Abstract
Objectives
• Gather a panel of Latin American experts in testing and treating BRAF-melanoma. • Describe the current landscape of BRAF-mutated melanoma in Latin America. • Outline the current gaps in testing and recommend improvements for testing and treating BRAF-mutated melanoma in the region.
Introduction
Melanoma prevalence in Latin America is lower than in high- and middle-income countries. However, recent data indicate that the region’s incidence and mortality are rising, with more stage IV patients being diagnosed. According to international clinical practice guidelines, conducting BRAF-mutation testing in patients with stage III or stage IV melanoma and high-risk resected disease is imperative. Still, BRAF-mutation testing and targeted therapies are inconsistently available in the region.
Methods
Americas Health Foundation convened a meeting of Latin American experts on BRAF-mutated melanoma to develop guidelines and recommendations for diagnosis through treatment.
Results and Conclusions
Some recommendations for improving diagnostics through improving access and reducing the cost of BRAF-mutation testing, enhancing efficiency in pathology laboratories, and creating country-specific local guidelines. The panel also gave treatment recommendations for neo-adjuvant therapy, adjuvant therapy, and therapy for patients with metastatic disease in Latin America.
Introduction
Clinicians’ understanding of BRAF-mutational status is essential to faster treatment and improved outcomes. This narrative review aims to describe the current landscape of BRAF-testing in melanoma cases in Latin America (LATAM) and provide expert recommendations for diagnosis and treatment. Understanding melanoma signaling pathways improves the characterization and development of targeted therapies. The World Health Organization (WHO) classifies melanocytic lesions into 9 categories based on genetic changes. Ultraviolet (UV) radiation determines chronic sun damage (CSD) and is a major risk factor. 1 Low-CSD melanomas, or superficial spreading melanomas, are the largest group and frequently have the BRAF (v-RAF murine sarcoma viral oncogene homolog B1) V600 mutation. 2 High-CSD melanomas, such as lentigo maligna and desmoplastic melanomas, often have numerous mutations and may carry NRAS, BRAF non-V600E, or NF1 mutations. Melanomas not associated with CSD, including acral and mucosal melanomas, typically lack BRAF, NRAS, or NF1 mutations (triple wild-type), but a subset may have KIT or SF3B1 mutations. 3 Half of the melanoma cases have BRAF-mutation (V600 E), causing 500-700 times increased kinase activity. This mutation is commonly linked to melanoma and is found in benign melanocytic tumors. Other gene changes are necessary for complete melanoma transformation. 4 Clinicians, laboratory scientists, and policymakers in Latin America (LATAM) must understand the current practice of diagnosing and treating BRAF-mutated melanoma to chart a course for improving access and providing patients personalized treatment options.
Methods
Americas Health Foundation (AHF) used PubMed, MEDLINE, and EMBASE to identify LATAM-based oncologists and pathologists who have published in melanoma, oncology, and BRAF testing. AHF used the following search terms: ‘BRAF,' ‘BRAFV600', ‘treatment,' and ‘melanoma' in combination with ‘Latin America' from January 1, 2017, to June 10, 2022. The identified articles were in English, Portuguese, and Spanish. Six experts from LATAM (Argentina, Brazil, Chile, Colombia, and Mexico) met for 3 days in April 2023, to examine the landscape of diagnosing and treating BRAF-melanoma in LATAM and to create recommendations for optimum diagnosis and treatment. AHF assigned each panel member a question on BRAF-melanoma in LATAM. Individual panel members answered their question based on the AHF literature review, their reviews, and personal knowledge. The panel reviewed and amended each answer during a 3-day meeting with many discussion rounds. Following the meeting, the panel evaluated and approved the final document.
Results
Melanoma Prevalence in LATAM
Melanoma prevalence in LATAM (1.7/100 000) 5 is lower than in high- and middle-income countries. However, recent data show that the region’s incidence and mortality are rising, with more stage IV patients being diagnosed. 6 BRAF-testing can be an effective method for melanoma mutation detection, but it has limitations. Treatment delays can occur while physicians await results. Additionally, heterogeneous genetic ancestry among LATAM of mixed Native American, European, and African descent accounts for differences in the genetic profiles of melanoma and other solid tumors. LATAM represents 10% of the global population living in 33 countries with social diversity and economic heterogeneity. 7 International data show that mortality from melanoma is higher in lower-income areas of the world (57.5%) compared with higher-income areas (16.3%). 8 Histological subtypes also differ in the region: most patients with acral melanomas live near the equator, and the highest incidence of patients with extensive superficial melanomas live in the south. The difference in histology also reflects the variability in the frequency of patients with mutant and non-mutant BRAF melanomas.9-13
Understanding of the prevalence of BRAF mutations in melanoma in LATAM is evolving. However, retrospective data suggest BRAFV600E is the most common mutation, accounting for approximately 50% of BRAF mutations in melanoma patients in LATAM. For example, a study conducted in México 14 reported a BRAF mutation prevalence of 54.6%, with the V600 E mutation being the most frequent. The reported prevalence varies across the region, with 50% in Argentina, 39% in Brazil, and 45% in Colombia. 15 The prevalence of BRAF mutations in melanoma can vary based on several factors, including geographic location, race, and ethnicity. Thus, it is essential to consider the local epidemiology of melanoma when evaluating the potential impact of BRAF-testing on clinical outcomes.
BRAF-Mutation Tests in LATAM
BRAF-mutation melanoma diagnosis is available through several tests, and immunohistochemistry (IHC), real-time PCR (RT-PCR), and next-generation sequencing (NGS) are the most common detection methods. Each testing method has unique strengths, weaknesses, and challenges that should be considered before testing a patient’s tumor sample. 16 Some diagnostic centers use IHC as a screening tool, while others prefer RT-PCR or NGS. In Europe, laboratories use VE1 monoclonal antibodies to detect BRAFV600BRAFE protein mutations efficiently in tissue samples. Although the antibody does not quantify expression, it is a qualitative tool with high sensitivity (98.6%) and specificity (97.7%). This test is relatively inexpensive and quickly produces results. RT-PCR testing compares the intensity of the mutant and wild-type signal with high sensitivity (96%) and specificity (100%). Despite the cost-effectiveness and speed of RT-PCR, this method frequently employs kits that only target the most prevalent mutations, including V600E, V600K, and V600D. NGS technology has high sensitivity (100%) and specificity (99%) for detecting all BRAF mutations.17-19 However, access in LATAM is limited because of high costs and delayed results. Although liquid biopsy is a non-invasive emerging tool for BRAF-testing, it is not widely used in clinical settings.
BRAF-Mutation Testing Conducted in Latin America
According to international clinical practice guidelines, it is imperative to conduct BRAF-mutation testing in patients with stage III or stage IV melanoma and high-risk resected disease. 20 BRAF-testing methods are typically limited in LATAM to pharmaceutical industry-sponsored diagnostic programs or melanoma detection campaigns conducted by private nonprofit organizations, such as the Venezuelan Anti-Cancer Society, the Colombian Centro Dermatologico Federico Lleras Acosta, the Brazilian Society of Dermatology, and the Fundación Cruzada Patagonica and Sociedad Argentina de Dermatología of Argentina.
Reflex molecular testing is attractive, particularly for less commonly ordered biomarkers. To improve BRAF-testing accessibility, regional governments should establish centralized testing facilities, public-private partnerships, and awareness campaigns for better laboratory and supplies access. Scientific associations should implement quality assurance programs for pathology. In Colombia, the national healthcare system covers PCR costs (USD300), and in Costa Rica, mutation analyses, including BRAF, are included in public healthcare. Unfortunately, access to BRAF-inhibitors is limited to those who can afford them financially. In Mexico, pharmaceutical companies provide BRAF-mutation tests, but access to targeted therapies remains a significant challenge throughout LATAM.
Metastatic samples are preferred for analysis, but in their absence, lymph node metastases or the primary tumor may be used, as there is a high level of concordance between the BRAF status of primary melanomas and their metastases. 21 Testing for BRAF-mutation at an earlier stage (IIB, IIC) is indicated for patients with limited access to appointments and specialists 22 to avoid needless delays in targeted BRAF/MEK inhibitor therapy. Early testing for BRAF mutations in melanoma patients can identify those likely to respond to targeted therapy with BRAF-inhibitors, improving overall survival (OS) rates.22-25 Patients diagnosed with BRAF-mutated melanoma stage I-III had worse OS than those without the mutation, suggesting that early detection of a BRAF-mutation in localized melanoma may be necessary for identifying patients at higher risk for disease progression and who may benefit from more aggressive treatment. 26
BRAF-Mutated Melanoma Treatment in LA
Access to standard-of-care treatment for BRAF-mutated melanoma is limited in LATAM, especially in the public healthcare system. New therapies are often accessible only through clinical trials. However, clinical trials come with delays, costs, and time limitations, limiting patients to short-term solutions. Previously, a consensus panel found at least 1 targeted therapy (TT) for BRAF-mutated melanoma is available and approved for treatment in Argentina, Brazil, Chile, Colombia, Mexico, and Uruguay. In some LATAM countries, TTs are unavailable to patients in the public healthcare system. TT offers significant advantages over chemotherapy, including a high response rate and rapid improvement of disease symptoms.27-30 Unfortunately, in some LATAM countries, chemotherapy is the only available treatment choice for patients with metastatic melanoma. (Figure 1) No evidence suggests it provides any advantage in OS with patients with metastatic melanoma. Approved systemic treatments and guidelines for countries in Latin America. The map reflects available medications; however, all countries do not guarantee insurance and healthcare coverage.
Ideal Treatment Decisions and Sequencing for Melanoma in LATAM
In precision oncology, identifying targets for personalized treatment is crucial for cancer patients. The BRAF-mutation rate and effectiveness of targeted therapies in LATAM melanoma patients remain unknown. Regional treatment decisions are primarily based on Western studies. Initially, BRAF and MEK inhibitors were used for metastatic melanoma, and more recently, they have been compared to immunotherapy in the first-line setting.
Clinical Trial Outcomes for BRAF-Inhibitors
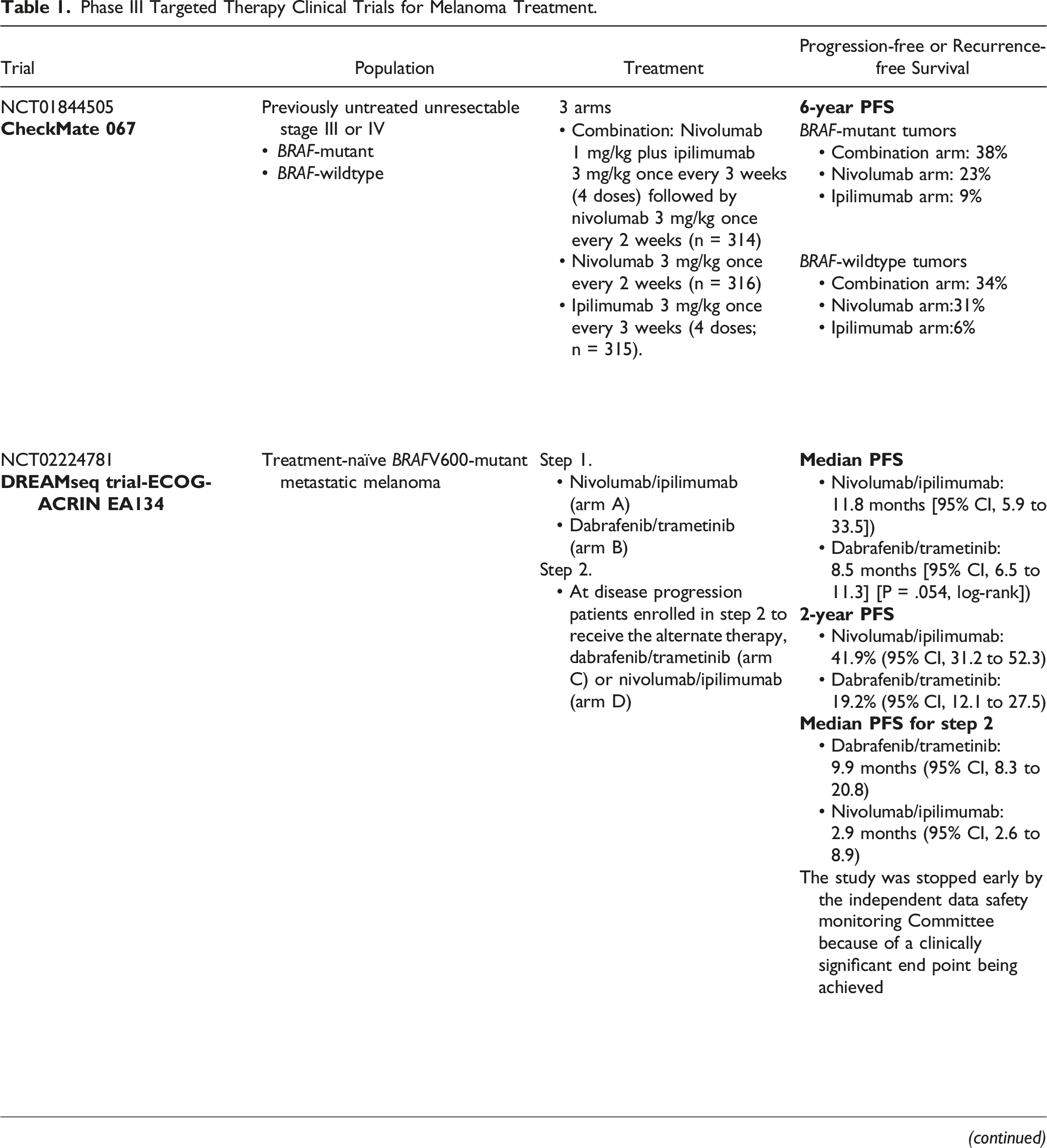
The phase III DREAMseq trial, published in January 2023, was the first to address the potential benefit of dual immunotherapy with nivolumab-ipilimumab (NI) in treatment-naïve patients, followed by the BRAF-MEK inhibitors, dabrafenib-trametinib (DT) upon progression. 19 Progression-free survival (PFS) was the study’s primary endpoint, with OS and objective response rate (ORR) as secondary endpoints. In this study, the median PFS was significantly longer in the NI group (11.5 months) than in the DT group (8.2 months); the ORR was also significantly higher in the NI group (58.9%) than in the DT group (46.4%). However, the study found no significant difference in OS between the 2 groups, with a median OS of 25.1 months (NI) and 21.2 months (DT). The authors conclude that while the NI combination therapy showed superior PFS and ORR compared to the DT combination therapy; it had a higher incidence of grade 3 or 4 adverse events. Therefore, the current suggestion is that physicians should base treatment options on individual patient factors and preferences and that further research is needed to identify biomarkers that may predict response to these treatments. Also, in the DREAMseq trial, immunotherapy was less effective after TT, with lower ORR achieved in patients exposed to immunotherapy after progression to first-line TT than in those who received immunotherapy in the first-line. This panel believes TT should be the first-line treatment option, mainly in patients who are ineligible for or who decline combination immunotherapy.
Patients who will receive immunotherapy prefer treatment with combination immunotherapy, either with NI (the only option available across LATAM currently) or with nivolumab-relatlimab (NR) over a single-agent PD-1 inhibitor. In the checkmate 067 trial, NI improved PFS and OS compared to nivolumab monotherapy.31,32 In the subgroup analysis performed at a 6.5-year follow-up, the 301 patients with BRAFV600-mutant tumors appeared to benefit more from the combination than nivolumab alone. Also supporting these findings, in the phase III RELATIVITY-047 trial, in which 275 patients had advanced unresectable or BRAFV600-metastatic melanoma, there was an improvement in PFS compared to nivolumab monotherapy, with a more favorable toxicity profile. 33 Although this combination is not approved in the region, some LATAM countries participated in the trial.
Additionally, the outcomes of KEYNOTE-001, KEYNOTE-002, and KEYNOTE-006 trials support anti-PD1 monotherapy with pembrolizumab use in BRAFV600 E/K-mutant melanoma. 34 Pembrolizumab achieved comparable rates of 4-year PFS (23% vs 20%) and OS (38% vs 35%) in BRAF-mutated melanoma patients compared to BRAF-wild type. Based on the above data, this panel recommends using single-agent PD-1 inhibitors as a first-line treatment only for patients unable to tolerate the potential toxicities of combination immunotherapy. Still, it should be preferred over TT for most patients.
Three TT of BRAF and MEK inhibitor combinations exist: DT, encorafenib-binimetinib (EB), and vemurafenib-cobimetinib (VC). Although they have not been directly compared in clinical trials, all appear to have similar efficacy, including ORRs of up to 70%. In a combined analysis of COMBI-d and COMBI-v, DT had a median PFS and OS of approximately 11 and 26 months, respectively. 35 Estimated PFS and OS at 5 years were about 19% and 34%, respectively. This combination could be suitable for patients with central nervous system metastases, as it can cross the blood-brain barrier.
The phase III trial (COLUMBUS) was performed in 577 patients with BRAFV600-mutated melanoma, comparing the combination of the BRAF inhibitor encorafenib plus the MEK inhibitor binimetinib vs either encorafenib alone or the single-agent BRAF inhibitor vemurafenib. 36 Encorafenib could be the preferred option for patient convenience. Patients can take it with or without meals, and it is stored at room temperature, as opposed to the combination of DT, which requires administration around meals and refrigeration for trametinib.
Phase III Targeted Therapy Clinical Trials for Melanoma Treatment.
Treatment of Patients with BRAF-Mutated Melanoma in LATAM
Guidelines Informing Treatment Decisions.

Abbreviation: SBOC, Sociedade Brasileira De Oncologia Clinica; AAOC, Argentinian Society of Clinical Oncology; AHCO, Asociación Colombiana de Hematología y Oncología
Treatment sequencing in the real world depends on the initial regimen and country approvals. Patients may receive single-agent anti-PD1 for low-volume disease or dual immune-checkpoint inhibitors for symptomatic/high tumor burden cases. Those with advanced mutant BRAF melanoma often switch to TT upon progression. LATAM-specific treatment decisions lack supporting data, so clinicians rely on institutional expertise and international guidelines. 41 Among the panel members, most patients with BRAF-mutated melanoma were prescribed first-line immunotherapy combinations or TTs compared to single-agent anti-PD1 therapies.
As in other areas of oncology, once a treatment proves its value in the metastatic scenario, it is used in earlier stages of the disease. Patients with non-metastatic high-risk melanoma (IIB-IIID), high-risk node-positive microscopic disease, and a BRAFV600 driver mutation can be treated with 1 year of either immunotherapy or TT since both have demonstrated improved outcomes. Nevertheless, access is heterogeneous in LATAM, and the optimal adjuvant therapy has not been established worldwide.
Adjuvant DT showed improved OS in a phase III trial. 42 Patients who cannot receive immunotherapy or are at greater risk for immune-related adverse events may benefit from adjuvant DT because toxicities from TT are reversible once treatment is discontinued, distinct from potential long-term toxicities, primarily endocrine, resulting from immunotherapy. In the COMBI AD trial, 870 patients with completely resected BRAFV600-mutation-positive stage III melanoma were randomly assigned to the combination of DT or matching placebos for 1 year. At a median follow-up of 5 years, the combination of DT was found to improve both 5-year recurrence-free survival (RFS) (52% vs 36%, HR .51) and 3-year OS (86% vs 77%, HR .57) compared with placebo. In a phase III trial (CheckMate 238), 1 year of adjuvant nivolumab improved RFS over ipilimumab among 906 patients with complete resection of AJCC seventh edition stage IIIB, IIIC, or IV melanoma. 43 Of 381 patients with BRAF-mutant tumors (42%), nivolumab trended toward improved 5-year RFS compared with ipilimumab (50 vs 42%, HR .8). The other anti-PD1 alternative is pembrolizumab. In the KEYNOTE-054, 44 a phase III trial, 1019 patients with completely resected stage III (including stage IIIA tumors with ≥1 mm of disease in the lymph nodes) received 1 year of adjuvant treatment. In the 440-patient subset with BRAFV600-mutated tumors, pembrolizumab improved 3-year RFS at a median follow-up of 3 years (62% vs 37%, HR .51).
Pros and Cons of Oral and IV Adjuvant Medications.

This panel recommends using adjuvant therapy, either with TT or immune therapy, according to accessibility. Still, published data shows higher rates of recurrent disease (25%–30%) during the adjuvant immunotherapy period compared to patients treated with TT (12%).44,46,47
Last, appropriate treatment in the neo-adjuvant setting is still being studied. Patients with resectable macroscopic stage IIIB-IV melanoma are currently given immunotherapy before surgery;48,49 the preliminary results showed a promising complete response. For BRAF-mutated patients who do not respond well and may be resistant to immunotherapy, neo-adjuvant therapies should only be considered in clinical trials. Access to certain treatment options is limited, and there is insufficient information regarding mutational profiles and other factors that may impact treatment response. Other regional obstacles are affordability and reimbursement of treatment, limited infrastructure, fragmented healthcare systems, lack of human resources, pitfalls in the sample journey, and quality assurance. 13 Adherence to guidelines in melanoma patients has been established as a good practice worldwide to ensure better patient outcomes.50-52 There is no prospective validation data of the international guidelines adapted for our region. Also, there are no publications regarding adherence to international guidelines.20,53 Improving access to effective melanoma treatments is a critical concern in LATAM. The availability of drugs varies, limiting medical care. Ensuring that all globally used medical technologies are accessible to patients in our region is essential. Some international approvals, such as TT triplet combinations (BRAF, MEK, and immunotherapy), T-VEC, Tils therapy, and tebentafusp for ocular melanomas, are unavailable in LATAM. 54 Not all LATAM countries can access the diversity of TT available in the US and Europe. The best practices for guiding patient treatment consider safety profiles, dosage frequency, storage, and demographic characteristics and should not be based on the only option available. Regarding the future trends in melanoma diagnosis and the evolving treatment landscape, LATAM must prepare for a sustainable approach to precision medicine to treat the right patient with the right approach. The preparation involves many areas, including:
Education
Improving knowledge dissemination in cancer care is crucial to addressing disparities in access to targeted treatments across LATAM regions. Education should cover technical topics, healthcare system structure, and patient care rights. Pathologists, oncologists, and surgeons need to be well-informed about genomic testing. Continued medical education is essential to increase awareness and promote equitable access to high-quality care.
Artificial Intelligence (AI)
Data intelligence in acquiring, organizing, and interpreting patient data will improve decision-making. AI visualization can optimize indicators like therapy indication, complication monitoring, and therapy response, significantly improving patient care. Radiomics and pathology may incorporate image-based algorithms, transforming diagnostics. AI will be an asset in diagnosing, testing, and treating BRAF-mutated melanomas. 55
Turnaround Time
Expediting the turnaround time of services in healthcare facilities and among diagnostic services such as radiology and pathology is crucial to optimize the length of the patient diagnosis journey, increasing their chances of being able to engage in proper treatment or clinical trials within the correct time frame, thus allowing the best treatment and best outcome.
Regulatory Agencies
LATAM needs to work on regulatory pathways for incorporating new technology and treatment, with lessons learned from the COVID-19 pandemic. The contentious issue of pay-per-performance strategy in the healthcare system needs careful consideration. Payers should collaborate with healthcare facilities to reduce financial toxicity for patients and themselves. Ensuring molecular testing accessibility through insurance coverage is also crucial.
Medical Societies
Local clinical practice guidelines are essential tools to disseminate cancer care knowledge and are a step toward delivering better and more homogeneous care in LATAM. Medical and academic societies might coordinate guidelines to match local technology, workflow, and treatment with local regulatory agencies to improve patient care.
Patient Access and Affordability
Access to Testing
Molecular laboratories are usually in urban centers, making access to testing more challenging for remote regions of many LATAM countries. Implementing a patient-centered approach to testing will provide patients with streamlined access to care through referrals to well-equipped centers.
Affordability of Testing
Affordability poses a challenge to precision medicine adoption in LATAM’s public and private healthcare systems. Public systems lack coverage for molecular testing in BRAF-mutant melanoma, while private insurances have limitations. Current testing focuses on specific genetic hotspots, but consensus, regulation, and integration of comprehensive biomarker panels may be necessary for optimal care in the future. Pharmaceutical companies offer testing programs, but these lack public policy support and may not be sustainable. Competition from centralized labs can impact the availability and pricing of local tests.
Research
Clinical Research
Affordable approaches for clinical trials, incorporating new technologies like AI and machine learning, are urgently needed for sustainable drug development from basic research to clinical validation, which could lead to improvements in population health. 56 In LATAM, institutions are engaged in international phase II and III trials but face challenges in finding eligible patients and transportation. Since April 2014, LATAM countries have enrolled fewer patients in clinical trials than North American countries. 57
Translational Research
Investing in translational research in BRAF-mutant melanoma in LATAM can lead to the development of affordable and accessible treatment options, including novel therapeutic targets and TT tailored to the local population. Translational research can also contribute to developing suitable diagnostic tools and biobanking initiatives in LATAM, improving early diagnosis and research resources. Furthermore, investing in translational research can help build local research capacity, fostering knowledge exchange and making LATAM a hub for BRAF-mutant melanoma research.
Collaborations
Using solely American and European genomic data causes biases. The Latin American Cooperative Oncology Group (LACOG), US-Latin-America Cancer Research Network, Latin American Federation of Cancer Societies, and Latin American Consortium for Lung Cancer Research (CLICaP) strive to improve clinical research in LATAM. Numerous initiatives in LATAM are working to increase genomic data reflecting underrepresented populations to improve treatment choice access and patient outcomes. Argentina created the Argentinian Population’s Reference and Genomics Biobank program (PoblAr) in 2014. 58 Some initiatives, such as the Online Brazilian Mutation Repository (Abraom), have been established in Brazil. 59 Incorporating precision medicine, data science, and AI may improve the cost-efficiency of many healthcare system processes. 60
Infrastructure and Quality Programs
Molecular testing lacks regulation in LATAM countries, posing patient risks because of inconsistent laboratory practices. Regional accreditation and proficiency testing programs in local languages are needed to improve the quality of molecular data generation. Collaborations among stakeholders, including the pharmaceutical industry, academia, medical associations, healthcare institutions, and the local public, can optimize resources for test implementation, drug development, and clinical trials for BRAF-mutant melanoma in LATAM.
Multidisciplinary Tumor Board
Integrated discussion of patients' clinical and molecular findings with a multidisciplinary board of specialists in cancer care improves the quality of care. Challenges are imposed in LATAM due to insufficient numbers of specialists. Virtual tumor boards provide collaboration opportunities among molecular scientists, pathologists, and clinicians to offer team-based diagnosis of patient tumors. LATAM clinicians may improve diagnosis accuracy and bolster scientific collaboration using virtual tumor boards with central and reference center support.
Discussion
This review is from an expert panel of LATAM doctors who are treating patients in hospitals and clinics in the region. The region’s diversity and a panel composed of a select group of practitioners limited the scope of knowledge. However, our methodology included experts from a range of countries that included Argentina, Brazil, Chile, Colombia, and Mexico. The panel relied on published literature and personal experience diagnosing and treating BRAF-mutated melanoma in LATAM. This review adds to the information from published papers addressing the unmet needs of patients with BRAF-mutated melanoma in LATAM.13,61
Conclusion
A multi-stakeholder approach is vital to enhance outcomes for BRAF-mutated melanoma in LATAM. Pathology labs can improve efficiency through automation and digital pathology integration in laboratories and teaching these techniques to residents. Medical societies should provide guidelines, education, collaboration, and research promotion. Governments should prioritize equitable patient navigation and resource allocation. Regulatory agencies must expedite new treatments and regulate molecular testing. Precision medicine should be a national strategy. Please see Box 1 and 2 for detection, diagnosis, regulation, and treatment recommendations.


Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The organization and implementation of the consensus conference were carried out by the AHF, a 501(c)3 nonprofit organization dedicated to improving healthcare throughout the Latin American Region and was supported by an unrestricted grant from the Pfizer Foundation. The funder had no influence on the design, implementation, and content of this manuscript.
