
Characteristics of the Studies Included in This Meta-Analysis

FMD, fasting-mimicking diet; ChT, chemotherapy; AC, adriamycin (doxorubicin) and cyclophosphamide; AC-T, adriamycin (doxorubicin) and yclophosphamide followed by taxol (paclitaxel); NACT, neoadjuvant chemotherapy; TEC, docetaxel, epirubicin, and cyclophosphamide; NAST, neoadjuvant systemic therapy, W, week.
Quality Assessment
This study used the Cochrane risk of bias assessment tool (RoB 1.0 version) to evaluate the quality of the included literature. The quality evaluation results showed that 75% of the studies were at low risk in terms of random sequence generation; 75% of the studies were at moderate risk in terms of allocation concealment; and all studies were at low risk in terms of reporting bias. For detailed evaluation results, please refer to Supplemental Figures 1 and 2.
Pathological Complete Response
A meta-analysis was conducted on the eight included studies. Compared with the control group, there was no significant difference in the pCR rate of the experimental group (RR = 1.21, 95% CI [0.85, 1.71], P = 0.28), and the heterogeneity was relatively low (I2 = 22%) (Figure 2). Forest Plot Showing the Impact of Adding Metformin to Neoadjuvant Treatment for Breast Cancer on the pCR Rate in Patients
Sensitivity analysis was performed by excluding each study one by one, and the results were stable. Meanwhile, publication bias was evaluated using a funnel plot, which was roughly symmetrical, and no significant publication bias was found. For detailed analysis results, please refer to Figure 3. Funnel Plot Showing the Effect of Metformin on the pCR Rate of Breast Cancer Patients
Subgroup Analysis
Subgroup analysis was conducted based on the presence or absence of metabolic syndrome. In the group without metabolic syndrome, there was no significant difference in pCR between the two groups (RR = 1.12, 95% CI [0.81, 1.55], P = 0.49). In the group with metabolic syndrome, there was also no significant difference in the pCR rate between the two groups (RR = 2.09, 95% CI [0.55, 7.85], P = 0.28). Refer to Figure 4 for details. Forest Plot Showing the Effect of Metformin on the pCR Rate in Breast Cancer Patients Under the Condition of Metabolic Syndrome (A) Metabolic Syndrome (B) Non-metabolic Syndrome)
Subgroup analysis was also performed based on geographical regions. In Eastern countries, there was no significant difference in the pCR rate between the two groups (RR = 1.15, 95% CI [0.63, 2.11], P = 0.65), and in Western countries, there was also no significant difference between the two groups (RR = 1.32, 95% CI [0.75, 2.32], P = 0.34). Refer to Figure 5 for details. Forest Plot Showing the Impact of Metformin on the pCR Rate in Breast Cancer Patients Under Regional Analysis (A) Eastern Countries (B) Western Countries)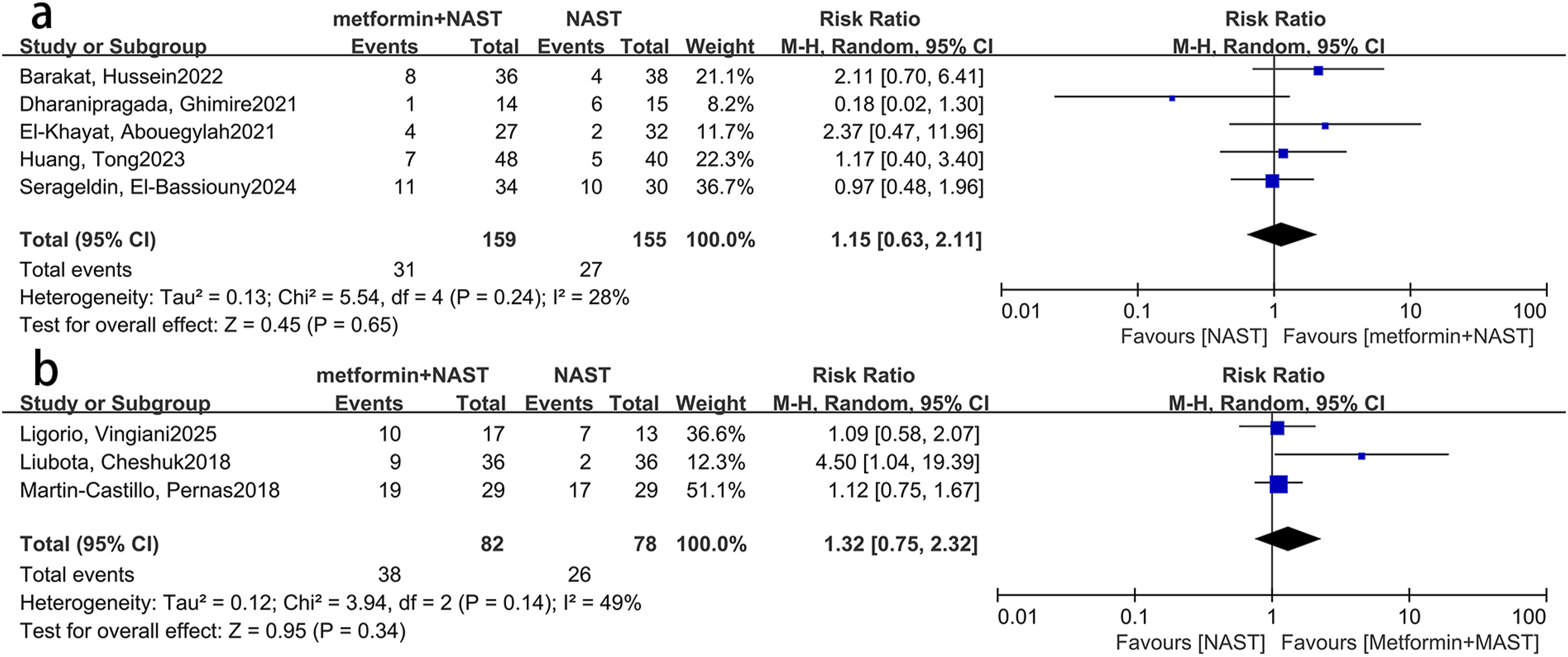
Discussion
This meta-analysis included eight RCTs and is the first systematic study to explore the impact of metformin in neoadjuvant therapy for BC. The results showed that adding metformin to the neoadjuvant treatment regimen for BC did not significantly improve the pCR rate of patients.
Metformin is a classic hypoglycemic drug. Its hypoglycemic effect is mainly achieved through mechanisms such as inhibiting hepatic gluconeogenesis and reducing intestinal glucose absorption. 17 Its potential anti-tumor effect can be attributed to two mechanisms, namely the indirect mechanism and the direct mechanism.
The indirect anti-cancer effects of metformin. The indirect anti-cancer effects of metformin are mainly reflected in the regulation of blood glucose, insulin and insulin-like growth factor (IGF) system, etc. It inhibits hepatic gluconeogenesis by antagonizing glucagon signaling pathways and inhibits the activity of key metabolic enzymes, thereby reducing circulating blood glucose levels 18 ; it can also improve insulin sensitivity and alleviate hyperinsulinemia. 6 Hyperinsulinemia can act as a pro-cancer growth factor, activating insulin receptors (IR) and insulin-like growth factor receptors (IGF-1R), and further activating PI3K and MAPK signaling pathways, promoting the growth and survival of cancer cells. 19 Metformin can inhibit these signaling pathways by reducing insulin and IGF-1 levels. In addition, cancer cells take up a large amount of glucose. 20 Metformin can indirectly cut off the energy supply of cancer cells by reducing blood glucose levels, thereby inhibiting tumor growth. However, this effect is still controversial. In the study by Bonanni, B. et al, in non-diabetic BC patients, metformin did not significantly regulate insulin and blood glucose levels, and its effect on Ki67 protein levels was not statistically different from that of the placebo. Moreover, this study also indicated that the effect of metformin on apoptosis was related to insulin resistance. The effect of metformin is subject to dual regulation based on the state of insulin resistance. In the subgroups of patients with insulin resistance or overweight (body mass index (BMI) ≥ 27 kg/m2), metformin treatment can reduce Ki67 levels, especially in the Luminal B molecular subtype; while in female patients with a homeostasis model assessment (HOMA) index ≤2.8 or BMI <27 kg/m2, the opposite trend was observed. However, none of the results were statistically significant. 21 This indicates that the indirect anti-tumor effect of metformin may not be stable and its effect may be influenced by individual differences, disease status and other factors of patients, making it difficult to exert the expected anti-tumor effect in practical applications. The effect of metformin on BC is also related to blood glucose concentration. The study by Zordoky, B. N. M., et al showed that in triple-negative BC, the anti-tumor effect of metformin was not obvious in a high-glucose environment, but was significantly enhanced in a low-glucose environment. 22 All these indicate that the indirect anti-cancer effect of metformin is influenced by multiple factors and may not necessarily work on BC.
The direct anti-cancer effect of metformin. Metformin exerts its effects by targeting multiple events and pathways, such as mammalian target of rapamycin (mTOR), cell cycle, inflammation, glucose metabolism, angiogenesis, and cancer stem cells (CSCs). Most of these effects are related to the activation of AMPK by metformin. 7
The AMPK pathway is the core regulatory mechanism for cellular energy metabolism. When the ratio of AMP to ATP in the cell increases or when upstream kinases (such as liver kinase b1 (LKB1) and Calcium/calmodulin-dependent protein kinase (CaMKK)) are phosphorylated, AMPK is activated. Activated AMPK restores energy homeostasis by inhibiting anabolic processes, promoting catabolic processes, and regulating the cell cycle. Metformin, on the one hand, indirectly activates AMPK by inhibiting the function of mitochondrial complex I and increasing the AMP/ATP ratio; on the other hand, it can also enhance the phosphorylation of LKB1 on AMPK, further strengthening its activity. This activation effect can inhibit the glycolysis and proliferation of cancer cells and induce autophagy.23,24 The AMPK pathway is an important mechanism by which metformin exerts its anti-cancer effects, but activating the AMPK pathway does not necessarily mean that it will produce anti-tumor effects. Park, J.H., et al pointed out that in TNBC, low-dose metformin activates the AMPK - acetyl-CoA carboxylase (ACC) - fatty acid beta-oxidation (FAO) axis, promotes Src protein phosphorylation, and enhances the proliferation and migration abilities of tumors; only high-dose metformin can inhibit mitochondrial function and produce anti-tumor effects. 25 Moreover, studies have shown that TNBC MDA-MB-231 cells, due to the lack of LKB1 kinase, cannot activate AMPK and have resistance to metformin, which may not exert anti-tumor effects. 26 Furthermore, Hampsch, R.A., et al demonstrated through in vitro and animal experiments that metformin, by activating the AMPK pathway, does not cause the death of tumor cells but instead promotes the survival of dormant estrogen receptor-positive (ER+) BC cells in a protective rather than inhibitory manner. 27 All of these indicate that the anti-tumor effects of metformin through the AMPK pathway are influenced by various factors, such as the dose of metformin, the type of BC, etc., and the process of its action is complex.
Multiple studies have shown that metformin has an anti-angiogenic effect, thereby exerting an anti-tumor effect.28,29 Firstly, metformin inhibits endothelial function and inflammatory mediator levels, down-regulates the expression of factors such as vascular endothelial growth factor (VEGF) and hypoxia-inducible factor 1-α (HIF-1α), thereby limiting tumor angiogenesis. This effect may be related to the inhibition of the mTOR signaling pathway. Secondly, metformin reduces HIF-1α activity by inhibiting the mTOR pathway, and subsequently reduces the expression of VEGF.7,30 The mechanism of metformin’s effect on angiogenesis in BC cells is complex. Dallaglio, K., et al's research indicates that metformin shows a complex dual effect on angiogenesis in BC cells. On the one hand, metformin may cause the upregulation of VEGF in BC cells, which is related to the pro-angiogenic effect. On the other hand, metformin can block the increase in cytochrome P450 1B1 (CYP1B1) levels in endothelial cells exposed to tumor cell-conditioned medium. CYP1B1 has a promoting effect on angiogenesis both in vivo and in vitro. 31 Metformin also shows a complex dual effect on melanoma. The effect of metformin on melanoma is usually cell-inhibitory in melanoma cells, but it does not have this effect in melanoma cells with BRAF mutations. It accelerates cell proliferation by upregulating VEGF-A through AMPK-dependent mechanisms. 32 These all indicate that the effect of metformin on angiogenesis is conditional and is influenced by multiple factors. Additionally, studies have shown that the anti-vascular effect of metformin is also related to geographical location. In the treatment of non-small cell lung cancer involving metformin, Mexican patients using metformin may benefit, while in Canadian patients, it may lead to an increased risk of death. 33 BC is a highly regionalized cancer with significant heterogeneity among types, making it difficult to confirm the effect of metformin on angiogenesis in BC cells. Therefore, this study conducts subgroup analysis based on geographical location. In this study, BC patients are divided into subgroups from Eastern and Western countries for analysis. The research results show that the addition of metformin has no significant effect on the pCR rate of patients. This suggests that the effect of metformin on BC treatment is independent of geographical location.
Metformin can also exert its effects by influencing the metabolism of breast cancer. It achieves this by inhibiting gluconeogenesis, improving insulin sensitivity, blocking cell growth-related signaling pathways; activating AMPK to inhibit lipid and protein synthesis and reverse the metabolic pattern of cancer cells; and simultaneously inhibiting mitochondrial complex I, causing an energy crisis in cancer cells, especially being more lethal to cancer cells that rely on oxidative phosphorylation.7,34 However, the specific effects of metformin are still controversial. Kalinsky, K., et al's research found that in overweight and obese BC patients, metformin administration had no significant effect on ki67. Although the patients' BMI, leptin and cholesterol levels all significantly decreased, and fasting insulin and HOMA index also showed a downward trend, the improvement of these metabolic indicators was not significantly related to the significant decrease in the proliferation index. 35 It is demonstrated that even though metabolism improves, the growth of tumor cells is not inhibited. Moreover, individual differences among patients may lead to inconsistencies in the metabolism, distribution and action mechanism of metformin in the body, thereby affecting its anti-tumor effect. This is consistent with the subgroup analysis results of this study. Whether BC patients have metabolic syndrome or not, adding metformin does not improve the pCR rate of the patients.
This meta-analysis still has limitations. Firstly, the number of studies and sample size are limited. A total of eight RCTs were included, involving 474 patients. This scale is insufficient in terms of effect size estimation and heterogeneity test, and there may be statistical errors, which may underestimate the true efficacy. Secondly, pCR can be regarded as a favorable prognostic indicator for more aggressive subtypes such as TNBC and HER2+ BC. However, this is not the case for Luminal B-type BC. There are certain limitations to it. In addition, the number of subgroups is limited. BC is a heterogeneous cancer, and factors such as obesity level, genetic phenotype, and BC subtypes may all affect the efficacy of metformin; especially, the biological characteristics of different BC subtypes vary, resulting in different treatment responses and response rates for each subtype, so each subtype needs to be evaluated separately. However, due to the limitation of the availability of original data, this study only conducted subgroup analysis based on two preset variables and was unable to further examine the effects of BC types, doses, courses, and other factors on the pCR rate. It is hoped that further research will be conducted in these areas in the future.
Conclusion
This study included eight RCTs and did not observe that adding metformin to neoadjuvant therapy for BC could improve the pCR rate.
Supplemental Material
Supplemental material - Enhancing the Efficacy of Metformin in Neoadjuvant Therapy for Breast Cancer: A Meta-Analysis Based on Randomized Clinical Trials
Supplemental material for Enhancing the Efficacy of Metformin in Neoadjuvant Therapy for Breast Cancer: A Meta-Analysis Based on Randomized Clinical Trials by Fei Xie, Ting Jiang, Mengyi Qian, Xinyan Yu, Jiani Ji, Endi Song in Cancer Control
Footnotes
Author Contributions
Each author contributed significantly to the conception and development of the present paper. E.S. designed the research process. M.Q. searched the database for corresponding articles and extracted useful information from the articles above. J.J., T.J. and X.Y. used statistical software for analysis. F.X. and T.J. drafted the meta-analysis. All authors have read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets supporting the conclusions of this article are included within the article itself. For further details, please contact the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
