Abstract
Transporters of the solute carrier family 25 (SLC25) regulate the intracellular distribution and concentration of nucleotides, amino acids, dicarboxylates, and vitamins within the mitochondrial and cytoplasmic matrices. This mechanism involves changes in mitochondrial function, regulation of cellular metabolism, and the ability to provide energy. In this review, important members of the SLC25 family and their pathways affecting tumorigenesis and progression are elucidated, highlighting the diversity and complexity of these pathways. Furthermore, the significant potential of the members of SLC25 as both cancer therapeutic targets and biomarkers will be emphasized.
Keywords
Introduction
Cancer is among the most daunting health challenges, and it always affects humanity as it causes millions of deaths annually. 1 The tremendous progress made towards the knowledge of the molecular basis of cancer and the development of targeted therapies is still not sufficient, as the disease is complex. Thus, the search for new therapeutic targets and prognostic markers is a process that never ends.2,3 In this instance, mitochondria, frequently considered as the cellular powerhouses, have become key players in cancer biology. They profoundly affect the substance metabolism and energy supply of cells through multiple metabolic pathways such as tricarboxylic acid cycle, fatty acid oxidation, oxidative phosphorylation and so on, affecting cell death, proliferation and metastasis. 4 The solute carrier family 25 (SLC25) is an element related to mitochondrial function, and its role in ion transport has attracted the attention of researchers.
The Mitochondrial Carrier Family was initially discovered in S. cerevisiae and is currently the largest human solute transport protein family, with 53 members (Figure 1).
5
The SLC25 family is mostly present in the inner mitochondrial membrane and transports fatty acids, amino acids, carboxylic acids, inorganic ions, and other metabolic products. It is highly conserved and features a three-domain structure with six transmembrane α-helices, along with a 3-fold repeated motif of hydrophobic and charged residues. This symmetry serves as the foundation for using analogous transport channels.
6
The primary mechanism by which the SLC25 family influences the cellular phenotype may be through the provision of a pathway that links cytoplasmic solutes and the mitochondrial matrix. This allows the SLC25 family to affect the distribution and concentration of transported substrates, thereby influencing the energy metabolism that occurs in the mitochondria. For example, SLC25A4 (ANT1) is responsible for transporting ADP and ATP across the mitochondrial inner membrane, which directly affects energy output from oxidative phosphorylation.
7
Other family members are involved in many key metabolic pathways, such as the citric acid cycle and iron metabolism.8,9 In addition, the SLC25 family may play a role in regulating downstream metabolic states by affecting WNT signaling pathways.10,11 Given the central role of mitochondria in cellular metabolism and the altered metabolic state of cancer cells, the SLC25 family provides a unique perspective for exploring cancer pathophysiology. Physiological Role of Solute Carrier Family 25 (SLC25). AAC: ADP/ATP carrier; APC: ATP-Mg/P(i) carrier; PIC: phosphate carrier; AGC: aspartate/glutamate carrier; GC: glutamate carrier; ORC: ornithine carrier; OGC: oxoglutarate carrier; DIC: dicarboxylate carrier; ODC: oxodicarboxylate carrier; CAC: carnitine/acylcarnitine carrier; MFRN: mitoferrin; DNC: deoxynucleotide carrier; SAMC: S-adenosylmethionine carrier; UCP: uncoupling protein; GSH: glutathione; 2-OA: 2-Oxyadipate; α-KG: α-ketoglutaric acid; GLU: glutamic acid; ASP: aspartate; TCA cycle: tricarboxylic acid cycle; PDH: pyruvate dehydrogenase; ALA: 5-aminolevulinic acid; mPTP: mitochondrial permeability transition pore.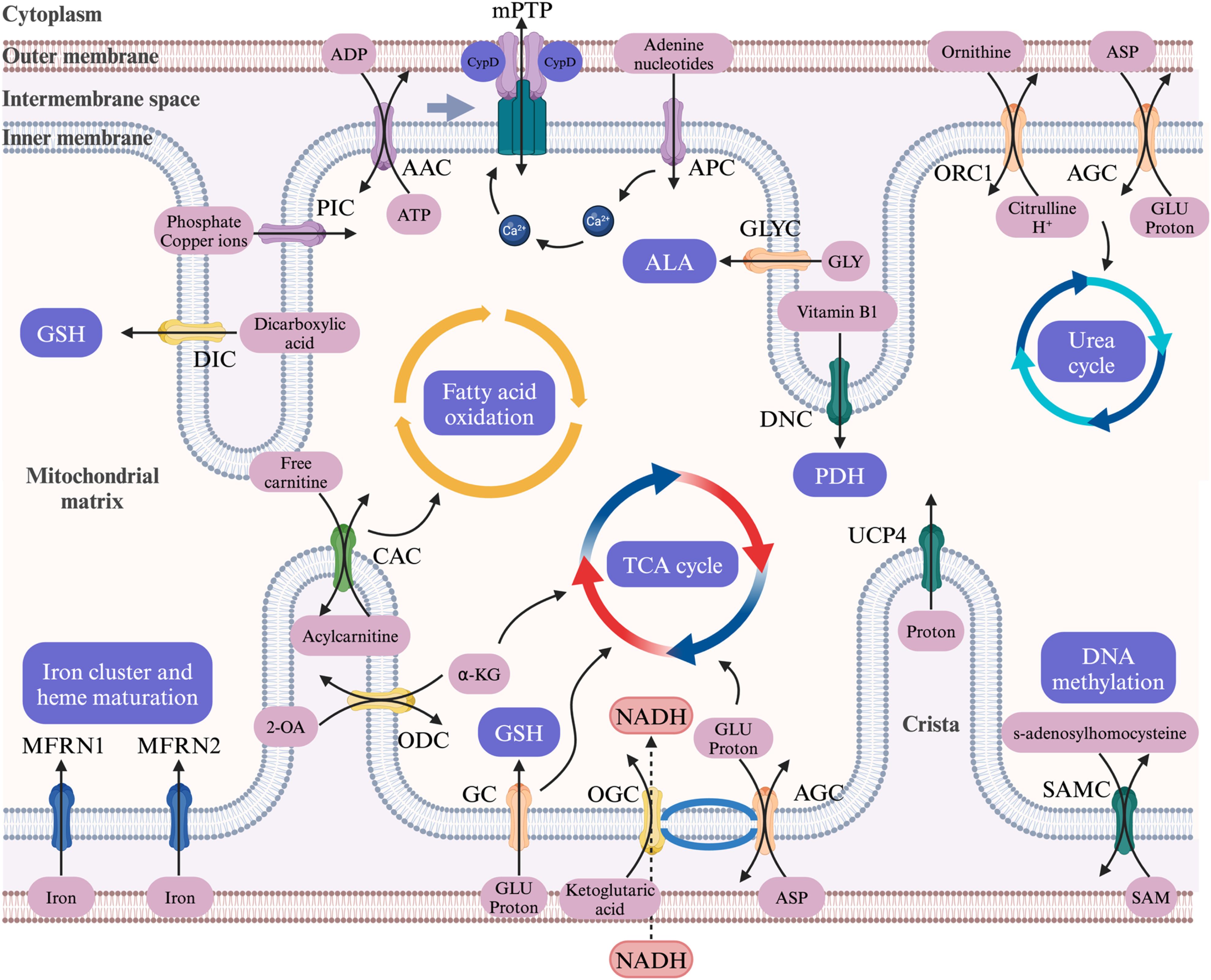
The last few years have seen significant developments in the understanding of the complicated interactions between SLC25 family members and cancer. This implies that disturbances in the expression and function of these transporters may be implicated in cancer formation. 12 For instance, a few SLC25 transporters promote cancer cell survival and increase cell growth by maintaining its metabolism. 13 However, other members of this family may inhibit tumor growth, and their deletion or loss of function can result in the progression of the cancer. 12 Other members are involved in the resistance of tumors to specific treatments. 14 Overall, the SLC25 family members are gaining importance in cancer treatment and prognosis because of their intricate associations with cancer. The expression levels of certain members of the SLC25 transporter family are associated with patient prognosis, tumor response to therapy, and disease recurrence. These findings indicate that they can be used as biomarkers for predicting cancer treatment outcomes. In addition, the regulation of SLC25 transporters may provide an opportunity for therapeutic intervention by targeting cancer cell metabolism and overcoming drug resistance, which is particularly challenging and rewarding in cancer treatment. Such dual responsibilities of the SLC25 family uncover cancer cell metabolic weaknesses and underline their remarkable role in oncology.15,16
Important Members of the SLC25 Mitochondrial Carrier Family and the Cancer Types in Which They May be Involved.

AAC: ADP/ATP carrier; ANT: adenine nucleotide translocator; APC: ATP-Mg/P(i) carrier; AGC: aspartate/glutamate carrier; GC: glutamate carrier; GLYC: Glycine Carrier; ORC: ornithine carrier; OGC: oxoglutarate carrier; ODC: oxodicarboxylate carrier; CAC: carnitine/acylcarnitine carrier; MFRN: mitoferrin; DNC: Deoxynucleotide carrier; SAMC: S-adenosylmethionine carrier; PIC: Phosphate carrier
Members of the SLC25 Family Related to Nucleotide Transport
Adenine Nucleotide Translocator (ANT)
In aerobic eukaryotic cells, ATP is primarily produced through oxidative phosphorylation in the mitochondria. ATP is exported to the cytoplasm via a specialized transporter, the ADP/ATP carrier (AAC), which provides energy for cells and mediates the entry of ADP into the mitochondrial matrix for ATP synthesis.7,17 These are heavy-duty and widespread transmembrane proteins from the mitochondria, also known as adenine nucleotide translocases. AAC comprises four known isoforms, AAC1, AAC2, AAC3, and AAC4, encoded by SLC25A4, SLC25A5, SLC25A6, and SLC25A31, respectively, all of which function in a tissue-dependent manner to meet various cellular energy needs.18,19 Beyond its role in the ADP/ATP antiport, it functions as a proton transporter involved in mitochondrial uncoupling and regulates the activity of the mitochondrial permeability transition pore (mPTP). 20
Recently, SLC25A4 (ANT1), a voltage-dependent anion channel (VDAC), and cyclophile D (CypD) were reported to make up a mPTP (mitochondrial permeability transition pore).21,22 The increased permeability of the inner membrane results from the quick opening of the mPTP, which allows the flow of molecules up to 1500 Da. This results in the formation of a swollen matrix and a collapse of the mitochondrial membrane potential (Δψm), which is hitherto known to regulate mitochondrial function and mediate apoptosis or necrosis. 7 Hirsutine promotes the interaction between ANT1 and CypD through the dephosphorylation of glycogen synthase kinase-3β (GSK3β); through this process, the ATP is depleted, which leads to apoptosis and subsequently inhibits tumor growth in A549 xenograft mice. 23 Furthermore, SLC25A4 upregulation is implicated in the suppression of glioblastoma (GBM) cell proliferation and migration of GBM cells by inhibiting the transcription factor REST (repressor element 1-silencing transcription factor). 24 In addition to this, overexpression of SLC25A4 has been reported to induce apoptosis in breast cancer cells both in vitro and in vivo. 25 The loss of SLC25A4 has been observed as a progressive event in cervical and prostate cancers. 25 In summary, overexpression of SLC25A4 has been shown to markedly reduce tumor cell growth, viability, and migration potential in various cancers, confirming its role in inhibiting tumor growth. However, recent studies have suggested that SLC25A4 is involved in the cGMP/PKG pathway, and the activation of this pathway is essential for promoting gastric cancer cell proliferation, metastasis, and chemotherapy resistance. 26
SLC25A5 is highly expressed in various cancers and may contribute to tumor development in multiple ways.27,28 High SLC25A5 expression in non-small cell lung cancer, ovarian cancer, and neuroblastoma is often associated with poor prognosis.14,29,30 A deficiency in SLC25A5 has been noted in mesothelioma, where the oxidative stress implicated by this defect may play a significant role in cancer progression. 31 However, it upregulates programmed cell death-related signals and inhibits cell proliferation by suppressing the EGFR and ERK-MAPK signaling pathways. 32
Although the specific mechanisms by which ANT promotes or inhibits tumor growth in various cancers require further investigation, its potential as a prognostic biomarker for numerous cancer types is increasingly being recognized. For instance, SLC25A4 has been identified as a biomarker for renal cell carcinoma and SLC25A5 is a crucial component of the prognostic model for cervical cancer.33,34
ATP-Mg/P(i) Carrier (APC)
ATP-Mg/Pi carriers can exchange phosphate for adenine nucleotides coupled with magnesium or protons in an electrically neutral manner. Unlike ANT, which does not alter the mitochondrial matrix adenosine nucleotide pool, APC transporters can achieve the net import and export of adenine nucleotides. This functionality allows them to indirectly influence adenosine nucleotide-dependent metabolic pathways in mitochondria, thereby adapting to changes in cellular energy demands.13,35 Four subtypes of the APC transporter are observed. Except for APC4 (SLC25A41), APC1 (SLC25A24), APC2 (SLC25A23), and APC3 (SLC25A25) are calcium-regulated.35,36 They share a similar structure, consisting of a C-terminal domain with six transmembrane helices homologous to mitochondrial carrier proteins and an N-terminal domain featuring a Ca2+-binding EF-hand, which confers Ca2+ sensitivity to the transporter. 37
APC1 (SLC25A24), also known as short Ca(2+)-binding mitochondrial carrier-1 (SCaMC-1), is overexpressed in various neoplastic conditions, notably breast carcinoma, colorectal cancer, and other forms of malignancy.38,39 For instance, in colorectal cancer, SLC25A24 is considered a potential tumor suppressor because its reduced expression appears to accelerate tumor malignancy. 40 Furthermore, studies have indicated that SLC25A24 contributes to fibroblast recruitment via tumor bud-derived C–C chemokine ligand 5 (CCL5) and plays a role in constructing the tumor microenvironment at the frontier of colorectal cancer (CRC) invasion. 41
Several studies have explored the mechanisms through which SCaMC-1 enhances cancer cell survival. Ca2+ in the cytoplasmic matrix can initiate the uptake of ATP/ADP by SCaMC-1, leading to an increase in adenine nucleotides within the mitochondria. This increase enhances the Ca2+-buffering capacity and ultimately mediates mitochondrial permeability transition (mPT) desensitization by Ca2+, thus improving cancer cell survival. 13 The enhanced Ca2+-buffering capacity of SLC25A24 protects cancer cells. 42 However, knocking out this transporter significantly reduces the calcium buffering capacity in the mitochondria, rendering cells sensitive to necrotic death mediated by mPT under oxidative stress and calcium overload. Under conditions of Ca2+ overload or oxidative stress, the formation of large non-specific pores in the inner mitochondrial membrane, known as mPTP, triggers mPT and leads to cell death. 13
Under ischemic conditions, the overexpression of SCaMC-3 (SLC25A23) serves as a survival mechanism by which cancer cells mitigate oxidative stress and avoid cell death induced by excessive Ca2+13,. 43 Concurrently, the combination of local hypoxia and increased SCaMC-3 expression during the early development of hemangiomas may safeguard cells against oxidative stress. 44 However, SLC25A23 is not merely a promoter of cancer; it is a silent target of H3K27me3 modification in epigenetics and is potentially involved in the mechanism by which the Enhancer of Zeste Homolog 2 induces astrocytoma. Typically, a low expression correlates with a lower survival rate. 45 In diffuse large B-cell lymphoma, the relationship between SLC25A23, MLH1, and p53 indicates that overexpression of SLC25A23 may be linked to a better prognosis. 46 Previous studies have underscored the beneficial role of SLC25A23 in regulating calcium uptake, which is critical for managing reactive oxygen species (ROS) and cell death. In cancer, the downregulation of calcium homeostasis proteins may lead to drug resistance.47,48 Therefore, targeting these calcium transporters to augment the apoptotic capacity of malignant cells may be an effective strategy for treating related cancers.
The ability of APC transporters to regulate the calcium-buffering capacity and influence mPTP activity underscores their significance in protecting cancer cells from apoptosis and necrosis induced by oxidative stress and calcium overload, which are essential for cell survival under stressful conditions. In addition, it regulates the distribution of nucleotides inside and outside the mitochondria, together with adenine nucleotide transporters, thus affecting energy metabolism and cell survival.
Members of the SLC25 Family Related to Amino Acid Transport
Aspartate/Glutamate Carrier (AGC)
The aspartate/glutamate antiporter, encompassing AGC1 and AGC2, and encoded by SLC25A12 and SLC25A13, respectively, mediates the efflux of aspartic acid and the influx of glutamate and protons into the mitochondria.49,50 These carriers are integral components of the malate-aspartate shuttle and are involved in maintaining the NAD+/NADH ratio in the cytoplasm and mitochondria. Aspartic acid and glutamic acid are interconvertible with key metabolic intermediates, such as those in the tricarboxylic acid cycle, thereby influencing the cycle’s efficiency and energy output. 51 The malate–aspartate shuttle primarily transports the reductive hydrogen of cytoplasmic NADH into the mitochondria, participates in the shuttle metabolic pathway of oxidative phosphorylation, and ultimately produces ATP to supply cellular energy. 52
Aspartic acid output mediated by AGC1 (SLC25A12) is crucial for supporting tumor cell proliferation and redox homeostasis, and the knockdown of AGC1 can significantly inhibit the growth of allograft tumors. 53 Furthermore, researchers have reported that AGC1 deficiency results in reduced cellular solute aspartic acid levels and an increased dependence on glutaminase. Therefore, combination cancer therapies targeting aspartic acid and glutamine metabolism may have synergistic effect. 53
Citrin, encoded by SLC25A13, is upregulated in many types of cancer and is essential for replenishing NAD+ for glycolysis and NADH for oxidative phosphorylation. Citrin overexpression in cancer cells boosts the supply of cytoplasmic aspartic acid, potentially disrupting the balance between cytoplasmic metabolites. This disruption can lead to increased CAD (carbamoylphosphate synthetase 2, aspartate transcarbamylase, dihydroorotase) activity, enhanced pyrimidine biosynthesis, and metabolic reprogramming, altering energy production, thereby promoting tumor cell proliferation.54,55 Thus, targeting citrin may constrain cancer progression by limiting mitochondrial respiration and glycolysis. Germline citrin mutations result in a rare urea cycle disorder known as citrullinemia type 2 (CTLN-II), which is associated with hepatocellular carcinoma (HCC) and may contribute to its development.56-58 Additionally, studies have indicated that high SLC25A13 expression is associated with a poor prognosis in neuroblastoma and colorectal cancer.54,59,60
Although the functions of the two aspartate/glutamate antiporters are largely similar, there are differences in their tissue expression. SLC25A12 is expressed in the brain, heart, and skeletal muscles but is typically inactive in normal liver tissue. However, studies have indicated that SLC25A12 is reactivated in the HCC of the HepG2 cell line via histone acetylation and CREB recruitment. Conversely, SLC25A13 is downregulated in the same cell line, potentially owing to the tumor-suppressive role of forkhead box protein A2(FOXA2).61-63 Additionally, SLC25A12 silencing has been linked to G1 cell cycle arrest in HepG2 cells, which significantly impacts cell proliferation. 61 AGC2 does not appear to be essential for cancer cell proliferation, as evidenced by the fact that silencing the SLC25A13 gene does not affect HCC cell proliferation, silencing the SLC25A12 gene markedly influences the growth and migration of hepatoma cells, highlighting a unique role for AGC1 in HCC.61,62
Glutamate Carrier (GC)
Mitochondrial inner membrane glutamate transporters, including GC1 and GC2, encoded by SLC25A22 and SLC25A18, respectively, facilitate the transport of glutamate from the cytoplasm to the mitochondrial matrix, accompanied by proton influx. 64 Glutamate is oxidized to alpha-ketoglutarate (α-KG) and ammonia, aiding in maintaining ammonia balance, acting as a critical intermediary metabolite in the tricarboxylic acid cycle, and contributing to energy supply. 51 Moreover, it may play a role in the synthesis of the antioxidant glutathione (GSH). 65
Several studies have reported that SLC25A22 promotes the progression of KRAS-mutant CRC through various mechanisms. KRAS, a critical oncogene in CRC, upregulates the expression of SLC25A22, which in turn enhances the biosynthesis of succinate from α-KG in an SLC25A22-dependent manner. The accumulation of succinate disrupts the activity of α-KG-dependent epigenetic regulators, inhibits the expression of a WNT repressor, procadherin, and ultimately activates WNT/β-catenin signaling. This pathway targets genes associated with CRC stemness, such as LGR5, thereby promoting the development of colorectal tumors. 66 Furthermore, studies have indicated that patients with CRC and elevated SLC25A22 expression have shorter survival times than those with lower levels. SLC25A22 directs glutamine-derived carbon towards aspartic acid biosynthesis through glutamine decomposition. Aspartic acid then stimulates the growth and metastasis of KRAS-mutant CRC via two distinct pathways: the first involves the GOT1-catalyzed conversion of aspartic acid to oxaloacetic acid, which is essential for cell survival and growth, while the second pathway is mediated by asparagine synthetase, promoting metastasis. 67 Aspartic acid, a component of pyrimidine synthesis, is crucial for cell proliferation. 67
SLC25A22 also mediates other mechanisms of resistance to tumor cell death. Ferroptosis is a type of oxidative cell death driven by lipid peroxidation. Downregulation of SLC25A22 expression is linked to increased sensitivity to ferroptosis.68,69 SLC25A22-dependent NADPH synthesis protects against oxidative damage during ferroptosis by facilitating the production of GSH, a hydrophilic antioxidant. 70 Additionally, SLC25A22 enhances the expression of stearoyl-CoA desaturase in pancreatic ductal adenocarcinoma cells (PDAC) in an AMP-activated protein kinase-dependent manner, leading to the production of monounsaturated fatty acids (MUFA).68,71 These fatty acids reduce lipid peroxide accumulation, thereby conferring resistance to ferroptosis in PDAC. In a therapeutic context, SLC25A22 confers radiation resistance to GBM by altering glutamate metabolism. In radiation-resistant GBM cells, the mitochondrial bidirectional glutamate transporter, SLC25A22, is upregulated and exhibits unidirectional transport from the mitochondria to the cytoplasm, resulting in the accumulation of cytoplasmic glutamate. This accumulation enhanced GSH production and proline synthesis in radiation-resistant GBM cells. Increased GSH levels protect cells from the damaging effects of ROS induced by ionizing radiation, whereas increased proline, a rate-limiting substrate of collagen biosynthesis, induces extracellular matrix remodeling, contributing to the invasive phenotype of GBM. 65
Glycine Carrier (GLYC)
Glycine Carrier (GLYC), encoded by SLC25A38, is a mitochondrial glycine transporter that introduces glycine into the mitochondrial matrix. During the initial stages of heme biosynthesis, glycine and succinyl coenzyme A are crucial for the synthesis of 5-aminolevulinic acid (ALA) within the mitochondrial matrix. 72
SLC25A38, positioned upstream in the HIF signaling pathway, has been shown to inhibit the production of angiogenic factors by reducing the levels of transcriptional coactivators and HIF-1α protein, thereby impeding angiogenesis and tumor progression. 73 The same study on uveal melanoma supported the idea that it may act as a tumor suppressor and participate in microvascular invasion.73-75 However, in endometrial carcinoma, estrogen and succinate enhance the expression of ALAS1 and SLC25A38 via the ER β/NCOA1 axis. Subsequently, ALAS1 catalyzes the reaction between succinyl-CoA and glycine to produce heme, leading to the increased proliferation and invasiveness of endometrial carcinoma cells.76,77
Ornithine Carrier (ORC)
Ornithine carrier1 and 2, encoded by SLC25A15 and SLC25A2, respectively, catalyze the exchange between ornithine in the cytoplasm and citrulline and hydrogen ions in the mitochondria. 78 Protons neutralize the positive charge of ornithine, resulting in electrically neutral transport. In addition, it facilitates the transport of lysine, arginine, and ornithine across the mitochondrial inner membrane. 79 Moreover, it plays a crucial role in this metabolic pathway by bridging cytoplasmic and mitochondrial reactions within the urea cycle. 80
SLC25A15 is upregulated in various cancers, including gastric cancer, bladder urothelial carcinoma, and melanoma, and is associated with cancer progression and a poor prognosis.81-84 For example, a study on prostate cancer revealed that SLC25A15 is a downstream target of circFOXO3. By activating the circFOXO3/miR-29a-3p/SLC25A15 axis pathway, circFOXO3 greatly increases SLC25A15 expression, significantly inhibits apoptosis, and advances the development of prostate cancer. 85
Amino acid transport-related transporters similarly promote metabolic reprogramming in cancer cells by regulating the exchange of key metabolites such as aspartic acid, glutamic acid, and glycine. These carriers help meet the metabolic needs of rapidly proliferating tumor cells. This process involves maintaining redox homeostasis, supplying NAD + for glycolysis and oxidative phosphorylation, and supporting the biosynthesis of pyrimidines and other macromolecules essential for cell growth, thereby underscoring the critical role of transporters in meeting the energy and biosynthetic demands of cancer cells.
Members of the SLC25 Family Related to Dicarboxylate Transport
Oxoglutarate Carrier (OGC)
The OGC encoded by SLC25A11 facilitates electrical neutral exchange between 2-ketoglutarate and other dicarboxylates and is integral to the malate-aspartate shuttle. 86 Researchers have identified its crucial role in ATP production in cancer cells as it regulates the transport of NADH from the cytoplasm to the mitochondria. Additionally, it is transported from the cytoplasm to the mitochondria, maintaining GSH levels and thus limiting ROS production, which protects tumor cells from oxidative damage. 87
In a study on HCC, OGC expression was upregulated to preserve cellular GSH levels against oxidative stress. However, its overexpression counteracts the impairment of mGSH transport caused by mitochondrial cholesterol accumulation. 88 This contradictory maintenance of mGSH levels enables OGC to support cell growth and tumor progression by facilitating hypoxia tolerance and stabilizing mitochondrial membranes through the inhibition of cholesterol in mitochondrial outer membrane permeabilization and cell death. 88 In another study on non-small-cell lung cancer and melanoma cells, it was demonstrated that the oxoglutarate carrier SLC25A11 plays a vital role in transporting NADH from the cytoplasm to the mitochondria as malate, which is crucial for ATP production in cancer cells. 89 Therefore, blocking SLC25A11 expression may curb cancer growth by reducing ATP production. However, in other cases, OGC may exhibit tumor suppressor effects. SLC25A11 interacts with SMIM26 and acylglycerol kinase (AGK) in clear cell renal cell carcinoma to form a complex (SMIM26-SLC25A11-AGK) that enhances mitochondrial GSH, ATP, and the oxygen consumption rate. This promotes mitochondrial oxidative phosphorylation and inhibits AGK/AKT signaling-mediated cancer metastasis. 90 SLC25A11 is considered a tumor suppressor gene in metastatic paragangliomas and pheochromocytomas, with mutations linked to an increased risk of metastatic disease. 91
Dicarboxylate Carrier (DIC)
Dicarboxylate Carrier (DIC), encoded by SLC25A10, catalyzes the transport of dicarboxylates, such as malonates, succinates, inorganic sulfur anions, and phosphates, across the mitochondrial inner membrane. 92 Similar to SLC25A11, it is involved in maintaining antioxidant GSH to protect against oxidative stress.
Studies have shown that chronic circulatory hypoxia upregulates the expression of mitochondrial dicarboxylate carriers SLC25A10 and SLC25A11 in hypoxia-tolerant cancer cells in vitro. This upregulation helps to maintain metabolic reprogramming and antioxidant defense in these cells.93,94 Genetic or pharmacological defects in SLC25A10 result in the loss of mitochondrial membrane potential, leading to the formation of mitochondrial ROS and triggering endogenous apoptosis due to the depletion of GSH. Consequently, inhibition of SLC25A10 can mitigate the increased radiation resistance caused by chronic circulatory hypoxia, both in vitro and in vivo, by disrupting the enhanced antioxidant capacity of hypoxia-tolerant cancer cells. This inhibition enhances the efficacy of chemotherapy and radiotherapy. 95 Additionally, SLC25A10-dependent malate transport may influence the regeneration of isocitrate and NADPH by affecting citric acid transport across the mitochondrial inner membrane and indirectly by affecting mitochondrial redox homeostasis.8,96
Oxodicarboxylate Carrier (ODC)
Oxodicarboxylate Carrier (ODC), encoded by SLC25A21, is responsible for the reverse exchange of 2-oxadipic acid (2-OA) and α-ketoglutarate (α-KG) across the mitochondrial inner membrane. 97 These substrates play a critical role in various intracellular metabolic pathways. 2-Oxyadipate (2-OA) enhances amino acid metabolism and urea synthesis, while α-KG is vital for the tricarboxylic acid cycle. At the same time, as a nitrogen transporter, α-KG can promote the synthesis of certain amino acids.98,99 SLC25A21 is crucial for the survival and progression of KRAS-mutant CRC. In these cancers, ODC expression is downregulated, and a decrease in SLC25A21 expression correlates with poor patient survival. This association is because it down-regulates the efflux of glutamine-derived α-KG in mitochondria, thereby increasing glutamine hydrolysis and enhancing downstream oxidative decarboxylation and GTP production. This process contributes to the sustained activation of KRAS in the KRAS mutant, CRC. 100 Furthermore, blocking α-KG outflow via SLC25A21 deletion reduces α-KG-dependent DNA demethylase activity, further diminishing SLC25A21 expression. Overexpression of SLC25A21 sensitizes KRAS-mutated CRC cells to cetuximab, highlighting its therapeutic potential. 100 In esophageal squamous cell carcinoma, the abnormal transfer of 2-oxo dicarboxylic acids by SLC25A21 disrupts the degradation pathways of lysine and tryptophan, leading to the accumulation of 2-oxanedioic acid and quinolinic acid. 98 2-OA is converted into acetyl-CoA via DHTKD1 (dehydrogenase E1, transketolase domain containing 1) and glutaryl-CoA dehydrogenase in the mitochondria, influencing mitochondrial metabolic activity and cell proliferation. 95 Recent findings indicate that low SLC25A21 expression predicts a poor prognosis in acute myeloid leukemia. 101 SLC25A21 serves as a marker for monitoring changes in methylation during glioma progression. 102
These amino acid transporters play crucial roles in enhancing the metabolic flexibility of cancer cells. For instance, OGC (SLC25A11) transports metabolites necessary for ATP production, such as NADH, to cater to the high energy demands of tumor cells. Moreover, these transporters are integral to the antioxidant defense mechanisms in cancer cells. By facilitating the movement of GSH and influencing the mGSH pool, they help reduce the production of ROS and shield tumor cells from oxidative damage, which is vital for their survival and proliferation, particularly under stressful conditions such as hypoxia. Consequently, miRNAs have emerged as potential therapeutic targets in cancer.
Members of the SLC25 Family Related to Fatty Acid Transport
Carnitine/Acylcarnitine Carrier (CAC), which is encoded by SLC25A20, mediates the electroneutral exchange of acylcarnitine with free carnitine across the mitochondrial membrane. 103 This transporter is crucial for the mitochondrial oxidation pathway as it transfers fatty acids in the form of acylcarnitine to the mitochondrial matrix, where they are activated by carnitine palmitoyltransferase 2 for fatty acid β oxidation. 104 With adequate oxygen, these fatty acids are oxidized to CO2 and H2O, releasing substantial energy. 105
In prostate cancer, fatty acid oxidation facilitated by SLC25A20 is a critical energy source, and CAC overexpression significantly supports cell survival. 106 However, its expression is often reduced in HCC cells, leading to diminished fatty acid oxidation, which in turn promotes the growth and metastasis of HCC. 107 Studies have indicated that SLC25A20 can inhibit the growth and metastasis of HCC in vitro and in vivo by blocking the G1-S cell cycle, EMT (epithelial-mesenchymal transition), and inducing apoptosis. 107 Additionally, the variable expression of SLC25A20 in low-grade and high-grade gliomas assists in the differential diagnosis. 108 Moreover, the expression of SLC25A20 in cancer may be extensively regulated by miRNA.106,107
The function of SLC25A20 in the service of acyl carnitine transport is of great importance to the mitochondrial fatty acid β-oxidation process and thus the energy substrate of malignant cells. The multiple manifestations of this transporter in diverse cancers and its regulation by miRNAs underscore the possibility of using it as a diagnostic tool and as a therapeutic target.
Members of the SLC25 Family Related to Iron Ion Transport
The SLC25 family includes two ferrous ion transporters, SLC25A37 (MFRN-1) and SLC25A28 (MFRN-2), which are key to the transfer of iron into the mitochondria, required for the synthesis of heme and iron-sulfur clusters. They, in turn, are important cofactors for enzymes implicated in the functioning of mitochondria to produce ATP, which is responsible for the maintenance of redox balance.109,110 SLC25A37 is predominantly located in red blood cells; however, its expression levels in other tissues are considerably lower. SLC25A28 is more widely distributed. 109
Research on the antitumor effects of PINK1 (PTEN-induced putative kinase 1) and PRKN/PARK2 (parkin RBR E3 ubiquitin protein ligase) has highlighted the significance of mitochondrial iron transport in suppressing pancreatic tumorigenesis. 111 PINK1- and PRKN-mediated autophagic degradation of SLC25A37 and SLC25A28 prevented iron accumulation in the mitochondria. This process supports the activation of inflammasomes, the release of HMGB1, and subsequent immune checkpoint expression, thereby inhibiting pancreatic tumor growth. 112 Further studies have shown that degradation of SLC25A37 and SLC25A28 via the PINK1-PARK2 pathway triggers a HIF1α-dependent Warburg effect and an AIM2-dependent inflammasome activation in tumor cells. Moreover, AIM2-mediated HMGB1 release induces the expression of CD274/PD-L1. 111 Low PARK2 expression and high levels of SLC25A37 and AIM2 are closely associated to a poor prognosis in patients with pancreatic cancer. 111 SLC25A37 and SLC25A38 exhibit low expression in HCC. 113 SLC25A37 serves as a critical target of miR-7 and participates in its tumor-suppressive effects in rhabdomyosarcoma. 114
In addition, iron-mediated apoptosis may play a significant role in the influence of iron transporters on tumor cells. 9 Research has shown that the BRD7-P53-SLC25A28 axis is crucial for regulating iron apoptosis in hepatic stellate cells. BRD7 overexpression promotes iron apoptosis in hepatic stellate cells, potentially because increased BRD7 expression facilitates the mitochondrial translocation of p53 through direct binding with the p53 N-terminal transactivation domain. Subsequently, mitochondrial p53 forms a complex with SLC25A28, enhancing its activity. This interaction leads to the abnormal accumulation of redox-active iron and hyperactivation of the electron transfer chain, ultimately inducing iron apoptosis. 115 This mechanism may inhibit the progression of advanced HCC fibrosis. 115 However, SLC25A22 inhibits iron-induced apoptosis-mediated tumor suppression. SLC25A22-dependent NAPDH synthesis shields PDAC cells from iron-induced cell death by facilitating the production of GSH, a crucial hydrophilic antioxidant. Moreover, SLC25A22 stimulates the expression of stearoyl-CoA desaturase in an AMP-activated protein kinase-dependent manner in PDAC cells, leading to the production of iron-resistant monounsaturated fatty acids. 68
Additionally, in the therapeutic context, lysosomal iron release combined with SLC25A28-dependent mitochondrial iron uptake triggers PDT-mediated and iron-dependent mitochondrial dysfunction, resulting in cell death. This suggests its potential utility for the treatment of head and neck cancer. 116
These two iron transporters, particularly SLC25A28, appear to play crucial roles in inhibiting tumor initiation and progression. They are involved in the activation of inflammasomes regulated by PINK1 and PRKN, the expression of immune checkpoints, and the induction of iron apoptosis through iron accumulation.
Members of the SLC25 Family Related to Vitamin Transport
Deoxynucleotide Carrier (DNC)
The DNC encoded by SLC25A19 facilitates the transport of thiamine diphosphate (vitamin B1) into the mitochondria. Thiamine serves as an essential cofactor for critical metabolic enzymes such as transketonolase, α-ketoglutarate dehydrogenase, and pyruvate dehydrogenase. 117 These enzymes are vital for cellular metabolism; for instance, pyruvate dehydrogenase catalyzes the transfer of hydrogen in pyruvate molecules within the mitochondria, converting pyruvate to acetyl-CoA. This reaction acts as a link in mitochondrial metabolism, connecting the aerobic oxidation of sugars to the tricarboxylic acid cycle and providing energy to the cell. 118
Studies have demonstrated that the upregulation of SLC25A19 significantly enhances the mitochondrial levels of vitamin B1, indicating its potentially critical role in supporting cancer cell metabolism. 119 The unique influence of SLC25A19 on the activity of key enzymes that indirectly affect the TCA (tricarboxylic acid cycle) and, consequently, lipid metabolism and energy supply has led researchers to speculate on its important role in prostate cancer. 120 In breast cancer, SLC25A19 is upregulated and potentially regulated by oncogenic microRNA-155 (miR-155). 121
S-Adenosylmethionine Carrier (SAMC)
S-Adenosylmethionine Carrier (SAMC), encoded by SLC25A26, facilitates the exchange of cytoplasmic S-adenosine-methionine (SAM) and mitochondrial S-adenosine-homocysteine, both of which are essential for intracellular methylation processes. 122 DNA methylation, a chemical modification of DNA, can alter DNA conformation, stability, and interactions with proteins without altering the DNA sequence, thereby regulating gene expression. 123
SAM is primarily generated in the cytoplasm by the methionine cycle. SLC25A26 enhances mitochondrial SAM levels and utilization, leading to the hypermethylation of mtDNA, reduced expression of mitochondrial respiratory complex subunits, and decreased mitochondrial ATP production. 122 For instance, studies on cervical cancer have shown that SLC25A26 acts as a tumor suppressor gene, and its overexpression in CaSki cells increases mitochondrial SAM availability, promoting mtDNA hypermethylation, which results in decreased expression of respiratory complex subunits and reduced mitochondrial ATP and cytochrome C release.122,124 Furthermore, increased SAM transport to the mitochondria impairs the methionine cycle, leading to homocysteine accumulation at the expense of GSH, which is significantly depleted. These changes halted the cell cycle in the S phase, induced apoptosis, and enhanced the chemosensitivity of CaSki cells overexpressing the SAM vector to cisplatin. 122 Moreover, SLC25A26 overexpression in HCC cells induces senescence by reducing TERT methylation levels. 125 Additionally, alterations in mtDNA methylation caused by rs11706832-regulated SLC25A26 may contribute to the development of low-grade gliomas.126,127
The impact on key metabolic enzyme activities and the extent of mitochondrial DNA methylation are the principle ways in which these vitamin transport-related carriers influence tumor cell viability, reaffirming the diverse pathways in tumor cells affected by transporters.
Other Members of the SLC25 Family
Phosphate Carrier (PIC)
Phosphate Carrier (PIC), encoded by SLC25A3, is an inorganic ion transporter located on the inner mitochondrial membrane that is capable of transporting phosphate or copper ions into the mitochondrial matrix.128,129 This transport pathway affects oxidative phosphorylation and is involved in ATP synthesis, and it plays a critical role in maintaining the mitochondrial matrix copper pool, which is essential for the assembly of the cytochrome C oxidase complex. 129
SLC25A3 is targeted by the carcinogenic factor CircCNST and may significantly contribute to the progression of osteosarcoma, where its high expression is often associated with a poor prognosis. 130 SLC25A3 plays a crucial role in the staging and prognosis of chronic myeloid leukemia. 131
PIC, together with ANT and APC, coordinates the complex balance between the intake and release of molecules essential for ATP production, engages in oxidative phosphorylation, and maintains cellular energy homeostasis. Given the altered metabolic demands of cancer cells, the regulation of ADP-ATP exchange and phosphate transport by SLC25 family members may directly influence energy availability in cancer cells, thereby impacting tumor proliferation and survival. This highlights their potential as targets for therapeutic intervention.
Uncoupling Protein (UCP)
The UCP subfamily belongs to the SLC25 family and includes five subtypes encoded by SLC25A7 (UCP1), SLC25A8 (UCP2), SLC25A9 (UCP3), SLC25A27 (UCP4), and SLC25A14 (UCP5), respectively.132-134 UCP family is responsible for exporting dicarboxylic acid intermediates from the Krebs cycle, exchanging phosphate and a proton across the mitochondrial inner membrane. This process, driven by mitochondrial power, influences glycolysis, glutaminolysis, and the GSH-dependent redox balance. 135 Additionally, it facilitates the continuous export of oxaloacetic acid and related tetracarboxylic acids from the mitochondrial matrix to the cytoplasm, negatively regulating the oxidation of acetyl-CoA through the Krebs cycle, thereby reducing the ATP/ADP ratio and the production of ROS. 136
Although the role of the UCP family of transporters in tumor tissues has not been extensively studied, emerging evidence suggests its significance. In neuroblastoma, UCP4 overexpression may enhance tumor survival by interacting specifically with complex II to increase ATP synthesis and ensure cellular energy supply. 137 Research on breast cancer supports the notion that UCP4 overexpression promotes cell proliferation and inhibits apoptosis and differentiation, likely due to its effects on ROS homeostasis and mitochondrial antioxidants. 138 Furthermore, SLC25A27 (UCP4) has been reported to differentiate endometrial carcinoma from normal endometrium, showing promise as a new biomarker. 139
Studies have reported that SLC25A8 (UCP2) may provide a substrate for glutamine metabolism mediated by oncogenic KRAS mutations by transporting aspartic acid into the cytosol, thus supporting human PDAC genesis. 140 Additionally, the overexpression of SLC25A8 (UCP2) in lung cancer may be related to its impact on the intracellular energy supply and the high energy demands of these cancer cells. 141 SLC25A14 (UCP5) is believed to regulate ROS production and plays a critical role in the pathogenesis of head and neck paraganglioma.142,143
Conclusion
Our study observed that SLC25 family transporters effectively affect the selection of cellular metabolic pathways and mitochondrial metabolic efficiency through a variety of pathways, making them important regulatory nodes of tumor metabolic reprogramming that have a profound impact on tumor growth, spread, metastasis, and response to anti-chemotherapy drugs (Figure 2). Therefore, abnormal expression and function of SLC25 family transporters are closely related to the occurrence and development of tumors. These changes often contribute to the formation of tumor cell-specific metabolic characteristics. For example, the ANT family members that transport nucleotides directly accelerate the production and supply rate of ATP.
19
AGC, GC, and ODC transport of amino acids and dicarboxylates and CACT for transporting carnitine can increase the supply of substrates involved in the tricarboxylic acid cycle, fatty acid oxidation, amino acid cycle, and oxidative phosphorylation.51,52,99,104 Accelerating material metabolism and energy production and providing sufficient energy for tumor cells accelerates the proliferation rate and migration ability of tumor cells. In addition, resistance to killing by tumor cells and enhancement of their survival ability are important mechanisms by which SLC25 family transporters promote cancer. OGC and DIC can resist ROS and oxidative damage caused by radiotherapy and chemotherapy by enhancing the maintenance of GSH levels.87,95 GC1 can resist ferroptosis in cells, and a few transporters are involved in resistance to specific drugs.
70
These studies indicate the great potential of SLC25 family members as anticancer drug targets. In addition to ferroptosis induced by ferrous ion transporters, such as MFRN-2, and apoptosis mediated by ANT family members through the mPTP, the anti-tumor role of the SLC25 family has been indicated.9,23 The influence pathways include regulating the activity of key metabolic enzymes in mitochondrial metabolism and affecting the methylation degree of mitochondrial DNA, which verifies the diversity and complexity of the intrinsic mechanisms of its influence on the occurrence and development of tumors, further increasing the difficulty of research and presenting a great challenge.
125
Substances, signaling pathways, physiological and biochemical processes involved in/regulated by SLC25 family in cancer. AAC: ADP/ATP carrier; ANT: adenine nucleotide translocator; APC: ATP-Mg/P(i) carrier; AGC: aspartate/glutamate carrier; GC: glutamate carrier; GLYC: glycine carrier; ORC: ornithine carrier; OGC: oxoglutarate carrier; ODC: oxodicarboxylate carrier; CAC: carnitine/acylcarnitine carrier; MFRN: mitoferrin; DNC: deoxynucleotide carrier; SAMC: S-adenosylmethionine carrier; PIC: phosphate carrier; REST: repressor element 1-silencing transcription factor; mPTP: mitochondrial permeability transition pore; CTLN-II: citrullinemia type 2; CAD: carbamoylphosphate synthetase 2, aspartate transcarbamylase, dihydroorotase; cGMP: cyclic GMP; CCL5: C–C chemokine ligand 5; HIF: hypoxia-inducible factor; GLU: glutamine; GLS: glutaminase; GSH: glutathione; AGK: acylglycerol kinase; TCA: tricarboxylic acid cycle; α-KG: α-Ketoglutaric acid; EMT: epithelial-mesenchymal transition; FAO: fatty acid oxidation.
Discussion
Recent studies have reported that SLC25 family members may exert their effects on tumor cells indirectly by affecting the state of immune cells in the tumor microenvironment. For example, in a cervical cancer study, SLC25A5 expression was positively correlated with M0 macrophages, CD4+T cells, neutrophils, and dendritic cells, and patients with high SLC25A5 expression generally had a better prognosis. 144 SLC25A5 may play a role by affecting the number of CD8+ cells; however, an inverse correlation was found between CD8 and SLC25A5, with patients in the high-risk group showing higher immune infiltration. 145 SLC25A22 promotes asparagine binding and activates SRC phosphorylation, which in turn recruits myeloid-derived suppressor cells through CXCL1 and inactivates cytotoxic T cells, leading to an immunosuppressive microenvironment and thus facilitating tumor evasion during immune surveillance.10 Besides, the expression of SLC25A11 is positively related to the recruitment of immune cells such as CD8+ T cells, CD4+ T cells, and dendritic cells. 11
For future research directions, several aspects warrant in-depth exploration: a comprehensive examination of the precise mechanisms by which SLC25 members affect carcinogenesis, progression, and response to treatment, which are sensitive aside from the gross effects on the phenotype of cancer cells by modulating mitochondrial metabolism, signal transduction, and energy conversion. In addition, the relationship between SLC25 members and patient survival, treatment response, and disease recurrence is strong; therefore, their chances of being biomarkers for cancer prognosis are high. Finally, the development of therapeutic strategies that modulate the activity of SLC25 transporters to precisely target cancer cell metabolism and address drug resistance represents a major advancement in cancer treatment. As our understanding of the relationship between the SLC25 family and cancer deepens, there will be an increased focus on translating these scientific discoveries into clinical applications, potentially improving the treatment outcomes and quality of life of patients with cancer. Furthermore, studying the complex interactions between mitochondrial function, cancer metabolism, and the SLC25 family is likely to offer new perspectives and strategies for cancer therapy. This research area is replete with challenges and opportunities, meriting sustained focus and thorough investigation.
Footnotes
Acknowledgments
Author Contributions
Conceptualization, R.G. and D.Z.; Methodology, Z.L. and D.L.; Resources, Z.L. and X.Y.; Data Curation, X.Q.; Writing—Original Draft Preparation, R.G.; Writing—Review & Editing, all authors; Visualization, J.Z.; Supervision, Z.L. and C.Q.; Project Administration, D.Y. and X.Y. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was funded by Natural Science Foundation Project of Jiangxi Province [No. 20232BAB206096], Science and Technology Research Project of Education Department of Jiangxi Province [No. GJJ2200112] and Science and Technology Project of Health Commission of Jiangxi Province [No. 202210480].
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
