Abstract
Background
Acute lymphocytic leukemia is a cancer affecting the blood and bone marrow and is the most frequently diagnosed cancer among children. In Ethiopia, it represents the predominant form of childhood leukemia, comprising approximately 80% of cases and serving as a leading cause of childhood cancer-related deaths. Therefore, the objective of this study is to examine the survival status and factors that may predict mortality in children admitted with acute lymphocytic leukemia at cancer treatment hospitals in Addis Ababa, Ethiopia.
Methods
A retrospective follow-up study was conducted at cancer treatment hospitals in Addis Ababa, focusing on children diagnosed with acute lymphocytic leukemia. The investigation covered records from January 1, 2017, to December 30, 2023, encompassing a sample of 230 study records. Variables with a P-value below 0.25 in the bivariate analysis were selected for entry into the multivariable analysis. Subsequently, variables demonstrating a P-value less than 0.05 in the multivariable Cox proportional hazards model were deemed statistically significant.
Results
The cumulative proportion of survival was 98.3% (95%CI: 94.8, 99.5), 89.2% (95%CI: 82.0, 93.6), and 24.1% (95%CI: 8.43, 44.1) at the end of the 20th, 40th, and 60th month, respectively. The incidence rate of mortality among cohort of children admitted with acute lymphocytic leukemia was 0.45 per 100 child months. History of relapse (AHR: 2.48; 95%CI: 1.01, 6.08) and infection (AHR: 2.34; 95%CI: 1.03, 5.31) were independent predictors of mortality among children admitted with acute lymphocytic leukemia.
Conclusion
The likelihood of mortality increased in the later stages of follow-up for children admitted with acute lymphocytic leukemia, and the incidence density rate of mortality in this group was lower compared to previous reports from other regions. Furthermore, independent predictors of mortality among children with acute lymphocytic leukemia included a history of relapse and infection.
Background
Acute Lymphocytic Leukemia is a devastating form of cancer that impacts the blood and bone marrow. While it is predominantly diagnosed in children, it can also manifest in adults. 1 Representing the most prevalent form of childhood cancer, acute lymphocytic leukemia constitutes approximately 20% to 30% of pediatric cancer cases. 2 The initial strides in improving survival were marked by the introduction of prophylactic cranial irradiation. 3 Subsequent advancements in risk stratification, adaptive treatment intensity modifications, and the management of acute adverse effects have been pivotal in further enhancing survival rates.4,5
The origin of acute lymphoblastic leukemia lies in a DNA mutation affecting stem cells, leading to an overproduction of immature white blood cells. 5 These premature white blood cells are released from the bone marrow before reaching full maturation and acquiring the ability to combat infections like their fully developed counterparts. The 5-year relative survival rate for acute lymphocytic leukemia stands at 68.8%. 6
In Ethiopia, acute lymphoblastic leukemia poses a considerable health challenge, especially among the pediatric population. 6 Despite advancements in treatment, mortality rates associated with this disease remain elevated. 7 Pinpointing predictors of mortality in Ethiopian children with acute lymphoblastic leukemia remains a complex task.6,7 Variables including patient characteristics, clinical manifestations, disease severity, treatment approaches, and health care accessibility collectively contribute to determining the outcomes for children diagnosed with acute lymphocytic leukemia.6–8
Ethiopia has adopted a comprehensive National Action Plan for the Prevention and Control of Chronic Non-Communicable Diseases, and the expansion of cancer treatment services is underway. 9 Epidemiological data are important for estimating and properly allocating resource needs and health policy prioritization. 1
Despite notable progress in treatment, the overall survival rate for acute lymphoblastic leukemia remains relatively modest. 1 To enhance outcomes for children with acute lymphocytic leukemia in Ethiopia, there is an urgent need to investigate the determinants contributing to mortality. A comprehensive understanding of these predictors can empower health care providers to devise tailored interventions, thereby improving outcomes and mitigating mortality rates.
In addition, despite the heightened incidence of Acute Lymphocytic Leukemia among children in Ethiopia, mortality rates persist at elevated levels, and the factors influencing mortality for this disease are not comprehensively elucidated. It is imperative to take proactive measures and invest in research endeavors aimed at developing targeted interventions that can ameliorate outcomes for children with acute lymphocytic leukemia in Ethiopia and beyond. The objective of this study is to assess the survival status and predictors of acute lymphocytic leukemia among children admitted to cancer treatment hospitals in Addis Ababa.
Methods and Materials
Study Design, Area and Period
A retrospective cohort longitudinal study was conducted at cancer treatment hospitals in Addis Ababa among records enrolled between January 1st and 15th, 2024. The research focused on three designated governmental hospitals; Tikur Anbessa Specialized Hospital, St. Paul Hospital Millennium Medical College, and Yakatit 12 Hospital Medical College.
Populations
The study participant records were enrolled as per the EQUATOR STROBE reporting guidelines. 10 The source population for this study comprised records of children admitted with acute lymphocytic leukemia at cancer treatment Hospitals in Addis Ababa. The study populations were randomly selected records of children admitted with acute lymphocytic leukemia at selected hospitals from January 1st, 2017, to December 30th, 2023. It was an open cohort study, allowing individuals to enter and exit from the cohort. The study’s commencement was defined by admission to the cancer treatment hospitals during the period. The study participants were exit from the cohort by either death or recovery, loss to follow-up, or the completion of follow-up time without any observed outcomes. In instances where children were lost to follow-up, discharged alive, and did not experience the outcome by the end of the study period, they were considered as censored data. Given the study’s single-arm nature, only children admitted with acute lymphocytic leukemia to cancer treatment hospitals within the study period were included, and the analysis involved comparing outcomes between those who died and those who survived.
Eligibility Criteria
Children diagnosed with acute lymphocytic leukemia and admitted to cancer treatment hospitals in Addis Ababa within the study period, were included in the study and incomplete records were excluded from the study.
Sample Size Determination and Procedure
The sample size was calculated using the double population proportion formula through the Epi Info version 7 stat calc program, considering the following parameters: a 95% confidence interval, 80% statistical power, an unexposed to exposed ratio of 1:2, a percentage outcome in the exposed group (48.7%), an adjusted hazard ratio (AHR) of 3.3, 5 and an additional 10% for potential incomplete records. This calculation resulted in a final sample size of 230.
All children admitted to the three selected Hospitals within the study period were identified from the database using their medical registration numbers. Utilizing a computer-generated method, random medical registration numbers were then selected. Each hospital’s sampling frame was established, and the total sample size was allocated to each hospital proportionally. Finally, individual participants were selected from each hospital using a simple random sampling method.
Variables of the Study
The time to death served as the dependent variable, while the independent variables encompassed socio-demographic factors (age, gender, and place of residence), hematologic factors (white blood cell count, hemoglobin level, platelet count, immune phenotype, leukemia subtypes, and complete remission), and pathologic factors (history of relapse, number of relapses, place of relapse, comorbidities, hepatomegaly, splenomegaly, malnutrition, and infection).
Operational Definition of Terms
Data Collection Tools and Procedures
A data collection format/checklist was devised to anonymously gather information from the medical charts of study participants at the chosen health facilities. The document was prepared in English. Trained nurses conducted the data collection process. All study participant records adhered to the eligibility criteria. Prior to data collection, both baseline and follow-up records underwent examination. Subsequently, health professionals retrospectively reviewed all records of patients diagnosed with acute lymphocytic leukemia. To confirm instances of death, medical death certificates were scrutinized at the hospitals.
Data Quality Control
To ensure data quality, a suitable data abstraction tool was developed. A pretest was conducted on 5% of the sample size at Zewuditu Memorial Hospital, utilizing a structured checklist, 2 weeks before the main study. This pretest aimed to assess commonly recorded variables in patient medical records. Data collectors and supervisors both experienced MSc nurses specializing in pediatric oncology care and currently working in this unit, underwent comprehensive 1-day training. Throughout the data collection process, supervisors and investigators were closely monitored to ensure high-quality data. Daily checks for data completeness were performed, and any encountered changes were promptly addressed. Additionally, during data administration, storage, and analysis, supervisors and investigators conducted double-checks for completeness and consistency by randomly selecting cards to evaluate overall data coherence.
Data Processing and Analysis
The data underwent cleaning, updating, and coding prior to analysis. Epi-Data version 4.6 was used for data entry, and analysis was conducted using SPSS version 26. Continuous data were presented as either mean and standard deviation or median, depending on the distribution. Frequency distribution was applied for categorical data. Subsequently, participant statuses were categorized as censored or death. The mean value substitution method was used to manage missing values. Throughout the study period, the incidence density rate (IDR) was determined. Akaike information criterion (AIC) was used to choose the model used for data analysis. Mean survival time and cumulative probability of survival were estimated using Kaplan-Meier survival curves, and log-rank tests were conducted to compare survival curves after admission between groups of patients grouped by predictors. Variables with a P-value <0.05 were considered to meet the assumption. Each explanatory variable underwent a bivariate Cox-proportional hazards regression model check. Hazard ratios with 95% confidence intervals and P-values were utilized to assess the strength of associations and determine the statistical significance of the results.
Ethical Considerations
Ethical clearance was obtained from the Yanet College IRB with a Reference Number YC/056/23. All collected data were coded and kept in a separate room before being entered into the computer and names were not included in the data collection format to maintain confidentiality. The data were locked by a secret code after being entered into the computer and were not disclosed to anyone except the principal investigator. This study used anonymized electronic patient data, and therefore the requirement to obtain informed consent was waived by the IRB.
Results
Socio Demographic Characteristics
Socio Demographic Characteristics of the Children Admitted With Acute Lymphocytic Leukemia at Cancer Treatment Hospitals in Addis Ababa, Ethiopia; 2024.

Medical and Related Characteristics
Medical and Related Characteristics of the Children Admitted With Acute Lymphocytic Leukemia at Cancer Treatment Hospitals in Addis Ababa, Ethiopia; 2024.
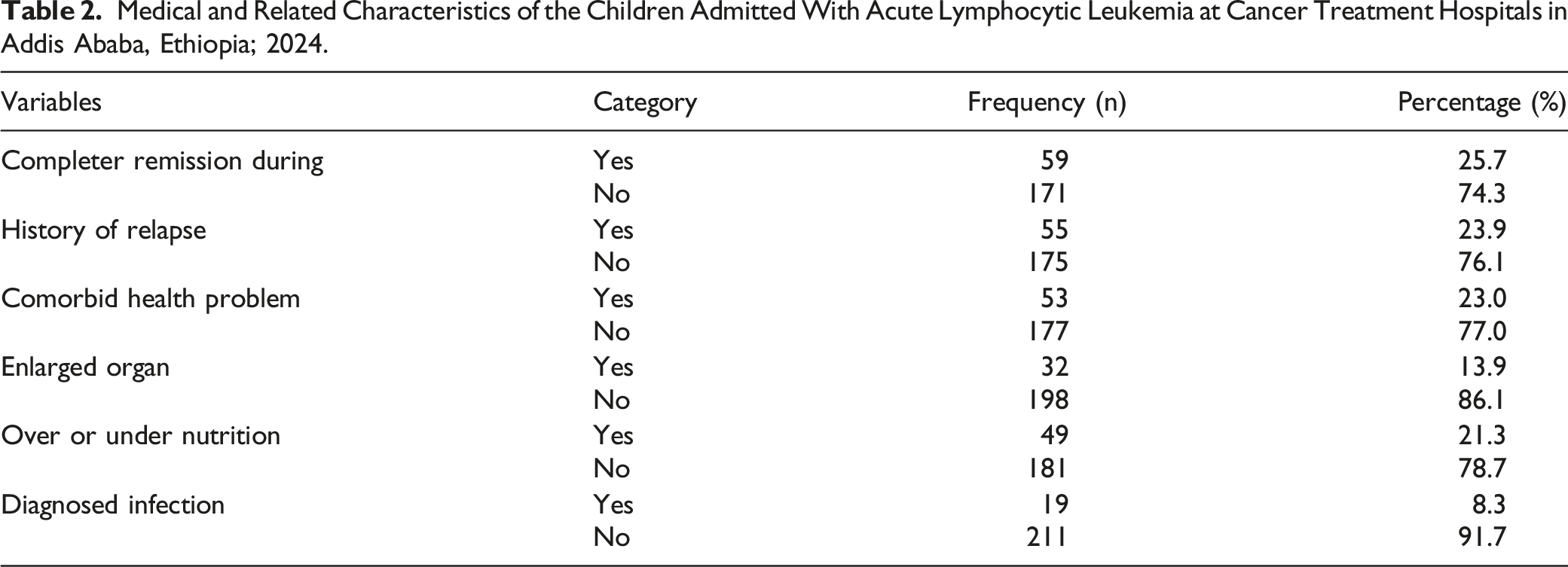
Time to Death Among Children Admitted with Acute Lymphocytic Leukemia
The cumulative proportion of survival at the end of 20th month was 98.3% (95%CI: 94.8, 99.5), 89.2% (95%CI: 82.0, 93.6), and 24.1% (95%CI: 8.43, 44.1) at the end of 20th, 40th and 60th month respectively. As it has been seen with the next graph, during the 20 months was a lower probability of death. While on the next 40 months (Month 21 to 40), the proportion of death has slightly increased and the graph fell down slowly and finally after 40th month the graph is decline rapidly indicates that there was higher number of deaths (Figure 1). The survival time to death among children admitted with Acute Lymphocytic Leukemia at cancer treatment Hospitals in Addis Ababa, Ethiopia; 2024.
Incidence of Mortality Among Children Admitted with Acute Lymphocytic Leukemia
In this study, children were followed for a total of 7508 months. Among the total children followed, 34 were died. Therefore, the incidence rate of mortality among cohort of children admitted with acute lymphocytic leukemia was 0.45 per 100 child months. This study revealed that, 3 (8.82%) of the dead children were died within the first 20 months and 14 (41.18%) in the first 40 months of initiation of follow up.
Predictors of Mortality Among Children Admitted With Acute Lymphocytic Leukemia
In this study, both bivariate and Multivariable analysis was conducted using cox proportional hazard model. Age, sex, place of residence, history of relapse, comorbidity, enlarged organ, over/under nutrition and infection were candidate variables for the multivariable analysis. In multivariable analysis, history of relapse and infection were statistically significant variables.
Predictors of Mortality Among Children Admitted With Acute Lymphocytic Leukemia at Cancer Treatment Hospitals in Addis Ababa, Ethiopia; 2024.

aIndicate variables which have P-value <0.25 in bivariate analysis and.
bIndicate variable which have P-value <0.05 in multivariable cox proportional hazard model.
Discussion
The observed incidence rate of mortality among the cohort of children admitted with acute lymphocytic leukemia was determined to be 0.45 per 100 child months. To provide context, the incidence of childhood mortality (0-14 years) associated with acute lymphocytic leukemia in Nordic countries is reported as 4.0 per 100,000 children per year. 8 In Switzerland, over 4 consecutive 4-year periods, the mean incidence of childhood mortality (<15 years) among admitted Acute Lymphocytic Leukemia cases was 3.8 per 100,000 children per year. 9 An analysis of Saudi databases identified an age-adjusted incidence of childhood mortality with Acute Lymphocytic Leukemia (14 years old or younger) at 2.35 per 100,000 populations in 2014. 10 According to findings from the Centers for Disease Control and Prevention (CDC) spanning from 2001 to 2014, the incidence of childhood mortality among diagnosed Acute Lymphocytic Leukemia cases is reported as 34 per 1,000,000 people per year in individuals <20 years old. Notably, this incidence can reach as high as 75.2 per 1,000,000 people per year in the age group of 1-4 years old. 11 The higher incidence observed in these studies can be attributed to three potential explanations. Firstly, our study includes children up to 18 years old, contributing to the elevated incidence.
A statistically significant predictor of mortality among children with acute lymphocytic leukemia was the history of relapse. This finding aligns with a study conducted in Sweden. 12 Relapsed acute lymphoblastic leukemia, or relapsed Acute Lymphocytic Leukemia, denotes the recurrence of Acute Lymphocytic Leukemia in patients previously treated for the disease. 13 Approximately 15 to 20% of children treated for Acute Lymphocytic Leukemia, who initially achieve complete remission, may experience a relapse. 14 In cases of relapsed Acute Lymphocytic Leukemia, lymphocyte stem cells (a type of blood stem cell) transform into immature white blood cells known as lymphoblasts or “blasts”. 12 Unlike healthy white blood cells, these blasts accumulate in the bone marrow, reducing space for healthy blood cells, including white blood cells, red blood cells, and platelets. Furthermore, these abnormal cells are ineffective in combating infections. 15
Acute lymphocytic leukemia is a rare blood cancer that affects white blood cells, crucial for the body’s immune response. 16 For children with acute lymphocytic leukemia, the presence of an infection increases the likelihood of mortality. This study’s findings are consistent with a study conducted in Sweden spanning from 1988 to 2017. 12 The increased risk is attributed to a defect in lymphocytes. In Acute Lymphocytic Leukemia, the bone marrow produces an excessive number of lymphocytes, a type of white blood cell crucial for infection defense. 13 However, in Acute Lymphocytic Leukemia, these cells become abnormal and less effective in combating infections. Additionally, they overcrowd healthy cells, leading to conditions such as infection, anemia, and increased susceptibility to bleeding. 15
In individuals with acute leukemia, the risk of developing infections is elevated due to both the leukemia itself and its treatment. 17 This susceptibility to infection may be linked to a decline in neutrophil count, with 26 (76.5%) of the deceased children exhibiting a neutrophil count below 500 cells/mm3. Neutropenia stands out as the primary risk factor for infection development, with the severity and frequency of infections increasing as the absolute neutrophil count drops below 500 cells/mm3. 17 Other contributing factors may include compromised cellular or humoral immunity, disruption of normal barriers like the skin and mucosal surfaces, and the presence of vascular access catheters and other medical devices. 18 Patients often exhibit multiple concurrent risk factors. Moreover, the frequent use of antimicrobial agents for various purposes, such as prophylaxis, empiric therapy, pre-emptive administration, specific or targeted therapy, and occasionally maintenance or suppressive therapy, can influence the nature and spectrum of infections, raising concerns about the emergence/selection of multidrug-resistant (MDR) organisms. 19 A limitation of this study is that, being record-based, it may not reflect the actual responses of the subjects but rather the data documented in the records. Consistently, the study did not assessed immunophenotype, leucocyte count of the study participants, detailed clinical data, such as information regarding treatment regimens, disease stages at diagnosis, and genetic characteristics of the leukemia.
Conclusion
The likelihood of mortality among children admitted with acute lymphocytic leukemia was elevated during the later follow-up period, and the incidence density rate of mortality in this cohort was comparatively lower than reported in previous studies from other regions. Furthermore, independent predictors of mortality among children with Acute Lymphocytic Leukemia were identified as a history of relapse and infection. It is recommended to implement strategies focused on providing training to the families of children with acute lymphocytic leukemia to enhance awareness regarding the predictors of Acute Lymphocytic Leukemia.
Footnotes
Acknowledgments
We would also like to thank Tikur Anbessa Specialized Hospital, Yekatit 12 Hospital Medical College and St. Paul Hospital Millennium Medical College administrative staffs for their unreserved effort throughout the research process.
Author’s Contribution
Samuel Dessu Sifer was involved in the conception, design, analysis, interpretation, report and manuscript writing, review of the design, analysis, interpretation and report writing and Milkiyas Solomon was involved in data analysis, interpretation and writing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
The data sets generated and/or analyzed are available with a reasonable request through the corresponding author. All data generated or analysed during this study are included in this published article [and its supplementary information files].
