Abstract
Background
Liquid biopsy, including the detection of circulating tumor cells (CTCs), has emerged as a promising tool for cancer diagnosis and monitoring. However, the prognostic value of CTCs in nasopharyngeal carcinoma (NPC) remains unclear due to the lack of phenotypic characterization. The expression of Excision Repair Cross-Complementation Group 1 (ERCC1) and CTCs epithelial-mesenchymal transition (EMT) have been associated with treatment efficacy. In this study, we aimed to evaluate the prognostic significance of ERCC1 expression on CTCs and their EMT subtypes before treatment in NPC.
Methods
We retrospectively analyzed 108 newly diagnosed locally advanced NPC patients who underwent CanPatrol™ CTC testing between November 2018 and November 2021. CTCs were counted and classified into epithelial, epithelial-mesenchymal hybrid, and mesenchymal subtypes. ERCC1 expression was divided into negative and positive groups. Clinical features and survival outcomes were analyzed.
Results
The positive rate of CTCs was 92.6% (100/108), with an ERCC1 positivity rate of 74% (74/100). Further analysis of the subtypes showed that positive ERCC1 on mesenchymal CTCs was associated with a later N stage (P = .01). Positive ERCC1 expression was associated with poor overall survival (OS; P = .039) and disease-free survival (DFS; P = .035). Further analysis of subtypes showed that the positive ERCC1 on mesenchymal-type CTCs was associated with poor OS (P = .012) and metastasis-free survival (MFS; P = .001).
Conclusion
Our findings suggest that ERCC1 expression on CTCs may serve as a new prognostic marker for NPC patients. Evaluating CTCs subtypes may become an auxiliary tool for personalized and precise treatment.
Plain language summary
Background
Liquid biopsy, including the detection of circulating tumor cells (CTCs), has emerged as a promising tool for cancer diagnosis and monitoring. However, the prognostic value of CTCs in nasopharyngeal carcinoma (NPC) remains unclear due to the lack of phenotypic characterization. The expression of Excision Repair Cross-Complementation Group 1 (ERCC1) and CTCs epithelial-mesenchymal transition (EMT) have been associated with treatment efficacy. In this study, we aimed to evaluate the prognostic significance of ERCC1 expression on CTCs and their EMT subtypes before treatment in NPC.
Methods
We retrospectively analyzed 108 newly diagnosed locally advanced NPC patients who underwent CanPatrol™ CTC testing between November 2018 and November 2021. CTCs were counted and classified into epithelial, epithelial-mesenchymal hybrid, and mesenchymal subtypes. ERCC1 expression was divided into negative and positive groups. Clinical features and survival outcomes were analyzed.
Results
The positive rate of CTCs was 92.6% (100/108), with an ERCC1 positivity rate of 74% (74/100). Further analysis of the subtypes showed that positive ERCC1 on mesenchymal CTCs was associated with a later N stage (P = .01). Positive ERCC1 expression was associated with poor overall survival (OS; P = .039) and disease-free survival (DFS; P = .035). Further analysis of subtypes showed that the positive ERCC1 on mesenchymal-type CTCs was associated with poor OS (P = .012) and metastasis-free survival (MFS; P = .001).
Conclusion
Our findings suggest that ERCC1 expression on CTCs may serve as a new prognostic marker for NPC patients. Evaluating CTCs subtypes may become an auxiliary tool for personalized and precise treatment.
Introduction
Nasopharyngeal carcinoma (NPC) is a malignancy that originates from the epithelial cells lining the nasopharyngeal mucosa. It exhibits a unique geographical distribution, with a high incidence observed in regions such as East and Southeast Asia, the Arctic, North Africa, and the Middle East.1,2 In 2020, the estimated global incidence of NPC reached 133 354 cases, with 85% occurring in Asia, resulting in 80 008 deaths. 3 In the cases of locally advanced NPC patients (those without metastasis), the 5-year survival rates post-treatment typically range between 70%–90%.2,4,5 However, a notable subset, accounting for 10%–25% of patients, experiences treatment failure characterized by poor outcomes such as distant metastasis and regional recurrence, which carry a bleak prognosis.6-8 Hence, the identification of biomarkers associated with treatment efficacy and prognosis is crucial.9,10
Circulating tumor cells (CTCs), being a liquid biopsy technique, have the capability to furnish precise insights pertaining to tumors.11,12 In contrast to the inherent risks associated with obtaining tumor tissue through traditional biopsies, such as bleeding and infection, liquid biopsy offers numerous advantages, including non-invasiveness, convenience, cost-effectiveness, and the ability to repeatedly sample. 13 Nevertheless, it is crucial to acknowledge that the manifestation of CTCs encompasses not solely quantitative variances but also functional heterogeneity within distinct cell subpopulations.14-16
The gene Excision repair cross-complementation group 1 (ERCC1) plays a pivotal role in nucleotide excision repair, responsible for repairing cellular damage induced by chemotherapy and radiotherapy in tumor cells. 17 However, it also promotes the survival of tumor cells following chemoradiotherapy, thereby elevating the likelihood of recurrence and metastasis. 18 Numerous prior investigations have consistently established a correlation between ERCC1 expression in primary tumors and the prognosis of patients with head and neck squamous cell carcinoma (HNSCC). 17 Nevertheless, there is currently a dearth of substantial evidence regarding the association between ERCC1 expression in CTCs and the prognosis. In current clinical practice, the epithelial-mesenchymal transition (EMT) subtype is the most widely accepted classification for CTCs subtyping. 27 EMT markers are pivotal in categorizing CTCs into distinct cellular phenotypes, including epithelial-type CTCs, hybrid-type CTCs, and mesenchymal-type CTCs. 19
In the present study, we evaluated the expression of the ERCC1 gene in the circulating CTCs within a cohort of treatment naïve laNPC patients. Besides, the current study explored the relationship between CTC EMT subtyping, ERCC1 expression, and their impact on clinical characteristics and prognosis.
Materials and Methods
Study Design and Patient Enrollment
In this retrospective study, we collected the clinical data from a cohort of 108 patients diagnosed with laNPC. The reporting of this study conforms to REMARK guidelines, 20 and have de-identified all patient details. These patients received treatment at the Department of Radiation Oncology, First Affiliated Hospital of Guangxi Medical University, from November 2018 to November 2021. The staging was performed using the eighth Edition of the Union for International Cancer Control/American Joint Committee (UICC/AJCC) TNM staging system. 21 Inclusion criteria encompassed the following: (1) a confirmed histopathological diagnosis of NPC, (2) classification as stage III-IVa according to the eighth UICC/AJCC staging system, (3) no prior history of anti-tumor treatment or other malignancies, (4) a Karnofsky Performance Score 22 ≥ 70) successful completion of prescribed radiotherapy regimen. Exclusion criteria comprised: (1) the presence of severe underlying diseases, such as cardiovascular conditions, uncontrolled diabetes, or mental disorders, and (2) radiotherapy interruptions or failure to adhere to the planned treatment due to unrelated reasons. This study adheres to the principles outlined in the Helsinki Declaration, and the research protocol was approved by the Ethics Committee of the First Affiliated Hospital of Guangxi Medical University (2023-E466-01) in September 07, 2023.
Treatment Regimen
A comprehensive assessment was conducted for all patients, including nasopharyngoscopy, magnetic resonance imaging (MRI) for nasopharynx and neck, chest computed tomography (CT), abdominal CT or ultrasound, bone scintigraphy (ECT), electrocardiogram, evaluation of Epstein-Barr virus (EBV) DNA copy number in peripheral blood, and blood biochemical tests. Positron emission tomography/computed tomography (PET/CT) was recommended to some patients, tailored to their clinical circumstances.
Treatment regimens for laNPC patients involved concurrent chemoradiotherapy combined with induction or adjuvant chemotherapy and complemented by immunotherapy and targeted therapy in some patients. Intensity-modulated radiation therapy (IMRT) technique was applied to all patients.
Isolation of CTCs
Peripheral blood samples were collected from patients before treatment using CanPatrol CTC technology.
The procedure of isolation of CTCs in peripheral blood involved the following steps: (1) Peripheral blood collection: A total volume of 10 mL of peripheral blood was collected from each patient using EDTA anticoagulant blood collection tubes (divided into 2 tubes). (2) Red blood cell lysis: 15 mL of lysis buffer (154 mM NH4Cl, 10 mM KHCO3, and .1 mM EDTA) was added and thoroughly mixed with the blood sample to remove the red blood cells. The mixture was left at room temperature for 30 minutes. Subsequently, a benchtop low-speed centrifuge TDZ5-WS (Xiangyi, Hunan) was used to centrifuge the samples at 500 G for 5 minutes to remove the supernatant. (3) Preprocessing: The resulting cell pellet was resuspended in phosphate-buffered saline (PBS) (Sigma, St. Louis,USA), and fixed with 4% formaldehyde to achieve a final concentration. The fixation process was carried out for 8 minutes. (4) Nanotechnology filtration: The preprocessed samples were subjected to filtration through specialized tubes containing a membrane characterized by an 8 μm diameter pore size. A vacuum filtration pump (Aotesains, Tianjin) was employed. The filtration step effectively removes small leukocytes while retaining circulating CTCs on the membrane. The retained CTCs were further fixed with 4% formaldehyde at room temperature for 1 hour.
RNA In Situ Hybridization Assay
Capture Probe Sequences.

Sequences for the bDNA Signal Amplification Probes.

The EMT Subtype and the ERCC1 Expression on CTCs
We employed 2 distinct classification methodologies, namely EMT classification and ERCC1 expression classification, to classify CTCs.
Previous research highlights the pivotal role of EMT markers in categorizing CTCs into various cellular phenotypes, including epithelial-type CTCs (positivity for epithelial markers), hybrid-type CTCs (positivity for both epithelial and mesenchymal markers), and mesenchymal-type CTCs (exhibiting positivity for mesenchymal markers) .
23
To visualize the expression of epithelial and mesenchymal genes in CTCs, we represented epithelial gene expression with red fluorescent signals, mesenchymal gene expression with green fluorescent signals, and the leukocyte marker CD45 gene expression with white signals (Figure 1). Immunofluorescence images of CTCs in different EMT classifications and ERCC1 expression.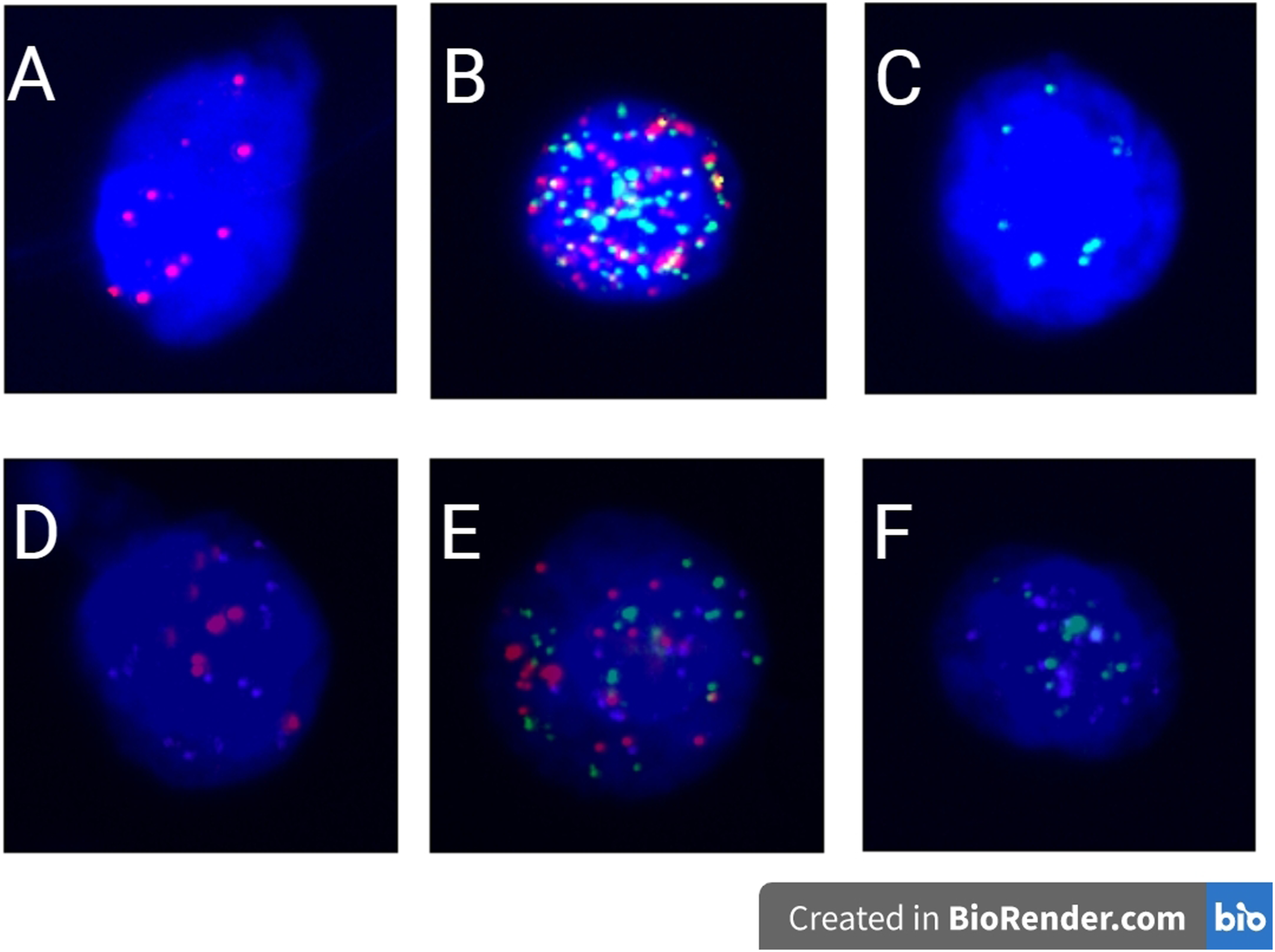
Concurrently, we examined the ERCC1 gene expression on each detected CTC, with purple signals indicating the expression of the ERCC1 gene (Figure 1). A fluorescent signal of 1 or higher are considered positive expression. Based on the fluorescence signal data, CTCs were categorized into 6 types (Figure 1): (1) Epithelial-type and ERCC1-negative CTCs (red fluorescence); (2) Hybrid-type and ERCC1-negative CTCs (red + green fluorescence); (3) Mesenchymal-type and ERCC1-negative CTCs (green fluorescence); (4) Epithelial-type and ERCC1-positive CTCs (red + purple fluorescence); (5) Hybrid-type and ERCC1-positive CTCs (red + green + purple fluorescence); (6) Mesenchymal-type and ERCC1-positive CTCs (green + purple fluorescence).
The red fluorescent signal dots represent epithelial markers (EpCAM, CK8/18/19). The green fluorescent signal dots represent mesenchymal markers (vimentin and twist). The white fluorescent signal dots represent leukocyte markers (CD45). The purple fluorescent signal dots represent ERCC1 gene markers. A represents epithelial-type CTCs and ERCC1 negative, B represents mixed-type CTCs and ERCC1 negative, C represents mesenchymal-type CTCs and ERCC1 negative. D represents epithelial-type CTCs and ERCC1 positive, E represents mixed-type CTCs and ERCC1 positive, F represents mesenchymal-type CTCs and ERCC1 positive.
Assessment of Treatment Response
Efficacy evaluations were performed on all patients both upon completion of treatment and 3 months post-treatment, utilizing the same diagnostic imaging modalities employed during initial diagnosis (including CT, MRI, US, and PET) to assess treatment response. The Response Evaluation Criteria in Solid Tumors (RECIST) were employed to categorize efficacy, distinguishing between complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). 24
Follow-Up of Study Subjects
Following the completion of treatment, study participants were scheduled a follow-up strategy with visits every 3 months over 2 years. The routine visits encompassed thorough physical examinations, blood biochemical tests, nasopharyngoscopy, MRI scans of the head and neck, chest CT, ECT, and abdominal ultrasound. Additional evaluations, such as pathological biopsy or PET/CT scanning were performed if any abnormalities were detected. Subsequently, follow-up appointments were adjusted to every 6 months in the period of 2 to 5 years after treatment completion.
Statistical Analysis
Statistical analysis was carried out using SPSS Statistics version 23.0 (IBM Corp., Chicago). The differences between groups categorized by clinical characteristics were tested using the chi-square test. Survival analysis was executed via the generation of Kaplan-Meier survival curves, with an assessment conducted using log-rank. In all statistical analyses, significance was determined at a threshold of P < .05.
Result
Patient Characteristics
Clinical Characteristics and the Expression of ERCC1 on CTCs of the NPC Patients.

Abbreviations: ERCC1,Excision Repair Cross-Complementation Group 1;CTCs, circulating tumor cells.
Expression of ERCC1 on CTCs and Clinical Characteristics
CTC EMT-Subye and ERCC1expression.

Abbreviations: ERCC1, Excision Repair Cross-Complementation Group 1; CTCs, circulating tumor cells.
Within this subgroup of 100 CTC-positive patients, the ERCC1 positive rate within this subgroup was determined to be 74% (74/100). In intergroup analysis, results suggest no significant associations were observed between ERCC1 expression and clinical characteristics, including gender, age, T stage, N stage, TNM stage, and smoking status (Table 3). ERCC1 expression was evaluated for each CTC EMT subtype. Notably, ERCC1 expression on mesenchymal-type CTCs demonstrated a significant association with the N stage of patients (P = .01). However, ERCC1 expression on epithelial and hybrid-type CTCs showed no significant association with the N stage of patients (P > .05). Besides, no significant association was observed between ERCC1 expression and other clinical features such as gender, age, T stage, TNM stage, and smoking status (Table 3).
Expression of ERCC1 on CTCs and Treatment Response
Among the cohort of 100 patients who tested positive for CTCs, 47 patients (47%) achieved a CR while 53 patients (53%) achieved a PR at the completion of treatment. Furthermore, 3 months post-treatment, 80 patients (80%) achieved a CR while 20 patients (20%) achieved a PR.
Correlation Between Clinical Efficacy and the Expression of ERCC1 on CTCs of the NPC Patients.

Abbreviations: ERCC1, Excision Repair Cross-Complementation Group 1; CR, complete response; PR, partial response; CTCs, circulating tumor cells.
Furthermore, a subgroup analysis was conducted to explore the potential relationship between ERCC1 expression on EMT subtype of CTCs and treatment response. The results indicated that at the completion of treatment, the CR rate in the ERCC1-positive subgroup of mesenchymal-type CTCs (8/29, 27.6%) was lower than that in the ERCC1-negative subgroup (39/71, 54.9%), with a statistically significant difference (P = .016). Similarly, 3 months post-treatment, the ERCC1-positive subgroup had a lower CR rate (19/29, 65.5%) compared to the ERCC1-negative subgroup (61/71, 85.9%), with a statistically significant difference (P = .028). No evidence of a correlation between ERCC1 expression and treatment response was observed in the epithelial-type and hybrid-type CTCs (Table 5).
The Expression of ERCC1 on CTCs and Prognosis
The 100 CTCs-positive patients were followed with a median follow-up duration of 32 months (ranging from 10 to 58 months). The observed rates of 3-year overall survival (OS), disease-free survival (DFS), recurrence-free survival (RFS), and metastasis-free survival (MFS) were 89%, 82%, 93%, and 85%, respectively.
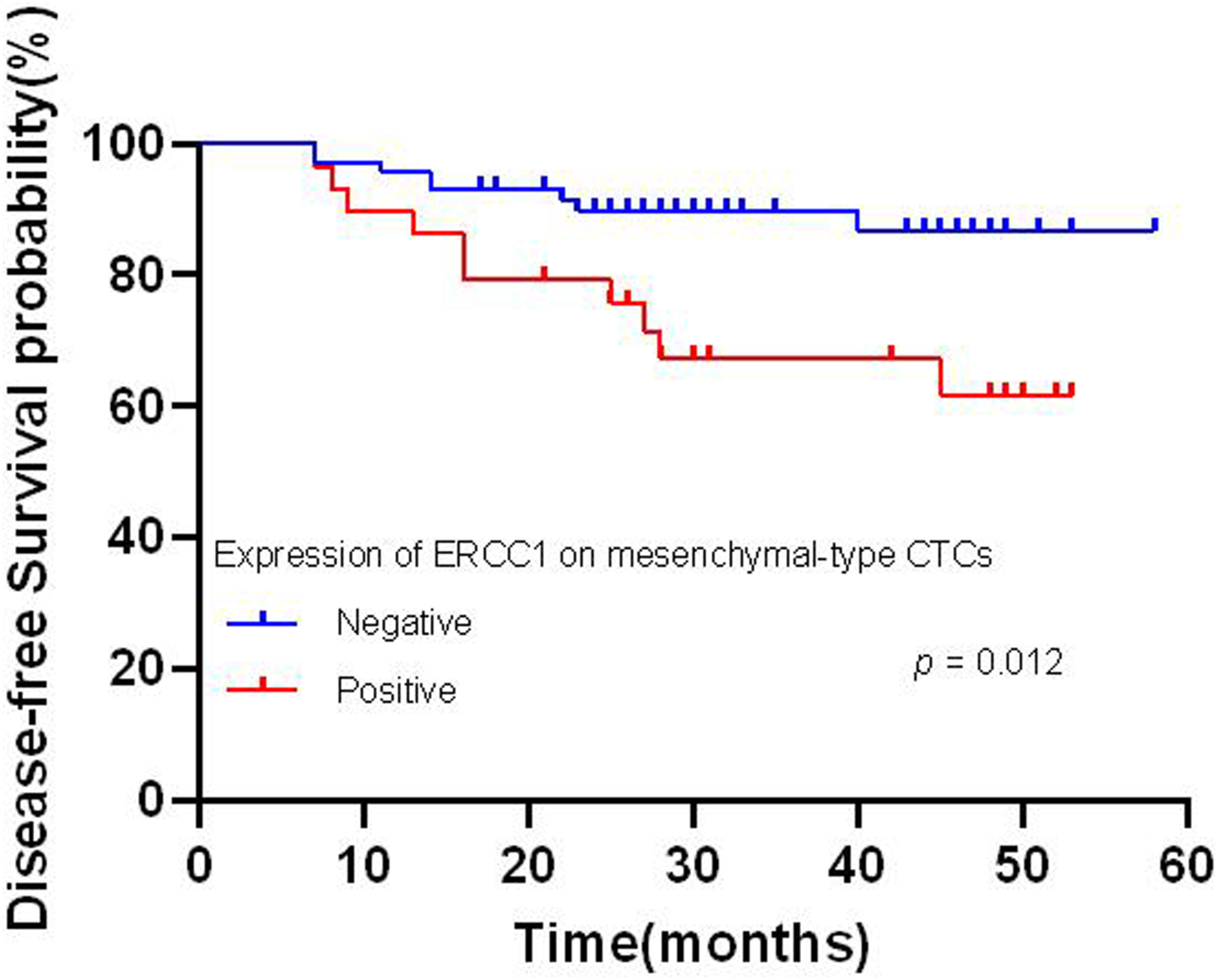
Further analysis was performed to explore the association between ERCC1 expression on CTCs and patient prognosis. We found a noteworthy association between ERCC1 expression on CTCs and worse OS (P = .039) (Figure 2), and DFS (P = .035) (Figure 3). No significant associations were found between ERCC1 expression and RFS or MFS. The association between ERCC1 expression and prognosis in EMT subtype was estimated. Results revealed significant associations between ERCC1 expression and poorer DFS (P = .012) (Figure 4), as well as MFS (P = .001) (Figure 5) in mesenchymal-type CTCs. Conversely, no significant association was observed with OS and RFS. Furthermore, no significant association was detected between T stage, N stage, TNM stage, ERCC1 expression on epithelial and hybrid-type CTCs and patient prognosis (Table 6). Kaplan–Meier’s survival curves of expression of ERCC1 on CTCs with over survival. Kaplan–Meier’s survival curves of expression of ERCC1 on CTCs with disease-free survival. Kaplan–Meier’s survival curves of expression of ERCC1 on mesenchymal-type CTCs with disease-free survival. Kaplan–Meier’s survival curves of expression of ERCC1 on mesenchymal-type CTCs with metastasis-free survival. Kaplan-Meier Analysis on Prognostic Factors of the NPC Patients. Abbreviations: ERCC1, Excision Repair Cross-Complementation Group 1; CTCs, circulating tumor cells.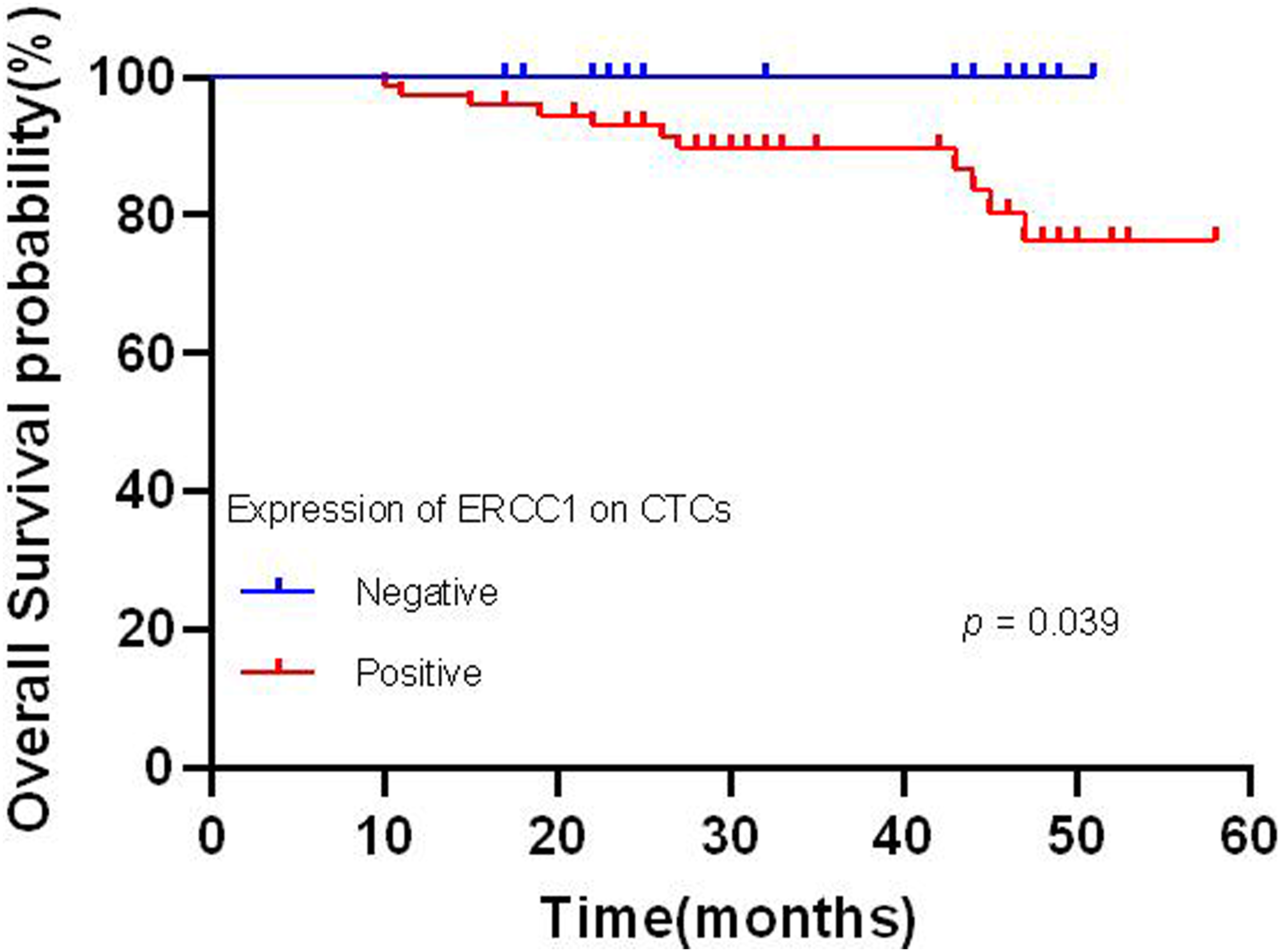



Discussion
In recent years, there have been notable advancements in the radiotherapy techniques, ushering in a new era of precision medicine in managing NPC. Nevertheless, developing personalized medicine by analyzing the genetic characteristics in liquid biopsies, such as CTCs, saliva, or urine in NPC, remains limited. Consequently, identifing accurate and effective biomarkers of genetic features in liquid biopsy is crucial to improve treatment outcomes and enhance the quality of life for NPC patients.9,25
The current study introduces a novel molecular biomarker, ERCC1, detected on CTCs in NPC before anti-cancer treatment. ERCC1, an integral component of the nucleotide excision repair system, is pivotal in multiple DNA repair pathways.26,27 Bišof et al conducted a meta-analysis on a sample size of 1288 patients, encompassing 1427 tissue samples. The results of this analysis revealed associations between ERCC1 expression and OS (HR = 2.72, 95 % CI: 1.79-4.13, P = .000) in NPC patients according to subgroup analyses. 17 Nevertheless, the biopsy procedure for primary tumor tissue presents inherent risks such as bleeding and infection, limiting the practical use of ERCC1 in clinical settings.
In current study, we conducted an investigation into the correlation between ERCC1 subtyping on CTCs and the clinical characteristics of patients. The findings indicated that the prevalence of ERCC1 on patients with N2-3 (83%) was higher compared to those with N0-1 (66%), although the P-value exceeded the threshold of .05 and did not achieve statistical significance. This observation is intriguing, as previous research has demonstrated the crucial role of ERCC1 in various DNA repair pathways, including homologous recombination, interstrand crosslink repair, and the mending of DNA double-strand breaks.27,28 Moreover, it has been implicated in evading recognition and elimination by the immune system during disease progression, often being associated with later clinical manifestations.27,28 Nevertheless, the present study did not find a noteworthy correlation between ERCC1 expression on CTCs. In the course of our investigation, we proceeded to categorize CTCs based on EMT markers and explored ERCC1 expression in different subtypes. The findings indicated that the prevalence of mesenchymal-type ERCC1 in patients with N2-3 (43%) was significantly greater than that in patients with N0-1 (17%), establishing a statistically significant disparity. However, this relationship was not observed in epithelial and hybrid types. These results align with previous research, as Wu et al have previously suggested, based on their comprehensive analysis of multiple studies, that advanced stages often exhibit the highest occurrence of mesenchymal CTCs. 23 This may be due to the acquisition of mesenchymal properties by tumor cells through EMT, which includes characteristics such as loss of cell polarity, reduced cell adhesion, induced cell migration, and increased intravascular permeability, significantly contributes to the progression and metastasis of malignant tumors.29,30 Remarkably, mesenchymal-type CTCs exhibit enhanced invasiveness due to their acquisition of mesenchymal phenotypic traits.30,31
No similar phenomenon was observed in the staging analysis of T stage and TNM classification. This could be due to the fact that the eighth UICC/AJCC TNM staging system is primarily defined based on the attributes of “Tumor" (T), “Nodes" (N), and “Metastasis" (M), which primarily describe the anatomical extent of the tumor. 21 Specifically, “T" delineates the extent of local invasion of the primary tumor, “N" indicates the presence and extent of regional lymph node metastasis, and “M" describes the presence of distant metastasis. The migratory behavior observed in mesenchymal-type CTCs is more strongly correlated with the N stage rather than the T stage. It is important to note that the TNM staging system considers not only the N stage, but also the combined assessment of T, N, and M stages. Consequently, investigating the association with the T stage and TNM classification alone may yield unfavorable outcomes.
In present study, we examined the association between ERCC1 expression and treatment response. Our findings revealed that patients expressing ERCC1 exhibited a lower CR rate compared to those without ERCC1 expression, with this difference being statistically significant. Notably, in survival analysis, we observed a significant association between elevated ERCC1 expression on CTCs and unfavorable OS and DFS. Our findings are consistent with those of previous scholars. Chebouti et al demonstrated that in ovarian cancer, the presence of ERCC1-positive CTCs post-chemotherapy is associated with platinum resistance (P = .01), as well as diminished PFS (P = .03) and OS) (P < .01). 32 Similarly, Das et al reported that in patients with metastatic non-small-cell lung cancer (NSCLC) undergoing platinum-based chemotherapy, a low expression of ERCC1 on CTCs correlates with prolonged PFS. 33 This association could be attributed to ERCC1’s role in repairing DNA damage induced by platinum-based treatment and actively mending DNA double-strand breaks caused by radiotherapy. 34 Consequently, tumor cells with heightened ERCC1 expression display enhanced survival capabilities following treatment, leading to an unfavorable prognosis.35,36 These findings are consistent with previous research on ERCC1 expression in primary tumors of NPC, 37 suggesting the potential utility of CTCs as a surrogate for assessing ERCC1 expression, circumventing the need for primary tumor tissue testing. Further analysis of CTCs revealed that increased ERCC1 expression on mesenchymal-type CTCs was associated with a lower CR rate and adverse outcomes in terms of DFS and MFS. These results may be stem from the heightened invasive and migratory characteristics exhibited by mesenchymal-type CTCs. 19 In the present study on survival analysis, the results suggest that T stage, N stage, and clinical TNM stage do not demonstrate prognostic significance. This outcome may be influenced by several factors, including the administration of immune maintenance or adjuvant chemotherapy primarily to stage IV patients, while stage III patients did not receive similar treatments.
This observation has prompted us to reflect on CTC applications’ present state and prospects. Currently, in clinical practice, the Food and Drug Administration (FDA) has approved the application of Cell Search technology to enumerate CTCs, primarily for stratifying patients with breast, colorectal, and prostate cancer. 38 Consequently, researchers have been investigating the integration of CTCs as novel biomarkers into the NPC staging system. Nevertheless, most previous research has predominantly concentrated on enumerating CTC counts, leaving the clinical application potential of CTCs in NPC largely unexplored. To better uncover the potential of CTCs, a paradigm shift is necessary, transitioning from simplistic enumeration toward meticulous molecular characterization. 39 Currently, numerous biomarkers for prognostic assessment in NPC have reached a relatively mature stage, 40 The ongoing investigation pertains to the examination of molecular markers present on CTCs in NPC. Thus far, researchers have identified a limited number of markers, namely COX-2, MMP-9, and FN1, all of which have demonstrated associations with patient prognosis.16,41-43 This study posits that ERCC1 may potentially emerge as a novel molecular marker, supplementing the existing markers. Nevertheless, the translation of CTC-based biomarkers into clinical applications remains a formidable and protracted endeavor.
In the present study, we acknowledge that the first-time documentation of ERCC1 expression on CTCs in NPC was analyzed only, without measuring the correlation between the ERCC1 expression patterns in primary tumor tissues and CTCs. This unresolved matter necessitates additional confirmation through subsequent studies involving paired samples of primary tumor and CTCs. Moreover, there has been extensive research conducted on various potential biological markers, such as CTCs, 23 EBV-related markers, 44 microRNAs, 45 and hypermethylation of tumor suppressor genes. 46 However, this study primarily focuses on exploring CTCs, while the discussion regarding EBV-related markers remains limited. This limitation stems from the fact that a significant proportion (92%) of the examined patients exhibited EBV-related markers, which lacks an adequate number of negative controls. Additionally, the study is a retrospective analysis conducted at a single center with a restricted sample size, particularly in subgroup analyses, necessitating cautious interpretation of the outcomes. The observation of ERCC1 has been limited to pre-treatment, with no examination of its dynamic fluctuations during and post-treatment to assess their influence on prognosis. More data are essential to reinforce future investigations. Consequently, further multicenter studies are indispensable to validate our discoveries and establish comprehensive clinical conclusions.
Conclusion
In summary, this study represents a comprehensive exploration of the clinical relevance of the molecular marker ERCC1 on CTCs in NPC. Our findings suggest ERCC1 expression on CTCs may offer a novel prognostic marker for NPC patients. Besides, examining CTC subtypes shows potential as an assistant for tailoring personalized and precision treatment strategies.
Supplemental Material
Supplemental Material - Prognostic Significance of Excision Repair Cross-Complementation Group 1 on Circulating Tumor Cells for Nasopharyngeal Carcinoma
Supplemental Material for Prognostic Significance of Excision Repair Cross-Complementation Group 1 on Circulating Tumor Cells for Nasopharyngeal Carcinoma by Ting Liu, Yuanqing Li, Junmei Song, Bo Li
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Youth Foundation of China (82202939, to T. Huang), and China Postdoctoral Science Foundation (2022MD723765, to T. Huang).
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
