Abstract
Introduction
Predicting prognosis before treatment in rectal cancer patients undergoing neoadjuvant therapy is essential for effective treatment strategy selection. This study evaluates the prognostic impact of the pretreatment pan-immune-inflammation value (PIV) on pathological response, local control, and overall survival.
Methods
We included 126 patients who received neoadjuvant treatment for rectal cancer from November 2020 to May 2024. This study was conducted retrospectively. PIV was calculated as (neutrophil count × platelet count × monocyte count)/lymphocyte count, with an optimal threshold identified at 475. Patients were categorized into low (PIV < 475) and high (PIV ≥ 475) groups. Pathological responses were analyzed using the chi-square test, while survival rates were assessed with the Kaplan-Meier method and log-rank tests. Cox regression analysis examined the influence of variables on outcomes.
Results
The high PIV group had a significantly higher prevalence of N2 disease (33.3% vs 13.7%, P = .02) and a greater incidence of abdominoperineal resection (35.9% vs 17.2%, P = .02). Pathological response was notably higher in the low PIV group (75% vs 33.3%, P < .001). Overall survival and local recurrence-free survival rates were significantly better in the low PIV group (P = .006 and P = .004, respectively). In multivariate analysis, PIV was the only significant factor affecting overall and local recurrence-free survival.
Conclusion
Higher pretreatment PIV values correlate with poorer pathological response and reduced overall and local recurrence-free survival in rectal cancer patients. These findings support the use of PIV for risk stratification and treatment personalization.
Introduction
Rectal cancer is among the most aggressive forms of cancer worldwide. It is estimated that in 2024, there will be 152 810 new diagnoses of colorectal cancer in the USA, resulting in 53 010 deaths. 1 The standard treatment for patients with ≥T3 or N+ disease is a multimodal approach that includes neoadjuvant therapy (either a short course of radiotherapy or a long course of chemoradiotherapy) followed by surgery.2,3 Recent studies have indicated that non-surgical follow-up, known as the “watch and wait” strategy, can be effectively implemented after total neoadjuvant treatment (TNT) in selected patients—particularly those who achieve a pathological complete response.4,5 Therefore, it is crucial to identify prognostic markers that can predict post-treatment recurrence and overall survival and help select candidates for the watch-and-wait approach.
Multiple studies have shown that the Systemic Index of Inflammation (SII), Neutrophil-to-Lymphocyte Ratio (NLR), Platelet-to-Lymphocyte Ratio (PLR), and Monocyte-to-Lymphocyte Ratio (MLR) can all serve as predictors of prognosis in cases of locally advanced rectal cancer.6-8 In 2020, a new inflammatory biomarker called the Pan-Immune-Inflammation Value (PIV) was introduced. Fuga et al defined PIV as: (neutrophil count × platelet count × monocyte count) /lymphocyte count. This trial investigated the prognostic significance of PIV in metastatic colorectal cancer patients who received chemotherapy. The findings indicated that patients with a high PIV had worse progression-free survival (PFS) and overall survival (OS) compared to those with a low PIV. 9 Similarly, Li et al. 10 Have demonstrated that elevated PIV is associated with poor survival outcomes in non-metastatic colorectal cancer patients following surgery. Numerous studies have been conducted to examine the prognostic significance of PIV in various cancer types, including breast cancer,11,12 esophageal cancer, 13 malignant melanoma, 14 and lung cancer.15,16
This study aims to evaluate the impact of PIV on pathological response, local control, and overall survival in rectal cancer patients undergoing neoadjuvant therapy. Identifying a marker that can be analyzed before treatment may help in selecting patients who are likely to benefit from a watch-and-wait approach.
Methods
Patients
This retrospective study analyzed patients with rectal adenocarcinoma, aged 18 to 90 years, an ECOG performance score of 0-2, and no history of immune disorders. To minimize selection bias, all patients meeting these specified criteria were included in the study. Patients with known immune disorders were excluded, and in addition, patients with elevated C-reactive protein (CRP) or neutrophil levels were also excluded from the analysis. These patients received neoadjuvant treatment between November 2020 and May 2024, using short-course radiotherapy, long-course chemoradiotherapy, or total neoadjuvant treatment. One hundred sixty seven patients were initially reviewed, and 126 patients who met the eligibility criteria were included in the study. Standard evaluations included pelvic MRI and FDG PET-CT scans prior to treatment. The study was approved by the Ethics Committee of Gulhane Training and Research Hospital (Approval No: 2025/129, Date: 12 June 2025). The reporting of this study conforms to STROBE guidelines. 17 All patient data were fully de-identified prior to analysis to ensure that no individual could be identified in any way, in accordance with ethical standards and data protection regulations.
Neoadjuvant Treatment Protocols
Patients received neoadjuvant treatment prior to surgery, with two main schedules identified: long-course chemoradiotherapy (50.4 Gy in 28 fractions or 50 Gy in 25 fractions with 825 mg/m2 capecitabine) and short-course radiotherapy (25 Gy in 5 fractions). Most patients also received consolidation or induction chemotherapy as total neoadjuvant treatment became standard during the study period. All patients then underwent surgery.
Pan-Immune-Inflammation Value
Pre-treatment PIV was calculated using the formula: (neutrophil count × platelet count × monocyte count) / lymphocyte count.
Follow-Up
Patients were observed in accordance with the institutional protocols following surgery. Physical examinations and laboratory assessments were conducted every 3 months during the first two years, followed by 6-month intervals thereafter. Imaging studies, including contrast-enhanced pelvic MRI or CT scans, were performed every 6 months or sooner if indicated by symptoms or abnormal findings.
Pathological Assessment and Outcomes
The study aims to identify the correlation between pre-radiotherapy PIV and local control, as well as to evaluate its relationship with pathological response and OS. Pathological complete response (pCR) indicates no residual tumor post-surgery, while OS is the time from diagnosis to death or last follow-up. Local recurrence-free survival (LRFS) measures the time from diagnosis to the first local recurrence or last follow-up.
Statistics
A statistical analysis was conducted using IBM SPSS 27 to determine the optimal cutoff value for pre-radiotherapy PIV distinguishing low and high PIV groups, utilizing ROC analysis. Patient characteristics were assessed with independent samples t-tests and chi-squared tests. The relationship between pre-radiotherapy PIV, pathological response, and local control was explored using chi-squared tests, while OS and LRFS were analyzed with Kaplan-Meier curves and log-rank tests. Finally, Cox regression analysis was performed for multivariate analysis.
All tests were performed with a 95% confidence interval, and a P-value of less than .05 was considered statistically significant.
Results
Patients Characteristics

PIV: pan-immune inflammation value; ECOG: eastern cooperative oncology group. Statistical significance at p < 0.05 are marked in bold.
The optimal pre-radiotherapy PIV cutoff, determined via ROC analysis, was 475 (AUC: 0.75, sensitivity: 0.80, specificity: 0.74, Youden index: 0.54). This value correlated significantly with local recurrence, leading to classification into low (PIV < 475) and high (PIV ≥ 475) groups. Only the N stage showed a significant difference, with the high PIV group exhibiting a higher prevalence of N2 disease (33.3% vs 13.7%, P = .02; see Table 1).
Treatment Characteristics

PIV: pan-immune inflammation value; TNT: total neoadjuvant treatment; ChRT: chemoradiotherapy; RT: radiotherapy. Statistical significance at p < 0.05 are marked in bold.
PIV and Pathological Response
Univariate and Multivariate Analyses of Pathological Response

ECOG: eastern cooperative oncology group; TNT: total neoadjuvant treatment; Cht: chemotherapy; LAR: low anterior resection; APR: abdominoperineal resection; RT: radiotherapy; PIV: pan-immune inflammation value. Statistical significance at p < 0.05 are marked in bold.
PIV and Survival Outcomes
A median follow-up period of 17 months (ranging from 4 to 48 months) was observed in the patient cohort. Of the patients, 80.2% were alive without disease, 9.5% had died, and 10.3% were alive with disease.
Figure 1 shows the relationship between PIV and survival outcomes. The cohort did not reach median OS, distant metastasis-free survival (DMFS), or LRFS times. The median LRFS times were not reached in either the low or high PIV groups, though local recurrence was significantly higher in the high PIV group (20.5% vs 2.3%, P = .001). In univariate analysis, consolidation chemotherapy was significant for LRFS (P = .04), but not in multivariate analysis (P = .08). In multivariate analysis, only PIV was a significant predictor for LRFS (P = .004, Table 4). The Relationship Between PIV and Survival Outcomes Univariate and Multivariate Analyses of LRFS LRFS: local-recurrence free survival; ECOG: eastern cooperative oncology group; TNT: total neoadjuvant treatment; Cht: chemotherapy; LAR: low anterior resection; APR: abdominoperineal resection; RT: radiotherapy; PIV: pan-immune inflammation value. Statistical significance at p < 0.05 are marked in bold.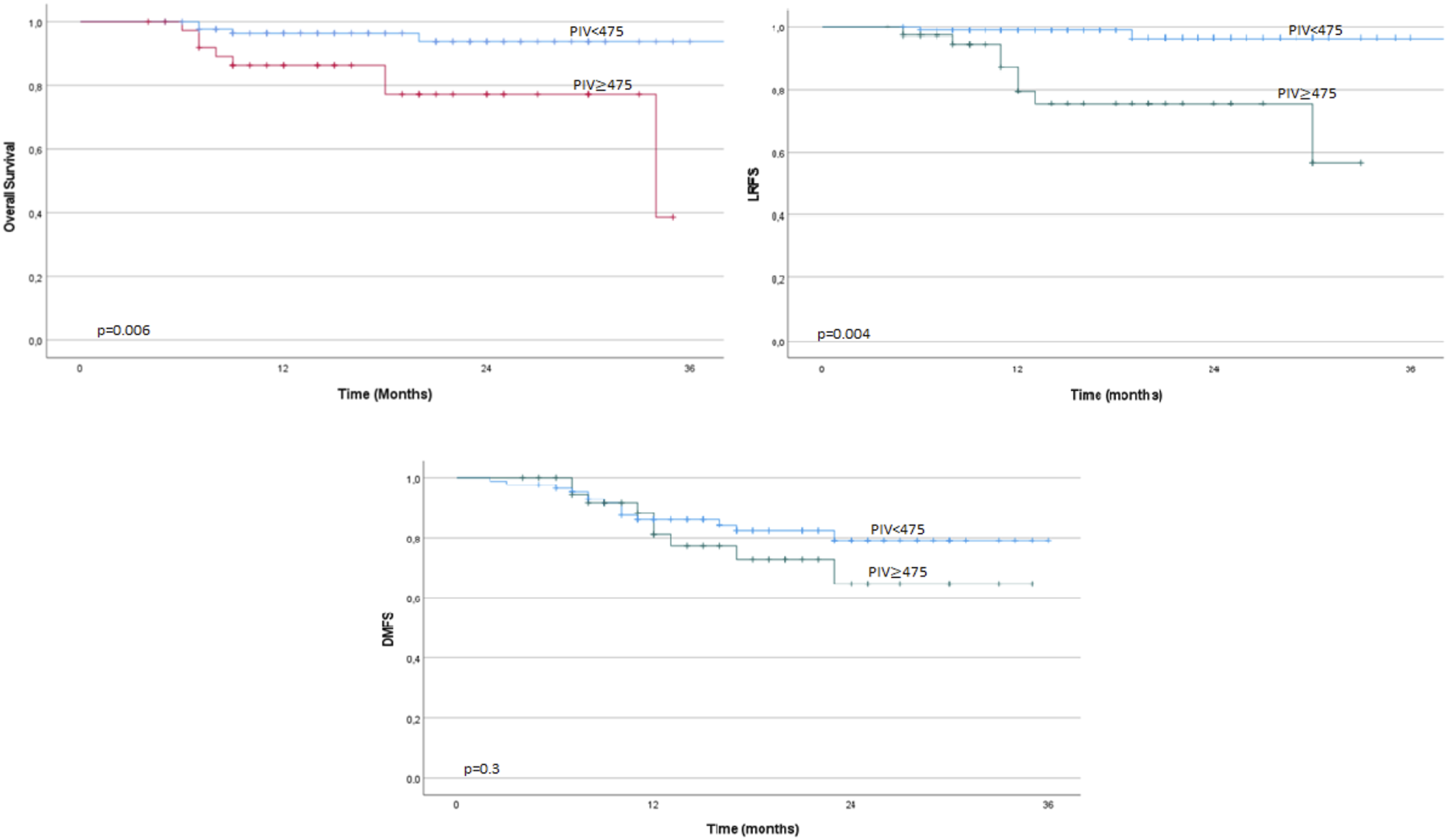

Univariate and Multivariate Analyses of Overall Survival

ECOG: eastern cooperative oncology group; TNT: total neoadjuvant treatment; Cht: chemotherapy; LAR: low anterior resection; APR: abdominoperineal resection; RT: radiotherapy; PIV: pan-immune inflammation value. Statistical significance at p<0.05 are marked in bold.
Although a significant correlation was found between the LRFS and OS rates and pre-radiotherapy PIV, no significant relationship was identified between PIV and DMFS (P = .30).
In addition to the primary analysis, subgroup and interaction analyses were conducted to evaluate whether the prognostic value of PIV differed by treatment type. In subgroup analysis, high PIV was found to be associated with inferior OS, both in patients who received TNT (HR: 3.49, P = .10) and in those who did not (HR: 6.21, P = .10). While these associations did not reach statistical significance-likely due to limited sample size, the direction and magnitude of these effects support the consistency of PIV’s prognostic value. Furthermore, in interaction analysis, the observed interaction was not found to be statistically significant (P = .76), thereby suggesting a consistent adverse prognostic impact of high PIV, irrespective of the treatment approach. These findings support the hypothesis that PIV is a robust and generalizable prognostic biomarker irrespective of treatment strategy.
Discussion
In 2020, Fuca et al 9 demonstrated that the PIV could serve as a novel index for predicting outcomes in patients with metastatic colorectal cancer. Since then, the relationship between PIV and prognosis has been explored across various cancer types.12,18-20 However, there is limited research examining the connection between PIV and prognosis, specifically in patients with rectal cancer undergoing neoadjuvant treatment. 21
This study evaluates the impact of PIV on outcomes for rectal cancer patients receiving neoadjuvant therapy. Results show a significantly higher pathological response in patients with PIV < 475 compared to PIV ≥ 475. Additionally, local control, LRFS, and OS were better in the low PIV group. No significant predictors for outcomes were found other than PIV levels before radiotherapy, indicating that PIV is a key predictive factor.
The discovery of leukocytes in tumor microenvironments (TME) in the 19th century sparked a growing body of evidence linking inflammation and cancer.22-24 Within the TME, platelets and monocytes promote a neo-angiogenic pro-tumor effect through neutrophils, while lymphocytes exhibit anti-tumor activity against inflammation.22,25,26 Therefore, it is not surprising that biomarkers such as the neutrophil/lymphocyte ratio (NLR) and the platelet/lymphocyte ratio (PLR) are effective predictors of patient outcomes. Numerous studies have evaluated the prognostic significance of these indices.27-30 Several investigations into these immune-inflammatory markers have focused specifically on patients with rectal cancer. A meta-analysis of 47 trials involving rectal cancer patients found that poor overall survival was significantly associated with high NLR, high PLR, and low lymphocyte/monocyte ratio (LMR). High NLR and low LMR were also linked to worse disease-free survival. 30 This meta-analysis included patients who underwent surgery for rectal cancer, regardless of whether they received neoadjuvant therapy.
Another meta-analysis examined patients with rectal cancer who underwent neoadjuvant chemoradiation followed by resection. This study documented a significant negative association between NLR and OS and disease-free survival (DFS). 31 However, these indices rely on only two immune-inflammatory cell counts. This led to the development of a new PIV index, which incorporates platelets, neutrophils, monocytes, and lymphocytes. 9
In a trial evaluating the PIV in 438 metastatic colorectal cancer patients, those with elevated PIV levels had poorer progression-free survival (PFS) of 9.5 months compared to 12.5 months for lower PIV levels. Median OS was also lower at 21.6 months for high PIV vs 34.4 months for low PIV (P < .001). 9 A further study was conducted in order to investigate the prognostic value of the pre-operative PIV in 650 colorectal patients. The results of this study demonstrated that patients in the low PIV group exhibited significantly better 5-year OS in comparison to the high PIV group. 32
A recent meta-analysis of six studies with 1879 colorectal cancer patients found that high PIV levels were linked to worse OS and PFS outcomes (P < .001). Similarly, our study showed high PIV levels (≥475) correlated with poorer OS and LRFS, while a trend was noted for DMFS that wasn’t statistically significant, likely due to a short follow-up period. 33
Despite the existence of numerous studies examining this subject in patients with colorectal cancer, only one study has evaluated the impact of PIV on outcomes in locally advanced rectal cancer patients who received neoadjuvant chemoradiotherapy. This trial demonstrated that patients with low PIV exhibited significantly higher rates of pCR (P = .029), OS (P = .002), and DFS (P = .001) compared to those with high PIV. 21 It is important to note, however, that the aforementioned trial exclusively included patients who had undergone neoadjuvant long-course chemoradiotherapy. In our study, patients who had undergone TNT or only short-course radiotherapy in addition to long-course chemoradiotherapy were included. However, multivariate analysis of pathological response, LRFS and OS revealed that the choice of neoadjuvant treatment was not statistically significant. In each form of neoadjuvant treatment, a significant correlation was found between pre-radiotherapy PIV and survival outcomes.
The watch and wait approach is a novel non-operative strategy for rectal cancer patients who have developed a pathological complete response following neoadjuvant treatment. 34 However, a debate persists regarding the selection criteria for this approach. The research does not ascertain whether only a pathological complete response is sufficient. Therefore, the identification of candidates for this approach necessitates the development of novel prognostic markers. The utilization of PIV is a viable option for this purpose. This study demonstrated that patients with low PIV exhibited a more favorable prognosis in comparison to those with high PIV. So, patients with low PIV levels may be selected for the watch and wait approach in cases of complete response following neoadjuvant treatment.
This study is the first of its kind in the existing literature to evaluate the pathological response and survival outcomes in patients with rectal cancer who have undergone total neoadjuvant therapy. However, it is important to acknowledge several limitations of the study. First, the follow-up period was relatively short. Although there is a trend observed in DMFS, we believe the lack of statistical significance is primarily due to this brief follow-up. Second, as the study was conducted at a single center, the number of patients was limited. Nonetheless, the patient cohort in this study is comparable to that of other studies in the literature. Thirdly, despite the exclusion of patients with immune disorders, elevated CRP or neutrophil levels in order to minimize the confounding impact of systemic inflammation on PIV, the presence of subclinical or undiagnosed inflammatory conditions could not be entirely ruled out.
Conclusions
In conclusion, our findings suggest that pre-radiotherapy PIV levels are a significant predictor of pathological response, LRFS, and OS in rectal cancer patients who have undergone neoadjuvant treatment. This metric may be useful in selecting candidates for a watch-and-wait strategy, especially for patients showing a complete response after neoadjuvant therapy. However, further research is required to confirm its routine application in clinical practice.
Footnotes
Ethical Approval
The study was approved by the Ethics Committee of Gulhane Training and Research Hospital (Approval No: 2025/129, Date: 12 June 2025). All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration. Since the study was conducted retrospectively, written or oral informed consent was not required.
Author Contributions
All authors contributed to the conceptualization of the study and the methodology. Ismail Kaan Sert led data curation. Aybala Nur Ucgul analyzed and wrote the manuscript. Huseyin Bora critically revised the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Research data are stored in an institutional repository and will be shared upon request to the corresponding author.
