Abstract
Objective
This study aimed to evaluate the expression levels of Shh, Gli1, and Cyr61 proteins in gastric cancer tissues and analyze the relationship between these three proteins and the clinicopathological factors and prognosis of patients.
Methods
This was a retrospective study. Four hundred gastric cancer tissue specimens from patients who underwent radical gastrectomy in Zhangye People’s Hospital affiliated to Hexi University between February 2013 and February 2021 underwent immunohistochemical analysis.
Results
The positive expression rates of Shh, Gli1, and Cyr61 in gastric cancer tissues were 55.5%, 56.5%, and 64.5%, respectively. The expressions of Shh, Gli1, and Cyr61 in gastric cancer tissues were significantly correlated with tumor size, depth of invasion, and degree of differentiation (P < .05). The expression of Shh protein was positively correlated with the expression of Gli1 protein (P < .01), and the expression of Gli1 protein was positively correlated with the expression of Cyr61 protein (P < .01). Univariate and multivariate analyses showed that the expression of Shh, Gli1, and Cyr61 could predict the prognosis of patients (P < .05). Receiver operating characteristic curve analysis combined with TNM staging could better predict the three-year overall survival of patients (P < .05).
Conclusion
Shh, Gli1, and Cyr61 proteins are significantly expressed in gastric cancer tissues and are risk factors for the prognosis of patients with gastric cancer.
Introduction
Gastric cancer (GC) is the fifth most common cancer worldwide, with nearly half of the cases occurring in China. 1 It is estimated that in 2022, there will be about 4,820,000 and 2,370,000 cases in China and the United States, respectively. GC accounts for several new cancer cases. The number of new cancer cases in China is about twice that of the United States, but the number of deaths is about five times that of the United States. Although many effective treatments, such as surgery, chemotherapy, and radiation therapy, are available, the prognosis of patients with GC remains poor.2,3 In China, the overall 5-year survival rate is as low as 40%. Most GCs are diagnosed at stage III or IV, with more than half of the patients having metastases; therefore, the disease burden remains high, and the prognosis is suboptimal. Research on GC has delved into the molecular level, but there is still a lack of biomarkers for diagnosis, disease progression, and prognosis. Studies have shown that the occurrence and development of GC are related to many signal transduction regulatory mechanisms, such as the sonic hedgehog (Shh) signaling pathway and Cyr61. Researchers have found that the Shh signaling pathway is activated in digestive tract tumors and plays an important role in tumor progression. 4 The activation of the Shh signaling pathway can promote the proliferation of GC cells and tumor development. 5 Zhong J et al. found that the zinc finger of the cerebellum can regulate the migration and invasion of GC cells through Shh, phosphoinositide 3-kinase (PI3K), and mitogen-activated protein kinase (MAPK). 6 Cyr61 not only increases tumor angiogenesis, but also participates in apoptosis and senescence, promotes tumor cell adhesion, proliferation and metastasis, and plays an important role in tumor occurrence and development. 7
In this study, the expressions of Shh, Gli1, and Cyr61 were simultaneously detected in GC tissue, and the relationship between these gene products and the clinicopathological factors of patients and the prognosis of patients was analyzed. This study has identified new biomarkers for the diagnosis and prognosis of GC, thus providing new reference targets for GC treatment.
Materials and Methods
Sample Collection
Clinical and pathologic characteristics of patients according to Shh, Gli-1, Cyr61.

Abbreviations: Shh, Sonic hedgehog; Gli-1, glioma associated oncogene homolog 1; Cyr61, cysteine-rich61.
Immunohistochemical Staining
Paraffin tissue sections were preheated in an incubator at 65°C for 5 h, dewaxed with conventional xylene, repaired in a water bath inside a pressure cooker, and blocked with a peroxidase blocker for 30 min. 9 The samples were then incubated overnight at 4°C with the primary antibodies rabbit anti-human Shh (ab53281, Abcam), rabbit anti-human Gli1 (ab217326, Abcam), and rabbit anti-human Cyr61 (ab228592, Abcam). The slides were then incubated with horseradish peroxidase conjugated goat anti-rabbit IgG secondary antibody for 30 min at 37°C. Finally, the diaminobenzidine (DAB) color reaction was observed. Two senior pathologists evaluated the immunohistochemical staining results. The staining intensity was scored as follows: 0 point for negative, 1 point for weak positive, 2 points for positive, and 3 points for strong positive. The staining intensity score was multiplied by the percentage of positive cells to obtain the final H-Score, and the median H-Score served as the positive-negative cut-off. 10 Overall survival (OS) was calculated from the surgical treatment date to the end of follow-up or date of death.
Statistical Analysis
Chi-square tests were used to compare categorical variables. Survival curves were generated using the Kaplan–Meier method, and a log-rank test was used to test for differences between survival curves. The multivariate Cox proportional hazard regression model was used for survival analyses. Model accuracy was assessed using the receiver operating characteristic (ROC). All P-values were two-sided, and <.05 was considered statistically significant.
Results
Relationship Between Shh, Gli1, and Cyr61 Expression and Clinicopathological Characteristics of Patients With GC
Immunohistochemical staining showed that Shh, Gli1, and Cyr61 were mainly expressed in the cytoplasm and cell membrane (Figure 1). The positive expression rate was 55.5% for Shh, 56.5% for Gli1, and 64.5% for Cyr61. Shh, Gli1, and Cyr61 expressions were correlated with tumor size, depth of invasion, and differentiation (P < .05). The expression of Shh protein was positively correlated with that of Gli1 (P < .01). Similarly, the expressions of Gli1 and Cyr61 were also positively correlated (P < .01) (Table 1). Shh, Gli1, and Cyr61 protein expression in gastric cancer tissues. High (A) and low (B) Shh expression in gastric cancer tissues. High (D) and low (E) Gli1 expression in gastric cancer tissues. High (G) and low (H) Cyr61 expression in gastric cancer tissues, negative control (C,F,I) in gastric cancer tissues. Original magnification: ×200.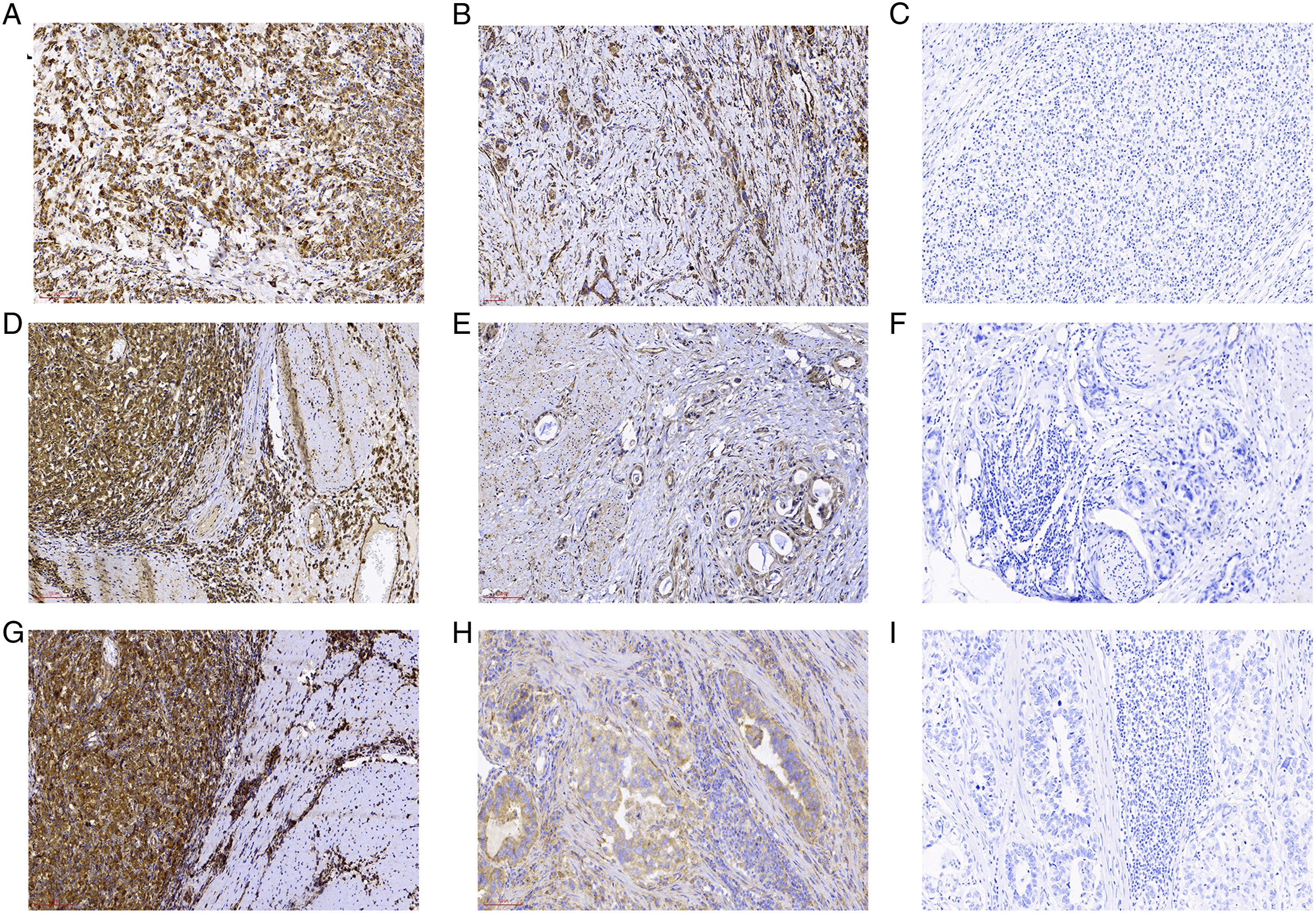
Relationship Between the Expression of Shh, Gli1, and Cyr61 and Prognosis of Patients With GC
All 400 patients with GC were followed up, and 209 survived, while 191 died. The results of patient survival analysis showed that patients with high expression of Shh, Gli1, and Cyr61 had a poor prognosis (Figure 2). The median OS of patients with low Shh expression was undefined (ratio=.3687, 95% confidence interval [CI] = .3047-.5661), while that of patients with high Shh expression was 100 months (ratio = 2.713, 95% CI = 1.767-3.282). The median OS of patients with low Gli expression was undefined (ratio = .2796, 95% CI = .2008-.3895), while that of patients with high Gli expression was 57 months (ratio = 3.576, 95% CI = 2.568-4.981). The medium OS for patients with low Cyr expression was undefined (ratio = .2883, 95% CI = .2070-.4014), while that for patients with high Cyr expression was 49 months (ratio = 3.469, 95% CI = 2.491-4.831). Univariate Cox regression analysis showed that Shh, Gli1, Cyr61 expression, patient age, tumor size, depth of invasion, degree of differentiation, lymph node metastasis, and TNM stage were significantly correlated with OS. These meaningful univariate variables were then included in the multivariate analysis. Cox proportional hazards regression analysis showed that patient age, Shh, Gli1, Cyr61 expression, TNM stage, and tumor differentiation were independent prognostic factors for OS in patients with GC (Table 2). The area under the curve (AUC) of three-year OS predicted by TNM stage alone was .711, but the combination of TNM with Shh, Gli1, or Cyr61 improved the prediction accuracy (AUC = .756, AUC = .778, AUC = .804, respectively, Figure 3). This demonstrates the advantages of using molecular biomarkers in the assessment of postoperative predictive survival in patients with GC. Kaplan–Meier curves according to Shh, Gli1, and Cyr61 protein expression level in patients with gastric cancer. Kaplan–Meier analysis of OS according to Shh expression (A), Gli1 expression (B), and Cyr61 expression (C). P-value was calculated using the log-rank test. Univariate and multivariate Cox regression analyses for OS of patients (n = 400). Abbreviations: CI, confidence interval; OS, overall survival. ROC analysis for the prediction accuracy of TNM combined with Shh (A), Gli1 (B), Cyr61 (C) model for 3-year OS and TNM alone. P-values show the statistical significance of the AUC.


Discussion
GC is a highly aggressive malignant tumor with a high mortality rate. 11 GC is easily missed because its clinical symptoms are similar to those of benign lesions such as chronic gastritis. Therefore, many GCs are already in the advanced stage when diagnosed. Even after surgery and chemotherapy, some patients still die due to metastasis and recurrence. Therefore, an in-depth study of the mechanism of occurrence and development of GC and the search for prognostic biomarkers is crucial for GC diagnosis and treatment. The Shh/Gli1 signaling pathway was first discovered in drosophila. During embryogenesis, Shh/Gli1 signaling is activated and regulates cell proliferation and differentiation. Excessive activation of Shh/Gli1 is associated with GC and other human tumors. 12 In particular, the Shh signaling pathway is abnormally activated in pancreatic, liver, and colorectal cancers. Shh is also closely related to the occurrence and development of basal cell carcinoma, medulloblastoma, pancreatic, breast, colon, ovarian, and small-cell lung carcinomas. 13 Studies have found that the activation of the Shh signaling pathway in Gli1 mediates lung cancer cell proliferation and Shh-dependent mesenchymal cell activation. 14 Our results showed that the expressions of Shh and Gli1 proteins were high in GC tissue, suggesting that the Shh signaling pathway was activated in GC. In addition, the expression of Cyr61 protein was also upregulated in GC.
We also found that the expression of Gli1 in GC was highly positively correlated with Shh and that the survival prognosis of patients with GC with high Gli1 expression was worse. You et al. confirmed that Gli1 is related to GC angiogenesis and that Gli1 promotes the expression of MMP14 and MMP2, thereby leading to GC progression. 15 Chakrabarti et al. showed that Shh signaling mediates the expression of programmed death-ligand 1 (PD-L1) in GC cells and promotes tumor proliferation, which may be the reason for the poor prognosis of patients with GC with high Shh expression. 16 Zhang et al. showed that Shh is highly expressed in cervical cancer. 17 When the SHH gene is silenced and the Shh signaling pathway is inhibited, the expressions of Shh signaling pathway-related factor Gli1 and vimentin were decreased, E-cadherin was increased, tumor cell migration and invasiveness were significantly reduced, and tumor growth was significantly slowed down in animal experiments. These findings indicate that the SHH gene is closely related to epithelial–mesenchymal transition (EMT) and that Shh causes EMT to promote tumor progression. Ke et al. showed that Shh/Gli1 expression was significantly increased in GC and that Gli expression was positively correlated with vimentin and negatively correlated with E-cadherin. 18 After adding recombinant Shh proteins to activate the signaling pathway, the migration and invasion of GC cells were significantly enhanced, the expression of Gli and vimentin was significantly increased, and the expression of E-cadherin was downregulated. The opposite result was obtained after adding Gli antagonist 61 to block the signaling pathway, indicating that Shh/Gli can promote GC EMT, reduce intercellular adhesion, enhance GC cell migration ability, and promote tumor cell invasion leading to tumor spread. In hypoxia-induced GC cells, Xu et al. confirmed that the hypoxia microenvironment causes Shh/Gli1 signaling pathway activation and promotes EMT. 19 After the siRNA treatment targeting Gli-1 inhibits the Shh/Gli1 signaling pathway, EMT was reversed, vimentin expression was significantly reduced, and E-cadherin expression was significantly increased. Yoo et al. obtained similar results, showing that the PI3K/Akt inhibition blocked the Shh-induced EMT, the activity of MMP-9, and lymphangiogenesis, reducing tumor invasiveness and metastasis in GC. 20 Their findings establish that Shh signaling promotes GC metastasis through the activation of the PI3K/Akt pathway, which leads to EMT and MMP-9 activation. Liang et al. found that TGF-β1 in SGC-7901 cells led to the increased expression of vimentin and Gli and decreased expression of E-cadherin. 21 After the addition of Gli inhibitor GANT61, the expression of vimentin was downregulated, the expression of E-cadherin was upregulated, and TGF-β1-induced EMT was blocked. It is suggested that the Shh/Gli1 pathway may promote the migration and invasion of GC cells by inducing EMT, resulting in a poor prognosis for patients with GC. We further analyzed the correlation between Shh, Gli1, and Cyr61 proteins and various clinicopathological factors. The results showed that the expression of Shh, Gli1, and Cyr61 in GC tissue was related to tumor size, depth of invasion, and degree of differentiation, and the differences were statistically significant. It is thus suggested that Shh, Gli1, and Cyr61 are related to GC progression and invasion.
Our study found that the expression of Cyr61 in gastric cancer is related to tumor size and T stage. Moreover, Shh, Gli1, and Cyr61 were not associated with TNM stage, although T stage itself is an important part of the TNM stage. In this study, all patients had localized gastric cancer without metastasis and underwent surgical treatment. Shh, Gli1, and Cyr61 expression were not associated with N stage. Therefore, N stage in Shh, Gli1, and Cyr61 expression may be associated with only T stage (i.e., the depth of invasion), and not the TNM stage. Wei et al. reported a relationship between metastasis and the expression of CYR61 protein. 22 N stage was not related to CYR61 protein; however, it was related with the TNM stage, indicating that T and M stage played an important role and the depth of invasion and metastasis were related to the expression of CYR61 protein. Our study is a single-center, small-scale retrospective study; a large multi-center study may present more accurate results. We also found that the expression of Shh, Gli1, and Cyr61 is related to the degree of tumor differentiation. The higher expression of Shh, Gli1, and Cyr61 in well-differentiated patients may be closely related to the tumor size of the patients. The tumor size of the well-differentiated patients was higher. A larger more consistent tumor may have led to more diverse sampling of sections. Moreover, patients with larger tumors had a higher expression of Shh, Gli1, and Cyr61, which may be related to more complete sampling. However, further studies with larger sample sizes are still needed.
Our study found that the prognosis of patients with high Cyr61 expression is worse. These results are consistent with the findings of Lin et al. 2 The survival time of patients with high levels of Cyr61 expression was significantly shorter than that of patients with low levels of Cyr61 expression.Liu et al. showed that in laryngeal squamous cell carcinoma, the expressions of vimentin and Cyr61 were higher in tumor tissue than in tumor-adjacent tissues and were closely related to lymph node metastasis and positively related to the T stage. 23 The E-cadherin result was the opposite, which proves that the expression of Cyr61 is closely related to EMT, leading to poor tumor prognosis. Knockdown of Cyr61 expression severely impairs the migration and invasion of GC cells, showing that the high expression of Cyr61 is closely related to GC progression. Studies have also shown that Cyr61 may activate Wnt, Shh, NF-κB, and COX2 signaling pathways in tumor cells.24-26 Our study found that the expression of Cyr61 is closely related to Shh, indicating that these two proteins may promote each other in the progression of GC. Multivariate Cox proportional hazards regression analysis showed that patient age, Shh, Gli1, Cyr61 expression, TNM stage, and tumor differentiation were independent prognostic factors for OS in patients with GC. Shh/Gli1 signaling pathway and Cyr61 are closely related to tumor proliferation, metastasis, survival and angiogenesis, and these promote tumor invasion and metastasis. Although the specific mechanisms still require further in-depth research, these proteins may be promising therapeutic targets in gastric cancer patients.
This study had some limitations. First, the study’s retrospective design and limited sample size may have imputed selection bias; we did not calculate the required sample size for this study. Second, we did not evaluate the progression-free survival of the patients. This is because it is difficult for many patients to come to the hospital when they know the surgery was not successful or they are about to die after the operation is not advanced, or about to die based on the current follow-up conditions at the grassroots level in China. Therefore, PFS data cannot truly reflect the real-world situation of the patients. Third, the determination of Shh, Gli1, and Cyr61 expression via immunohistochemistry using various antibodies makes it difficult to clearly define the threshold. Further large research is needed to validate our findings.
Conclusion
Our findings suggest that Shh, Gli1, and Cyr61 proteins expression have positive intercorrelations. Gastric cancer patients with high levels of Shh, Gli1, and Cyr61 proteins have a poor prognosis. Shh, Gli1, and Cyr61 proteins are independent risk factors for the prognosis of patients; however, combining these proteins and TNM staging may better predict three-year postoperative OS of gastric cancer patients.
Abbreviations
American Joint Cancer Committee
area under the curve
diaminobenzidine
epithelial-mesenchymal transition
Gastric cancer
mitogen-activated protein kinase
overall survival
phosphoinositide 3-kinase
programmed death-ligand 1
sonic hedgehog
Footnotes
Author Contributions
Xiaoling Quan, Jun Qian, Jiaxi Yao designed this work. Zhenming Zhang, Yujie Qin wrote the manuscript and make statistical analysis. Xin Gai, Qiling Tian, Yaqiong Guo performed the immunohistochemical staining. Jun Qian and Jiaxi Yao performed the data review. All authors have read and approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (2019PT320005), Open Fund Project of NHC Key Laboratory of Diagnosis and Therapy of Gastrointestinal Tumor (NLDTG2020015), 12th Science and Technology Innovation Project (No:135).
Ethical Approval
Ethical ratification was authorized by the Ethics Committee of Hexi University Affiliated Zhangye People’s Hospital with the approval number B2019-012 and written informed consent was obtained from all patients.
