Abstract
Phytochemicals, chemicals from plants, have garnered huge attention for their potential ability to prevent cancer. In vivo and preclinical models show that they do so often by affecting the hallmarks of cancer. Phytochemicals affect key pathways involved in the survival, genome maintenance, proliferation, senescence, and transendothelial migration of cancer cells. Some phytochemicals, namely antioxidants, can scavenge and quench reactive oxygen species (ROS) to prevent lipid peroxidation and DNA damage. They also trigger apoptosis by stopping the cell cycle at checkpoints to initiate the DNA damage response. Numerous in vitro and in vivo studies suggest that phytochemicals hinder cancer onset and progression by modifying major cell signaling pathways such as JAK/STAT, PI3K/Akt, Wnt, NF-kB, TGF-β, and MAPK. It is a well-known fact that the occurrence of cancer is in itself a very intricate process involving multiple mechanisms concurrently. Cancer prevention using phytochemicals is also an equally complex process that requires investigation and understanding of a myriad of processes going on in the cells and tissues. While many in vitro and preclinical studies have established that phytochemicals may be potential chemopreventive agents of cancer, their role in clinical randomized control trials needs to be established. This paper aims to shed light on the dynamics of chemoprevention using phytochemicals.
Keywords

Introduction
Cancer is a disease caused by abnormal growth and uncontrolled division of cells. It can initiate at any organ and can metastasize to remotely located organs. A diverse array of causative agents aid in causing cancer, ranging from physical agents, namely ultraviolet rays and ionizing radiation, to chemical agents like asbestos, alcohol, and tobacco smoke, and biological agents such as certain bacteria, viruses, and parasites. 1 According to the International Agency for Research on Cancer (IARC), cancer is among the leading causes of death worldwide in 2022 resulting in 9.7 million fatalities. About 1 in 5 people develop cancer in their lifetime, and approximately 1 in 9 males and 1 in 12 females die from the cancer. 2 In recent times, the mortality rate from cancer has decreased considerably owing to the discoveries in molecular biology, recombinant DNA technology, and immunology.3,4 Reduction in the number of smokers, maintenance of healthy eating habits, enhancement of living standards and environmental facilities, and focus of attention towards one’s fitness are a few contributors to this heartening trend.
Healthy eating practices have been linked with lower cancer rates for a long time. While no conclusive evidence suggests any particular food can cure cancer, a nutritious diet encompassing vegetables, fruits, whole grains, and legumes can help lower the risk factors associated with cancer. 5 Different foods have different components that aid in safeguarding against various types of cancer. Some foods, such as tomatoes and grapes, contain chemicals like lycopene and resveratrol, which help fight against prostate, breast, and pancreatic cancers.6,7 Other foods like whole grains have dietary fibers that protect from colon and digestive tract cancer. 8 Phytochemicals are naturally occurring bioactive compounds in plants that have several health benefits. Cancer cells have 6 distinctive traits defined as hallmarks of cancer. These signature attributes, unique to the cancer cells, are sustained proliferative signaling, evasion of growth suppressors, resistance to apoptosis, replicative immortality, induction of angiogenesis, and metastasis. 9 The phytochemicals in plant-based foods work as chemopreventive agents by mitigating the hallmarks of cancer. These chemicals such as isoflavones, catechins, and lycopene exert inhibitory effects against cancer. 10
There are generally 2 types of chemopreventive agents of cancer. The blocking agents that suppress the initiation of cancer in healthy individuals reduce DNA damage by blocking endogenous or exogenous carcinogens. They prevent general genomic instability and neoplastic transformation. The mechanism might be decreased uptake of or increased metabolism of procarcinogens; enhanced detoxification of free radicals and electrophiles, and finally, upregulation of DNA repair activity. Other modes of action include the downregulation of inflammatory responses and the production of reactive oxygen and nitrogen species. In some instances, blocking agents act by obstructing epigenetic mechanisms of cancer progression such as methylation and deacetylation. The second type of chemopreventive agent is called a suppressing agent. They act on 1 or more signal transduction pathways that lead to cell proliferation. They also induce increased apoptosis of cancerous lesions and lead to inhibition of angiogenesis. 11
Based on the stage at which the chemopreventive agent acts, chemopreventive agents can be grouped into 3 categories. The first category of chemoprevention, the primary chemoprevention, involves the prevention of cancer in high-risk and other healthy individuals. These agents prevent cancer biomarkers from appearing. The second category, secondary chemoprevention, includes the prevention of premalignant lesions from developing into invasive cancer. These include agents such as non-steroidal anti-inflammatory drugs (NSAIDs) in patients with colorectal cancer. Primary and secondary chemoprevention sometimes fall under the umbrella of primary chemoprevention. Finally, tertiary chemoprevention involves administering agents that prevent the recurrence of invasive cancer. 11
Phytochemicals can act both as suppressing agents or blocking agents in chemoprevention. They can also be primary, secondary, or tertiary chemopreventive agents. In vitro and in vivo studies have identified potential modes of action of certain phytochemicals. Most phytochemicals act on multiple signaling pathways and nodes of cancer development. The role of select phytochemicals–lycopene, resveratrol, sulforaphane, isoflavones, catechin, quercetin, curcumin, luteolin, and apigenin– in chemoprevention has been discussed in this paper. 11
Lycopene
Lycopene is a bright-red organic pigment called carotenoid, which gives fruits such as tomato, watermelon, red guava, and papaya their distinctive red color (Figure 1). It is a powerful antioxidant known for health benefits against cardiovascular diseases, hypertension, diabetes, and cancer.
12
When heated, the trans-isomer form of lycopene converts to cis-form and is readily absorbed by the body due to its increased solubility in bile acids.
13
This makes cooked tomatoes an ideal source of lycopene. Lycopene shows a dual impact on oxidative processes, acting as a pro-oxidant at high doses and an antioxidant at low doses. This dual nature of lycopene is vital in studying its potential chemopreventive properties. At low concentrations, it protects against DNA damage from reactive oxygen species, thereby reducing the chance of mutation and cancer.
6
At high doses, it helps destroy diseased cells. Thus, it can act in both primary and tertiary chemoprevention. In in vivo studies, lycopene has been shown to regulate oxidative and inflammatory processes, apoptosis, cell division, angiogenesis, and metastasis.
14
(A) Lycopene, (B) Modulation of cell signaling pathways by lycopene.
Lycopene helps contain cancer cells by the arrest/apoptosis of tumor cells and inhibition of invasion. It reduces phosphorylation in Rb, allowing the active Rb to bind with E2F transcription factors, which helps arrest the cell cycle at the G1/S transition. When DNA damage is detected, ATM/ATR kinases are activated, and p53 is phosphorylated, triggering p21 and ultimately leading to cell cycle arrest by blocking cyclin D1, CDC2, and cyclin E. The arrested cell can have 2 fates; it can either undergo a DNA repair process or inactivate Bcl-2 proteins which leads to a series of steps in apoptotic cascade causing apoptosis. Lycopene also obstructs the PI3K/AKT, enabling FOXO3 stimulation, which promotes the expression of p15, p21, and p27 to trigger cell cycle arrest and apoptosis. Lycopene confines the invasive cancer cells by inhibiting the MAPK and NF-kB pathways. By limiting the ERK, p38, and ROS, it thwarts JNK and MAPK kinase kinases (MAPKKKs) to halt MAPK and NF-kB pathways (Figure 1B).15,16
Lycopene has been long studied for its chemopreventive and anticancer properties, especially against prostate cancer. It accumulates at a much higher concentration in prostate tissues than in other tissues. 17 This tendency provides a reasonable explanation as to why the anticancer activities of lycopene are higher for prostate cancer than for other cancers. According to a study, lycopene subdues the phosphorylation effects on the Retinoblastoma protein (Rb), activates the tumor suppressor protein, p53, and inhibits the expression of cyclin D1 in G0/G1 phases, thereby arresting the cell cycle. 18 Additionally, the research suggests that lycopene inhibits the growth of nonneoplastic PrEC (Prostate epithelial cells) in vitro. Various papers indicate that this action of lycopene against prostate cancer might also be due to the alteration and inhibition of multiple pathways, including the PI3K/Akt pathway, 17 NF-kB signaling pathway, 14 AKT/mTOR pathway, MAPKs pathway, and JAK/STATs pathway. 19 A study by Yang et al demonstrated that lycopene supplementation suppresses prostate tumor cell growth by decreasing the VEGF levels in plasma. The results of this study revealed that these effects are probably linked to the reduction of proliferation and the interference of insulin-like growth factor 1 signaling. Furthermore, inhibiting VEGF by lycopene could mean that antitumor activities of lycopene involve anti-angiogenesis. 20
While a majority of the research indicates that lycopene mainly works wonders in prostate cancer, some papers mention that it has beneficial effects against other forms of cancer, such as breast cancer, by acting synergistically with other anticancer drugs like quinacrine that work by increasing the levels of ATP -binding cassette (APC), DAB2, GSK-3β and Axin, decreasing β-Catenin, p-GSK3β (ser 9) and CK1 and inhibiting Wnt-TCF signaling.
21
Other sources suggest that it has positive results in cancers associated with inflammation such as
Immunomodulatory Effects of Lycopene
It has been demonstrated that lycopene modifies immune response through various mechanisms. According to preliminary data on immunomodulation, lycopene modifies dendritic cell responsiveness by downregulating the expression of common surface dendritic cell markers such as CD80, CD86, and MHC II molecules. 23 Regulatory T cells are crucial for the destruction of malignant cells. Increased T cell counts secrete inhibitory cytokines such as IL-10 and TGF-β, which suppress effector cell function. In vitro studies have demonstrated a significant decrease in IL-10 and TGF-β levels upon increasing the lycopene concentration from 0.5 μmol l−1 to 5 μmol l−1. 24 Additional alterations in the concentrations of TNF-α, IL-1, IL-6, IL-8, and TIL-2, which are important T cell stimulants, were also noted.23,24 Moreover, lycopene may improve T cell activation and differentiation by increasing the ratios of CD4+/CD8+, IFNγ+/CD8+, perforin+/CD8+, and granzyme B+/CD8 + while having little effect on DNMT1 and DNMT3b. 23 Another study observed that lycopene may enhance the generation of serum immunoglobulin G and spleen B lymphocytes, improving cells’ overall immunological function. 16
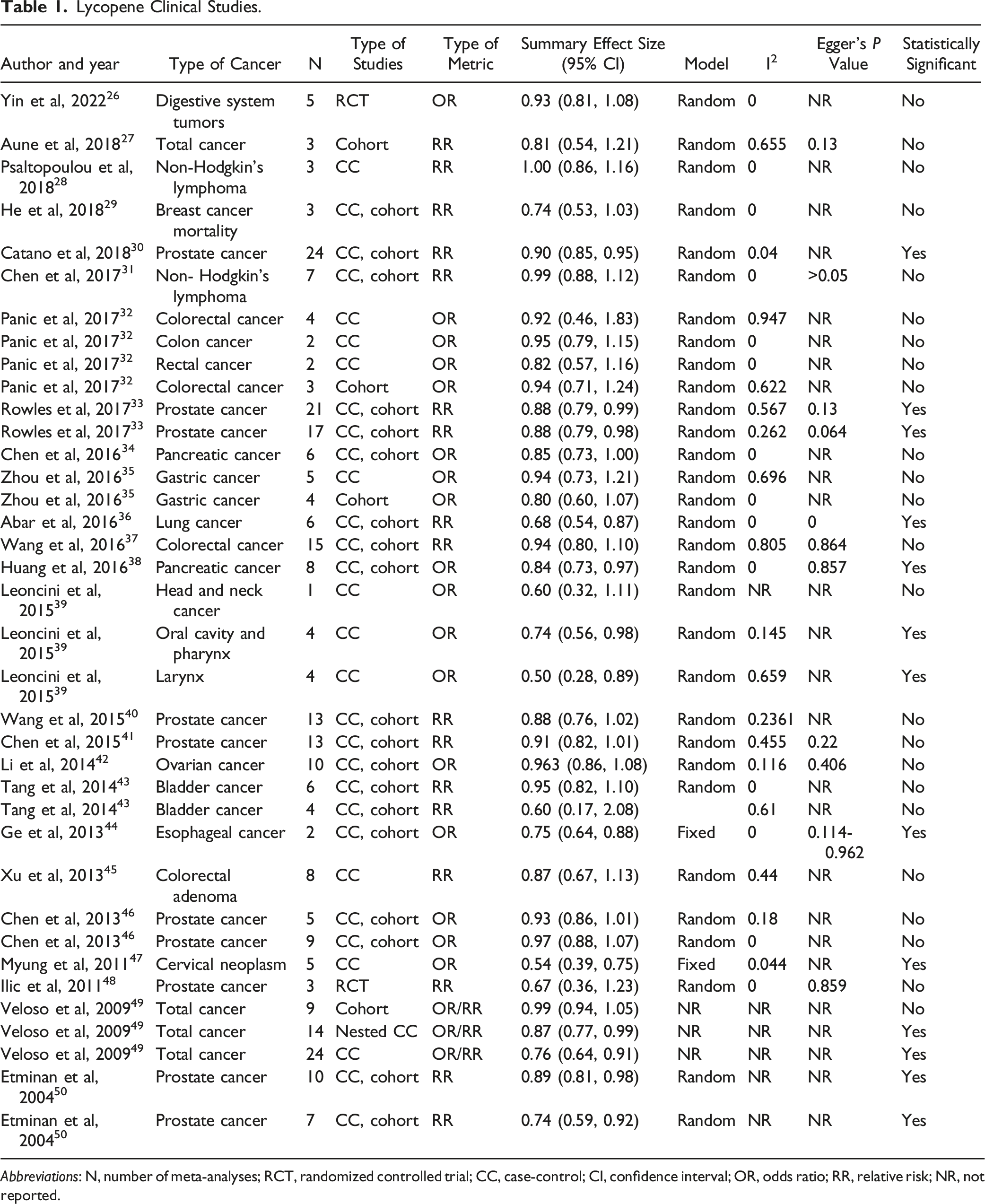
Lycopene Human Studies
Lycopene Clinical Studies.
Resveratrol
Resveratrol is a bioactive compound belonging to the class of polyphenols called stilbenes (Figure 2). Usually occurring in the trans-isomeric form in plants, the trans-resveratrol converts into a more bioactive form of dihydro-resveratrol when consumed orally.
51
The compound found predominantly in the skin of red grapes is produced as a protective agent by the grapes in response to environmental stressors, infection, or injury.
52
This phytoalexin antioxidant, produced primarily by red grapes to combat the damage done by UV radiation, shows promising results in the fight against skin cancer usually triggered by ultraviolet radiation and oxidative damage.
52
Several in vitro studies have shown that resveratrol has cytotoxic effects against skin, colon, breast, stomach, reproductive organs, liver, and thyroid cancer cells.
53
Resveratrol has been observed to reverse drug resistance in various cancer cells by making them responsive to anticancer drugs.
54
(A) Resveratrol, (B) Modulation of cell signaling pathways by resveratrol.
Resveratrol blocks Phase I enzymes (CYP450) to reduce the activation of carcinogens and induces Phase II enzymes (GST, UGT) to enhance the detoxification of carcinogens, thereby thwarting the cancer initiation process. It inhibits the PI3K/Akt pathway by inhibiting PI3K and, consequently inhibiting NF-kB/AP-1 and FOXO3a, leading to apoptosis and inhibition of invasion, respectively. Moreover, resveratrol halts the proliferation of cancer cells by disrupting the Wnt/β-catenin and TGFβ-Smad pathways. It checks proliferation by suppressing β-catenin, which restricts the transcription of c-myc and MMP-7, proteins necessary for proliferation. Similarly, the obstruction of Smad2/3 upregulates E-cadherin to stop the proliferation. Resveratrol also inhibits MAPKs, which hinders HIF-1ɑ to help prevent angiogenesis (Figure 2B).53,55,56
Resveratrol safeguards against cancer by scavenging and quenching the DNA-damaging reactive species, and preventing tumor initiation, progression, and metastasis. Resveratrol impedes the accumulation of ROS after exposure to oxidative agents like tobacco smoke condensate (TAR) and hydrogen peroxide. 57 It shows a protective action against lipid peroxidation and DNA damage by scavenging and quenching the ROS such as hydroxyl, superoxide, metal/enzymatic induced radicals, and radicals of cellular origin. 58 Attia further demonstrated the radical scavenging activity of resveratrol in their study on the effects of resveratrol on oxidative damage in genomic DNA and apoptosis induced by cisplatin. They observed that the cisplatin-induced genotoxicity and apoptosis were lessened in mice’s resveratrol-treated somatic and germinal cells. 59 Induction of apoptosis mediated by the alteration of cyclin and cyclin-dependent kinase to halt the cell cycle is another mechanism by which resveratrol protects against cancer. 60 Resveratrol downregulates the cyclin D1/Cdk4 complex, resulting in the arrest of the cell cycle at the G0/G1 phase. Meanwhile, it enhances the expression of cyclin E and cyclin A, which stops the cell cycle at the G2/M and S phases. 61 A similar study by Kim et al on lung carcinoma A549 cells demonstrated the effects of resveratrol in S phase arrest of the cell cycle by reducing Rb protein phosphorylation and inducing p21 and p53 protein expression. 62 Effective expression of these proteins provides the cell ample time to halt the cell cycle at various checkpoints and repair the DNA damage.
The antitumor activities of resveratrol are also attributed to various mechanisms by which it affects different signal transduction pathways responsible for cell growth, division, apoptosis, angiogenesis, and metastasis. 53 A study by Aziz et al observed that resveratrol inhibits the activation of phosphatidylinositol 3’-kinase/Akt, which ultimately results in modulations in the Bcl-2 family proteins, promoting the apoptosis of human prostate carcinoma LNCaP cells. 63 Resveratrol also induces apoptosis by modulating the mitogen-activated protein kinase pathway (MAPK) 64 and inhibiting NF-kB activation. 65 Similar studies noted that resveratrol alters pathways like Wnt/β-catenin, NF-kB, and AKT/GSK-3β/Snail, which play a significant role in cancer metastasis.66,67 Specifically, resveratrol has been shown to inhibit colon cancer’s invasion and metastasis by reversing epithelial-mesenchymal transition via the AKT/GSK-3β/Snail signaling pathway. 67
Immunomodulatory Effects of Resveratrol
Resveratrol can function as an immunomodulator in a variety of pathways. Increased splenic proliferation, which leads to a larger cell population of T and B cells, is one of the mechanisms promoted by resveratrol. In a study by Lai et al, mice treated with resveratrol showed a significantly higher spleen index than mice treated with levamisole or control. 68 In separate in vivo experiments, comparable increases in IL-2, TNF-α, and NF-kB were noted, although PGE2 production was suppressed. When resveratrol was used, production of TNF-α gradually decreased up to 25 μM, with the highest production reported at about 12.5 μM PGE2. 69 Antitumor cytokines are primarily produced by activated CD8+ T cells. Breast tumor-derived T cells treated with resveratrol showed a significant rise in the CD8+/CD4 + ratio—a necessary ratio for a longer survival rate for cancer patients—without causing any discernible change in CD4+ levels. 70 Resveratrol promotes the release of anticancer cytokines such as IFN-γ and inhibits the release of TGF-β. It is also observed that resveratrol stimulates the polarization of CD4+ T cells toward anticancer cells and decreases infiltration and polarization of immunosuppressive cells. 71 These immunomodulatory properties of resveratrol make it a suitable candidate for immunotherapy. Still, its poor bioavailability and low potency have made it difficult to be used in clinical settings. Nonetheless, new methods such as combination therapy and nanotechnology are being studied to overcome the limitations.
Resveratrol Human Studies
Resveratrol Clinical Studies.

Sulforaphane
Sulforaphane is a naturally occurring plant compound of the isothiocyanate family found in cruciferous vegetables such as broccoli, cabbage, cauliflower, and kale (Figure 3). It is not always present in its active form but is formed when myrosinase transforms its precursor glucoraphanin after any damage to the plant. Plants produce sulforaphane as a defense mechanism against pathogens, insects, and herbivores.
81
Numerous studies highlight sulforaphane’s health benefits, including protection against cardiovascular diseases,
82
diabetic effects,
83
deterioration of brain health,
84
and gastrointestinal malfunctions.
85
Sulforaphane is also labeled a chemopreventive phytonutrient due to its cancer-preventive properties in vitro and in vivo.
86
Evidence suggests that sulforaphane intake is linked with lowering the risk of colon,
87
lung,
88
and prostate cancer.
89
(A) Sulforaphane, (B) Modulation of cell signaling pathways by sulforaphane.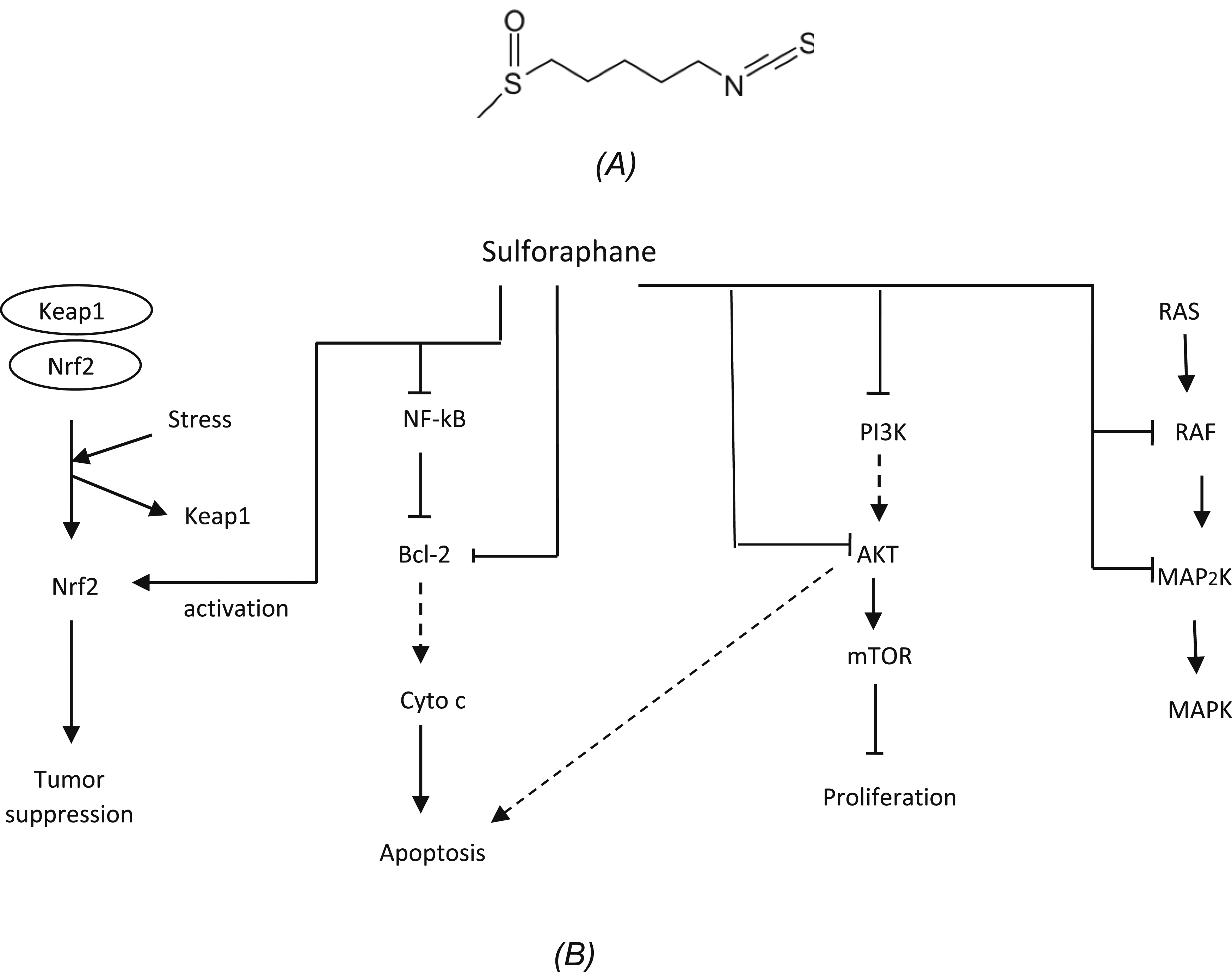
Sulforaphane acts as a tumor suppressor agent by promoting the transcription of Nrf2, a known tumor suppressor protein. It also displays apoptotic potential by blocking NF-kB, leading to Bcl-2 inactivation or direct inactivation of Bcl-2, triggering a series of steps that activate caspase 3, necessary for apoptosis. Moreover, sulforaphane subdues the PI3K/AKT pathway by hampering the PI3K and AKT. This disruption by inhibition of AKT stimulates the apoptotic pathways and disturbs mTOR, which is necessary for tumor survival and proliferation. Sulforaphane impedes the MAPK pathway by targeting multiple points: RAF and MAP2K. MAPK pathway is a crucial oncogenic pathway vital for cell proliferation (Figure 3B).90-92
The chemopreventive activity of sulforaphane is due to the inhibition of phase I enzymes responsible for activating pro-carcinogens and the promotion of phase II enzymes crucial in mutagen elimination. 93 It also alters cancer-related events like cell cycle, cell death, angiogenesis, metastasis, and invasion. The apoptotic action of sulforaphane is related to affecting the expression of apoptosis-related genes like p53, p21, Cdk2, Bax, 94 modulating the Bcl-2 family proteins, 95 activating caspases, and modulating cyclins and Cdks. 96 It is also noted that sulforaphane prevents proliferation, angiogenesis, and metastasis, the crucial steps in developing a full-blown cancer. A case in point is provided by an in vitro study conducted by Carrasco-Pozo et al, where only 10 μM sulforaphane could suppress the increased proliferation rate of lymph node carcinoma of the prostate (LnCaP) cells containing stimulated androgen receptors and prostate-specific antigen (PSA). 97 A similar in vitro study by Bertl et al, conducted using immortalized human microvascular endothelial HMEC-1 cells, suggested that sulforaphane significantly decreases microcapillaries’ formation, inhibits capillary-like tubes on the basement membrane matrix, and hinders cell migration. 98
Sulforaphane’s apoptotic, antiproliferative, and antimetastatic properties are intricately linked to its ability to modulate various signal transduction pathways involved in cancer development and progression. Extensive studies have shown that sulforaphane regulates numerous oncogenic signaling pathways like the Nrf2 - Keap1 pathway, NF-kB pathway, Akt pathway, signal transducer and activator of transcription 3 and 5, and other survival proteins. 99 Administration of sulforaphane inhibited the proliferation of CRC cells by upregulating the expression of UDP glucuronosyltransferase 1A (UGT1A) in CRC cells via extracellular signal-regulated kinase/Nuclear factor erythroid 2-related factor 2 (ERK/Nrf2) signaling pathway. 87 Moreover, sulforaphane inhibited the activities of sonic hedgehog (Shh), smoothened (Smo), Glioma-associated oncogene homolog 1 (Gli1), and Polyhomeotic-like protein 3 (PHC3) in CD133+ lung cancer cells by modulating the Shh signaling pathways and PHC3 to constrain the self-renewal of lung cancer stem cells. 88
Immunomodulatory Effects of Sulforaphane
Sulforaphane is a potent immunomodulator. It inhibits the NF-kB signaling pathway, which thwarts the expression of proinflammatory cytokines such as TNF-α, COX-2, IL-1β, and IL-18. 100 Similarly, it enhances the production of IL-2 and IFN-γ along with the proliferation of splenocytes, bone marrow cells, and thymocytes by stimulating various mitogens such as concanavalin A, phytohaemagglutinin, pokeweed mitogen, and lipopolysaccharide. 101 Similar attributes related to Nrf2 activation by sulforaphane enhanced cytoprotection by mitigating DNA damage and suppressing lipogenesis. 102 It also induces ROS-mediated upregulation of NKG2D ligands, increasing the susceptibility of tumor cells to NK cell-mediated lysis. 103 Furthermore, sulforaphane increases dendritic cell stimulatory capacity by modulating regulatory molecules, JAK/STAT3 - and microRNA - signaling, suggesting the potential for immunotherapy. 104 However, sulforaphane’s effects on cancer cells are intricate because, in primary human T cells, it acts in a pro-oxidative manner, potentially hindering T cell activation and their effector functions. 105 This dual nature of sulforaphane on cancer cells suggests that even though it shows promise in cancer immunotherapy, its combination with T cell-mediated therapy requires serious considerations to avoid any mishaps in the treatment.
Sulforaphane Human Studies
Sulforaphane Clinical Studies.

A 2021 paper by Kaiser et al evaluated several small clinical trials to assess the chemoprotective role of sulforaphane against various cancers. 115 Most of these trials tested whether the compound could inhibit type I enzymes, promote type II enzymes, repair DNA damage breaks, decrease deacetylase activity, and upregulate and downregulate important cancer biomarkers. Although around twenty preliminary clinical trials demonstrated positive outcomes, larger clinical trials with more robust primary and secondary endpoints need to be included.
Isoflavones
Isoflavones are naturally occurring flavonoids produced by the bean family members (Figure 4). Legumes, grains, and some vegetables contain isoflavones, but soybeans are the richest source of isoflavones. Some of the major isoflavones in soybeans are genistein, daidzein, and glycitein, ranging from 1.2 to 4.2 mg/g dry weight.
116
Isoflavones are considered phytoestrogens as they have estrogen-like properties and are derived from plants.
117
These phenolic plant compounds exert estrogenic/anti-estrogenic effects, influencing hormonal balance. Initially present in inactive forms in plants as glycosides, isoflavones are later converted to active aglycone forms by gut bacteria in the intestine.
118
(A) Genistein, (B) Modulation of cell signaling pathways by isoflavones.
Isoflavones have numerous potential health benefits, particularly concerning hormone regulation, and are used to treat hormone-dependent health conditions like menopause, breast cancer, and CVD. 119 Isoflavone maintains healthy endothelium and prevents endothelial cell dysfunction by inducing nitric oxide production to protect from CVD. 120 Isoflavones’ increased nitric oxide bioavailability also imparts antihypertensive effects by reducing endothelial cell oxidative stress or modulating vascular ion channel activity. 121 One study by Guo et al highlights the ability of isoflavones to exert estrogen-like effects in modulating the Nrf2 signaling pathway, which improved atherosclerosis and oxidative stress in in vivo and in vitro experiments. 122 Isoflavones also prevent diabetes by altering several cellular pathways crucial for maintaining glucose homeostasis. They help prevent type 2 diabetes by modulating nuclear receptor activity and non-receptor signaling on the cells necessary to maintain glucose homeostasis. 123 Since isoflavones have a similar chemical structure to endogenous estrogens, they interact with intracellular estrogen receptors, reducing the accumulation of lipids and the distribution of adipose tissue, thereby helping reduce obesity. They inhibit adipogenesis and lipogenesis by interacting with various transcription factors and upstream signaling molecules, aiding in maintaining metabolism and balance to treat obesity. 124
Isoflavones show tumor suppression ability by activating the Nrf2 protein. Genistein is observed to bind readily with ERβ receptors and thwart oncogenic progression in breast cancer by triggering apoptosis and restraining angiogenesis. The G0/G1 arrest of tumor cells is achieved by blocking NF-kB and AKT, which are crucial for upregulating the expression of cyclin D. Tumor cells undergo a DNA repair process in this quiescent stage and die off by apoptosis if the damage is irreversible. A three-way obstruction of PI3K, AKT, and STAT3 helps impede tumor proliferation. Isoflavones downregulate FAK, which is necessary to form focal adhesions between tumor cells and ECM required for migration, thereby containing the invasion. Furthermore, isoflavones modulate the JAK/STAT pathway by hindering the phosphorylation of STAT1 and STAT3, which reduces the production of inflammatory mediators like TNF-ɑ and COX-2 (Figure 4B).125-127
Soy isoflavones, like genistein, exhibit notable anticancer properties against breast cancer, 119 prostate cancer, 125 cervical cancer, 128 ovarian cancer, 129 lung cancer, 130 and renal cancer. 131 This chemopreventive nature of genistein is associated with its anti-inflammatory, 118 anti-oxidant, 132 anti-angiogenic, 133 anti-metastatic, 134 and anti-proliferative properties. 135 Isoflavones are observed to suppress prostate cancer cell growth by disrupting the expression of 2 copper transporter genes, CTR1 and ATP7A. Since copper levels are increased drastically in cancers, isoflavones cause pro-oxidant signaling by targeting endogenous copper, leading to ROS-mediated cell death. 136 After 50 μM genistein treatment, superoxide anion is generated, which quickly converts into hydrogen peroxide(H2O2) before forming hydroxyl (HO-) through the oxidation of reduced copper. This accumulation of ROS induces irreversible DNA damage, causing cell death in breast cancer cells. 137
Genistein downregulates cytokine-induced signal transduction events in the immune cells to work as an anti-inflammatory agent. 118 The anti-inflammatory effects of isoflavones are further demonstrated by a human trial where markers of inflammation (IL-18 and C-reactive protein) were decreased, and plasma nitric oxide levels were increased when postmenopausal women with metabolic syndrome were fed a soy nut diet (340 mg isoflavones/100 g soy nut) for 8 weeks. 138 Another study illustrated the anti-oxidative properties of isoflavones by observing a significant decrease in thiobarbituric acid reactive substances (TBARS) in plasma, liver, and brain by 33%, 18%, and 12%, respectively, after treatment with 2.5 mg/kg body weight isoflavones for 5 weeks. 139 The anti-angiogenic activity of genistein is caused by inducing apoptosis in VEGF-loaded endothelial cells, attributed to inhibition of MMP-2,-9 production and activity. Additionally, the study showed that genistein exposure decreased the activation of JNK and p38 induced by VEGF. 133 A similar study observed genistein’s anti-angiogenic and anti-metastatic effects by inhibiting c-erbB-2, MMP-2, and MMP-9 in breast carcinoma. 140
Isoflavones demonstrate chemoprevention by modulating various signaling pathways responsible for causing full-blown cancer. These pathways may be related to apoptosis, cell proliferation, inflammation, and hormone regulation. The activation of both the nuclear transcription factor NF-kB and Akt signaling pathway is inhibited by genistein, which is known to regulate the balance between cell survival and apoptosis. 140 A study by Xu et al noted the promotion of apoptosis in genistein-treated lung cancer A549 cells by modulating the IMPDH2/AKT1 pathway. 130 Genistein is also shown to arrest the MCF-7 and MDA-MB-231 breast cancer cells in the G0/G1 phase, thereby inducing an apoptotic pathway. 134 It has been demonstrated that isoflavones bind to ERα and ERβ, with a higher affinity towards ERβ receptors, to exert various anti-estrogenic, anti-apoptotic, and anti-inflammatory properties. 126 Genistein alters the PI3K/Akt pathway to induce differentiation in breast cancer stem cells by interactions with ER + cells. 141 The abundance of such evidence provides ample ground for further investigation of the chemopreventive abilities of this phytochemical.
Immunomodulatory Effects of Isoflavones
While isoflavones have a variety of physiological actions, the majority of them suppress NF-kB pathways and limit the generation of pro-inflammatory cytokines such as IL-6, TNF-α, IL-1β, and IFN-γ. 142 One of the most physiologically active isoflavones in soy, genistein, inhibits the growth of cancer cells by p-JNK, SGK1, Wnt, Akt, and Shh signaling pathways. According to a study by Jian Qin et al, the HCT-116 cell group treated with genistein at 50 and 100 μM showed a substantial decrease in Akt and SGK1 mRNA expression compared to normal cells. 143 Similar suppression of the p-MEK, p-ERK, p-JNK, and Shh pathways was observed in independent investigations by Qi Zhang and K. Li.144,145 Another frequent soy component, daidzein, enhances the BMP-2/Smads pathway while downregulating the expression of the STAT3 and STAT5 signaling pathways.146,147 Additionally, when isoflavones were applied to malignant cells, the CD4+/CD8 + ratio was considerably reduced. 148 Genistein is also being studied as an adjuvant in combination therapy with other drugs. One such study conducted by Liu et al to observe the effect of a genistein and cisplatin combination on CaSki human cervical cancer cells showed enhanced anticancer effects of cisplatin in CaSki cells. It was noted that the p-ERK1/2 and Bcl2 expression levels were decreased by 37% and 69%, respectively, and cleaved caspase-3 and p53 expression levels were increased by 115% and 304%, respectively, in the cisplatin plus genistein group as compared to that in cisplatin group. 128 Isoflavones’ significant immunomodulatory properties make them a promising candidate for cancer therapy.
Isoflavones Human Studies
Analyses of Isoflavone Intake and the Risk of Breast Cancer.

Catechins
Catechins are naturally occurring powerful antioxidants found primarily in green tea, cocoa, and berries. Classified as flavonols, catechins include compounds like epicatechin(EC), epigallocatechin(EGC), and epigallocatechin gallate(EGCG) (Figure 5). Their concentration in a typical brewed green tea beverage (250 mL) may vary from 50-100 mg depending upon preparation methods.
160
Catechins are found to exert beneficial effects in neurodegenerative diseases, CVD, cancer, and diabetes.
161
They enhance nitric oxide production, reduce LDL cholesterol levels, and inhibit platelet aggregation to benefit vascular endothelium.
162
It is observed that catechins can prevent diabetes by alleviating ER stress and promoting anti-inflammatory pathways.
163
Similarly, Brown adipose tissue (BAT) thermogenesis is noted in mice, suggesting catechin’s anti-obesity properties.
164
Additionally, catechins exert significant anti-inflammatory properties to prevent inflammatory bowel disease by regulating the activation and deactivation of inflammation-related oxidative stress-related cell signaling pathways such as NF-kB, MAPKs, and STAT1/3.
165
One of the most potent anti-cancer and anti-inflammatory catechins is EGCG.
166
EGCG is observed to be efficient in preventing prostate cancer, breast cancer,
167
lung cancer,
168
gastric cancer,
169
lymphomas,
170
and leukemia.
171
(A) Epigallocatechin gallate, (B) Modulation of cell signaling pathways by catechins.
Catechin affects anti-inflammatory signals to promote cell cycle arrest and apoptosis. It increases TNF-ɑ, which decreases NF-kB activity, causing apoptosis. Another anti-inflammatory signal, IL-10 is lessened, thereby downregulating the cyclin D1, leading to cell cycle arrest. TNF-ɑ increment also has the same effect on cyclin D1. Catechin inhibits the PI3K/AKT pathway, causing cell cycle arrest and reduced proliferation by decreasing cyclin D1 and blocking mTOR. It modulates the Wnt signaling pathway by degrading β-catenin, halting the transcription of MMPs, and obstructing the migration of tumor cells. One major mechanism by which catechin curbs tumor angiogenesis is blocking the EGFR, which downregulates a series of proteins, namely, ERK1/2, AP1, VEGF, PKB, and IL-8, halting angiogenesis. Moreover, catechin modulates the JAK/STAT pathway by limiting STAT3 phosphorylation, causing a rise in antitumor immune response (Figure 5B).172,173
Catechins work as both pro- and antioxidants to prevent cancer. In low doses, they act as antioxidants, scavenging the DNA-damaging ROS. They also chelate transition metals to prevent them from catalyzing oxidation reactions. Alternatively, catechins function as pro-oxidants at higher concentrations, triggering apoptosis and ferroptosis.174,175 Many articles note that EGCG’s anticancer abilities are due to its pro-oxidant nature, which promotes the Fenton reaction that generates radicals. EGCG elevates ROS levels to induce cell cycle arrest and apoptosis. It induces mitochondrial damage, ROS level elevation, DNA damage, and JNK activation to induce apoptosis in pancreatic cells, 176 glioblastoma cells, 177 and lung cancer. 178
Numerous studies have suggested that EGCG exerts significant efficacy against various forms of cancer by the mechanism of apoptosis and inhibition of proliferation and metastasis. EGCG alters both the caspase-dependent (intrinsic) pathway and death receptor (extrinsic) pathway by modulation of Bcl-2 family proteins 170 and downregulation of Mcl-1 and XIAP 171 to induce programmed cell death. In addition, EGCG also achieves apoptosis by the inhibition of TNF-ɑ activity. 179 The key antitumor mechanism of EGCG is attributed to its ability to suppress metalloproteinase activity. Many studies claim matrix metalloproteinases aid in tumor progression by allowing tumor invasion and metastasis. 180 A study on hepatocellular carcinoma cells (HCCLM6) demonstrates that the antimetastatic activity of the EGCG is due to its ability to inhibit MMP-2 and MMP-9 activity. 181 EGCG may either directly bind to the MMP-2 and MMP-9 to inhibit their activities 182 or alter some signaling pathways, including NF-kB, 183 MAPK/ERK, 184 and PI3K/Akt 185 to inhibit MMP expression. Similar mechanisms involving the modulation of several signaling pathways are responsible for EGCG’s antiproliferative abilities. One study puts forward the idea that EGCG lowers the expression of phosphorylated Akt (p-Akt) and phosphorylated mTOR (p-mTOR) through PTEN to modulate the PI3K/Akt/mTOR pathway, resulting in the decreased proliferation and apoptosis of pancreatic cancer cells linked with the expression of PTEN. 186 Another study demonstrates that EGCG subdues the proliferation of gastric cancer cells by silencing the wnt/β-catenin signaling pathway. 169 A similar study suggests that EGCG can reduce the proliferation and invasiveness of breast tumors by blocking the Wnt pathway with the help of the HBP1 gene. The downregulation of the Wnt pathway increases the expression of G1 regulators, c-MYC, and cyclin D1 genes, causing a reduction in invasive and migratory properties of the tumor. 187
Immunomodulatory Effects of Catechins
Catechins exert strong immunomodulatory effects on cancer cells. It is observed that the proinflammatory cytokines generated by malignant cells, such as IL-1β, IL-6, and TNF-α, dramatically decrease after being treated with catechins. 188 Catechins can also inhibit the NF-kB pathway to reduce the amount of proinflammatory cytokines. A study by Mackenzie et al found that NF-kB binding activity was suppressed dose-dependently in Jurkat cells pretreated with catechins, with the lowest binding occurring at 17.2 μM concentration of catechin. 189 Rawangkan et al suggested use of green tea catechin as an immune checkpoint inhibitor allowing cancer prevention and treatment based on the findings that EGCG partially restores T cell activity by inhibition of PD-L1/PD-1 signaling. 190 Catechins have shown potential to be used in combination therapy with other chemotherapeutic drugs for immunotherapy. A case in point is provided by the observation that catechins limit toxicity induced by a chemotherapeutic drug, irinotecan, by reducing its side effects, such as diarrhea, neutropenia, leucopenia, and non-alcoholic fatty liver disease. The same study also noted that it works as an effective anticancer agent as it mitigates the histopathology of colon adenocarcinoma. 191
Catechin Human Studies
Catechins Clinical Studies Against Prostate Cancer.

Quercetin
Quercetin is a plant flavonol belonging to the flavonoid group of polyphenols (Figure 6). It is found in many fruits, vegetables, and grains including capers, red onions, kale, and shallots. It has anti-oxidant and anti-inflammatory potential, showing great promise in treating and containing chronic health conditions, including CVDs and cancers.
198
Constant administration of quercetin decreases vascular smooth muscle cells (VSMC) and hampers the progression of CVDs. It also reduces NADPH oxidase, ROS, superoxide anion, and free radicals to exert anti-inflammatory and anti-hypertensive properties.
199
At the same time, it enhances oral glucose tolerance and pancreatic β-cell function while suppressing the activity of α-glucosidase and DPP-IV enzymes to impart protective effects against type-2 diabetes mellitus.
200
The anti-inflammatory and hypoglycemic nature of quercetin helps prevent obesity by decreasing fat deposition.
201
Quercetin also demonstrates potential chemopreventive properties against breast cancer,
202
liver cancer,
203
colon cancer,
204
cervical cancer,
205
blood cancer, prostate cancer, and lung cancer.
206
(A) Quercetin, (B) Modulation of cell signaling pathways by quercetin.
Quercetin triggers apoptotic pathways through multiple routes to safeguard against cancer. It can phosphorylate p53 or inhibit Bcl-2 to release cytochrome c and induce apoptosis through a series of steps. Quercetin conducts cell cycle arrest and apoptosis by phosphorylating ERK and JNK in the MAPK pathway. Disrupting the PI3K/AKT pathway by phosphorylating PI3K, AKT, and S6K contributes to its antiproliferative and apoptotic effects. It modulates the Wnt/β-catenin pathway by obstructing the nuclei’s β-catenin translocation and hampers the production of MMPs necessary for tumor cell migration. Quercetin also impedes the JAK/STAT pathway by halting the formation of p-STAT to promote its antiproliferative nature (Figure 6B).206,207
The combination of antioxidant, anti-inflammatory, apoptotic, antiproliferative, anti-angiogenic, and anti-metastatic effects of quercetin collectively endows it with chemopreventive qualities. Quercetin boasts antioxidant quality, thanks to its ability to hunt down and scavenge ROS such as peroxynitrite and hydroxyl radicals. 208 Rac1 is a GTPase that promotes cell migration and invasion by the production of ROS. Quercetin is found to target Rac1 with high affinity to make a stable complex and modulate the Rac1-p66Shc pathway to control ROS generation within the cells. 209 Likewise, its anti-inflammatory ability allows it to inhibit inflammatory enzymes and mediators to help prevent cancer. In vitro studies have shown reduced levels of inflammatory mediators, such as NO-synthase, COX-2, and CRP, in human hepatocyte-derived Chang liver cell lines after treatment with quercetin. The same study observed that quercetin blocks NF-kB activation resulting in the downregulation of pro-inflammatory genes. 210 Furthermore, another in vitro study discovered that quercetin lessens the levels of other inflammatory mediators like TNF-α, IL-6, and IL-1β in RAW264.7 macrophages by inhibiting the PI3K/Akt signaling pathway. 211 The apoptotic nature of quercetin is due to its ability to arrest the cell cycle, activate caspase proteases, and modify signaling pathways. Quercetin causes a G2 phase arrest in HPV-positive human cervical cancer-derived cells triggering apoptosis. 212 In human T-cell acute lymphoblastic leukemia Jurkat clones (J/Neo cell lines), quercetin is found to activate caspase-3 and caspase-9 to induce apoptosis in a dose-dependent manner. 213
Numerous studies have emphasized quercetin’s apoptotic potential through its ability to modulate signaling pathways like NF-kB/IκB, p38 MAPK, Bcl-2/Bax, 214 PI3K/Akt/mTOR, Wnt/β-catenin, 215 and FOXO3a. 216 Inhibition of angiogenesis by quercetin treatment was achieved by targeting the VEGFR-2 mediated angiogenesis pathway, suppressing the expression of the downstream regulatory factor AKT, and restraining tumor growth.217,218 Another study demonstrated quercetin’s anti-invasive and anti-metastatic effects on lung cancer cells by inhibiting the Snail-dependent Akt activation pathway. This study further observed that the expression of N-cadherin, vimentin, ADAM9, and MMPs-related proteins were notably downregulated and the expression of E-cadherin was significantly increased after quercetin treatment, which helped contain the invasiveness of the lung cancer cells. 219 Similarly, in a different study, the administration of 100 μM quercetin in CD44+/CD24− CSCs resulted in the restriction of proliferation of both cells to a great extent. 202
Immunomodulatory Effects of Quercetin
Quercetin functions as an immunomodulator for malignant cells. It acts as an anti-inflammatory agent by decreasing the levels of pro-inflammatory cytokines such as TNF-α, IL-β, and IL-6 in a dose- and time-dependent manner. 220 Quercetin administration causes rapid NK cell proliferation as shown by a study where 100 μg/mL of quercetin administration dramatically enhanced the proliferation of the NK-92 cell line. 221 Additionally, quercetin promotes the susceptibility of cancer cells to NK cell-mediated killing by inducing natural killer group 2D (NKG2D) ligands and suppressing heat shock protein (HSP70). 222 However, the low bioavailability of quercetin makes it challenging to use it alone in clinical settings which highlights the need for nano-formulation developments. It was observed, in a study, that nanoparticle-delivered quercetin has enhanced antitumor potential compared to quercetin alone. 223 Quercetin, when combined with conventional cancer therapies, acts as a sensitizer and protects non-cancer cells from the side effects of chemotherapy and radiotherapy. 224 Zhang et al best summarized the use of quercetin in immunotherapy where they introduced a codelivery of nano-formulated quercetin and alantolactone, and observed the promotion of antitumor immune response through synergistic immunogenic cell death in microsatellite-stable colorectal cancer. 225
Quercetin Human Studies
Clinical trial data on quercetin against cancer is not abundant. A clinical trial, involving 433 men with primary prostate cancer and 538 population-based controls, to study the effects of quercetin on prostate cancer revealed the potential chemopreventive attributes of quercetin. The results of the study showed a 27% reduction in the risk of prostate cancer from an intake of at least 24 μg of quercetin. 226 This clinical trial outcome hinted at promising chemotherapeutic and chemopreventive prospects of quercetin however, further clinical trials are required to confirm the results.
Curcumin
Curcumin is a bright yellow-colored polyphenol belonging to a group of natural compounds called curcuminoids (Figure 7). It is the primary bioactive compound in turmeric, with a concentration reaching up to 31.4 mg/g in pure turmeric powder.
227
Curcumin exhibits many therapeutic qualities including antioxidant,
228
anti-inflammatory,
229
chemopreventive, and immunity-enhancing properties.
230
A lower occurrence of cancer is associated with a dietary intake of curcumin.
231
The risk and incidence of various forms of cancer such as breast cancer,
232
lung cancer,
233
prostate cancer,
234
brain cancer,
235
pancreatic cancer,
236
and endometrial cancer,
237
is reduced by curcumin. (A) Curcumin, (B) Modulation of cell signaling pathways by curcumin.
Curcumin inhibits Bcl-2 and releases cytochrome c, Caspase 9, and Apaf 1 to induce apoptosis. It can suppress angiogenesis by inhibiting the NF-kB pathway and inflammatory mediators like COX-2. It modulates the JAK/STAT pathway by obstructing JAK to restrict the stemness of the tumor. Curcumin enhances E-cadherin levels while reducing the levels of N-cadherin, vimentin, fibronectin, snail, and slug through the repression of the TGF-β/Smad2/3 pathway to check metastasis. It modulates the Wnt signaling pathway by degrading β-catenin, halting the transcription of MMPs, and obstructing the migration of tumor cells. Moreover, curcumin can alter PI3K/AKT/mTOR pathway to increase the production of key proteins (e.g. Beclin 1) involved in autophagy (Figure 7B).238,239
The antioxidant potential of curcumin comes from its ability to scavenge free radicals such as ROS and RNS, 240 and modulate the activity of GSH, catalase, and SOD enzymes necessary for the neutralization of free radicals. 241 Working as an anti-inflammatory agent, curcumin reduces the expression levels of pro-inflammatory cytokines: TNF-α, IL-6, and IL-1β by inhibiting the NF-kB signaling pathway. 242 The study further concluded that curcumin inhibits the TGF-β1/Smads signaling pathway by reducing mRNA expression levels of fibrotic factors α-SMA, Smad2/3, and TGF-β to exert anti-inflammatory effects. Scientific research has established curcumin as an apoptotic, anti-angiogenic, anti-proliferative, and anti-metastatic phytochemical. Curcumin achieves apoptosis via various mechanisms, including ROS-mediated pathway, 243 downregulation of apoptosis suppressor proteins, 234 and alteration in signaling pathways such as induction of FOXO1 and inhibition of the PI3K/Akt pathway. 236 Meanwhile, an in vivo study claims curcumin suppresses angiogenesis by downregulating VEGF, CD31, and αSMC expression levels. 244 Another similar study using human ovarian cancer cell lines SKOV3ip1, HeyA8, and HeyA8-MDR in athymic mice revealed that oral intake of 500 mg curcumin per kg body weight was the ideal dose to suppress NF-kB and signal transducers and activators of transcription 3 (STAT3) activation and to mitigate angiogenic cytokine expression. 245 Curcumin modulates cell cycle and signaling pathways to subdue proliferation and perform apoptosis of the tumor cells. G2 phase accumulation of head and neck cancer SCC-9 cells was achieved by downregulating the PI3K/Akt/mTOR pathway that arrested the cell cycle on G2/M transition. 246 In addition, similar effects of curcumin in the modulation of signaling pathways were seen in an experiment involving non-small cell lung cancer cells (NSCLC) where tumor proliferation was inhibited and apoptosis was regulated by the upregulation of microRNA-192-5p (miR-192-5p) and the downregulation of the PI3K/Akt signaling pathway. 233 It is a well-known fact that epithelial-mesenchymal transition is involved in cancer progression and metastasis, and curcumin acts as an anti-metastatic substance by blocking the same. Curcumin achieves this by altering several expression pathways, including inhibiting c-Met expression, 247 upregulating NKD2, which downregulates the Wnt signaling pathway, 248 and phosphorylating Smad2/3, which suppresses the TGF-β2 signaling pathway. 249 Furthermore, curcumin was shown to decrease N-cadherin, twist, snail, and vimentin, while increasing E-cadherin in colorectal cancer SW480 cells, suggesting that it could suppress the EMT process by inhibiting CDX2/Wnt3a/β-catenin pathway. 250
Immunomodulatory Effects of Curcumin
Curcumin has been investigated frequently as an immunomodulator against malignant cells. Curcumin downregulates proinflammatory cytokines such as IL-1 and TNF-α 251 whose activation mostly depends on the NF-kB pathway. 252 Curcumin inhibits IKK to downregulate the NF-kB pathway. According to a study by Shih et al curcumin treatment of CT26 cell lines resulted in a dose-dependent decrease in the expression of PD-L1 and NF-kB. 253 Additionally, curcumin can repolarize cancer-associated macrophages into a tumoricidal form and can provoke NK cells to target tumor cells and cancer stem cells indirectly. 254 Challenges similar to those of other phytochemicals, like poor solubility and bioavailability, have made clinical application of curcumin a daunting task. Curcumin is currently being studied as a chemo-sensitizing agent and shows potential to be used as an adjuvant to cancer immunotherapy by enhancing the immune system’s ability to combat tumors. 255 Farghadani et al highlight the chemo-sensitizing ability of curcumin in a study where they noted that curcumin enhanced the anticancer efficacy of chemotherapy drugs like doxorubicin, paclitaxel, 5-fluorouracil, and cisplatin in breast cancer. 256
Curcumin Human Studies
Curcumin Clinical Studies.

Luteolin
Luteolin is a flavone, belonging to a group of plant compounds called flavonoids (Figure 8). It is a yellow-colored compound found primarily in celery, thyme, parsley, chamomile tea, broccoli, carrots, and several other foods. This polyphenol protects plants from UV radiation, fluctuating temperatures, insects, and microorganisms.
265
Luteolin exhibits several pharmacological properties, including antioxidant, anti-inflammatory,
266
neuroprotective,
267
and analgesic effects.
268
Numerous studies demonstrate the health benefits of luteolin in protection against Alzheimer’s disease,
269
CVDs,
270
and various cancers. Luteolin’s anticancer effects have been observed across a variety of cancers, ranging from breast cancer to colon cancer
271
and lung cancer
272
to gastric cancer.
273
(A) Luteolin, (B) Modulation of cell signaling pathways by luteolin.
Luteolin can trigger apoptosis by regulating both extrinsic and intrinsic pathways. It modulates the extrinsic apoptotic pathway by promoting the expression of death receptors (Fas, DR4, DR5), which induce apoptosis by caspase cascade induction. It can regulate the intrinsic apoptotic pathway by inhibiting antiapoptotic proteins such as Bcl-2. The inhibition of Bcl-2 is also linked to the activation of p53 as a result of DNA damage by ROS. Luteolin suppresses Ras, Raf, and MEK1/2, thereby impeding the expression of ERK1/2 and cyclin B1 to thwart cell cycle progression and angiogenesis. Moreover, it also disrupts the PI3K and AKT in the PI3K/AKT pathway to suppress GSK3β, downregulating cyclin D1 and causing cell cycle arrest. The inhibition of AKT also suppresses the mTOR obstructing cell cycle progression and angiogenesis. Luteolin also modulates the Wnt/β-catenin pathway, causing inhibition of GSK3β and β-catenin, and resulting in the inhibition of EMT (Figure 8B). 274
Chemoprevention by luteolin is a multifaceted approach that acts at multiple points by obstructing invasion, metastasis, angiogenesis, cell cycle regulation, and induction of apoptosis. The upregulation of pro-apoptotic proteins such as Bax and Caspase 3, combined with the downregulation of the anti-apoptotic protein Bcl-2, enhances luteolin’s apoptotic potential. 274 Luteolin can scavenge and neutralize ROS and protect the cells from lipid peroxidation and DNA damage. A recent study by Fernando et al demonstrated that luteolin scavenged intracellular ROS dose-dependently. They observed that ROS decreased by 31% at 0.625, 51% at 1.250, 58% at 2.500, 68% at 5.000, and 75% at 10.000 μg/ml luteolin concentration in lung fibroblast cells. 275 Cell cycle regulation is another mechanism by which luteolin safeguards against cancer. Luteolin causes cell cycle arrest and halts the proliferation of tumor cells by downregulating cyclin D1 and Survivin, and upregulating p21. 276 The antimetastatic character of luteolin is due to its ability to suppress epithelial-mesenchymal transition, a critical process of cancer metastasis. Several studies in breast cancer cells have highlighted this potential of luteolin.273,277 A case in point is provided by a study conducted by Cao et al, where they revealed that luteolin significantly inhibited YAP/TAZ activity by promoting YAP/TAZ degradation in Triple-Negative Breast Cancer (TNBC) cells to suppress their migration. 277
Luteolin modulates numerous cell signaling pathways to disrupt tumor initiation and progression. It can inhibit the growth, migration, and inhibition of SW620 and SW480 colon cancer cells by acting on the IL-6/STAT3 pathway. 278 A study on A549 lung cancer cells revealed that luteolin restricts tumor migration by suppressing focal adhesion development and limiting the FAK-Src signaling. 279 Another research reported the inhibition of viability, migration, angiogenesis, and invasion in vascular endothelial cells of NSCLC via miR-133a-3p upregulation. 280 This study also noted that luteolin decreased purine-rich element-binding protein B (PURB) and showed the inhibitory effects of the compound on tumors by miR-133a-3p/PURB- mediated MAPK and PI3K/Akt pathways. 280 Moreover, luteolin also inhibits YAP/TAZ activity by modulating the Hippo signaling pathway to suppress the migration of TNBC cells. 277 The effects of luteolin on the modulation of the PI3K/AKT signaling pathway to inhibit cancer have been studied extensively. One such research demonstrated that luteolin could contain the proliferation of melanoma cells and trigger apoptosis by reducing MMP-2 and MMP-9 expression through the mediation of the PI3K/AKT pathway. 281 A recent study also observed the ability of luteolin to modulate the PI3K/AKT pathway in HeLa cells. The findings suggest that luteolin suppresses proliferation and promotes apoptosis by altering AKT/mTOR/PI3K and MAPK pathways. 282
Immunomodulatory Effects of Luteolin
Luteolin has garnered attention in cancer immunotherapy because of its immunomodulatory effects. It suppresses inflammation by modulating cytokine secretion involved in immune responses. Luteolin promotes NK cell secretion of type I cytokines, especially IL-2 and IFN-γ, critical for providing antitumor immunity. 283 A similar study on the immunomodulatory effects of luteolin on lung cancer observed the increment of NK cell cytotoxicity and granule secretion at 12.5 μg/ml and 25 μg/ml luteolin concentrations. 284 As stated earlier, luteolin has antioxidant and anticancer properties along with its immunomodulatory properties, making it a potential candidate for cancer immunotherapy. A review by Shang et al explores the possibility of using nanocarriers as a delivery system to enhance solubility, circulation time, and targeting ability of luteolin, addressing its poor bioavailability. 285 Modifying luteolin nanocrystals with SDS enhanced the bioavailability of luteolin by 3.48-fold, according to a study by Liu et al. 286 Luteolin is also being studied as an adjunct in cancer therapy. In a study, luteolin has been shown to increase the antitumor activity of oxaliplatin in colorectal carcinoma. When 50 mg/kg BW/day luteolin was administered with 10 mg/kg BW/day oxaliplatin 3 times a week for 3 weeks in mice implanted with HCT116 colorectal carcinoma, a synergistic suppression of tumors was observed. 287 Another recent research by Chen et al notes that luteolin effectively inhibits cervical tumor proliferation and growth when combined with Asiatic acid. 288 Evidence and research such as this provide a foundation to explore the use of luteolin in cancer therapy, either alone or in combination with other drugs.
Luteolin Human Studies
While multiple preclinical studies involving in vivo and in vitro research techniques have already demonstrated the potential anticancer properties of luteolin, including its ability to induce apoptosis, inhibit proliferation, and disrupt cell migration, these findings are yet to be translated to clinical research.273,274,289 One of the primary reasons for the inability to conduct clinical trials is the poor bioavailability of luteolin, mainly due to its low solubility in water. 290 Another factor contributing to its poor bioavailability is the excessive glucuronidation of luteolin by enzymes such as uridine diphosphate glucuronosyltransferases 1 As (UGT1As). 291
Apigenin
Apigenin is a naturally occurring yellow-colored phytochemical of the flavone class found in various plants and vegetables (Figure 9). Dried chamomile flowers have up to 5000 μg/g of apigenin.
292
Parsley, celery, spinach, grapefruits, etc. are other sources of apigenin. This flavonoid has attracted significant attention recently owing to its beneficial effects in ameliorating diverse health complications. Apigenin has various biological and pharmacological properties, such as antioxidant, anti-inflammatory, neuroprotective, and cardioprotective abilities.
293
It decreases oxidative stress and neuroinflammation to protect against neurodegenerative diseases.
294
Apigenin shows beneficial effects against various forms of cancer, including breast,
295
prostate,
296
colorectal,
297
and hepatocellular carcinoma.
298
(A) Apigenin, (B) Modulation of cell signaling pathways by apigenin.
Apigenin induces apoptosis through the regulation of the TRAIL signaling pathway by preventing ANT-2-mediated deactivation of the DISC complex, resulting in the downregulation of antiapoptotic proteins such as Bcl-2, XIAP, and IAP 1/2. It also blocks the downstream activation of transcription factors, such as FOXO3a, that promote the expression of pro-apoptotic proteins like p21, KIP1, and WAF, thereby triggering apoptosis. Apigenin obstructs the phosphorylation of PI3K, AKT, and STAT3, causing the downregulation of HiF-1ɑ and VEGF to contain angiogenesis. Moreover, it suppresses the NF-kB signaling pathway to thwart the expression of genes involved in cell migration and invasion, such as MMP-2, MMP-9, Twist1, and β-catenin (Figure 9B).299,300
Apigenin monitors the hallmarks of cancer by inducing apoptosis and autophagy, and inhibiting cell proliferation, angiogenesis, and metastasis. The antioxidant mechanism of apigenin helps in cancer prevention by the inhibition of oxidant enzymes, modulation of redox signaling pathways, and free radical scavenging. 301 Apigenin can regulate the cell cycle at various cell cycle checkpoints to obstruct cell proliferation. It can halt the cell cycle at the G2 phase by reducing mRNA and protein levels of the key regulators that control the G2-M transition in prostate cancer cells. 302 Research by Bao et al concluded that apigenin also arrests renal cell carcinoma (RCC) at the G2-M transition by reducing the expression levels of cyclin A, B1, D3, and E. 303 Apigenin induces apoptosis in cancer cells by triggering both intrinsic and extrinsic pathways. It can trigger apoptosis intrinsically by altering mitochondrial membrane potential leading to the release of cytochrome c and activating a caspase cascade or by impeding the levels of anti-apoptotic proteins, such as Bcl-2 and Bcl-xL, while increasing pro-apoptotic proteins like Bax.304,305 Extrinsically, apigenin induces apoptosis by upregulating the mRNA expressions of caspase-3, caspase-8, and TNF-ɑ. 305 Furthermore, it may induce other cell death pathways besides apoptosis, such as autophagic cell death, ferroptosis, necroptosis, etc., by inducing endogenous ROS generation. 306
Modulation of signaling pathways is a critical process in cancer chemoprevention by apigenin. Apigenin alters numerous signaling routes to disrupt cancer initiation, proliferation, angiogenesis, and metastasis processes. It inhibits AKT phosphorylation, a key regulator of the cell cycle, growth, and survival, by suppressing AKT function or directly repressing PI3K activity by blocking its ATP-binding site. 307 Apigenin causes cell cycle arrest and apoptosis of human prostate cancer cells by inhibiting the PI3K/AKT/FOXO signaling pathway. 308 A mixture of apigenin and chrysin suppressed the activity of the p38 MAPK/Akt pathway to impede the proliferation, migration, and invasion of colorectal cancer cells. 309 Another recent study by Naponelli et al highlighted the role of apigenin in suppressing tumor angiogenesis by targeting HiF-1ɑ/HIF signaling pathways. 310 The same study also noted that apigenin inhibits multiple signaling pathways--Wnt/β-catenin, PI3K-AKT, and Hippo-YAP/TAZ to inhibit EMT, a key step for cancer metastasis. 310 Similarly, vitexin, a glycosylated form of apigenin, reduced the stemness of human endometrial cancer by downregulating Oct4 and Nanog through the inhibition of the PI3K/AKT pathway. 311 Moreover, apigenin reduces the expression of STAT3 and JAK2 to prevent tumor growth and proliferation by triggering apoptosis in HER2-overexpressing breast cancer cells.312,313
Immunomodulatory Effects of Apigenin
Apigenin modulates the immunological response via multiple routes to boost immunity. It enhances anti-tumor immunity by stimulating natural killer cells and CD8+ T lymphocytes while suppressing immunosuppressive cells like regulatory T cells and myeloid-derived suppressor cells. 314 It enhances the ability of NK cells to kill pancreatic cancer cells by upregulating the expression of NKG2D, perforin, and granzyme B. 315 Apigenin’s immunomodulatory effects in cancer are not limited to the stimulation and enhancement of the proliferative ability of immune cells, as it can also modulate immune checkpoint pathways in cancer. A recent study aiming to highlight the immunomodulatory potential of apigenin in triple-negative breast cancer (TNBC) concluded that it overcomes the inhibitory effects of PD-L1 inhibitors by halting the AKT/mTOR pathway. 316 This finding underscores the potential for developing combination therapies and personalized treatment strategies to boost the immune response in combating cancer. Combination treatments of apigenin and other chemotherapeutic agents are being studied in vivo and in vitro. One such study observed that apigenin sensitizes leukemia cells to the topoisomerase inhibitors, etoposide and cyclophosphamide, by promoting apoptosis and reducing ATP levels. 317 Apigenin also enhanced the action of BTK inhibitor abivertinib by the downregulation of BclX through the suppression of PI3K/p-AKT in diffuse large B-cell lymphoma cell lines. 318 A recent study by Nimal et al also noted that apigenin combined with vorinostat showed anticancer activities by modulating the expression of epigenetic and apoptotic regulators, leading to reduced cell migration and increased cell death in TNBC models. 319 In light of these findings, apigenin has gathered much attention from the medical community as a possible cancer chemopreventive agent.
Apigenin Human Studies
Chemoprevention using apigenin has not been successful because no clinical trial data have been published on the compound to date. Hence, there is a need to conduct clinical trials in controlled settings to identify and label the chemopreventive attributes of apigenin accurately. Nonetheless, the success in preclinical settings surely gives a foreground for designing and developing clinical trials.
Limitations of Chemopreventive Compounds in Cancer Therapy and Future Directions
Summary of Phytochemicals and Their Corresponding Molecular Targets and Therapeutic Use.

Early epidemiological studies showed that micronutrient deficiency results in cancer risk. There was a higher risk of esophageal cancer in populations of northern China, central Asia, and northern Iran that depended on cereals for diet and consumed very few fruits and vegetables. With US-China cooperation, the idea that micronutrients prevent cancer was tested in large clinical trials in the 1980s. A factorial design that tested the role of retinol, zinc, riboflavin, niacin, ascorbate, molybdenum, alpha-tocopherol, beta-carotene, and selenium was conducted. The study showed that a diet consisting of beta-carotene, alpha-tocopherol, and selenium reduced the rate of gastric and total cancer mortality. The effect was especially pronounced in the young population. 320
Carotene and alpha-tocopherol are vitamins, whose role has been extensively tested through clinical trials. In a factorial design, the role of the 2 chemicals in preventing lung cancer was tested in smokers with a high risk of lung cancer in the 1980s and surprisingly, the
The reasons why these phytochemical nutrients failed in clinical trials may be severalfold. In an already nutritionally well-nourished population, the nutrients might not prevent cancer. Additionally, these trials used very high doses of the nutrients, which might lead to untoward consequences. Moreover, the preventive effects might be prominent in early life and the population sample in these studies did not select young subjects. The selection of agent may have been inappropriate leading to negative results. 320
Besides nutritive phytochemicals, non-nutritive phytochemicals have also been tested in human experiments. While some experiments show positive results, others show negative results. The studies give very inconsistent results. Lifestyle factors, genetic polymorphisms, and other interfering factors may reduce the power of epidemiological studies compared to animal studies where the results are often promising. The inconsistencies may lie in the interpretation of cell lines and animal studies. In animal studies, very high levels of phytochemicals are used to get results, while in human intervention, much lower doses are used which might not give the same result. Differences in the bioavailability of agents in animal studies vs human studies might lead to result discrepancies. It is not always that high doses are required for effect. For example, for resveratrol, the lowest dose suppressed cancer much better than the higher dose. 320
The positive results shown by in vitro studies and animal models are obtained using a much higher dose of these compounds than the amount gained from a normal diet. This makes it difficult to acquire the same health benefits as the in vitro studies promised. One needs to take them in larger concentrations to compensate for their poor solubility and inability to penetrate the plasma membrane. Individual genetics, metabolic patterns, and genetic diversity due to cultural and geographic locations make it even more challenging to pinpoint these compounds’ bioavailability accurately. Using non-nutritive phytochemicals, in the long run, might be toxic if the dosage is very high. The presumed advantage of phytochemicals due to their nontoxicity would then be negated. Especially if the chemicals have to be taken for a long duration as chemopreventive agents, this aspect would come to the forefront.
Some phytochemicals impart benefits in certain forms of cancer while increasing the risk associated with other forms of cancer at the same time. For instance, isoflavones, although beneficial in breast and lung cancer, increase the risk associated with advanced forms of prostate cancer. 321 Flavonoids work as mutagens, pro-oxidants, and inhibitors of drug-metabolizing enzymes in addition to being an anticancer agent. 322 The lack of convincing evidence and ambiguous findings from the clinical trials have made it even more daunting to label specific phytochemicals with their cancer-mitigating abilities.
For most of the non-nutritive phytochemicals, there is a plethora of cell line and animal studies but very few high-powered human intervention trials. This can be attributed to a lack of resources as well. Furthermore, some of the large clinical trials using beta-carotene, alpha-tocopherol, and selenium were not very successful. Based on these results, the community is reluctant to spend a lot of time and resources conducting trials with other phytochemicals unless very promising and consistent results are derived from smaller clinical trials. It is very easy to choose a phytochemical of interest, select a population, plan dosage, and conduct a large clinical trial but the result has to be worth the effort.
One possible suggestion for conducting a worthwhile large-scale clinical trial is to combine the usage of multiple non-nutritive phytochemicals of different chemical classes and different targets. The trial can measure not only the effects on cancer chemoprevention but also the effects on other chronic diseases and health parameters. Evaluating the effect of combination phytochemicals is by no means an easy task. First, you have to start with 2 phytochemicals and measure their synergistic and antagonistic effects. Once you start adding third or fourth chemicals, the number of combinations increases tremendously. Analyzing synergism and antagonism in in vivo studies of all combinations might be very time-consuming and not feasible (perspective, a positive cocktail effect of bioactive components in the diet). One option is to try the hit-and-trial method with multiple chemicals at appropriate dosages determined by a panel of experts. After carrying out in vivo studies, bioavailability and toxicity studies in humans can be carried out using multiple chemicals at designated dosages. If no untoward effects are observed then efficacy studies can be conducted.
Alternatively, to test if combinations of phytochemicals have a synergistic chemopreventive effect, a randomized control trial where different dietary phytochemical index foods can be supplied to treatment vs control groups can be conducted and cancer risk measured. The dietary phytochemical index measures the amount of calories obtained from plant food, excluding potatoes. To devise a more accurate scale that measures total phytochemicals consumed, a different scale that also takes into account phytochemicals consumed from low-calorie food such as green tea can be used. Given the beneficial effects of a high phytochemical diet on cardiovascular and chronic diseases, the use of a high phytochemical diet should be promoted. If further chemopreventive effects on cancer were observed due to the intake of phytochemicals, the promotion of a phytochemical diet would get priority. 323
Biomarker discovery, so that endpoints can be properly measured, is crucial for cancer chemoprevention trial design. The success of cardiovascular chemoprevention has largely depended on finding biomarkers such as lipid levels and hypertension. In cases where large clinical trials using thousands of individuals are not possible, smaller clinical trials using high-risk individuals can be designed. 11
Conclusion
Cancer poses a severe public health concern globally. As the scientific community shifts its attention toward a preventive approach, phytochemicals can have a pivotal role in the fight against cancer. With proven therapeutic properties, many phytochemicals have been used in our households since ancient times. However, dosage, potential side effects, and negative interactions of these chemopreventive compounds had been overlooked previously, which somewhat decreases their true potential. As more research and findings surface, the use of phytochemicals in cancer prevention is becoming more evident. Clinical and pre-clinical studies of phytochemicals show encouraging results in cancer prevention. While having immense potential for the battle against cancer, these phytochemicals have limitations in dosage and bioavailability, as described in the limitations section. It is only through the evidence-based promotion of phytochemicals from studies with robust methodologies that a preventive approach against cancer can be effectively implemented and widely accepted in the medical fraternity.
Footnotes
Acknowledgments
We are deeply grateful to Mr. Sandesh Gaudel for his valuable insights into the immunomodulatory effects of phytochemicals. His guidance and thoughtful input significantly enriched this work. We would also like to express our sincere gratitude to Ms. Shristina Shrestha and Ms. Ruchie Shrees for their meticulous proofreading; their language suggestions and edits were instrumental in refining the article. Lastly, special thanks to Mr. Nirav Lekhak for his creative assistance in developing the figures and for his helpful language edits.
Author Contributions
NL and HKB conceptualized the paper. NL wrote on the preclincal aspects. HKB wrote on the clinical aspects.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
This manuscript is a review article and does not involve a research protocol requiring approval by the relevant institutional review board or ethics committee.
