Abstract
Introduction
Information about survival outcomes in metastatic biliary tract cancer (BTC) is sparse, and the numbers often quoted are based on reports of clinical trials data that may not be representative of patients treated in the real world. Furthermore, the impact of more widespread adoption of a standardized combination chemotherapy regimen since 2010 on survival is unclear.
Methods
We performed an analysis of the Surveillance, Epidemiology, and End Results database to determine the real-world overall survival trends in a cohort of patients with metastatic BTC diagnosed between the years 2000 and 2017 with follow-up until 2018. We analyzed data for the entire cohort, evaluated short-term and long-term survival rates, and compared survival outcomes in the pre-2010 and post-2010 periods. Survival analysis was performed using the Kaplan–Meier method, and Cox proportional hazard models were used to evaluate factors associated with survival.
Results
Among 13, 287 patients, the median age was 68 years. There was a preponderance of female (57%) and white (77%) patients. Forty-one percent died within 3 months of diagnosis (short-term survivors) and 20% were long-term survivors (12 months or longer). The median overall survival (OS) for the entire cohort was 4.5 months. Median OS improved post-2010 (4.5 months) compared to pre-2010 (3.5 months) (P < .0001). On multivariate analysis, age <55 years, intrahepatic cholangiocarcinoma, surgical resection, and diagnosis post-2010 were associated with lower hazard of death.
Conclusion
The real-world prognosis of metastatic BTC is remarkably poorer than described in clinical trials because a large proportion of patients survive less than three months. Over the last decade, the improvement in survival has been minimal.
Keywords
Introduction
In the United States, biliary tract cancers (BTCs) involving the gall bladder, extrahepatic, perihilar, and intrahepatic bile ducts are rare. Approximately 15,000 new cases are diagnosed yearly. 1 Despite the small numbers, the prognosis is grim. Five-year survival for resectable BTC is about 20%–35%. However, only about 25% of BTC presents at a resectable stage and given a 70% 5-year recurrence rate, an argument can be made that most BTCs are incurable.2–5
Literature is surfeit with data on the long-term outcomes and prognostic determinants of resectable BTC.2,6,7 These data, generated from hospital-based registries and public databases across decades, give us a reliable picture of the clinical outcomes in this cohort of BTC. In contrast, the real-world clinical outcome data for metastatic BTC is limited. Information is drawn mainly from the ABC-02 trial. This reported a median survival of 11.6 months with the combination of gemcitabine and cisplatin (gem-cis). 8 The addition of immune checkpoint inhibitor, Durvalumab, to this combination improved median survival to 12.8 months. 9 A scattering of single-institution studies suggests similar outcomes.10–12 It is recognized that clinical trial data overestimate the real survival of patients with metastatic disease, as these trials tend to enroll a robust and motivated patient population compared to patients treated in everyday practice. This potentially hides the true severity of the disease and impairs our understanding of the scale of the problem. For example, in metastatic pancreatic cancer, another recalcitrant cancer similar to BTC, an analysis of the Surveillance, Epidemiology, and End Results (SEER) database revealed a median survival of only two months. 13 This contrasts with clinical trial data that suggests a median survival of 9–11 months.14,15
We hypothesized that a similar situation prevails in metastatic BTC. We therefore sought to determine real-world trends in the survival of metastatic BTC by analyzing the SEER database. We evaluated short- (less than 3 months) and long-term (at least 12 months) survival of the subtypes of BTC and compared survival outcomes before and after publication of the pivotal ABC-02 study (2010) and increased adoption of gemcitabine and cisplatin as standard palliative chemotherapy.
Patients and Methods
Data Source and Study Population
We utilized case records from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) registry (18 registries, including Atlanta, Connecticut, Detroit, Hawaii, Iowa, New Mexico, San Francisco-Oakland, Seattle-Puget Sound, Utah, Alaska Native Tumor Registry, San Jose-Monterey, Los Angeles, rural Georgia, greater California, Kentucky, Louisiana, New Jersey, and greater Georgia) for the years 2000–2018. Representing approximately 28% of the U.S. population, and having wide geographic coverage, the registry is a quality data source for cancer statistics. SEER is a population-based cancer registry and commonly used for retrospective observational cohort studies. We chose the year 2000 as our starting point for the analysis to obtain 10 years of data prior to 2010 when the ABC-02 trial results were published. 8 We used de-identified patient-level data; thus, our study was granted exempt status by the Institutional Review Board of the University.
The biliary tract cancer cases were selected based on the TNM_7_CS_ v0204 schema recode in the SEER database. We identified all cases coded as “BileDuctsDistal,” “BileDuctsIntraHepat,” “BileDuctsPerihilar,” and “Gallbladder.” For the final analysis, we included distant cases based on “SEERCombinedSummaryStage2000200 code 7.” We also abstracted information on age at diagnosis, sex, race/ethnicity, marital status, year of diagnosis, anatomic location (perihilar bile duct cases and distal cholangiocarcinoma were categorized together as extrahepatic bile duct cancers), and whether patients had previously undergone surgery for BTC (recurrent disease). Missing values for covariates were excluded in the analysis. All the covariates had a missing rate of less than 5%.
Survival Outcomes
Our main outcome of interest was overall survival (OS). We accounted for the vital status and time to event (i.e., time from the cancer diagnosis to death, censoring, or last follow-up) measured in months for the examination of the survival outcomes. The SEER database reports survival in complete months and rounds down fractions to the nearest month. Additionally, cases with survival <30.4 days are accorded 0 survival month. To account for this artifact, we assigned a survival time of .5 months for cases coded with 0 survival time and added .5 months across the board to all survival times.13,16 The survival outcome analysis assessed the differences in the two groups: diagnosis before 2010 and diagnosis after 2010. We allowed at least 12 months for the follow-up post-diagnosis; thus, our study included patients with the diagnosis years from 2000 with last follow-up on December 31, 2018. For the patients who were diagnosed in 2017, we included their data throughout 2018 to allow the least 12-month follow-up. We chose to exclude cases diagnosed in 2010 to avoid indeterminate impact of the milestone ABC-02 clinical trial.
We defined long-term survivors as patients who lived for 12 months or longer following diagnosis of BTC, while short-term survivors were defined as those that lived for less than three months.
Statistical Analysis
We used descriptive analyses to examine the demographic and clinical characteristics of the patients. Comparisons of the patient characteristics between subgroups (e.g., patients surviving for less than 3 months vs patients surviving for 12 months or more) were assessed by standard statistical tests. The
The Kaplan–Meier method was used to investigate the survival outcomes within these two periods and the log-rank test to examine the differences between the two periods. Also, we conducted multivariable analyses using Cox proportional hazards regression modeling the OS. The effect of the diagnosis period before or after 2010 on the OS was examined with the presence of all covariates. To assess the proportional hazard assumption for the Cox models, we used Schoenfeld residual analysis. Because we assigned a survival time of .5 months for cases coded with 0 survival time, we conducted sensitivity analysis which excluded patients with 0 month survival. The results from the sensitivity analyses were congruent with results for the primary analysis except for patients with ECC.
We conducted all analyses in SAS 9.4 (SAS, Cary, NC). A P-value less than .05 was considered statistically significant.
Results
Patient Characteristics
Characteristics of Patients Diagnosed With Metastatic Biliary Tract Cancer From 2000 to 2018.

P-value for the comparison between the two groups: OS < 3 months group and OS ≥12 months group. SD, standard deviation.
aAmerican Indian/Alaskan/Asian Pacific ethnicity.
bExtrahepatic = distal hepatic and perihilar bile duct.
Short- and Long-Term Survivors
Forty-one percent of patients (n = 5475) were short-term survivors, and there were 2683 (20.2%) long-term survivors (Table 1). Compared to short-term survivors, long-term survivors were younger, with a median age of 64 years vs 72 years (P < .0001). They were also more likely to be married/with a domestic partner 65.1% vs 50% (P < .0001) and less likely to be separated/divorced or widowed 22.3% vs 34.2% (P < .0001). There were no differences in the racial distribution of short- and long-term survivors.
The proportion of short-term survivors decreased after 2010 (39%) compared to the pre-2010 period (45.2%). Conversely, the proportion of long-term survivors increased post-2010 to 21.4% compared to 17.9% pre-2010.
Overall Survival and Survival Trends
In the entire cohort, OS improved marginally between 2000 and 2017. This improvement in survival appears to be largely driven by those younger than 65 years and was most apparent with intrahepatic cholangiocarcinoma. (Figure 1). OS hovered around four months from 2004 to 2009, and around 5 months from 2011 to 2017. With the exclusion of short-term survivors, the survival numbers moved from around 8 months from 2004 to 2009 to 9 to 10 months from 2011 to 2017 (Supplementary Figures 1(A) and 1(B)). Kaplan–Meier survival estimates of patients for the entire population (A) and for those who survived ≥3 months (B).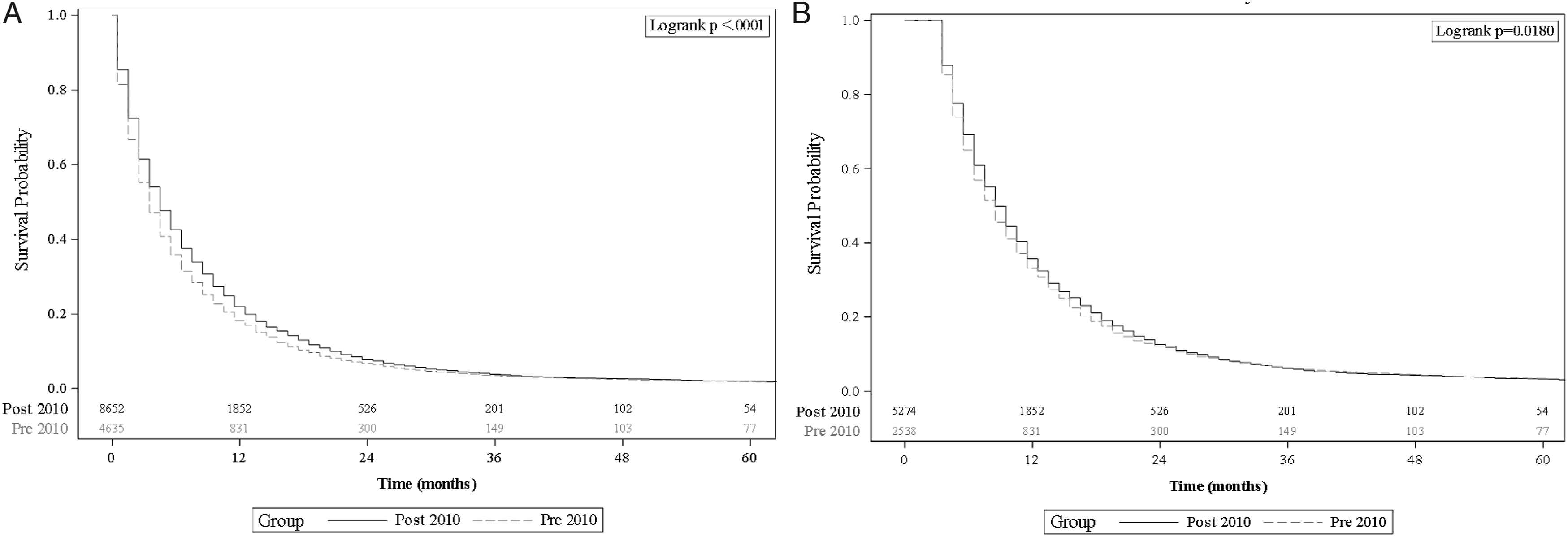
The median OS for the entire cohort was 4.5 months (range .5–222.5 months). The median OS among short-term survivors was 1.5 months (range .5–2.5 months), while median OS for long-term survivors was 19.5 months (range 12.5–222.5 months) (Table 1). There was a statistically significant improvement in median OS from 3.5 months pre-2010 to 4.5 months post-2010 (P < .0001) (Figure 1(A)). This improvement in median survival across the two time periods was not consistent across age groups. There was a 2-month improvement in median OS (5.5 months vs 7.5 months, P < .0003) among those <55 years and 1-month improvement among those >65 years at diagnosis (2.5 vs 3.5 months, P < .0001). However, there was no significant improvement in OS among 55–65-year-olds (P = .472). There was also a 1-month improvement in median OS among anatomic categories (P = .0006 for GBC, P = .046 for EHC and P < .0001 for ICC) pre- and post-2010 (Figures 2 and 3). When we excluded short-term survivors (41.2%), the median OS increased to 8.5 months and improved by 2 months from 7.5 months pre-2010 to 9.5 months post-2010 (P < .018) (Figure 1(B), Supplementary Figure 2(A)). Similar to the data from the entire cohort, there was no improvement in OS across the two time periods in the 55–65-year-old cohort after exclusion of short-term survivors (P = .385). Interestingly, there was no significant difference in survival among the anatomic subtypes between the two time periods after the exclusion of short-term survivors (Supplementary Figure 3). In addition, there was no difference in OS among long-term survivors between the two time periods, with a median OS of 19.5 months pre- and post-2010 (P = .6929) (Supplementary Figure 4). Kaplan–Meier survival estimates (3 year) of patients showing differences pre- and post-2010 based on age at diagnosis. (A): all age groups; (B): diagnosis age <55 years; (C): diagnosis age 55–65 years; (D): diagnosis age >65 years. Kaplan–Meier survival estimates of patients showing differences pre- and post-2010 based on age at diagnosis. (A): all anatomic subtypes. (B): Gall bladder cancer. (C): Extrahepatic cholangiocarcinoma. (D): Intrahepatic cholangiocarcinoma.

Factors Associated With Survival
Multivariate Cox Regression Model Assessing the Effects of Patient Characteristics on Overall Survival for the Entire Cohort.
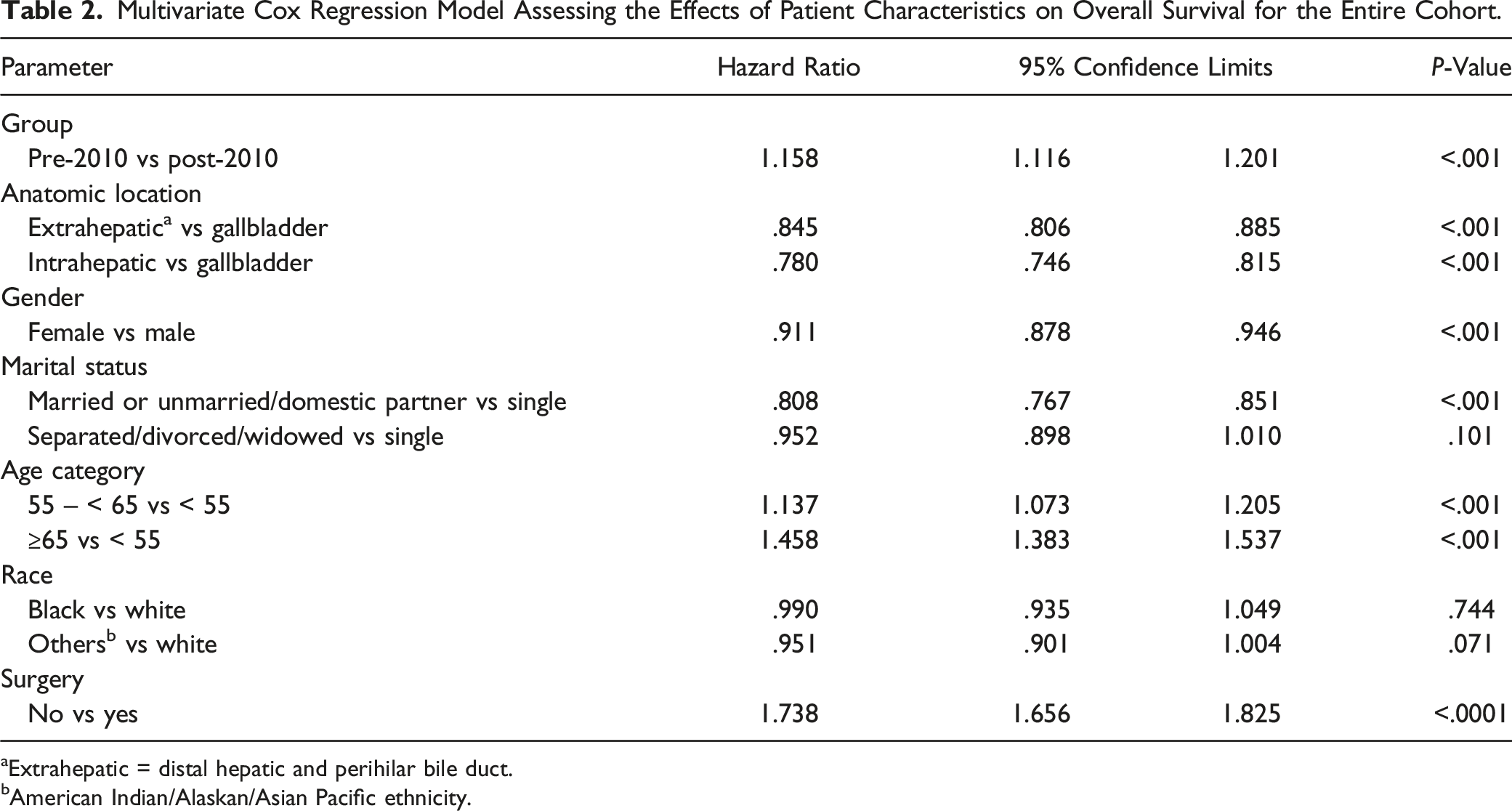
aExtrahepatic = distal hepatic and perihilar bile duct.
bAmerican Indian/Alaskan/Asian Pacific ethnicity.
Multivariate Cox Regression Model Assessing the Effects of Patient Characteristics on Overall Survival After Removing Short-Term Survivors.

aExtrahepatic = distal hepatic and perihilar bile duct.
bAmerican Indian/Alaskan/Asian Pacific ethnicity.
Multivariate Cox Regression Model Assessing the Effects of Patient Characteristics on Overall Survival for Long-Term Survivors.

aExtrahepatic = distal hepatic and perihilar bile duct.
bAmerican Indian/Alaskan/Asian Pacific ethnicity.
Discussion
Data on the real-world outcomes of the various subtypes of metastatic BTC are rare. Our analysis of the SEER database suggests that 41% of patients with metastatic BTC are alive for less than 3 months and that OS for all patients has only improved, from 3.5 months in the decade before 2010 to 4.5 months in the years after 2010. Although this represents a 22% improvement in survival rate, a 1-month improvement is clinically marginal and disappointing.
Survival estimates in our study are significantly lower than the often-quoted median survival for patients with BTC which is based on clinical trial and hospital-based registry data.10,17–19 It is recognized that data from clinical trials over-estimate survival compared to general clinical practice.20,21 Furthermore, the hospital-based data available are limited by relatively small sample size and cancer population heterogeneity, usually including patients with both locally advanced and metastatic disease. In addition, as opposed to population-based registries, hospital-based registry studies also tend to include a more robust patient population with Eastern Cooperative Group performance status of 0–2.
As previously noted, Golan and colleagues reported a median survival of only 2 months for metastatic pancreatic cancer patients reviewed in their SEER database analysis. 13 Others have reported a 3-month survival of 40%–60% for stage III and IV pancreatic cancer in the US, and European data suggests that in the modern era up to 45% of patients with pancreatic cancer do not survive up to 3 months.22,23 As far as we know, ours is the largest study to report population-based survival outcomes for metastatic BTC. Based on our analysis, a substantial proportion (41%) of patients with metastatic BTC do not survive up to 3 months and this cohort is responsible for the low survival numbers presented in this report. These patients are not represented in the discussions around the prognosis of BTC and are usually excluded from clinical trials. 24 Accordingly, when short-term survivors were excluded from our analysis, we noted an improvement in median survival to 8.5 months. This suggests that younger, more robust patients with potentially less aggressive biology are offered and get the most benefit from combination systemic therapy. In concordance with our results, Wang and colleagues, in their SEER database analysis of the metastasis patterns and the impact of chemotherapy in patients with metastatic BTC, also reported median OS between 8 and 9 months. 25 While it is not clear how they assessed the impact of chemotherapy based on SEER data, our study goes beyond their findings to assess the change in survival across almost 2 decades for the different subtypes of BTC.
The improvement in the survival of metastatic BTC post-2010 compared to the earlier decade is likely related, among other factors, to the improvements in management of metastatic BTC potentially occasioned by more widespread adoption of standardized combination chemotherapy, including gemcitabine and cisplatin. 26 This may also reflect improvements in diagnostic processes, adjunct treatments, and supportive care measures. Regardless of the underlying reasons, 1-month improvement in median OS is disappointing and underlines the extent of work that needs to be done to improve outcomes in metastatic BTC.
Our definition (determined prior to data analysis) of long-term survival at 12 months or more based on our clinical experience was justified, as only about 20% of patients fell into this category. On multivariate analysis, only ICC and surgical resection (suggesting low-volume disease) were associated with long-term survival. More interestingly, despite an increase in the proportion of long-term survivors post-2010, the period of diagnosis was not associated with long-term survival. This suggests that at least until 2018, the advancements in cancer diagnosis, management, and palliation have not impacted survival in this group. This raises the possibility that the biology of metastatic BTC in long-term survivors may be distinct. Based on more recent molecular information, this may be driven by a preponderance of tumors with fibroblast growth factor receptor (FGFR) genomic alterations which have a better prognosis. 27 The fact that a diagnosis of metastatic ICC was associated with long-term survival strengthens this point as this more favorable molecular profile is commoner in this anatomic subtype.28,29
Anatomical categorization of biliary tract cancers, though convenient, is inadequate. And it is becoming clearer that the molecular underpinnings of the different anatomic subtypes of BTC may play a role in the aggressiveness of each subtype and the potential response to systemic therapy. For example, compared to advanced ICC, metastatic ECC and GBC have worse prognosis. 30 As previously stated, the presence of FGFR fusion events limited only to ICC (about 15% of cases) portends better prognosis, and small molecule inhibitors targeting FGFR-2 fusions have been approved for management of this subtype of ICC based on significant improvements in objective response rates for chemotherapy refractory disease. 31 Alterations in the isocitrate dehydrogenase-1 (IDH 1) enzyme are reported in about 20% of ICC, and targeting this isoenzyme also provides a survival benefit after chemotherapy. 32 This and other molecular markers will allow more individualized treatment of BTC. On the other hand, a majority of BTCs do not carry a readily targetable molecular or chemical alteration and chemotherapy will remain an important component of the management of this subtype. Again, given the different molecular characteristics of different subtypes of BTC, it is important to query the optimal regimen for BTC subtypes. For example, ECC shares similar molecular alterations with pancreatic cancer (compared to ICC) with alterations in KRAS and TP53, 33 and it is possible that a fluoropyrimidine-based regimen may be more appropriate in the first line for ECC while a gemcitabine-based regimen may be better suited for ICC. The PRODIGE 38 study compared modified FOLFIRINOX to gem-cis in advanced and metastatic biliary tract cancer. The combination of gemcitabine and cisplatin provided superior PFS and OS over the experimental arm. Sixty percent of enrolled patients had ICC, and subgroup analysis showed that gem-cis was superior to mFOLFIRINOX in these patients.34,35 Also, the NIFE study compared 5-fluorouracil and liposomal irinotecan to gem-cis in a Phase II study. Responses to either combinations differed based on the anatomic subtype with improved responses and a progression-free survival benefit with 5FU and liposomal irinotecan in the ECC subset. 36 While this is interesting data, the numbers are small and are derived from subset analysis. More work needs to be done to validate this prospectively.
Our study shows a pronounced increase in the hazard of death among patients without surgery (HR 1.74, P < .001). The proportion of experiencing surgery for long-term survivors was also almost three times higher than short-term survivors. Surgical resection is not a standard practice with metastatic BTC. However, on a case-by-case basis, patients with low-volume, relatively slow growing disease may be offered resection. 37 The SEER dataset does not allow us a concrete understanding of the indications for resection for metastatic BTC.
Our study focused on patients with metastatic BTC, and this certainly contributed to the overall lower survival numbers reported. Reports of outcomes in advanced BTC often include patients with metastatic and locally advanced BTC. A previous retrospective study has reported poorer prognosis with metastatic BTC than locally advanced disease. 12 Furthermore, a post hoc analysis of studies of gem-cis in the ABC trials suggested an improvement in OS for ICC with liver-only disease compared to metastatic ICC although this was not statistically significant. 30 The goal of treatment in metastatic disease is generally palliative, and patients will most likely receive systemic therapy or best supportive care. However, in the locally advanced setting, depending on a variety of factors, patients may be offered liver-directed therapy in addition to systemic therapy with the goal of achieving a cure. Including this potentially more heterogenous group (in terms of extent of disease and treatments offered) would make it more difficult to establish real trends in survival especially after 2010 and the potential impact of standardized chemotherapy. We recognize that some patients with liver-only metastatic disease may receive locoregional therapy.
Another limitation of our study is its retrospective nature and the fact that the SEER database does not collect extensive information about the type of systemic therapy used. We are therefore unable to make any conclusions, only inferences, about a main sub-text of our analysis, that the adoption of standardized chemotherapy may have contributed to improvements in outcomes after 2010. Finally, based on the time period queried, our analysis does not capture the effect of recent advances in the management of BTC. The impact of adoption of capecitabine in the adjuvant setting based on the BILCAP trial, or the integration of FGFR2, IDH1-directed therapy, and immune checkpoint inhibitor therapy on real-world survival outcomes, will need to be assessed in the next few years.9,31,32
Conclusions
Among all patients with metastatic biliary tract cancers, there has only been a marginal improvement in median survival since the turn of the 21st century. About 4 in 10 patients die within 3 months of diagnosis. This represents a significant challenge and opportunity for improving outcomes in metastatic BTC. Clinical and research databases containing prospectively collected information are necessary for a more thorough clinicopathologic and molecular characterization of BTC. The understanding gained from analysis of these databases will be key to understanding short- and long-term survivors and improving outcomes of metastatic BTC.
Supplemental Materials
Supplemental Materials - Short- and Long-Term Survival of Metastatic Biliary Tract Cancer in the United States From 2000 to 2018
Supplemental Materials for Short- and Long-Term Survival of Metastatic Biliary Tract Cancer in the United States From 2000 to 2018 by Van Nghiem, Sarah Wood, Rekha Ramachandran, Grant Williams, Darryl Outlaw, Ravikumar Paluri, Young-il Kim, and Olumide Gbolahan in Cancer Control.
Footnotes
Acknowledgments
We acknowledge Drs Olatunji Alese, Neda Hashemi, and Abayomi Ogunwale for critical insights and comments during manuscript development.
Author Contributions
V.N and O.G contributed significantly to design of work, acquisition, analysis and interpretation of the data, and writing. R.R and Y.K contributed to data acquisition, analysis, interpretation, and writing. G.W, D.O, and R.P contributed significantly to the interpretation of data and writing. All authors contributed to the drafting and approved the final version to be published. All authors agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
