Abstract
Objectives
Radiation-induced oropharyngeal injury is a dose-limiting toxicity in head and neck cancer patients. Delineation of the “oropharyngeal mucosa” and limiting its dose to spare the oropharynx was investigated.
Methods
In this retrospective study, computed tomography imaging from eight patients with previously untreated head and neck cancer was employed. An adaptive contouring brush within the planning software Monaco was used to create an air cavity within the oropharynx, and then the air cavity was expanded uniformly 2 mm to create the “oropharyngeal mucosa”. Three plans were independently generated for each patient: Plan1: dose constraint was applied for the oropharynx; Plan2: dose constraints were applied for the oropharynx and the “oropharyngeal mucosa”; Plan3: dose constraint was applied for the “oropharyngeal mucosa”. T-tests were used to compare the dosimetry variables.
Results
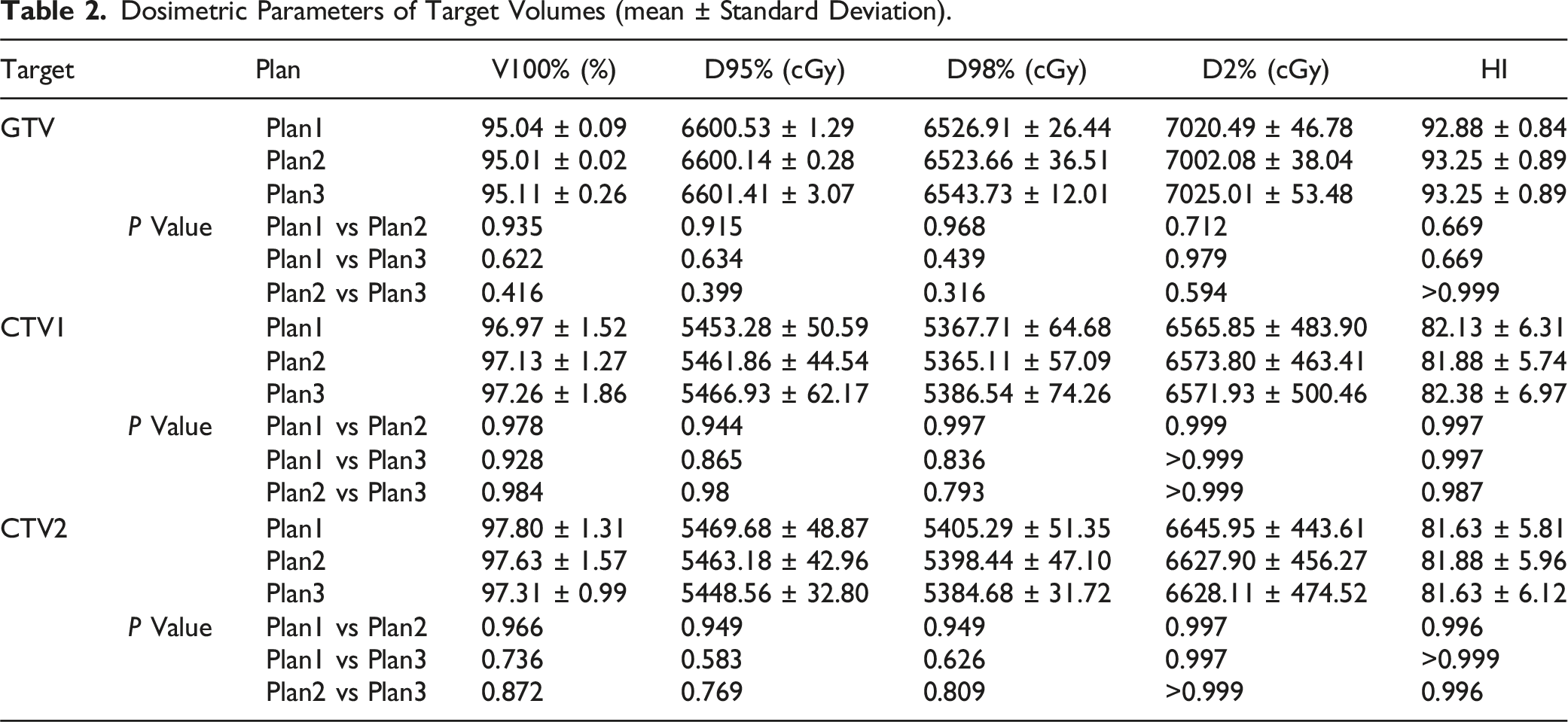
All plans had adequate target coverage and there were no statistical differences among plans. The mean dose, D30%, D45%, D50%, D85%, D90%, D95%, D100%, V25 Gy, V30 Gy, V35 Gy, V40 Gy, and V45 Gy of the oropharynx and “oropharyngeal mucosa” in Plan1 were significantly higher than those in Plan2 and Plan3. There were no significant differences between Plan2 and Plan3. There were no significant differences in the dosimetric parameters of any other organs at risk.
Conclusion
Delineation of the “oropharyngeal mucosa” and limiting its dose should be an easy and effective method to spare the oropharynx.
Plain language summary
Radiation-induced oropharyngeal injury is dose-limiting toxicity in head and neck cancer patients. Delineation of “oropharyngeal mucosa” and limiting its dose should be an easy and effective method to spare the oropharynx.
Keywords
Introduction
Head and neck cancer (HNC) is a heterogeneous group of malignancies, including cancer of the nasopharynx, hypopharynx, glottic larynx, lip mucosa, tongue, tonsil, et al. It remains one type of commonly diagnosed cancer worldwide.1-3 In general, HNC management is complex and includes surgery, radiation therapy, chemotherapy, or a combination of these modalities. Therefore, HNC requires a multidisciplinary team (MDT) approach involving different professional figures from all relevant specialties. 4 In case of radiation therapy, all patients should be treated by intensity-modulated radiation treatment (IMRT) or variant volumetric modulated arc therapy [I, A]. 5 Radiotherapy, as a single modality or in combination with chemotherapy/surgery, has been widely used in the treatment of HNC, and this has resulted in excellent local control and the possibility of cure with functional preservation.2,3,6,7 Radiotherapy remains one of main treatment modalities for neck metastases, too.7,8 At present, IMRT is becoming the standard radiotherapy technique for the treatment of HNC or neck metastases. Several studies reported that IMRT potentially improves local–regional control, reduces side effects, and improves quality of life in HNC, for IMRT is a highly conformal radiation technique that allows delivery of high radiation doses to the gross tumor, bulky lymph nodes, and high-risk areas while sparing adjacent organs.7,9-11
Even though, radiation-induced oropharyngeal injury is inevitable in more than half HNC patients because retropharyngeal, level II, III and Va of cervical lymph node regions are usually at high to intermediate risk and are covered within clinical target volumes (CTVs).7,10,12,13 Radiation-induced edema, mucositis of the oropharynx and swallowing dysfunction/dysphagia are common and dose-limiting toxicities.14,15 The risk and degree of radiation-induced injury are strictly correlated with radiation dose and volume delivered to organs at risk (OARs). 15 Sparing dose and volume of the oropharynx in HNC is not easy because the oropharynx is geometrically close to CTVs usually covering retropharyngeal, level II, III and Va of cervical lymph node regions.7,10,12,13 At the same, delineation of the oropharynx accurately on CT sections often proves challenging because anatomical boundaries of the oropharynx on CT are not always clear, especially among patients with HNC.13,16 MRI is superior than CT. Unfortunately, even in developed countries, most of hospitals are not equipped with MRI-guided linear accelerators. 17 Inaccuracy of oropharyngeal delineation directly and artificially impacts the delivered dose to the oropharynx. 18 Here an optimized method to reduce oropharyngeal dose without compromising target coverage by delineation of “oropharyngeal mucosa” and limiting its dose was reported.
Methods
Patient Selection
In this retrospective study, 8 consecutive patients with previously untreated of HNC who received definitive IMRT at our institution from December 2021 to December 2022 were included. We have de-identified all patient details. The reporting of this study conforms to STROBE guidelines. 19 The study has been reviewed and approved by the Ethics Committee of the Hebei Province Hospital of Chinese Medicine (Shijiazhuang, China); Ethics approval number: HBZY2023-KY-070-01.
Before treatment, relevant examinations were performed to determine the tumor extent and clinical stage. 7 Computed tomographic (CT) imaging from the vertex to 5 cm below the clavicle was obtained with the patient immobilized in the treatment position. Slice thickness and spacing were 3 mm throughout imaging.
Delineation
An experienced radiation oncologist and a radiation therapist delineated the corresponding target(s), normal tissue(s) and OARs, as previously described.7,12,16,20
The gross tumour volume (GTV) was delineated for each patient based on images (CT and MRI), endoscopic examination and other clinical examinations. 20 The clinical target volumes (CTVs) correspond to the GTV plus a 5 mm margin, and covered structures with substantial risk of microscopic infiltration and cervical lymph node regions at high to intermediate risk. The planning target volumes (PTVs) correspond to the CTV plus a 5 mm margin. They were delineated according to guidelines.7,12,20 To evaluate the impact of oropharyngeal or/and “oropharyngeal mucosa” delineation on CTV, CTV was divided into CTV1 and CTV2, with the superior end of the hyoid bone as the boundary. The spinal cord, parotid, mandible, oral cavity, larynx and oropharynx were delineated as OARs for all patients, as previously described.12,16
The Oropharynx was delineated according to the consensus guidelines,
16
as shown in Figure 1. An example of target delineation and isodose distributions. (A) oropharynx, “oropharyngeal mucosa” and CTV delineation. (B) Isodose lines. (C) dose–volume histograms.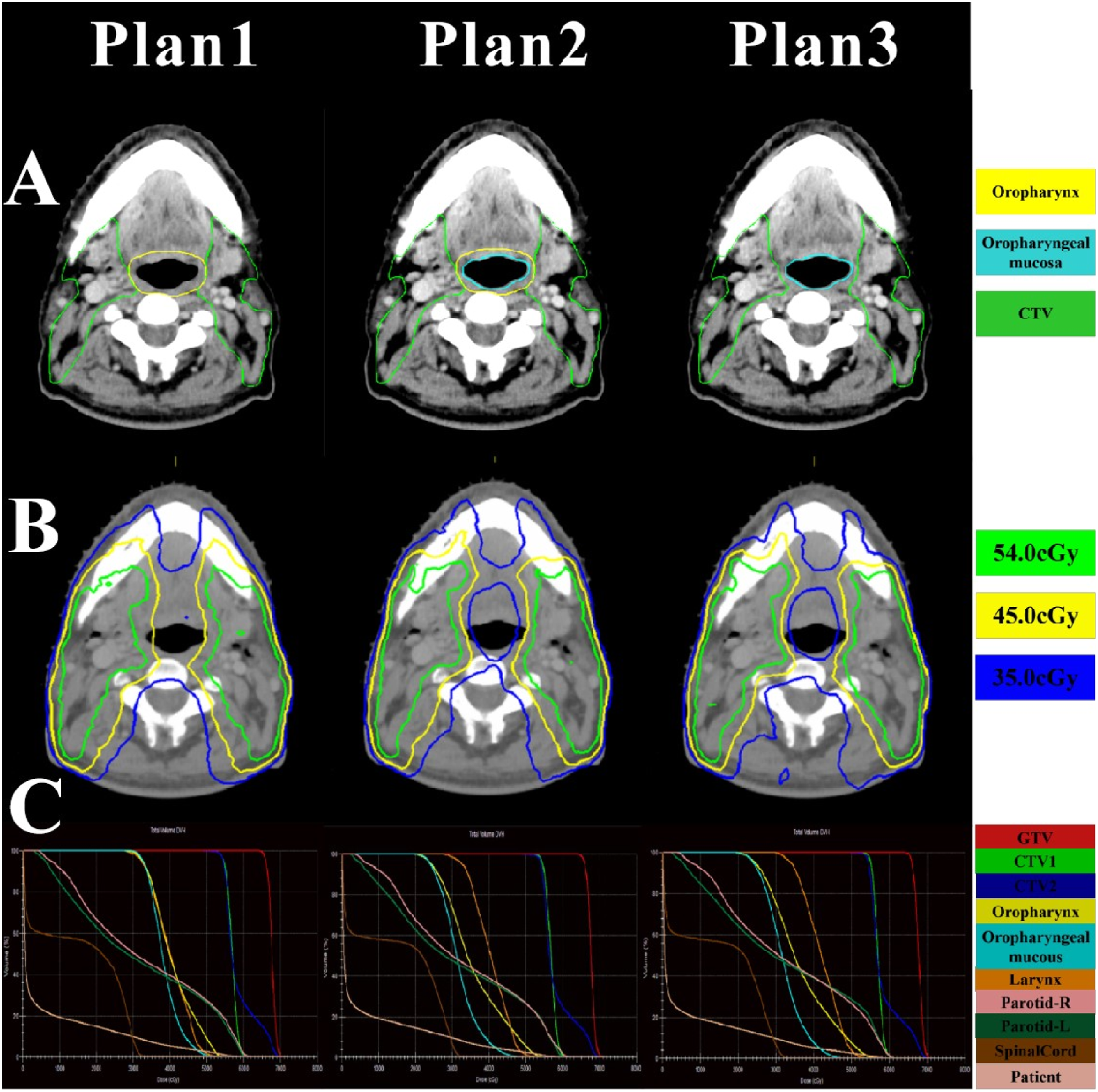
“Oropharyngeal mucosa” was delineated as OARs for all patients as below. An adaptive contouring brush within Monaco (1.1.0.0) (Elekta, Sweden) was used to create an air cavity within the oropharynx, and then, the air cavity was expanded uniformly 2 mm to create the “oropharyngeal mucosa”, as shown in Figure 1.
Radiation Planning
Patients were planned with GTV and CTV receiving 66 Gy and 54 Gy, respectively, in 30 fractions. The IMRT plan was completed with the Monaco 5.11.03 treatment planning system, 8 planning – fields, dynamic. Using the same optimization objectives for the corresponding target(s), 3 treatment plans were independently generated for each patient.
Plan1: The oropharynx was delineated as an OAR. The maximum dose was <55 Gy and the mean dose was <45 Gy. No dose constraint was applied for the “oropharyngeal mucosa”.
Plan2: Both the oropharynx and “oropharyngeal mucosa” were delineated as OARs. The maximum dose of the oropharynx was <55 Gy and the mean dose was <45 Gy. The maximum dose of the “oropharyngeal mucosa” was <25 Gy.
Plan3: The “oropharyngeal mucosa” was delineated as an OAR. The maximum dose was <25 Gy. No dose constraint was applied for the oropharynx.
All IMRT plans were designed and optimized by a medical physicist and a radiation oncologist, and all final plans were confirmed by the radiation oncologist. All final plans met criteria as previously described, such as that 95% or more of target volumes (GTV and CTV) received 95% of the prescribed dose.11,21
Dose volume parameters of all plans were analyzed. Various dosimetric parameters of GTV/CTV1/CTV2 (including V100% [volume receiving the prescription dose], D95% [minimum dose received by 95% of the volume], D98%, D2%, homogeneity index (HI, defined as D98%/D2%)) were calculated and collected. Dosimetric parameters of the oropharynx or “oropharyngeal mucosa”, including the maximum dose, the mean dose, V20 Gy (percent volume of an organ receiving no less than 20 Gy), V25 Gy, V30 Gy, V35 Gy, V40 Gy, V45 Gy, V50 Gy, V55 Gy, D25% (minimum dose received by 25% of the volume), D30%, D45%, D50%, D85%, D90%, D95% and D100% were calculated and collected.
Statistics
The data shown represent the mean ± standard deviation. Student’s T-test was used to compare dosimetry variables. P < 0.05 was considered statistically significant.
Results
Patient Characteristics
Main Patient and Tumor Characteristics.
Delineation of the “Oropharyngeal Mucosa” and Limiting its Dose Does not Compromise Target Coverage
Dosimetric Parameters of Target Volumes (mean ± Standard Deviation).
Delineation of the “Oropharyngeal Mucosa” and Limiting its dose Reduces Oropharyngeal Dose
Dosimetric Parameters of the Oropharynx (mean ± Standard Deviation).

Dosimetric Parameters of the “oropharyngeal Mucosa” (mean ± Standard Deviation).


The dosimetry indices of D25%, D45%, D85%, D95% (A) and the V20 Gy, V30 Gy, V40 Gy, V50 Gy (B) for the oropharynx. *P < 0.05, **P < 0.005, ****P < 0.0001.

The dosimetry indices of D25%, D45%, D85%, D95% (A) and the V20 Gy, V30 Gy, V40 Gy, V50 Gy (B) for the “oropharyngeal mucosa ”. ****P < 0.0001.
Maximum doses of the oropharynx in Plan1 to Plan3 were 6036.54 ± 494.52 cGy, 6018.74 ± 449.17 cGy and 6294.39 ± 353.73 cGy, respectively. There was no significant difference among them. But, mean doses of the oropharynx in Plan2 and Plan3 were significantly lower than that in Plan1. Mean doses in Plan1 to 3 were 4318.34 ± 108.70 cGy, 3784.29 ± 145.10 cGy and 3838.04 ± 130.30 cGy, respectively (Table 3). Plan2 and Plan3 had significant lower D25%, D30%, D45%, D50%, D85%, D90%, D95%, D100% of the oropharynx than Plan1 (Figure 2(A), Table 3). Plan2 and Plan3 had significant lower V20 Gy, V25 Gy, V30 Gy, V35 Gy, V40 Gy and V45 Gy of the oropharynx than Plan1, too (Figure 2(B), Table 3). These unanimously indicate that irradiation doses of the oropharynx in Plan2 (Delineation of both the oropharynx and the “oropharyngeal mucosa”) and in Plan3 (Delineation of the “oropharyngeal mucosa” alone) are significantly lower than that in Plan1 (Delineation of the oropharynx alone).
Maximum doses of the “oropharyngeal mucosa” in Plan1 to Plan3 were 5765.38 ± 535.74 cGy, 5615.00 ± 601.63 cGy and 5844.35 ± 648.57 cGy, respectively. There was no significant difference among them. But, mean doses in Plan2 and Plan3 were significantly lower than that in Plan1. Mean doses in Plan1 to 3 were 4057.84 ± 152.62 cGy, 3378.76 ± 103.88 cGy and 3403.99 ± 92.37 cGy, respectively (Table 4). Plan2 and Plan3 had significant lower D25%, D30%, D45%, D50%, D85%, D90%, D95%, D100% of the “oropharyngeal mucosa” than Plan1 (Figure 3(A), Table 4). Plan2 and Plan3 had significant lower V20 Gy, V25 Gy, V30 Gy, V35 Gy, V40 Gy and V45 Gy of the “oropharyngeal mucosa” than Plan1, too (Figure 3(B), Table 4). These unanimously indicate that irradiation doses of the “oropharyngeal mucosa” in Plan2 (Delineation of both the oropharynx and the “oropharyngeal mucosa”) and Plan3 (Delineation of the “oropharyngeal mucosa” alone) are significantly lower than that in Plan1 (Delineation of both the oropharynx alone). All data of dosimetric parameters of the oropharynx and “oropharyngeal mucosa” unanimously indicate that delineation of the “oropharyngeal mucosa” and limiting its dose reduces “oropharyngeal mucosa” dose.
There were no significant differences in any dosimetric parameters of the oropharynx or “oropharyngeal mucosa” between Plan2 and Plan3. Together with aforementioned data, these results indicate that delineation of the “oropharyngeal mucosa” and limiting its dose alone reduces oropharyngeal dose than delineation of the oropharynx and limiting its dose alone. Delineation of the “oropharyngeal mucosa” and limiting its dose alone reduces oropharyngeal dose similarly as delineation of both the oropharynx and the “oropharyngeal mucosa” and limiting their doses.
Delineation of the “Oropharyngeal Mucosa” and Limiting its dose Does not Increase Doses of OARs
Dosimetric Parameters of OARs (mean ± Standard Deviation).

Discussion
The oropharynx is the common part of the digestive and respiratory tracts. Its wall is covered by an epithelial mucosa and it is composed of an interior longitudinal layer of muscles and an external circular layer of muscles. 22 The epithelium is radiosensitive. More than 80% of patients will develop radiation-induced mucositis during head and neck radiotherapy, and more than half of patients will even develop grade 3-4 mucositis. Oropharyngeal mucositis may occur from a few days after initiation of radiotherapy to a long period after completion of radiotherapy. It causes pain and odynophagia, resulting in malnutrition which leads to poor quality of life and shortens overall survival.23,24 Swallowing dysfunction is another radiation induced toxicity in HNC and it is an acute and late adverse effect. During the course of HNC, approximately 60% to 75% of patients develop swallowing dysfunction. 25 Acute swallowing dysfunction may be a consequential effect of acute mucositis and edema. 26 Late swallowing dysfunction is largely related to radiation of the pharyngeal musculature responsible for the initiation and completion of swallowing. 22 Swallowing dysfunction leads to lots of sequelae, such as poor quality of life, malnutrition, aspiration of food into lungs which contributed to 19% of non-cancer deaths.21,22,27 These may result in the requirement for a permanent gastrostomy tube and long-term supportive care.21,22 Oropharyngeal radiation may also cause phonation dysfunction, dryness of the oropharynx, et al. 28
Prevention and treatment of radiation induced oropharyngeal injury are far from satisfactory. Reduction in radiation dose to the oropharynx is the main effective way to prevent adverse effects because of the strong association between radiation dose and radiation induced adverse effect endpoints. 21 Dr Sonis et al reported that ulcerative mucositis was present by 30 Gy with incremental doses of radiation to the GTV over a 7 week period (2 Gy per day to 70 Gy). 29 Accordingly, Dmax of 25 Gy was used as oropharyngeal mucosal dose constraint in our study. Feng found a strong association between the mean dose and dysphagia endpoints. 21 Doornaert reported a steep dose–effect relationship beyond 45 Gy to the pharyngeal wall. 9 Tiziana Rancati et al recommend that minimizing the volume of the oropharynx receiving ≥60 Gy, and reducing the volume receiving ≥50 Gy, are associated with reduced dysphagia/aspiration. 30 All the above studies have demonstrated a strong correlation between the radiation dose to the oropharynx and its adverse effects. Unfortunately, but not surprisingly, there were not many recommendations for prevention or treatment that could actually be made. This means that the bulk of patients undergoing head and neck radiotherapy still does not have satisfactory way to prevention or alleviate their mucositis. Hence, a method to reduce oropharyngeal dose by delineation of “oropharyngeal mucosa” and limiting its dose was explored here. “Oropharyngeal mucosa” was delineated by a 2 mm expansion margin from the interior wall of the oropharynx (Figure 1). Data showed that compared to delineation of the oropharynx alone, delineation of the “oropharyngeal mucosa” and the oropharynx not only significantly decreased the mean dose of “oropharyngeal mucosa” (3378.76 ± 103.88 cGy vs 4057.84 ± 152.62 cGy, P < 0.0001), but also significantly decreased the mean dose of oropharynx (3784.29 ± 145.10 cGy vs 4318.34 ± 108.70 cGy, P < 0.0001). Delineation of “oropharyngeal mucosa” and limiting its dose significantly improved dosimetric parameters of “oropharyngeal mucosa” and oropharynx, including V45 Gy to V25 Gy, D20% to D100%. On the other side, it did not change dosimetric parameters of target volumes or other OARs. These unanimously indicate that delineation of “oropharyngeal mucosa” and limiting its dose reduces doses of “oropharyngeal mucosa” and oropharynx without changing dosimetric parameters of target volumes and other OARs. As mentioned above, reduction in radiation dose effectively decreases adverse effects, our optimized method should theoretically decrease mucositis, swallowing dysfunction/dysphagia and other adverse effects. It should be noted that the oropharyngeal mucosa sparing depends on the primary location of the tumor. Thus, it can not be applied in oropharyngeal cancer.
Delineation of the oropharynx accurately on CT sections often proves challenging because anatomical boundaries of the oropharynx on CT are not always clear, especially among patients with HNC.13,16 Several guidelines for delineation of the oropharynx have been published. However, the definition, selection and delineation vary widely among different publications and authors.9,13,16 At the same time, delineation of the oropharynx is a time- and labor-consuming work. The interior wall of the oropharynx is clear on CT because air is in the cavity. So, it is easy to delineate “oropharyngeal mucosa” by a 2 mm expansion margin from the interior wall of the oropharynx (Figure 1). It is remarkable that compared to delineation of the oropharynx alone, delineation of the “oropharyngeal mucosa” alone significantly decreased the mean dose of “oropharyngeal mucosa” (3403.99 ± 92.37 cGy vs 4057.84 ± 152.62 cGy, P < 0.0001), the mean dose of oropharynx (3838.04 ± 130.30 cGy vs 4318.34 ± 108.70 cGy, P < 0.0001), significantly improved dosimetric parameters of “oropharyngeal mucosa” and oropharynx. On the other hand, delineation of the “oropharyngeal mucosa” alone did not change dosimetric parameters of target volumes and other OARs. There were no significant differences in dosimetric parameters of “oropharyngeal mucosa”, oropharynx and target volumes between the method of delineation of both “oropharyngeal mucosa” and the oropharynx and the method of delineation of the “oropharyngeal mucosa” alone, either. These unanimously indicate that delineation of the “oropharyngeal mucosa” alone is an easy and effective method to spare the oropharynx and “oropharyngeal mucosa” without compromising target coverage or other OARs, which should theoretically decrease mucositis, swallowing dysfunction/dysphagia and other adverse effects. Besides there are some limitations to our study. First, only 8 patients are included in the study and they are heterogeneous with respect to primary site. Second, the study has no evidence of clinical efficacy.
Conclusions
In conclusion, in HNC radiotherapy plans, delineation of the “oropharyngeal mucosa” and limiting its dose, with or without delineation of the oropharynx and limiting its doses, spare the “oropharyngeal mucosa” and oropharynx without compromising target coverage or other OARs. Delineation of “oropharyngeal mucosa” alone and limiting its dose should be a much easier and more effective method to spare the oropharynx than delineation of the oropharynx and limiting its dose.
Footnotes
Author Contributions
conceptualization, Jianming He and Diancheng Li; methodology, Xueqi Wang; validation, Xueqi Wang; formal analysis, Xueqi Wang; investigation, Xueqi Wang and Jianming He; data curation, Xueqi Wang, Jianming He and Yanyan Han; data analysis, all authors; writing—original draft preparation, Xueqi Wang and Jianming He; writing—review and editing, Xueqi Wang, Diancheng Li and Jianming He; visualization, Xueqi Wang; supervision, Jianming He and Diancheng Li; project administration, Jianming He. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Hebei Province Administration of Traditional Chinese Medicine (grant number. 2023020), Foundation of Health and Family Planning Commission of Hebei (grant number. 20180686, 20180688), Hebei Province Key Research and Development Program (grant number. 19277770D).
Ethical Statement
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
