Abstract
The current standard treatment for locally advanced squamous cell carcinoma of the head and neck (LASCCHN) comprises concurrent radiotherapy (CRT) alongside platinum-based chemotherapy. However, innovative therapeutic alternatives are being evaluated in phase II/III randomized trials. This study employed a Bayesian network meta-analysis (NMA) using fixed effects to provide both direct and indirect comparisons of all existing treatment modalities for unresectable LASCCHN. Methods: We referenced randomized controlled trials (RCTs) from January 2000 to July 2023 by extensively reviewing PubMed, EMBASE, and Web of Science databases, adhering to the Cochrane methodology. Relevant data, including summary estimates of overall survival (OS) and progression-free survival (PFS), were extracted from these selected studies and recorded in a predefined database sheet. Subsequently, we conducted a random effects network meta-analysis using a Bayesian framework. Results: Based on the Surface Under the Cumulative Ranking (SUCRA) values, the league table organizes the various treatments for OS in the following order: IC + RT&MTT, MTT-CRT, IC + CRT&MTT, CRT, IC + CRT, MTT-RT, IC + MTT-RT, and RT. In a similar order, the treatments rank as follows according to the league table: IC + CRT&MTT, MTT-CRT, IC + CRT, IC + RT&MTT, CRT, IC + MTT-RT, MTT-RT, and RT. Notably, none of these treatments showed significant advantages over concurrent chemoradiotherapy. Conclusion: Despite concurrent chemoradiotherapy being the prevailing treatment for LASCCHN, our findings suggest the potential for improved outcomes when concurrent chemoradiotherapy is combined with targeted therapy or induction chemotherapy.
Plain Language Summary
The current standard treatment for advanced head and neck cancer involves combining radiation therapy with chemotherapy. However, there are ongoing trials exploring alternative therapies. In this study, we conducted a comprehensive analysis of existing treatments using a statistical method called network meta-analysis. Our analysis included data from randomized controlled trials published between January 2000 and July 2023. We focused on overall survival and progression-free survival as key outcome measures. The results of our analysis showed that none of the alternative treatments demonstrated significant advantages over the standard concurrent chemoradiotherapy. Nevertheless, there is potential for improved outcomes when targeted therapy or induction chemotherapy is combined with concurrent chemoradiotherapy.
Introduction
Squamous cell carcinoma of the head and neck (SCCHN) develops from squamous epithelial cells. It can occur in different sites within the head and neck region, such as the pharynx, oral cavity, and larynx. 1 According to statistics, in 2020, an estimated 740,000 new cases of lip/oral cavity cancer, laryngeal cancer, oropharyngeal cancer and hypopharyngeal cancer were reported worldwide, with approximately 360,000 deaths. 2
A significant number of patients with squamous cell carcinoma of the head and neck (SCCHN) are diagnosed at an advanced stage, where the disease has already locally advanced. This is often due to the relative inconspicuousness of the lesion and the atypical nature of its early symptoms. A single treatment mode tends not to produce satisfactory results, and typically, a multidisciplinary treatment approach is relied upon. 3 While current guidelines for first-line therapy are concurrent chemoradiotherapy (CRT) with platinum-based chemotherapy for locally advanced squamous cell carcinoma of the head and neck (LASCCHN), 4 there have been significant advancements in the development and evaluation of novel treatment regimens. These include monoclonal antibodies targeting the epidermal growth factor receptor (EGFR), innovative radiotherapy fractionation schedules, as well as induction or adjuvant chemotherapy regimens. It is important to note, however, that not all treatment modalities have been directly compared in clinical trials. Therefore, traditional meta-analyses, which allows comparison of multiple treatments to be considered in a single analysis by combining direct with indirect evidence. 5 In cases where head-to-head comparisons are lacking, a network meta-analysis (NMA) can be employed to synthesize the available evidence from randomized controlled trials and indirectly compare different treatments. By integrating these direct and indirect comparisons, statistical models are used to estimate the relative effectiveness of different treatments. 6 In this study, we utilized network meta-analysis to compare and rank eight treatment strategies, including radiotherapy alone (RT), concurrent chemoradiotherapy (CRT), targeted therapy concomitant with radiotherapy (MTT-RT), targeted therapy concomitant with concurrent chemoradiotherapy (MTT-CRT), induction chemotherapy followed by concurrent chemoradiotherapy (IC + CRT), induction chemotherapy followed by concomitant radiotherapy with targeted therapy (IC + MTT-RT), concurrent targeted therapy and radiotherapy after induction chemotherapy and targeted therapy (IC + RT&MTT), and concurrent targeted therapy and radiotherapy after induction chemotherapy and targeted therapy (IC + CRT&MTT)for locally advanced head and neck squamous cell carcinoma. NMA allowed us to determine the optimal treatment approach and provide a ranking of treatment strategies based on their effect sizes. This analysis aimed to understand the relative efficacy of different treatment methods and provide valuable insights for future research and treatment directions.
Methods
The protocol was registered in the Prospective Register of Systematic Reviews (PROSPERO CRD42023452704). The review protocol can be found on the website https://www.crd.york.ac.uk/PROSPERO.
Search Strategy
According to the PRISMA-NMA checklist, 6 we executed a comprehensive network meta-analysis, extensively searching databases including Web of Science, PubMed, and Embase. The search period spanned from January 1st, 2000 to July 1st, 2023. Employing MESH terms, we formulated a targeted search strategy incorporating key phrases such as “head and neck squamous cell carcinoma” and “randomized controlled clinical trials”. The exhaustive search strategies utilized are detailed in Supplemental Figure 1. To identify any other potentially relevant studies, we manually examined the reference lists of included trials and reviews. We adhered to Cochrane’s Highly Sensitive Search Strategy, imposing no restrictions on language or publication type, ensuring thoroughness and inclusivity in our research approach.
Review Strategy
To reduce redundancy, a review strategy was executed by two independent investigators. They first screened and evaluated the abstracts and titles of retrieved studies. Following this preliminary assessment, a comprehensive review of the full texts was carried out. Any disputes or disagreements were settled with the involvement of a third reviewer. The entire process is graphically represented in Figure 1. Flow diagram of study selection.
Study Inclusion and Exclusion Criteria
Our study specifically targeted at randomized controlled trials comparing different systemic interventions for locally advanced squamous cell carcinoma of the head and neck (LASCCHN). Following the PICOS principle (Population, Intervention, Comparison/Control, Outcome, Study Design), the study population (P) included patients diagnosed with stage III or IV head and neck squamous cell carcinoma. The considered interventions (I) or comparisons (C) incorporated any two or more out of eight identified treatment options. We restricted the study design (S) to randomized controlled trials. Primary and secondary outcomes were defined as Overall Survival (OS) or Progression-Free Survival (PFS), respectively. Our primary objective was to collate survival outcomes and conduct a comprehensive assessment of these results.
Data Extraction
Data extraction was performed independently by two investigators. In cases of duplication, only the latest study was considered for data extraction. However, if the latest study offered limited information, we referred to the full results publication.
We utilized a standardized data collection form to gather essential information such as the year of publication, first author, recruitment period, treatment arms, hazard ratio (HR) for OS and PFS, study inclusion and exclusion criteria, number of assessable patients in each arm of the trial, and key baseline characteristics of participants. This systematic approach ensured consistent documentation of pertinent details across all studies. If the 95% confidence intervals (95%CI) and HR were not reported in the articles, we extracted data from survival curves using Engauge Digitizer 11.1 software, following the methods described by Tierney et al. 7 Our analysis mainly focused on stage II or III randomized controlled trials, prioritizing the use of the most recent and comprehensive data. To assess the risk of bias in the included studies, we followed guidelines outlined in The Cochrane Handbook for Systematic Reviews of Interventions. The selection of studies, data extraction, and risk of bias assessment were conducted by two researchers. This approach that enhances the process’s reliability and precision by minimizing potential biases and errors.
Statistical Analysis
In this study, the primary outcome measures were OS and PFS. To assess the effectiveness of different interventions, we combined the hazard ratios for OS and PFS. For each outcome, the corresponding effect size estimate (HR), along with its 95% confidence interval (95% CI), was calculated. This allowed for a comprehensive evaluation of the interventions included in our analysis.
The primary outcome measures were primarily analyzed using the Gemtc package of R 4.3.1 software and STATA.16. Initially, data pre-processing was executed in STAT16 software, followed by the generation of network diagrams for OS and PFS, as depicted in Figure 2. These diagrams provided a visual representation of both direct and indirect comparisons among various treatment regimens. Network diagrams representing reported survival data (OS and PFS) for multiple treatment strategies were constructed, adopting a consistency model for fitting.
8
In these network diagrams, different visual elements were utilized to represent distinct aspects of the data. The size of the endpoint circle was used to visually represent the number of patients associated with each treatment strategy, with larger circles indicating a larger patient cohort. Furthermore, the thickness of connecting lines conveyed the number of clinical trials associated with specific treatment comparisons. Thicker lines represented a greater number of trials, signifying robust evidence supporting that particular comparison. Network of treatments available for comparison of Overall Survival (A). Network of treatments available for comparison of progression-free survival (B). RT:radiotherapy alone; CRT:concurrent chemoradiotherapy; MTT-RT: targeted therapy concomitant with radiotherapy; MTT-CRT: targeted therapy concomitant with concurrent chemoradiotherapy; IC + CRT:induction chemotherapy followed by concurrent chemoradiotherapy; IC + MTT-RT:induction chemotherapy followed by concomitant radiotherapy with targeted therapy;IC + RT&MTT:concurrent targeted therapy and radiotherapy after induction chemotherapy and targeted therapy;IC + CRT&MTT:concurrent targeted therapy and radiotherapy after induction chemotherapy and targeted therapy.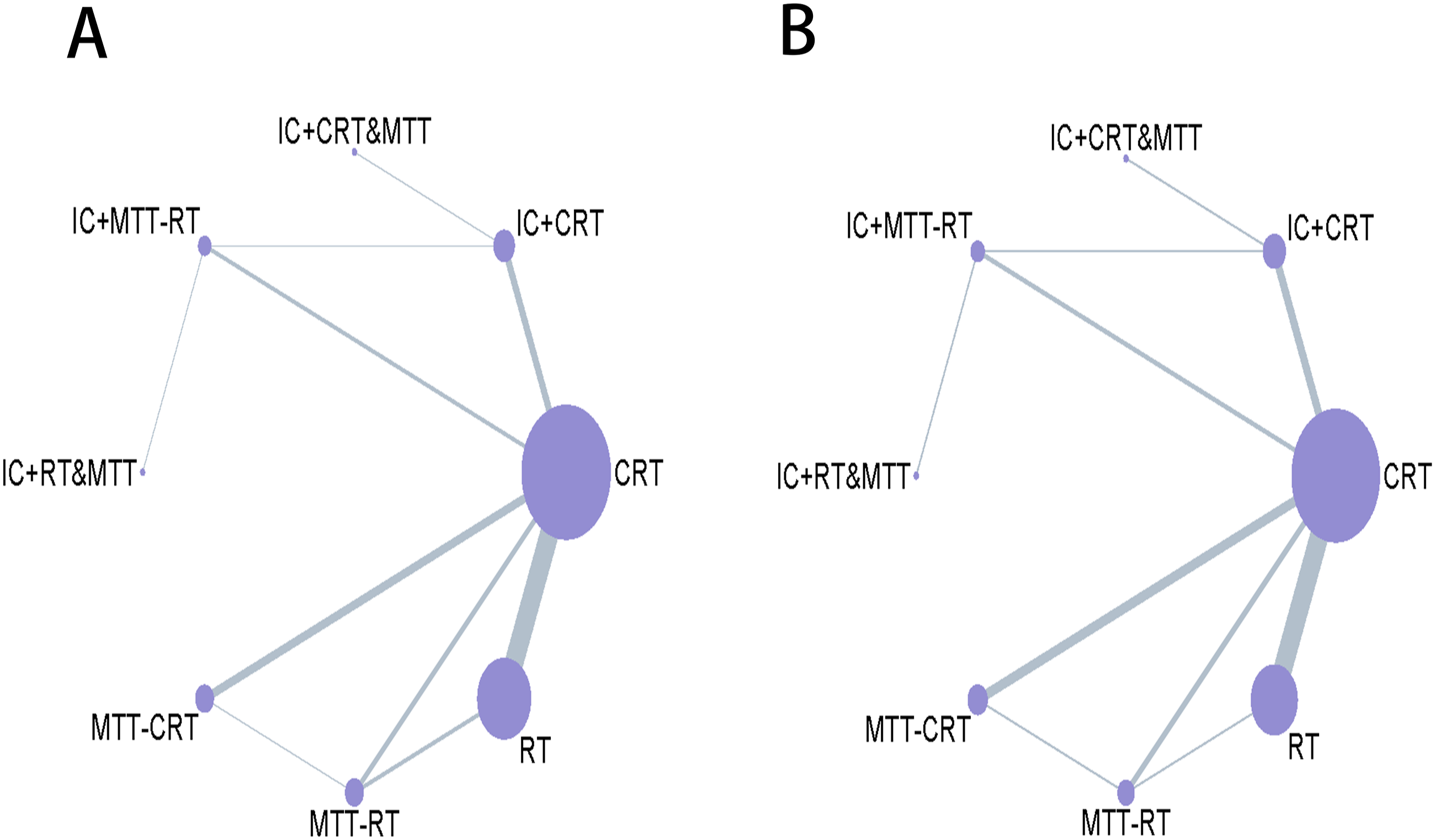
Subsequently, we conducted statistical calculations using the Gemtc software in R 4.3.1, which is an R extension package specifically designed for Bayesian network meta-analysis. Gemtc provides a suite of functionalities and tools for data analysis and visualization. With the application of Bayesian statistical methods through Gemtc, we aimed to achieve more accurate results. For a comprehensive analysis of the included clinical trials, we created a forest plot mapping the study outcomes. We assessed trial heterogeneity using the I2 statistic, 9 If the I2 statistic indicated low or moderate heterogeneity (I2<50%), we employed a fixed-effects model for the analysis. On the other hand, if the I2 statistic indicated high heterogeneity (I2 ≥ 50%), we employed a random-effects model for the analysis. Moreover, we employed SUCRA values to evaluate the efficacy of various treatment regimens, with higher scores indicating better effectiveness. 10 A network meta-league table was created to rank the treatments based on their SUCRA values. This approach facilitated the evaluation of each treatment’s relative effectiveness, enabling the establishment of a comprehensive treatment ranking. To enhance the accuracy of the results, a sensitivity analysis was performed, excluding studies with limited relevant data.
Results
Study Characteristics
Characteristics of included studies.

Quality Assessment
Our analysis predominantly comprised high-quality multicenter trials. In terms of bias risk, we identified 28 studies with low randomization bias, 19 with low allocation concealment bias, 29 with low bias concerning incomplete outcome data, and 31 with low selective reporting bias. The included studies showed high quality regarding randomization, allocation concealment, and complete outcome data reporting. This low bias risk implies our statistical analysis results are less likely to be skewed, making these studies suitable for our analysis. However, it’s important to note some studies had unclear bias risk due to insufficient methodological details reported, particularly around randomization and allocation concealment. Furthermore, three studies were classified as “unclear risk” due to selective reporting, as they failed to incorporate PFS, a crucial outcome measure for evaluating treatment efficacy. Despite uncertainties in methodological reporting, the included studies still demonstrated high quality on other evaluation criteria like sample size and clinical relevance of the interventions. Therefore, they were considered suitable for inclusion in our statistical analysis. For a comprehensive bias risk assessment, please refer to Figure 3. . Risk of bias assessment for all included studies.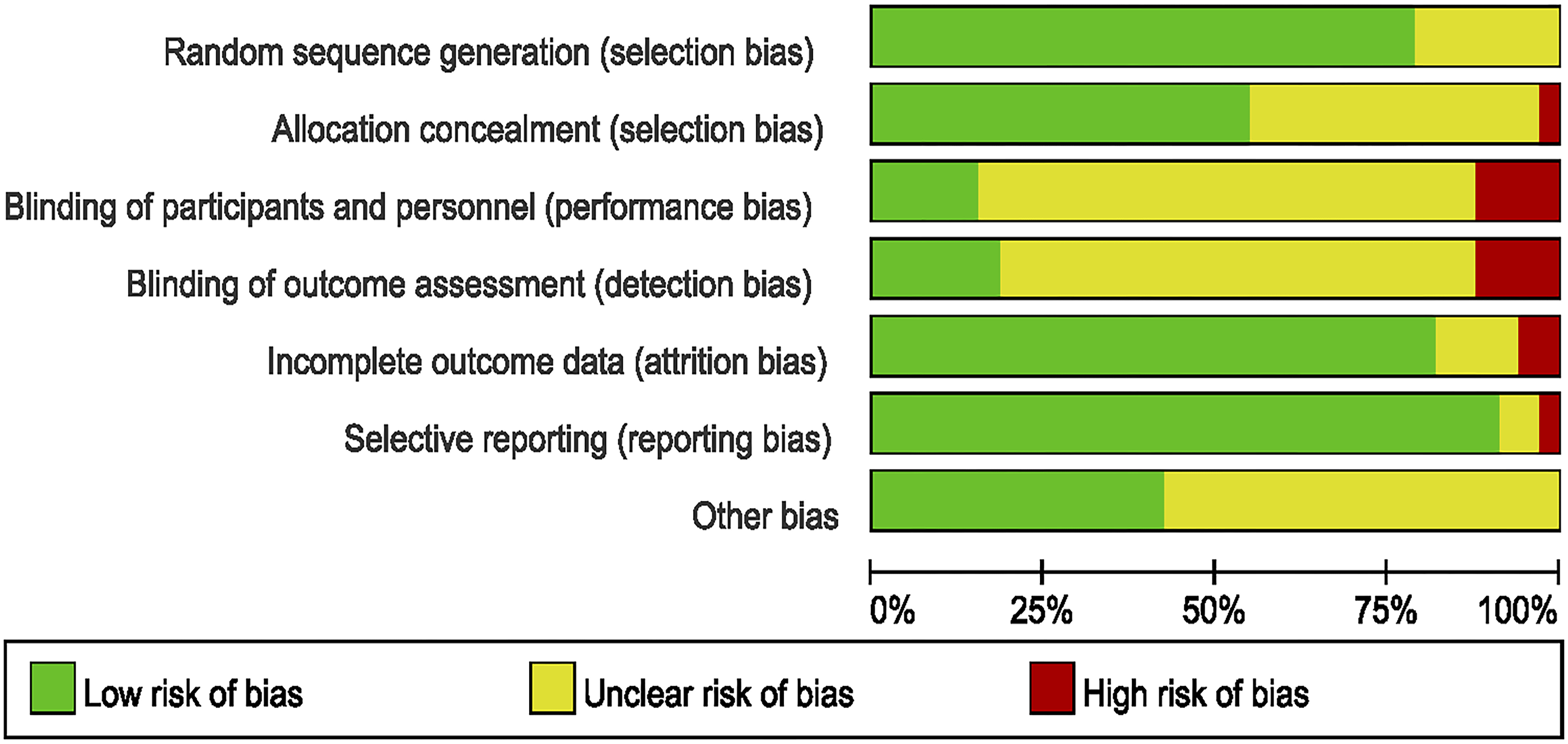
Sensitivity Analysis
Given the limited number of studies available for comparing the efficacy of IC + CRT&MTT and IC + RT&MTT, we conducted a sensitivity analysis by excluding these two articles. This decision was made due to potential lack of statistical power resulting from the small number of comparisons.30,31 A sensitivity analysis of 31 published studies11-43 demonstrates similar results to the full network meta-analysis (33 published studies) (Supplemental Figure 2). Regarding OS, MTT-CRT and CRT are in the top two positions of the SUCRA league table, indicating better patient survival compared to radiation therapy alone. For PFS, upon excluding IC + CRT&MTT and IC + RT&MTT, the rankings of the remaining treatments align with the results from the comprehensive network meta-analysis. The rankings are as follows: MTT-CRT, IC + CRT, CRT, IC + MTT-RT, and RT. Additionally, all four treatments demonstrate a PFS benefit compared to radiation therapy alone.
Network Meta-Analysis
In terms of overall survival, the forest plot illustrates that IC + RT&MTT (HR .68, 95% CrI 0.25-1.8), IC + CRT&MTT (HR .72, 95% CrI 0.071-7.4), and MTT-CRT (HR .93, 95% CrI 0.80-1.1) show a favorable trend over CRT. Yet, this trend fails to reach statistical significance, as shown in Figure 4. In addition, the league table ranks the treatments for OS based on the SUCRA values in the following sequence: IC + RT&MTT, MTT-CRT, IC + CRT&MTT, CRT, IC + CRT, MTT-RT, IC + MTT-RT, and RT, as shown in Figure 5. Among these, MTT-CRT and CRT demonstrate significant improvements in OS compared to RT. The I2 for inconsistency analysis regarding OS is 40%, indicating low heterogeneity among the literature included in the meta-analysis. Forest plot of (A) progression-free survival (PFS), (B) overall survival (OS). CRT:concurrent chemoradiotherapy;IC + CRT:neoadjuvant chemotherapy followed by concurrent chemoradiotherapy;RT:radiotherapy alone;IC + MTT-RT:induction chemotherapy followed by concomitant radiotherapy with targeted therapy; MTT-CRT:targeted therapy concomitant with concurrent chemoradiotherapy;MTT-RT:targeted therapy concomitant with radiotherapy;IC + RT&MTT:concurrent radiotherapy and targeted therapy following chemotherapy and targeted Induction;IC + CRT&MTT:concurrent targeted therapy and radiochemotherapy following chemotherapy and targeted induction. Efficacy of the Bayesian network meta-analysis in patients with LASCCHN Hazard ratios and 95% CIs for progression-free survival (upper triangle) and overall survival (lower triangle), and a hazard ratio <1.00 provides better survival benefits.

In terms of progression-free survival, the HR in IC + CRT&MTT (HR .68, 95% CrI 0.22-2.1), IC + RT&MTT (HR .83, 95% CrI 0.40-1.8), MTT-CRT (HR .90, 95% CrI 0.79-1.0), and IC + CRT (HR .91, 95% CrI 0.78-1.1) show trends towards an extension of PFS compared to CRT, as revealed by the forest plot. However none of these treatments exhibit significant superiority over concurrent chemoradiotherapy. According to the league table, the ranking for PFS is as follows: IC + CRT&MTT, MTT-CRT, IC + CRT, IC + RT&MTT, CRT, IC + MTT-RT, MTT-RT, and RT. Moreover, compared to RT, MTT-RT, MTT-CRT, and IC + CRT all exhibit significant benefits in improving PFS. MTT-CRT also proves to be more effective in enhancing PFS compared to MTT-RT. The I2 for the inconsistency analysis regarding PFS is 3%, indicating low heterogeneity among the included literature in the meta-analysis. However, neither the forest plot nor the league table display any treatment demonstrating statistically significant superiority over CRT in terms of both overall survival and progression-free survival.
Discussion
Our network meta-analysis did not find significant effects when including the treatment regimen with concurrent chemoradiotherapy (CRT) in locally advanced head and neck squamous cell carcinoma. However, our findings indicate that employing a combination of targeted therapy and induction chemotherapy with CRT might be a more promising treatment approach in the future.
For progression-free survival (PFS), the rankings are as follows: IC + CRT&MTT takes first place, followed by MTT-CRT, IC + CRT, IC + RT&MTT, CRT, IC + MTT-RT, MTT-RT, and RT. Our results suggest that the advantage of IC + CRT&MTT over CRT might be attributed to the inclusion of targeted therapy and induction. Anti-EGFR targeted drugs can suppress tumor cell proliferation, enhance apoptosis, reduce angiogenesis, invasiveness, and metastasis by inhibiting EGFR receptor activation. It is worth noting that EGFR is overexpressed in approximately 80%–90% of head and neck squamous cell carcinoma cases. 44 Hence, the role of targeted drugs in treating this cancer type is widely recognized. Patil et al 38 demonstrated that adding nivolumab to cisplatin-based concurrent chemoradiotherapy significantly improved patients’ progression-free survival (PFS), locoregional control (LRC), and disease-free survival (DFS). This benefit is particularly noticeable in patients with unfavorable prognostic factors. Additionally, nivolumab exhibits higher specificity for tumor binding, leading to more effective inhibition of tumor cell proliferation. This might explain the improved survival outcomes when nivolumab is added to concurrent chemoradiotherapy and supports the superior performance of IC + CRT&MTT over IC + CRT, especially in terms of PFS. On the other hand, induction chemotherapy has proven effective in reducing tumor size, eliminating micrometastases, and decreasing local-regional recurrence rates. As a result, it can be a suitable treatment option for patients who are not suitable for surgery or require organ preservation.45–49 Additionally, one study indicated that induction chemotherapy before concurrent chemoradiotherapy improves local recurrence risk, enhances local control rates, and reduces the need for salvage surgery. 37 According to these results, the inclusion of induction therapy in concurrent chemoradiotherapy is clinically meaningful in the treatment of advanced-stage head and neck squamous cell carcinoma. Furthermore, Lee et al 31 found significant improvement in PFS when cetuximab was added to induction chemotherapy and concurrent chemoradiotherapy, particularly in patients who completed three induction chemotherapy cycles. These findings contribute to the superior efficacy of IC + CRT&MTT over MTT-CRT seen in recent studies. In summary, our results indicate that the adding induction or targeted therapy trends to improve PFS compared to standard chemoradiotherapy (CRT) treatment. However, these outcome differences did not reach statistical significance.
For OS, the ranking is as follows: IC + RT&MTT, MTT-CRT, IC + CRT&MTT, CRT, IC + CRT, MTT-RT, IC + MTT-RT, and RT. Firstly, when comparing IC + RT&MTT to CRT, an overall improvement in OS is observed. This improvement can be attributed to the addition of targeted therapy and induction chemotherapy. A literature review demonstrated that sequential treatment with cetuximab and radiotherapy following induction chemotherapy had similar short-term efficacy to cisplatin plus radiotherapy. However, it resulted in better success rates, safety, and compliance in salvage surgery. Ultimately, these factors contribute to improved OS. 47 Additionally, it is noteworthy that our results indicate IC + RT&MTT outperforms IC + CRT&MTT in terms of OS. This suggests that an all-encompassing treatment approach involving induction therapy, targeted therapy, and concurrent chemoradiotherapy may lead to physical deterioration in patients and reduced tolerance to salvage treatment. Therefore, we speculate that omitting concurrent chemotherapy might be a feasible option when targeted therapy and induction chemotherapy are included. In addition, the MTT-CRT regimen also demonstrated a survival benefit regarding OS, similar to what was observed for PFS. This improvement is likely attributable to the inclusion of targeted therapy. A recent network meta-analysis 50 has shown that adding nivolumab to concurrent chemoradiotherapy improves treatment outcomes. However, our study differs from this meta-analysis as we classified treatments into different categories. For example, in the category of targeted therapy, we included various targeted drugs such as cetuximab, nivolumab, and erlotinib. Although these targeted drugs exert their effects through the EGFR target, we grouped them together. Despite these differences, we arrived at a similar conclusion: combining targeted therapy with concurrent chemoradiotherapy is likely more effective than CRT alone.
The above results show that adding targeted therapy and induction chemotherapy offers an OS benefit for patients with locally advanced head and neck squamous cell carcinoma compared to CRT alone.
However, the existing literature on IC + CRT&MTT and IC + RT&MTT is limited, warranting careful interpretation of these results. To mitigate this limitation, we performed a sensitivity analysis by excluding these two studies. The subsequent findings continued to suggest that including targeted therapy or induction chemotherapy with CRT may provide improved efficacy compared to CRT alone. Although no statistically significant superiority was observed, there remains a favorable trend in terms of efficacy for targeted therapy and induction chemotherapy when compared to CRT, as indicated in the sucra league table.
Our study has several limitations. Firstly, while our findings suggest an improvement in survival compared to CRT, these results do not reach statistical significance. However, we can observe the relative advantages of targeted and induction chemotherapy combined with concurrent chemoradiotherapy over concurrent chemoradiotherapy alone. Secondly, toxicities were not included as an outcome measure in our analysis, and safety differences across studies were not compared. Additionally, the literature we reviewed may lack detailed information on factors such as HPV status or primary tumor site. Consequently, our assessment of treatment regimens might have been limited in terms of comprehensively evaluating both efficacy and side effects. Thirdly, our study focused on comparing treatment categories rather than distinguishing specific drugs and treatments within each category. Thus, assessing which drug is more effective was not possible. Moreover, the efficacy of the immune checkpoint inhibitor PD-L1 in recurrent and metastatic SCCHN has been confirmed. 51 Whether adding immune checkpoint inhibitors to CRT or radical radiotherapy benefit patients with locally advanced SCCHN has become a research hotspot recently. However, due to the current lack of detailed literature on the combination of immune checkpoint inhibitors with CRT vs CRT alone, the efficacy of immune drugs was not included in this network meta-analysis.
Conclusion
Comparatively, the combination of targeted therapy or induction chemotherapy with chemoradiotherapy has the potential to improve treatment efficacy for patients with locally advanced squamous cell carcinoma of the head and neck (LASCCHN) when compared to concurrent chemoradiotherapy (CRT) alone.
Supplemental Material
Supplemental Material - A Network Meta-Analysis of the Systemic Therapies in Unresectable Head and Neck Squamous Cell Carcinoma
Supplemental Material for A Network Meta-Analysis of the Systemic Therapies in Unresectable Head and Neck Squamous Cell Carcinoma by Jinling Yuan, Kexin Shi, Guanhua Chen, Weilin Xu, Lei Qiu, Yinjiao Fei, Yuchen Zhu, Mengxing Wu, Yurong Li, Xinchen Sun, Yuandong Cao, and Shu Zhou in Cancer Control.
Footnotes
Authors’ Note
As our study is a network meta-analysis, ethics statement is not required.
Acknowledgments
We would like to thank JinLing Yuan and WeiLin Xu for the literature search.
Author Contributions
Study conception and design: Xinchen Sun; Yuandong Cao; Shu Zhou. data collection: Jinling Yuan; Weilin XU; Yinjiao Fei. analysis and interpretation of results: Guanhua Chen; Lei Qiu;Jinling Yuan; draft manuscript preparation: Mengxing Wu;Yuchen Zhu; Yurong LI. All authors have read and agreed to the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
With publication, the data set used for this meta-analysis will be shared upon request from the study authors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
