Abstract
Introduction
Cigarette smoking and alcohol drinking are well-known risk factors for various cancers. We aimed to determine a comprehensive profile of cancer risk associated with these lifestyle factors in predominantly low-income Americans.
Methods
We prospectively investigated the associations between cigarette smoking, alcohol drinking, and the risk of twelve cancer types among over 74 000 low-income Black and White adults from the Southern Community Cohort Study in the United States. We used the Cox proportional hazards models to estimate the hazard ratios (HRs) and corresponding 95% confidence intervals (CIs) for these associations.
Results
Compared to never smokers, current smokers had an increased HR for cancers of lung (HR: 14.14, 95% CI: 11.47-17.42), liver and bile duct (HR: 3.19, 95% CI: 2.40-4.25), kidney (HR: 1.47, 95% CI: 1.10-1.96), pancreas (HR: 1.88, 95% CI: 1.41-2.50), oral and pharynx (HR: 3.83, 95% CI: 2.70-5.42), and bladder (HR: 2.81, 95% CI: 1.92-4.11), and a reduced risk of prostate cancer (HR: 0.78, 95% CI: 0.68-0.89) and uterine cancer (HR: 0.45, 95% CI: 0.32-0.63); former smokers also exhibited elevated risks for cancers of lung, liver and bile duct, kidney, and bladder; however, a decreased risk for the lung, liver and bile duct, and bladder cancers was observed with longer durations of smoking cessation, with HRs from 9.71, 2.26, and 2.28 for a duration of <10 years down to 4.28, 1.58, and 1.42 for a duration of 10-19 years, respectively. Compared to never-drinkers, participants who consumed more than 2 drinks per day had increased risks of liver and bile duct cancer (HR: 1.66, 95% CI: 1.29-2.13) and oral and pharynx cancer (HR: 2.15, 95% CI: 1.58-2.91).
Conclusion
Cigarette smoking and alcohol drinking were associated with an increased risk of multiple cancers. Our findings support efforts to control cigarette and alcohol consumption for cancer prevention in low-income U.S. populations.
Introduction
Cancer is a leading cause of global disease burden and mortality. Cigarette smoking and alcohol drinking are well-established risk factors for both incidence and mortality of various types of cancer.1-7 The association of smoking with cancer risk varies by cancer type and differs across racial groups. Smoking is most strongly linked to lung cancer and poses a significantly elevated risk for cancers of the mouth, esophagus, liver, and kidney. In contrast, its association with other cancers is generally weak, inconsistent, or even inverse.1,8-16 Notably, racial/ethnic disparities exist in both the risks and outcomes of smoking-related cancer. For example, African American smokers have a higher risk of lung cancer than white smokers, 17 even when smoking patterns are similar. Factors such as socioeconomic status, access to healthcare, and genetic differences may contribute to these disparities. Additionally, alcohol consumption is linked to an increased risk of several cancers, particularly those of the mouth, esophagus, and liver.2,3 When alcohol is combined with smoking, cancer risk is especially elevated, as the two can act synergistically to increase cellular damage.18,19
Although numerous observational studies have investigated the impact of cigarette smoking and/or alcohol consumption on cancer risk,1,2,11,13,14,20-26 few have simultaneously investigated and compared the associations of smoking and alcohol drinking with the risk of multiple cancers within the same population or cohort. For instance, among 572 studies analyzing the association between alcohol consumption and cancer risk, only four and three studies examined cancers across 5-10 and more than 10 anatomical sites, respectively. 2 Few large-scale prospective cohort studies have been conducted in populations of African ancestry, although this group may be more vulnerable to cancer risk factors, such as cigarette smoking, compared to European or Asian populations, 1 and has higher mortality of cancers, including several smoking-associated cancers. 27
In this study, we evaluated the associations of cigarette smoking and alcohol consumption (drinking) with cancer at 12 primary sites among Black and White American adults enrolled in the Southern Community Cohort Study (SCCS).
Methods
Study Participants and Data Collection
The SCCS is an ongoing prospective cohort study, with the aim to investigate risk factors of chronic diseases and health disparities between Black and White adults in a low-income, low-education population across 12 southeastern U.S. states: Alabama, Arkansas, Florida, Georgia, Kentucky, Louisiana, Mississippi, North Carolina, South Carolina, Tennessee, Virginia, and West Virginia, as described in detail elsewhere.28,29 In brief, More than 85 000 adults, two-thirds Black, between 40 and 79 years of age were enrolled from 2002 to 2009. Approximately 86% of the participants were recruited from community health centers (CHCs) and institutions providing basic health care and preventative services in underserved areas, so the cohort encompassed a substantial number of individuals characterized by low income, low education, and lack of insurance. The remaining 14% of cohort participants were recruited through mail-based general population sampling. Sociodemographic characteristics, lifestyle factors including tobacco exposure and alcohol consumption, and personal medical history were collected at cohort enrollment. Data collection methods varied by recruitment source: CHC participants completed standardized computer-assisted personal interviews, while individuals from the general population responded to self-administered mailed questionnaires. In this study, we excluded individuals who self-identified as non-Black or non-White, those with a prior cancer diagnosis, and those missing information on cigarette smoking or alcohol drinking, resulting in a final analysis of 74 286 participants. The SCCS was reviewed and approved by the institutional review boards (IRB) at Vanderbilt University and Meharry Medical College (#010345). All participants including cancer patients in this study provided written informed consent and have been de-identified.
Measurement of Exposures and Covariates
Participants were asked whether they had smoked at least 100 cigarettes (5 packs of cigarettes) in their entire lifetime and, if they had, whether they still smoked at the time of enrollment in the study. They were also asked the age of smoking onset and the average number of cigarettes they smoked (or used to smoke) in 1 day. Data on age at enrollment, age of smoking initiation, and age at cessation (for former smokers) were available to calculate smoking duration. Pack-years of smoking were calculated by dividing the number of cigarettes smoked per day by 20 to determine the number of packs per day and then multiplying by smoking duration in years. Participants were divided into categories according to smoking status (never, former, and current), pack-years of both current and former smokers (never, <10, 10 to 19, and ≥20), age of smoking initiation (never, <20, 20 to 29, and ≥30), and years since smoking cessation (never, <10, 10 to 19, 20 to 29, and ≥30). The smoking information was also collected at each of the four follow-up surveys conducted in the SCCS.
Alcohol consumption was assessed using the baseline data, which captured detailed intake information for various alcoholic beverages over the past year. These beverages included light beer; regular beer, ale, malt liquor, or stout; white wine or wine coolers; red wine or red wine coolers; liquor or mixed drinks. Participants reported their drinking frequency, in categories ranging from never to two or more times per day, as well as the usual number of drinks per drinking occasion. The intake for each beverage type was determined by multiplying the reported frequency by the usual number of drinks consumed, and the total alcohol consumption was calculated as the sum of daily intake across all beverage categories. Participants were categorized into three groups based on their total alcohol consumption at the baseline: 0, 1 drink, and ≥2 drinks per day.
The baseline variables included in the Cox proportional hazards (CPH) regression models were age, sex, race, educational level, and annual household income. Missing values for education and household income (<0.1% and 1.3% of participants, respectively) were imputed using the sex- and race-specific mode.
Assessment of Cancer
Incident cancer was ascertained via linkage to state cancer registries and was classified according to the primary site and histology categories defined by the Surveillance, Epidemiology, and End Results (SEER) Program. Cancers were classified using the International Classification of Diseases (ICD)-O-3 code version. In this study, we investigated 12 cancers of primary sites, each with more than 175 incidence cases, including lung, colorectum, breast, prostate, liver and bile duct, kidney, pancreas, uterine (including endometrial and other tissues of the uterus), oral and pharynx, bladder, non-Hodgkins lymphoma (Non-Hodgkin), and multiple myeloma (M. Myeloma).
Statistical Analysis
We compared baseline characteristics of participant groups by smoking and alcohol drinking status. For each exposure group, we calculated the proportions of selected categorical demographic variables, including age, sex, race, education, and household income. Follow-up time started at age at baseline survey and ended at the time of cancer diagnosis, death, or the last date of state cancer registry linkage which differed by state (December 31 of the fiscal year of 2016, 2017, 2018, or 2019). We used the Cox proportional hazards model as the primary analysis to estimate the association of smoking and alcohol drinking with cancer risk using age as the time scale. Smoking information was updated with the four follow-up surveys; thus, it was treated as a time-dependent variable in the Cox model analysis. The potential confounders that were adjusted for in the Cox models included age at enrollment, sex, race, education, and household income. We conducted cause-specific Cox regression analysis to account for the competing risk of multiple cancer events. We evaluated the interaction of smoking and alcohol drinking with sex and race, and smoking-alcohol drinking interaction as well.
We estimated the population attributable risk (PAR) for smoking and alcohol using an approach in the context of survival analysis. 30 The PAR represents the percentage of cancer cases that could have been prevented if people avoided smoking or drinking.
To account for multiple testing, an association with a P-value <0.004 (0.05/12 cancers) was considered statistically significant. The reporting of this study conforms to STROBE guidelines. 31
Results
Characteristics of the Study Participants
Baseline Characteristics of the Study Cohort (%).

Cigarette Smoking and Cancer Risk
Adjusted Associations of Smoking Status With Cancer Risk in the SCCS, Overall and by Race a .

aDerived from models treating smoking status as a time-dependent variable, adjusted for sex, age at enrollment, race, education, and household income.
bAccording to smoking status at the baseline.
cPopulation attributable risk (PAR) will be reduced (positive percentage) or increased (negative) if people don’t smoke.
The associations between smoking status and cancer risk observed in the time-dependent CPH models were consistent with those from the baseline CPH models across all study participants (Supplemental Table S2). Therefore, we did not report cancer risk associations for other cigarette smoking-related variables under time-dependent CPH models.
Associations between smoking pack-years, initiation age, or duration of cessation and the risk of 12 types of cancer, both in the combined and within each racial group, are summarized in Supplemental Tables S3-S14. A greater number of smoking pack-years or a younger age of starting were generally associated with higher increased risks for lung, liver and bile duct, kidney, oral and pharynx, and bladder cancers. However, there is no clear trend pattern in the associations between these two smoking variables and risks of prostate and uterine cancers. Prolonged smoking cessation among former smokers was associated with a significantly reduced risk for lung, liver and bile duct, and bladder cancers when compared with never smokers. Notably, the HRs for lung cancer decreased with longer durations of smoking cessation: HRs were 9.71 (95% confidence interval [CI]: 7.51-2.55) for <10 years, 4.28 (95% CI: 3.09-5.94) for 10-19 years, 2.86 (95% CI: 1.96-4.18) for 20-29 years, and 1.25 (95% CI: 0.76-2.05) for ≥30 years. In contrast, a significantly reduced risk of uterine cancer was observed only among participants who had quit smoking within 10 years (Figure 1 and Supplemental Tables S3, S7, S10 and S13). Cancer Risk Among Former Smokers With Never Smokers as Reference Using the Baseline Cox Proportional Hazards Regression Models. The Duration of Smoking Cessation was Categorized by Years From Quitting Smoking to Baseline Interview.
Alcohol Drinking and Cancer Risk
Adjusted Associations of Alcohol Drinking With Cancer Risk in the SCCS, Overall and by Race a .
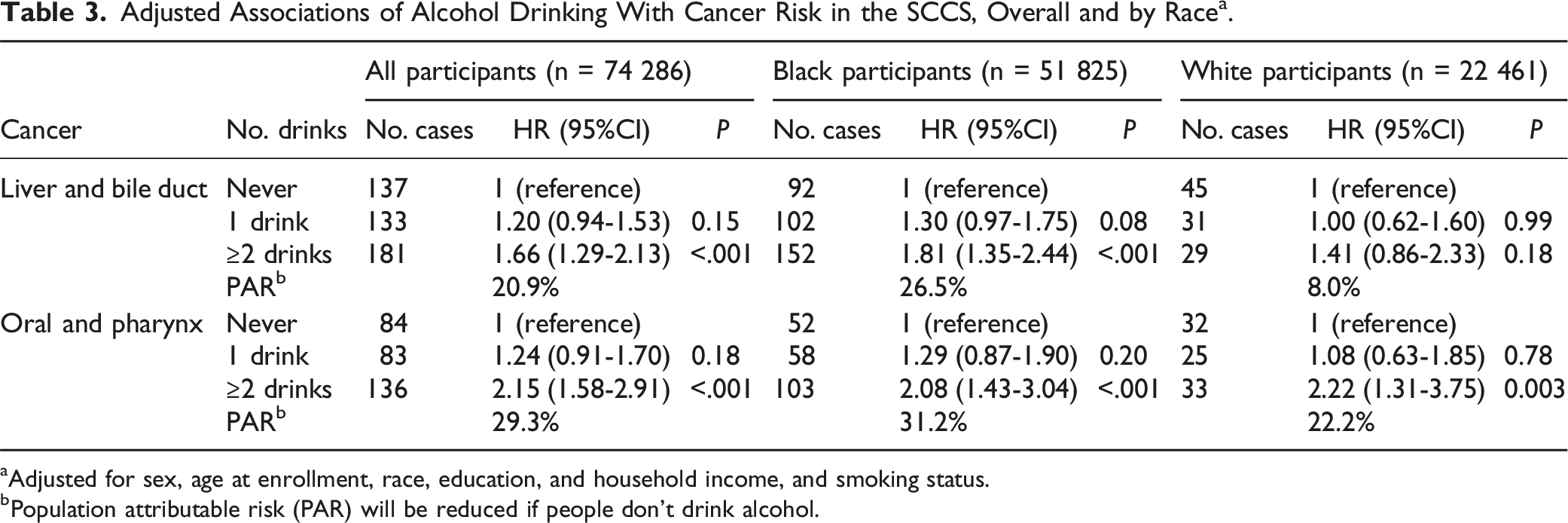
aAdjusted for sex, age at enrollment, race, education, and household income, and smoking status.
bPopulation attributable risk (PAR) will be reduced if people don’t drink alcohol.

Cancer Risk Among Alcohol Drinkers With Never Drinkers as Reference. The Results Were Based on the Baseline Cox Proportional Hazards Regression Analyses Among all Selected Participants.
Interaction between Race, Sex, Age at Enrollment, Smoking, and Alcohol Drinking
We evaluated the interaction of smoking and alcohol drinking with race, sex, and age at enrollment, and the interaction between smoking and alcohol drinking on the risk of all cancers investigated in this study, and no significant interaction was observed (all P > 0.05).
Discussion
In this study involving 74 286 low-income Black and White American adults, we found that cigarette smoking was associated with an increased risk of lung, liver and bile duct, kidney, pancreas, oral and pharynx, and bladder cancers, while it was linked to a lower risk of prostate and uterine cancers. Prolonged smoking cessation was associated with a reduced risk of lung, liver and bile duct, and bladder cancers. Additionally, consuming two or more alcoholic drinks per day was associated with an increased risk of liver and bile duct cancer, as well as oral and pharynx cancer. Similar associations were observed across both racial groups.
The significant associations between cigarette smoking status and cancer risk observed in this study are consistent with previous meta-analyses1,9,10,15,32-35 (also see Supplemental Table 15). Compared with earlier studies, the SCCS reported a much higher risk for lung cancer and liver and bile duct cancer among current smokers, with HRs of 14.1 vs 9.0, and 3.2 vs 1.5, respectively.1,10 Our analysis revealed that up to 85.9% of lung cancer cases and 54.6% of liver and bile duct cancer cases could have been avoided if all SCCS participants had abstained from cigarette smoking. In addition, current smokers of the SCCS had double the HRs of liver and bile duct cancer compared to previous meta-analysis studies on liver cancer (3.2 vs 1.5-1.6).1,10 These findings may reflect a higher proportion of current smokers or less healthy cancer-related lifestyles, such as combined cigarette smoking, alcohol drinking, and overweight/obesity in this low-income population.20,36 Conversely, we found that current smoking was inversely associated with the risk of uterine and prostate cancers, consistent with the results from previous meta-analyses and pooled analyses.15,32,35,37 The significant inverse associations observed among former smokers in the previous meta-analysis 35 was not detected in this study or the pooled analysis, 37 although a risk reduction of approximately 15% was not noted. While the lower prostate cancer risk among smokers may be attributed to factors such as reduced adherence to cancer screening, increased mortality from smoking-related diseases, or other confounding variables,15,32 smoking may confer a protective effect against uterine cancer, particularly in postmenopausal women. This potential protective effect is thought to be mediated through several anti-estrogenic mechanisms, including lower body fat among smokers (resulting in reduced estrogen conversion from androstenedione in adipose tissue), increased production of the potentially anti-carcinogenic metabolite 2-hydroxyestrone, higher circulating progesterone levels, and earlier menopause onset due to oocyte depletion. 38 Nevertheless, the precise biological mechanisms underlying the inverse associations between smoking and the incidence of prostate and uterine cancers remain unclear and warrant further investigation to inform cancer prevention strategies.
Notably, prolonged cessation among former smokers in SCCS was associated with a reduced risk of several cancers, especially lung cancer. The associations between smoking cessation and lung cancer risk in SCCS align with findings from previous studies, which demonstrate increased HRs among former smokers (vs never smokers), even 15-30 years after cessation, with HRs decreasing as the duration of cessation increases.39,40 These findings support the extension of lung cancer screening for former smokers beyond the 15-year quitting window 41 and highlight the significant benefits of earlier cessation.
Our study confirmed that moderate to heavy alcohol drinking (eg, ≥2 drinks per day) is a risk factor for liver and bile duct cancer, as well as oral and pharynx cancer, but not for colorectal, breast, kidney, prostate, or other cancers (Supplemental Table 15).2,3 Interestingly, a suggestive inverse association with M. Myeloma was also found in the previous dose-risk analysis showing an approximately 15% reduced risk for those consuming two to four drinks per day (∼25-50 g ethanol/day). 42 Furthermore, unconditional multivariable logistic regression analysis indicated a protective effect associated with heavy alcohol intake (ie, >20 g ethanol/day). 43 The biological mechanisms underlying the potential protective role of moderate to heavy alcohol drinking on the risk of M. Myeloma require further investigation. Proposed explanations include the immunomodulatory and antioxidant effects of alcohol, which may contribute to the observed inverse association.42,44,45 However, multiple mechanistic pathways, such as immune system impairment and the induction of oxidative stress and inflammation, are also linked to alcohol-driven carcinogenesis. 46
One significant strength of this study is its prospective investigation of the long-term effects of cigarette smoking and alcohol drinking on 12 types of cancer in a large, low-income population of Black and White Americans. In addition, comprehensive smoking exposure variables, including status, intensity, initiation age, and duration of cessation, were assessed. However, several limitations of this study should be acknowledged. Firstly, measurement errors may arise from self-reported data, including variables related to cigarette smoking and alcohol drinking. In addition, a mean of 12.1 years of follow-up may lead to biased association effects due to possible changes of smoking and drinking behaviors. 47 Secondly, residual confounding may exist due to unmeasured variables and uncontrolled factors, such as the comorbidity of chronic obstructive pulmonary disease in the association between smoking and lung cancer48,49 and obesity in the association between smoking and uterine cancers. 37 Obesity and diabetes are highly prevalent Southern states and among Black individuals of U.S.50-52 These two conditions are also associated with risk of several cancers including postmenopausal breast, endometrial, colorectal, liver, and pancreatic cancer,53-55 therefore further research is needed to clarify whether potential causal relationships between cigarette smoking and drinking and risk of these obesity/diabetes-associated cancers. Thirdly, the differential impact of cigarettes and alcohol on men and women needs further clarification, despite some studies observing similar risks for both sexes in certain types of cancer, such as lung cancer. 8 Fourthly, limited cases of several cancers in this study may limit power to validate their associations identified before1-3 (Supplemental Table 15). Fifthly, additional research may be warranted to assess the combined consumption patterns of cigarettes and alcohol, as well as their synergistic effect on cancer risk.18,19 Finally, other forms of tobacco use and the dose-response relationships for these substances should also be interrogated.
Conclusion
Cigarette smoking was found to increase the risk for lung, liver and bile duct, kidney, pancreas, oral and pharynx, and bladder cancers, while being associated with lower incidence of prostate and uterine cancers. Additionally, alcohol drinking was associated with an increased risk of liver and bile duct cancer, as well as oral and pharynx cancer. These findings underscore the critical importance of controlling cigarette smoking through reduction, cessation, and abstinence of smoking, and avoiding heavy alcohol drinking (ie, ≥2 drinks per day). These preventive interventions are particularly essential for reducing the risk of several cancer types in low-income populations in the United States.
Supplemental Material
Supplemental Material - Association of Cigarette Smoking and Alcohol Drinking With Risk of 12 Common Cancers Among Low-Income American Adults in the Southeastern United States
Supplemental Material for Association of Cigarette Smoking and Alcohol Drinking With Risk of 12 Common Cancers Among Low-Income American Adults in the Southeastern United States by Jiajun Shi, PhD, Wanqing Wen, MD, MPH, Qiuyin Cai, MD, PhD, Martha J Shrubsole, PhD, Xiao-Ou Shu, MD, PhD, and Wei Zheng, MD, PhD in Cancer Control
Supplemental Material
Supplemental Material - Association of Cigarette Smoking and Alcohol Drinking With Risk of 12 Common Cancers Among Low-Income American Adults in the Southeastern United States
Supplemental Material for Association of Cigarette Smoking and Alcohol Drinking With Risk of 12 Common Cancers Among Low-Income American Adults in the Southeastern United States by Jiajun Shi, PhD, Wanqing Wen, MD, MPH, Qiuyin Cai, MD, PhD, Martha J Shrubsole, PhD, Xiao-Ou Shu, MD, PhD, and Wei Zheng, MD, PhD in Cancer Control
Footnotes
Acknowledgments
The authors thank SCCS participants for their contributions to the study.
Ethical Statement
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the National Cancer Institute of the National Institutes of Health (USA) under award number U01CA202979. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
