Abstract
Introduction
Colorectal cancer (CRC) screening is an important strategy to reduce morbidity and mortality of cancer. However, evidence on CRC screening and outcomes among Métis people is limited and results are often conflicting.
Methods
This retrospective study examined CRC screening participation and retention rates, abnormal fecal test results, follow-up colonoscopy rates and wait times, and invasive CRC detection rates and distribution according to Métis status (Métis, non-Métis) and sex (male, female) from 2014 to 2022 among adults living in Alberta. Multiple administrative health databases were linked to investigate study outcomes. Adults aged 50 to 74 years who were eligible for CRC screening were included. Chi-square tests of independence and
Results
CRC screening participation rates among Métis males and females remained slightly higher than, or similar to, their non-Métis counterparts. However, retention rates among Métis people were lower compared to non
Conclusion
Findings from this study highlight the need for ongoing collaboration among Indigenous leaders, researchers, and healthcare services to support ongoing participation in CRC screening among Métis people.
Plain language summary
Keywords
Introduction
In Canada, colorectal cancer (CRC) is projected to be the fourth most diagnosed cancer (excluding non-melanoma skin cancers), affecting an estimated 25 200 individuals in 2024. 1 CRC is the second and third leading cause of cancer-related death in males and females, respectively. 1 While CRC incidence remains high, CRC incidence and mortality have significantly declined since the introduction of CRC screening in Canada in 2007.2,3 Screening can detect precancerous polyps and early-stage cancer, increasing the treatment options available and their effectiveness, thus reducing CRC incidence, morbidity, and mortality.4-6 Toward Optimized Practice (TOP) Clinical Practice Guidelines recommends CRC screening in adults aged 50 to 74 years through fecal immunochemical tests (FIT) every one to two years or sigmoidoscopy every 10 years. 7 Despite publicly funded screening programs across most of Canada, 8 socioeconomic inequities in CRC screening remain, 9 particularly among Indigenous people.10,11
The Canadian Constitution recognizes three Indigenous groups, including First Nations, Inuit, and Métis people. 12 Métis people trace their roots to the unions of First Nations people and European fur traders in the 18th century and share a unique culture, identity, and self-governance system.13-15 There is currently a lack of Métis-specific cancer data, 16 limiting understanding of CRC screening participation and outcomes. The underrepresentation of Métis people in research is in part due to a lack of Métis or other Indigenous identifiers in administrative health databases. While some Canadian studies have identified inequities in CRC screening participation among Métis people, 17 others have shown no difference in screening rates between Métis and non-Indigenous people.18,19 Likewise, evidence on CRC incidence among Métis people is varied. One Canadian study showed similar CRC incidence among Métis and non-Indigenous males, while CRC incidence among Métis females was lower than their non-Indigenous counterparts. 20 Conversely, Canadian studies from Alberta, Ontario, and Manitoba found no significant differences in CRC incidence between Métis people and their non-Métis counterparts.21-23
Most previous studies are limited by the small number of cancer cases diagnosed among Métis people and the limited ability to identify all Métis in the population.21-23 Moreover, these studies were conducted nearly a decade ago and, thus, may not reflect current CRC incidence among Métis people. Finally, some studies used self-reported screening data, which are subject to overreporting due to social desirability and recall bias.24,25 There is also a lack of evidence on sex-specific CRC screening rates among Métis people. Studies have shown that females are more likely to undergo CRC screening than males9,18,26,27; however, evidence remains unclear.28,29 In a qualitative study conducted in Ontario, Canada, females reported barriers to CRC screening such as bodily intrusion, anxiety regarding bowel perforation (eg, from colonoscopy), and embarrassment. On the other hand, barriers reported by males included procrastination with underlying fatalism, belief that screening was unnecessary or doubt that screening could reduce or prevent CRC, and being in a position of vulnerability. 30 Given the sex-specific differences in CRC screening uptake and perceptions, CRC screening rates and outcomes among Métis males and females should be considered separately to develop CRC screening interventions.
Canada has committed to the Truth and Reconciliation Commission for identifying and reducing health inequities among Indigenous people, 31 yet there is a lack of adequate and accessible data on the Métis population, including data on CRC screening and outcomes.16,31 Consequently, there may be unmet CRC screening needs within the Métis population. This study represents a collaboration between the Alberta CRC Screening Program and the Otipemisiwak Métis Government of the Métis Nation within Alberta (MNA). Using a system-level approach to screening program data, the current study examined the following outcomes according to Métis status (Métis, non-Métis) and sex (male, female): (1) CRC screening participation and retention rates, (2) the proportion of individuals with an abnormal FIT result (positivity rate), (3) the proportion of individuals who underwent a follow-up colonoscopy after an abnormal FIT result, (4) the wait time for a follow-up colonoscopy, and (5) the rates of invasive CRC detection and stage distribution.
Methods
Study Design and Population Sample
This retrospective study included data from adults living in Alberta aged 50 to 74 years from 2014 to 2022, as per the Alberta CRC screening guidelines in effect during this time. 7 Screening data were obtained from the Alberta Colorectal Cancer Screening Program (ACRCSP) database. The ACRCSP database contains dates and lab results for individual-level fecal tests (FIT from 2014 to present). Diagnostic and screening colonoscopy data were obtained from the Discharge Abstract Database, National Ambulatory Care Reporting Systems databases, and Alberta Practitioner Claims databases and linked to the ACRCSP database. The ACRCSP database is also routinely linked with the Alberta Cancer Registry (ACR), Vital Statistics, and the Alberta Health Person Directory (PD). Data obtained from the ACR included cancer stage, method of diagnosis, and date and facility of diagnosis. The PD database includes all Alberta residents registered for healthcare and was used to identify adults eligible for screening according to age. Provincial Healthcare Numbers (PHN) were also obtained from the PD database for deterministic linkage. Individuals who were previously diagnosed with invasive CRC or deceased were ineligible and, thus, excluded from the study. Quality assurance measures are routinely run on the various data sources received by the ACRCSP database as well as regular checks and reconciliation of each data load. Algorithms used by the Screening Program analysts to create the screening indicators were not available to the study team.
The MNA is the governing body for Métis people in Alberta, and advocates for the rights and self-determination of Métis Albertans. 32 Additionally, the MNA develops policies and implements programming to advance the socioeconomic and cultural well-being of its citizens. 32 The MNA collects, uses and discloses identifying Métis data to advance the health and wellness priorities of MNA Citizens through data governance agreements, including the agreement established with Alberta Health Services Screening Programs for the current study. Alberta Health generated PHN identifiers using the names and contact information of MNA Citizens obtained from the MNA Identification Registry. ACRCSP analysts then used the PHN identifiers to create Métis and non- Métis subpopulations. The various databases linked for this study are shown in Supplemental Figure 1. The MNA Identification Registry included more than 63 000 Citizens in 2023 and is the only registry in Alberta that recognizes people as Métis according to the National Definition.33,34
Study Outcomes
The current study used screening indicators and definitions based on the Canadian Partnership Against Cancer (CPAC) benchmarks for CRC screening program evaluation metrics (Supplemental Table 1). 35 The CRC screening program indicators for eligible individuals included participation rates (completed a fecal screening test within the measurement timeframe), retention rates (completed a subsequent CRC screening test within a specified timeframe), CRC screening test results (abnormal test results), wait times for follow-up diagnostic tests (when 90% of tests are completed), and CRC outcomes (invasive cancer detection rates and stage distribution). Participation and retention rates and abnormal test results (positivity rates) are reported as percentages as per the CPAC definitions. ACRCSP analysts generated the screening indicator values for each subpopulation by year for males and females, separately by Métis status and provided these aggregate values to the study team for analysis.
Research Ethics and Participant Consent
This project adhered to both Western and Métis ethical research practices. These included adopting the ethical principles and practices in alignment with the Tri-Council Policy Statement: 2 (2022), Chapter 9: Research Involving the First Nations, Inuit and Métis Peoples of Canada 36 and the Six Principles of Ethical Métis Research.36,37 Non-Métis project team members also completed the First Nations ownership, control, access, and possession (OCAP®) training. Research ethics approval (HREBA.CC-21-0145) for this study was granted from the Health Research Ethics Board of Alberta—Cancer Committee on September 13, 2021. Consent for this project for the Métis people was through our partnership with Otipemisiwak Métis Government. For non-Métis people, existing administrative data was accessed and aggregated. Screening participation implies consent as obtaining individual consent was deemed to be impractical, unreasonable, or not feasible to obtain, so consent was waived. The RECORD guidelines have been followed in this study. 38
Statistical Analyses
Descriptive and analytical statistical methods compared screening indicators between Métis and non-Métis people and between males and females within each subpopulation. These methods included chi-square tests of independence (stage distribution), and
Results
The number of females and males eligible for colorectal cancer screening (target population) since 2014 for the three age groups by Métis status is shown in Supplemental Table 2. The percentage of the Métis population in the total target population increases over the study time period in all age groups but is always substantially smaller than the non-Métis population. The percentage of the Métis population in the total target population decreases with increasing age group, confirming the importance of age standardization and the noted small sample sizes for the 70- to 74-year-olds.
Métis versus Non-Métis Females
The age-standardized participation rates among Métis females averaged over 2014 to 2022 were marginally higher than among non-Métis females from 2014 to 2022 (2.09%, 95% confidence interval (CI) = (−0.54%, 4.71%)). The APC in the age-standardized participation rates among Métis females was −1.14 ( (A) Participation Rates Among Females From 2014 to 2022 Inclusive by Age Group, (B) Retention Rates Among Females From 2014 to 2020 Inclusive by Age Group, (C) Abnormal FIT Results (Positivity Rates) of the Entry-Level Screening Test Among Females From 2014 to 2022 Inclusive by Age Group. The All-Ages Panel Represents Age-Standardized Rates (ASR). MN, Métis; Non-MN, Non-Métis
Métis vs Non-Métis Males
Similar patterns were observed when comparing Métis males and non-Métis males. The age-standardized participation rates were slightly higher among Métis males averaged over 2014 to 2022 than non-Métis males in all age groups (Figure 2A, 2.62%, 95% CI = (−0.47%, 5.71%)). The APC of the age-standardized participation rates for Métis males was −1.85 ( (A) Participation Rates Among Males From 2014 to 2022 Inclusive by Age Group, (B) Retention Rates Among Males From 2014 to 2020 Inclusive by Age Group, and (C) Abnormal FIT Results (Positivity Rates) of Entry-Level Screening Tests for Males From 2014 to 2022 Inclusive by Age Group. All Ages Panel Represents Age-Standardized Rates for Males. MN, Métis; Non-MN, Non-Métis.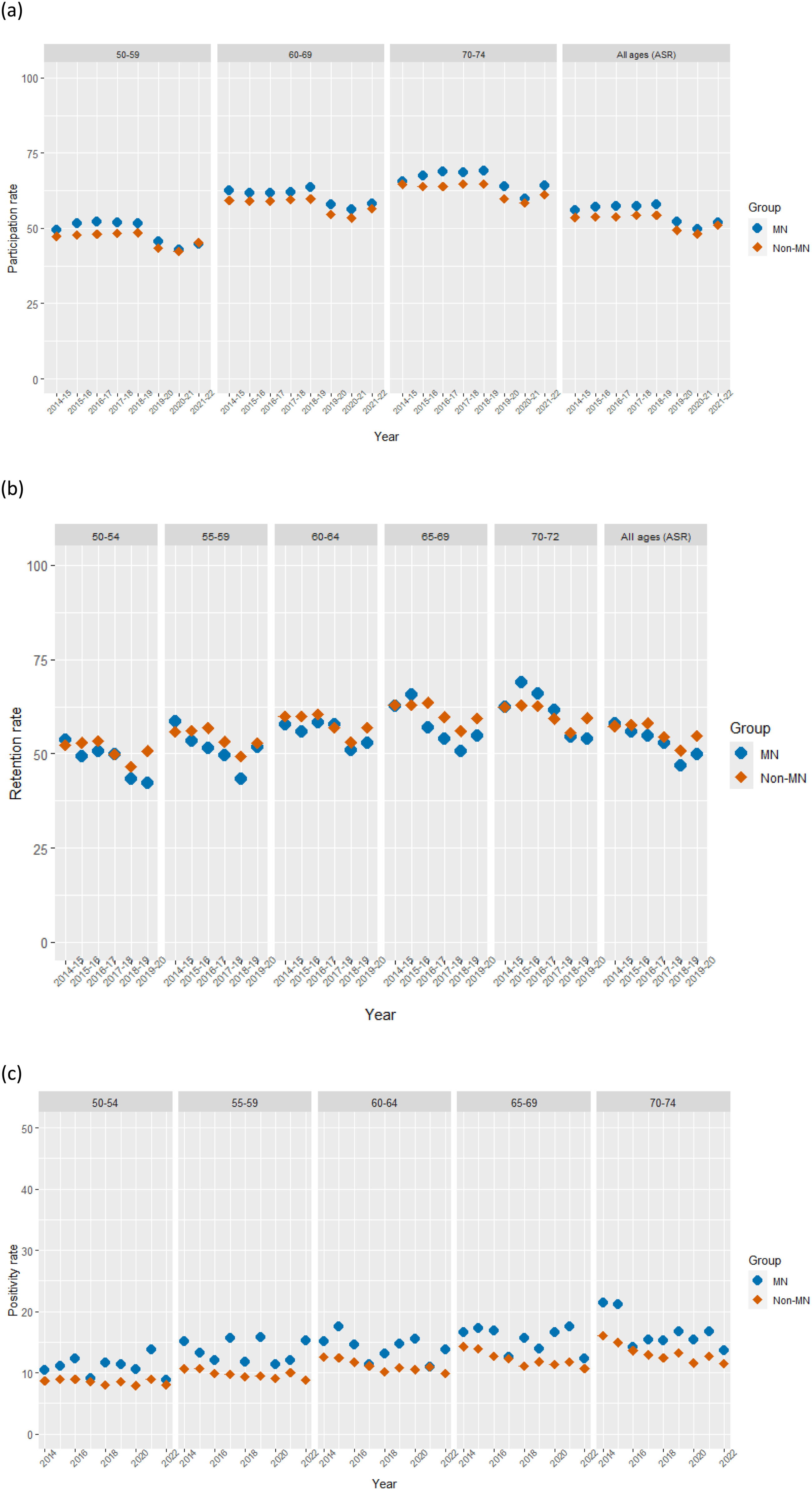
Similar increasing and decreasing patterns were found in Métis and non-Métis males as with females in the follow-up colonoscopy test rates following an abnormal FIT result (Supplemental Figure 4). The average difference between Métis males and non-Métis males for all the age groups and over all years combined was non-significant −2.65%. All test rates were well below the national benchmark of 85% within 180 days 39 for both groups of males, except for Métis males aged 70-74 years in 2017. Supplemental Figure 5 shows the 90th percentile wait times in days from an abnormal FIT result to a follow-up colonoscopy, which ranged from 88 to 180 days for Métis males and from 124 to 154 days for non-Métis males. Similar to females, these results are substantially higher than the national standard of 90% within 60 days of an abnormal fecal test result for a follow-up colonoscopy. 35
Métis Versus Non-Métis, Males and Females Combined
Average Invasive Colorectal Cancer Detection Rate per 1000 People (95% Confidence Interval) From 2014 to 2021 Among Métis and Non-Métis People Aged 50 to 74 Years

Abbreviations: CRC, colorectal cancer; FIT, fecal immunochemical test.
Average Detected Invasive Colorectal Cancer Stage From 2014 to 2021 Among Métis and Non-Métis People

*number suppressed.
Males vs females by Métis status groups
Within the Métis population, the average age-standardized participation rate among females was slightly higher than among males (2.22% higher, 95% CI = (−0.81%, 5.25%)). However, the age-standardized retention rates among males were slightly higher than among females averaged over time (3.29% higher, 95% CI = (−1.72, 8.32)). There were significant downward trends for both participation (APC males = −2.0, Abnormal FIT Results (Positivity Rates) of Entry-Level Screening Test From 2014 to 2022 Inclusive by Age Group Comparing Métis Males to Métis Females (F, Females; M, Males)
In the non-Métis population, the age-standardized participation rate was similar to that of Métis people, whereby females had slightly higher average participation rates than males (2.76% higher, 95% CI = (0.053%, 5.46%)). However, the age-standardized retention rates were comparable between non-Métis males and females (−0.07% difference, 95% CI = (−3.39%, 3.54%)). Both males and females had significant downward APC trends for participation rates (APC males = −1.4, Abnormal FIT Results (Positivity Rates) of Entry-Level Screening Tests From 2014 to 2022 Inclusive by Age Group Comparing Non-Métis Males to Non-Métis Females (F, Females; M, Males)
Discussion
CRC screening participation rates among Métis females and males remained slightly higher than, or similar to, their non-Métis counterparts throughout the study period. Among Métis and non-Métis people, CRC screening rates were higher among females than males, except for those aged 70-74 years, where participation was slightly higher among males. Upon examining trends, CRC screening participation significantly decreased among Métis males and non-Métis males and females, notably from 2019 to 2021, which was likely due to COVID-19 public health measures that paused CRC screening.
40
CRC screening retention rates among Métis females and males were lower compared to their non
Contrary to our findings, most previous Canadian studies showed lower CRC screening participation rates among Métis people compared to non-Métis people; however, these differences were non-significant. For instance, aggregated data from the Canadian Community Health Survey indicated higher rates of CRC underscreening among Métis people living in Ontario compared to their non-Indigenous counterparts.18,19 Similarly, in Calgary, Alberta, FIT screening rates among First Nations and Métis people were lower than their non-First Nations and non-Métis counterparts, respectively.
17
In a recent study, we also found significantly lower CRC screening rates among First Nations people in Alberta compared to non-First Nations people.
41
Therefore, the higher screening rates of Métis people found in the current study may be partly attributable to strategies implemented specifically for Métis people. For instance, the MNA developed
The Alberta CRC Screening Program, in collaboration with Indigenous representatives, co-developed strategies to enhance CRC screening among Indigenous people, such as providing screening education and developing culturally safe resources. 47 Alberta Health Services (AHS) Screening Programs also implemented strategies to improve screening awareness and participation among residents of rural and remote communities in Alberta, including Indigenous populations. 47 Notably, in 2020, the Integrated Access to Cancer Screening (IACS) pilot initiative integrated colorectal and cervical cancer screening with a mobile mammography service known as Screen Test, 48 which offers breast screening to women in 120 rural communities across Alberta.49,50 Results showed over 90% of eligible adults (n = 653) attended IACS clinics, with nearly half (n = 317) eligible for CRC screening. 48 The IACS initiative effectively increased CRC screening uptake, where over half of patients (n = 178) were overdue and 37% (n = 96) had never been screened. 48 However, because the IACS initiative was integrated with Screen Test, uptake was primarily among female patients (99% of patients), even though CRC screening was also available for males.
Findings from the current study showed slightly higher CRC screening participation rates among females than males; however, these differences were not significant. While some studies conducted in Alberta found lower 17 or similar28,29 CRC screening among females, studies conducted in Canada9,18 and the United Kingdom 26 found higher screening rates among females. Similarly, a meta-analysis found higher screening uptake among females in European studies, while uptake was similar for males and females in studies conducted in Asia. 27 These differences in CRC screening participation by sex, race/ethnicity, and geographic location point to the importance of considering social and cultural factors to promote and support screening among different population groups.9,27,29
Despite similar screening rates among Métis and non-Métis people, retention rates were lower among Métis males and females, though the difference was only significant among Métis females. Métis males had slightly higher retention rates than Métis females for most years; however, this gap closed over time. These findings indicate that Métis people are less likely to return for CRC screening following a normal screening result. Previous Canadian studies have shown that approximately half of Métis people are overdue or in need of CRC screening; although, these rates did not significantly differ from non-Indigenous people.18,19 Métis and other Indigenous populations experience significant barriers to accessing cancer screening and care, many of which are the result of the ongoing and lasting impacts of colonialism, a potent social determinant of Indigenous health.51,52 Mistrust in healthcare providers, experiences of racism and discrimination and lack of cultural competency among healthcare providers are among the many barriers that may hinder regular CRC screening participation among Métis people.51,52 Socioeconomic factors such as poverty and geographic isolation further impede access to cancer screening services (eg, lack of transportation and challenges in taking time away from work).51,53 Ongoing efforts are needed to develop interventions and leverage existing programs, resources, and partnerships to improve access to and ongoing participation in CRC screening among Métis and other Indigenous people.46,51
The current study showed that a significantly higher proportion of Métis males had positive FIT results than non-Métis males. A higher proportion of Métis females also had positive FIT results than their non-Métis counterparts; however, this difference was non-significant. Sensitivity analyses that omitted the 2014 – 2015 data found the average differences between the groups were marginally lower (results not shown). A recent investigation similarly showed higher rates of abnormal fecal tests in First Nations people compared to non-First Nations people.
41
The current study also found that slightly fewer Métis males underwent a follow-up colonoscopy within the recommended time frame than non-Métis males, although this gap narrowed or closed in recent years. The follow-up colonoscopy rate among Métis females remained similar to, or higher than that of non-Métis females overall; however, there was a large variation from year to year, given the small sample size. The 90th percentile wait time in days from an abnormal fecal test result to a follow-up colonoscopy among Métis and non
Finally, no significant differences in invasive CRC detection rate or stage at diagnosis were detected through screening between Métis and non
Some limitations should be considered. First, this study includes only registered citizens of the MNA. Therefore, the sociodemographic characteristics of MNA Citizens may differ from other Métis people living in Alberta. However, nearly half of Métis people in Alberta are registered with the MNA and, thus findings should be generalizable to most Métis people in Alberta. Second, the smaller number of screen-eligible Métis people and CRC cases resulted in wide confidence intervals for most estimates and should be interpreted accordingly. Third, factors such as the type of FIT used and differences in screening program features (eg, FIT testing frequency and abnormal FIT thresholds) across Canada may affect positivity rates, as well as CRC (and adenoma) detection rates.60,61 Notably, the abnormal FIT threshold in Alberta (75 ng/ml) is lower than that in most other provinces or territories in Canada (ranging from 100 to 150 ng/ml). 61 Therefore, it is possible that the between-group differences in abnormal FIT results identified in this study could be smaller under higher thresholds.60,61 Finally, the current study was limited to age and sex as covariates, and thus, we were unable to conduct analyses that examined other pertinent socioeconomic and health-related factors such as income and geographic location.
Despite these limitations, the current study is the first to examine CRC screening and outcomes among Métis and non-Métis males and females in Alberta using the MNA Identification Registry linked to administrative health data in Alberta. Data linkage provides consistent and reliable data that can be used to inform equitable, appropriate, and accessible cancer control strategies for Métis people. 16 Notably, access to and use of Métis-specific data was made possible through the collaboration and partnership between the MNA, Alberta Health and Alberta Health Services Screening Programs. This partnership also ensured that the information collected aligned with the MNA’s research goals and mandate to improve the health and well-being of Métis people in Alberta.
Conclusion
The current study showed similar or higher CRC screening rates among Métis males and females living in Alberta, Canada compared to their non
Supplemental Material
Supplemental Material - Colorectal Cancer Screening Among Métis and Non-Métis Males and Females in Alberta, Canada
Supplemental Material for Colorectal Cancer Screening Among Métis and Non-Métis Males and Females in Alberta, Canada by Michelle L. Aktary, Reagan Bartel, Momtafin Khan, Chinmoy Roy Rahul, Bonnie Chiang, James Newsome, June Kima, Ashton James, Amanda Andrew, Angeline Letendre, Karen A. Kopciuk, Huiming Yang in Journal of Cancer Control.
Footnotes
Acknowledgments
Project leaders are grateful to the Otipemisiwak Métis Government staff for their support of this project. From AHS Screening Programs, Monica Schwann (Director, Screening Programs), Linan Xu (Lead, Analysts) and Jin Chen and Yi Guo (statisticians for colorectal cancer screening data), and Melissa Potestio from the Department of Community Health Services, University of Calgary all contributed substantially to our project.
Ethical Considerations
Research ethics approval (HREBA.CC-21-0145) for this study was granted from the Health Research Ethics Board of Alberta—Cancer Committee on September 13, 2021.
Author Contributions
Conceptualization, A.L., R.B., A.A., H.Y. and K.K.; methodology, A.L., R.B., B.C., A.A., J.K., J.N., A.M.A., H.Y. and K.K.; software, C.R.; formal analysis, C.R., and K.K.; data curation, C.R., K.K.; writing—original draft preparation, M.A., M.K. and K.K.; writing—review and editing, all authors; visualization, C.R.; supervision, R.B., A.A, H.Y. and K.K.; project administration, K.K.; funding acquisition, A.L., R.B., A.A., H.Y. and K.K. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes for Health Research (Knowledge Holders Angeline Letendre and Reagan Bartel and Primary Investigators Karen Kopciuk and Huiming Yang) under grant number PGG-175360. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Due to the nature of the research, including the adoption of the Principles of Ethical Métis Research and the CARE Principles for Indigenous Data Governance, supporting data are not available.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
